uric acid metabolism and insulin resistance
Reading File
Finding Sources
Finding Sources
Searching PubMed
"uric acid" AND "insulin resistance"
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Finding Sources
I now have comprehensive material from multiple authoritative textbooks. Let me compile the full answer.
Uric Acid Metabolism and Insulin Resistance
These two phenomena are deeply intertwined — each promotes the other, and both converge in the metabolic syndrome.
1. Uric Acid Metabolism: Overview
Uric acid is the end product of purine catabolism in humans. The pathway:
Purines → Hypoxanthine → Xanthine → Uric acid
The conversion of hypoxanthine to xanthine, and then to uric acid, is catalyzed by xanthine oxidase, an enzyme highly active in the liver and intestines. Humans (and other hominoids) uniquely lack uricase, the enzyme that would further oxidize uric acid to the highly soluble allantoin — a mutation that occurred ~18 million years ago. This makes uric acid accumulation possible in humans in a way that is not possible in most other mammals.
Under normal conditions:
- ~2/3 of daily urate turnover is excreted by the kidney
- ~1/3 is eliminated by the intestine (via ABCG2)
Uric acid is freely filtered at the glomerulus. The proximal convoluted tubule then reabsorbs ~90% of filtered urate through a complex set of transporters (URAT1, OAT1, OAT3, GLUT9/SLC2A9, ABCG2, NPT1, NPT4) — collectively called the transportasome.
In >90% of gout patients, hyperuricemia results from renal underexcretion, not overproduction. — Goldman-Cecil Medicine
2. How Insulin Resistance Causes Hyperuricemia
There are two major mechanisms:
A. Impaired Renal Urate Excretion
Insulin normally promotes uricosuria (urinary uric acid excretion). In insulin-resistant states, this effect is lost: the kidney retains more uric acid, raising serum urate levels.
"Hyperuricemia reflects defects in insulin action on the renal tubular reabsorption of uric acid." — Harrison's Principles of Internal Medicine, 22nd Ed.
Specifically, insulin resistance impairs the normal suppression of tubular urate reabsorption, leading to net urate retention.
B. Impaired Ammoniagenesis → Low Urine pH → Uric Acid Crystallization
Insulin resistance also impairs renal ammonium production and excretion. Ammonium is the main urinary buffer for hydrogen ions. When ammonium excretion falls, the kidney excretes proportionally more titratable acid and less citrate — resulting in a persistently low urine pH (typically <5.5).
Uric acid has a pKa of ~5.4. At low pH, urate exists predominantly in the undissociated, insoluble uric acid form rather than the soluble urate ion, dramatically increasing the risk of uric acid crystallization and stone formation. — Campbell-Walsh-Wein Urology
This was elegantly demonstrated by Abate et al., who used hyperinsulinemic euglycemic clamps to measure insulin sensitivity in normal subjects and uric acid stone formers: urinary pH inversely correlated with insulin resistance, and uric acid stone formers showed the most severe insulin resistance among all groups.
3. How Hyperuricemia Worsens Insulin Resistance
The relationship is bidirectional:
A. Endothelial Dysfunction
Uric acid adversely affects nitric oxide synthase (NOS) in the macula densa and stimulates the renin-angiotensin-aldosterone system (RAAS), contributing to hypertension. There is evidence that uric acid reduction normalizes blood pressure in hyperuricemic adolescents with hypertension. — Harrison's, 22nd Ed.
Elevated uric acid also increases asymmetric dimethylarginine (ADMA), an endogenous NOS inhibitor, further impairing endothelial function and reducing insulin-mediated vasodilation in muscle — a key pathway by which insulin promotes glucose uptake.
B. Oxidative Stress
Uric acid has paradoxical antioxidant effects extracellularly but pro-oxidant effects intracellularly — it activates NADPH oxidase, generating reactive oxygen species that impair insulin signaling cascades (particularly the PI3K-Akt pathway).
C. Inflammation
Hyperuricemia promotes proinflammatory cytokines (TNF-α, IL-1β, IL-6), which themselves cause insulin resistance.
4. The Fructose Connection
High-fructose corn syrup consumption drives both insulin resistance and uric acid production simultaneously. Fructose metabolism in the liver:
- Is insulin-independent, bypassing normal feedback controls
- Generates AMP → inosine → hypoxanthine as a byproduct, directly fueling xanthine oxidase activity and uric acid production
- Promotes de novo lipogenesis, dyslipidemia, and hepatic insulin resistance (MAFLD/MASH)
This explains the parallel epidemiological rise in gout, metabolic syndrome, obesity, and uric acid nephrolithiasis in Western populations. — Goldman-Cecil Medicine, Harrison's
5. Uric Acid in Metabolic Syndrome
Hyperuricemia is considered a component manifestation of the metabolic syndrome, reflecting pathway-specific insulin resistance. Uric acid stone formers share the full metabolic syndrome phenotype:
| Metabolic Syndrome Feature | Shared by Uric Acid Stone Formers |
|---|---|
| Obesity / visceral adiposity | ✓ |
| Hypertriglyceridemia | ✓ |
| Hyperglycemia / T2DM | ✓ |
| Hypertension | ✓ |
| Low urine pH / hyperuricemia | ✓ |
"Uric acid stone formers have been found to share many of the characteristic features of metabolic syndrome, including hypertriglyceridemia, hyperglycemia, obesity, and hypertension." — Campbell-Walsh-Wein Urology
Among patients with type 2 diabetes, 34–36% of kidney stones are pure uric acid stones, versus ~11% in non-diabetics. — Goldman-Cecil Medicine
6. Pathophysiology Diagram: Uric Acid Nephrolithiasis
The following diagram illustrates the three pathophysiologic mechanisms leading to uric acid nephrolithiasis, with obesity ↔ insulin resistance as the central driver of low urinary pH:
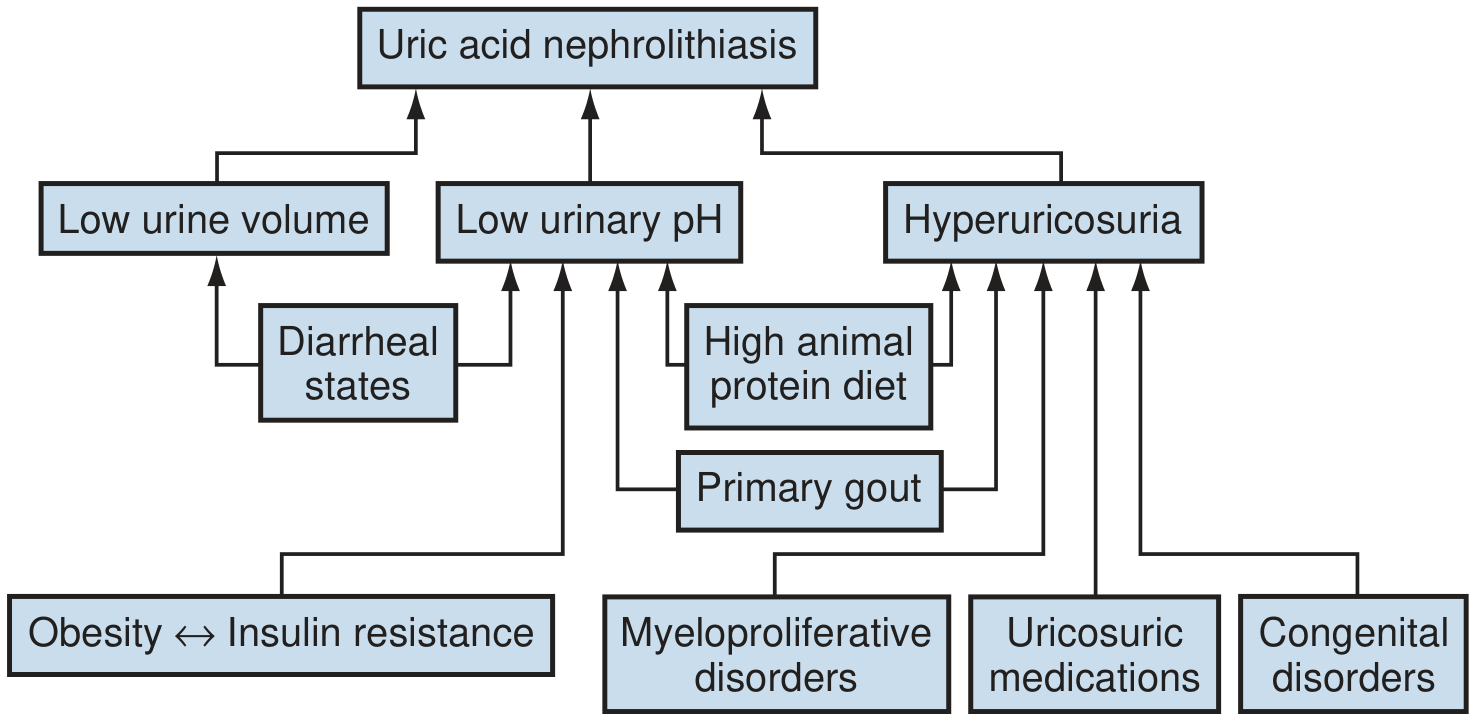
Fig. Pathophysiology of uric acid nephrolithiasis. Obesity ↔ insulin resistance drives low urinary pH through impaired ammoniagenesis, the most critical pathogenic factor. — Campbell-Walsh-Wein Urology
7. Clinical Implications
| Aspect | Clinical Relevance |
|---|---|
| Serum uric acid as biomarker | Elevated urate should prompt evaluation for metabolic syndrome, T2DM, hypertension |
| Uric acid stones | Treat with urinary alkalinization (potassium citrate to pH 6.0–6.5) + address insulin resistance |
| Xanthine oxidase inhibitors | Allopurinol/febuxostat reduce urate production; may also have anti-inflammatory metabolic benefits |
| Weight loss | 5–10% weight reduction improves insulin sensitivity, reduces hyperuricemia, raises urinary pH |
| Dietary fructose | Reduction of fructose intake addresses both insulin resistance and uric acid overproduction |
Sources:
- Harrison's Principles of Internal Medicine, 22nd Ed. — Metabolic Syndrome chapter
- Goldman-Cecil Medicine — Gout Pathobiology chapter
- Campbell-Walsh-Wein Urology — Uric Acid Nephrolithiasis chapters
- Brenner and Rector's The Kidney — Uric Acid Transport section
- Comprehensive Clinical Nephrology, 7th Ed.
insulin resistance
Finding Sources
Searching PubMed
"insulin resistance" AND mechanisms
Reading File
Reading File
Reading File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.