Tell me about glycognesis
Reading File
Finding Sources
Searching the Web
glycogenesis biochemistry pathway regulation
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of glycogenesis — the synthesis of glycogen from glucose.
Glycogenesis: Synthesis of Glycogen
Glycogen is the primary storage form of glucose in humans, found mainly in liver (for blood glucose homeostasis) and skeletal muscle (for local energy use). Glycogenesis occurs in the cytosol and requires energy from both ATP and UTP.
Step 1 — Glucose Activation to UDP-Glucose
Glucose must first be "activated" before it can be incorporated into glycogen:
- Glucose → Glucose 6-phosphate via hexokinase (muscle) or glucokinase (liver), using ATP
- Glucose 6-phosphate → Glucose 1-phosphate via phosphoglucomutase (glucose 1,6-bisphosphate is an obligatory intermediate)
- Glucose 1-phosphate + UTP → UDP-glucose + PPi via UDP-glucose pyrophosphorylase
- Pyrophosphate (PPi) is immediately hydrolyzed to 2 Pi by pyrophosphatase, making the reaction irreversible and driving UDP-glucose production
UDP-glucose is the active, high-energy glucosyl donor for all subsequent steps.
Step 2 — Primer Requirement (Glycogenin)
Glycogen synthase cannot initiate chains on free glucose — it requires a primer. The protein glycogenin (37 kDa) serves as the primer:
- Glycogenin auto-glucosylates itself at tyrosine-194 using UDP-glucose
- It then extends this chain to at least 8 glucose residues in α(1→4) linkage
- This oligosaccharide primer is then handed off to glycogen synthase
- Glycogenin remains permanently at the core of the glycogen granule
Step 3 — Chain Elongation by Glycogen Synthase
Glycogen synthase is the key regulated enzyme of glycogenesis:
- Transfers glucose from UDP-glucose to the nonreducing end of the growing chain
- Forms α(1→4) glycosidic bonds, releasing UDP
- Can only elongate existing chains — hence the need for the glycogenin primer
Step 4 — Branch Formation
Without branching, the result would be an unbranched chain (amylose). Glycogen is highly branched, which:
- Greatly increases water solubility
- Multiplies nonreducing ends (sites for both synthesis and degradation), enabling much faster mobilization
Branching enzyme (amylo-α(1→4)-α(1→6)-transglycosylase) transfers a block of 6–8 glucosyl residues from the nonreducing end to an internal residue via an α(1→6) linkage, creating a branch point. Branches occur roughly every 8 glucosyl residues.
Pathway Diagram
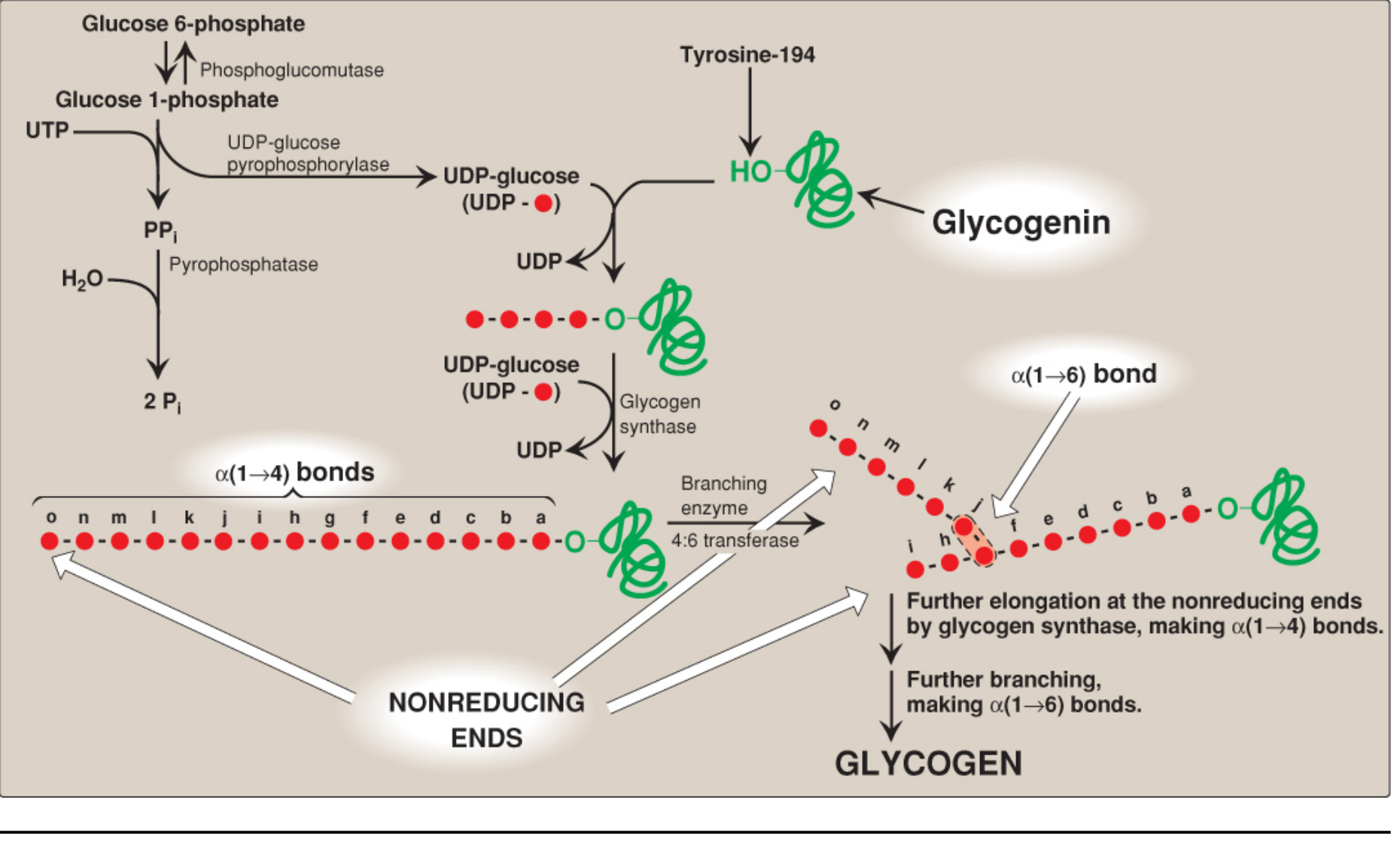
Lippincott Biochemistry 8e, Figure 11.5 — Full glycogen synthesis pathway
Regulation of Glycogenesis
Regulation occurs at two levels: hormonal (covalent) and allosteric.
Hormonal (Covalent) Regulation
| State | Hormone | Effect on Glycogen Synthase |
|---|---|---|
| Fed / high insulin | Insulin | Activates glycogen synthase (promotes dephosphorylation via PP1) |
| Fasted / stress | Glucagon, Epinephrine | Inactivates glycogen synthase (via PKA-mediated phosphorylation) |
- Active glycogen synthase "a" = dephosphorylated form
- Inactive glycogen synthase "b" = phosphorylated form (at multiple sites; the more phosphorylated, the more inhibited)
- This is opposite to glycogen phosphorylase (which is active when phosphorylated)
The mechanism: glucagon/epinephrine → GPCR → adenylyl cyclase → ↑cAMP → PKA activation → glycogen synthase phosphorylation → glycogenesis inhibited
Insulin reverses this by activating protein phosphatase-1 (PP1), which dephosphorylates and activates glycogen synthase.
Allosteric Regulation
| Effector | Effect |
|---|---|
| Glucose 6-phosphate (high, well-fed state) | Allosterically activates glycogen synthase b → promotes glycogenesis |
| Glucose 6-phosphate (high) | Also inhibits glycogen phosphorylase → prevents glycogenolysis simultaneously |
| ATP (high energy state) | Inhibits glycogen phosphorylase (favors glycogenesis) |
| AMP (low energy, muscle only) | Activates glycogen phosphorylase b without phosphorylation |
Tissue Context
| Tissue | Primary stimulus for glycogenesis | Purpose |
|---|---|---|
| Liver | Post-meal hyperglycemia, insulin | Buffer blood glucose |
| Skeletal muscle | Rest after exercise, insulin | Local fuel reserve |
Summary of Key Enzymes
| Enzyme | Reaction | Notes |
|---|---|---|
| Hexokinase / Glucokinase | Glucose → G6P | Low Km (HK) vs. high Km (GK) |
| Phosphoglucomutase | G6P ↔ G1P | Reversible, G1,6-BP intermediate |
| UDP-glucose pyrophosphorylase | G1P + UTP → UDP-glucose + PPi | Not a regulatory step |
| Glycogenin | Self-glucosylation, primer formation | Core protein of glycogen granule |
| Glycogen synthase | UDP-glucose → α(1→4) chain | Rate-limiting regulated enzyme |
| Branching enzyme | α(1→4) → α(1→6) transfer | Creates branch points every ~8 residues |
Sources: Lippincott Illustrated Reviews: Biochemistry 8e, pp. 377–392; Harper's Illustrated Biochemistry 32e, pp. 184–185
This is a shared conversation. Sign in to Orris to start your own chat.