I have to write a 10 mark answer: A 25 year old male presents with tender xanthomas and a family history of premature cardiovascular disease. His LDL cholesterol levels are markedly elevated. (A) Discuss the metabolism of lipoproteins with emphasis on LDL metabolism (6M) (B) Explain the above biochemical defect and its clinical consequence (2M) (C) Add a note on diagnostic and therapeutic approaches of hyper lipoprotienemia (2M)
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
I now have all the material needed. Let me compose the complete 10-mark answer.
10-Mark Answer: Lipoprotein Metabolism, Familial Hypercholesterolemia, and Hyperlipoproteinemia
(A) Metabolism of Lipoproteins with Emphasis on LDL Metabolism (6 Marks)
Overview of Lipoproteins
Lipoproteins are macromolecular complexes that transport hydrophobic lipids (triacylglycerol, cholesterol esters, phospholipids) through the aqueous plasma. Each particle has a hydrophobic core surrounded by a hydrophilic shell of phospholipids, free cholesterol, and apolipoproteins. The major classes — chylomicrons, VLDL, IDL, LDL, and HDL — are distinguished by density, size, lipid composition, and apolipoprotein content.
1. Exogenous (Dietary) Pathway — Chylomicron Metabolism
Dietary lipids are absorbed in the small intestine, re-assembled into chylomicrons within enterocytes, and secreted into the lymphatics.
- Chylomicrons contain apo B-48, apo C-II, and apo E.
- In the capillaries of adipose and muscle, lipoprotein lipase (LPL) — activated by apo C-II — hydrolyses the core triacylglycerol (TAG), releasing free fatty acids for cellular uptake.
- As >90% of TAG is degraded, the particle shrinks into a chylomicron remnant. The C apolipoproteins (but not apo B-48 or apo E) are returned to HDL.
- Chylomicron remnants are taken up by the liver via receptor-mediated endocytosis mediated by apo E, through the LDL (apo B-100/E) receptor and LDL receptor-related protein-1 (LRP-1). Lysosomal hydrolysis releases amino acids, free cholesterol, and fatty acids; the receptor is recycled.
(Lippincott Illustrated Reviews: Biochemistry, 8e, p. 644)
2. Endogenous Pathway — VLDL and IDL Metabolism
The liver produces VLDL to export endogenous TAG to peripheral tissues.
- Nascent VLDL contains apo B-100 and acquires apo C-II and apo E from circulating HDL.
- LPL in peripheral tissues hydrolyses VLDL TAG, progressively reducing the particle to IDL (intermediate-density lipoprotein).
- IDL may be:
- Taken up directly by the liver via the LDL (apo B-100/E) receptor, or
- Further processed by hepatic lipase → LDL
- Importantly, each LDL particle is derived from a single precursor VLDL particle, as only one molecule of apo B-100 is present and is conserved throughout the cascade. (Harper's Illustrated Biochemistry, 32e, p. 263)
3. LDL Metabolism — Receptor-Mediated Endocytosis ⭐
LDL is the primary cholesterol-carrying lipoprotein in humans, containing ~70% of total plasma cholesterol. Its metabolism is the central focus of this question.
Step-by-step LDL receptor pathway:
| Step | Event |
|---|---|
| [1] | LDL binds to the LDL receptor (a negatively charged glycoprotein) via apo B-100 in clathrin-coated pits on the cell surface |
| [2] | The LDL–receptor complex is internalized by endocytosis as a coated vesicle |
| [3] | The vesicle loses its clathrin coat and fuses with endosomes |
| [4] | Endosomal ATPase lowers pH → LDL dissociates from the receptor; the receptor recycles to the cell surface |
| [5] | LDL is transferred to lysosomes → hydrolysed by lysosomal acid hydrolases → releases free cholesterol, amino acids, fatty acids, and phospholipids |
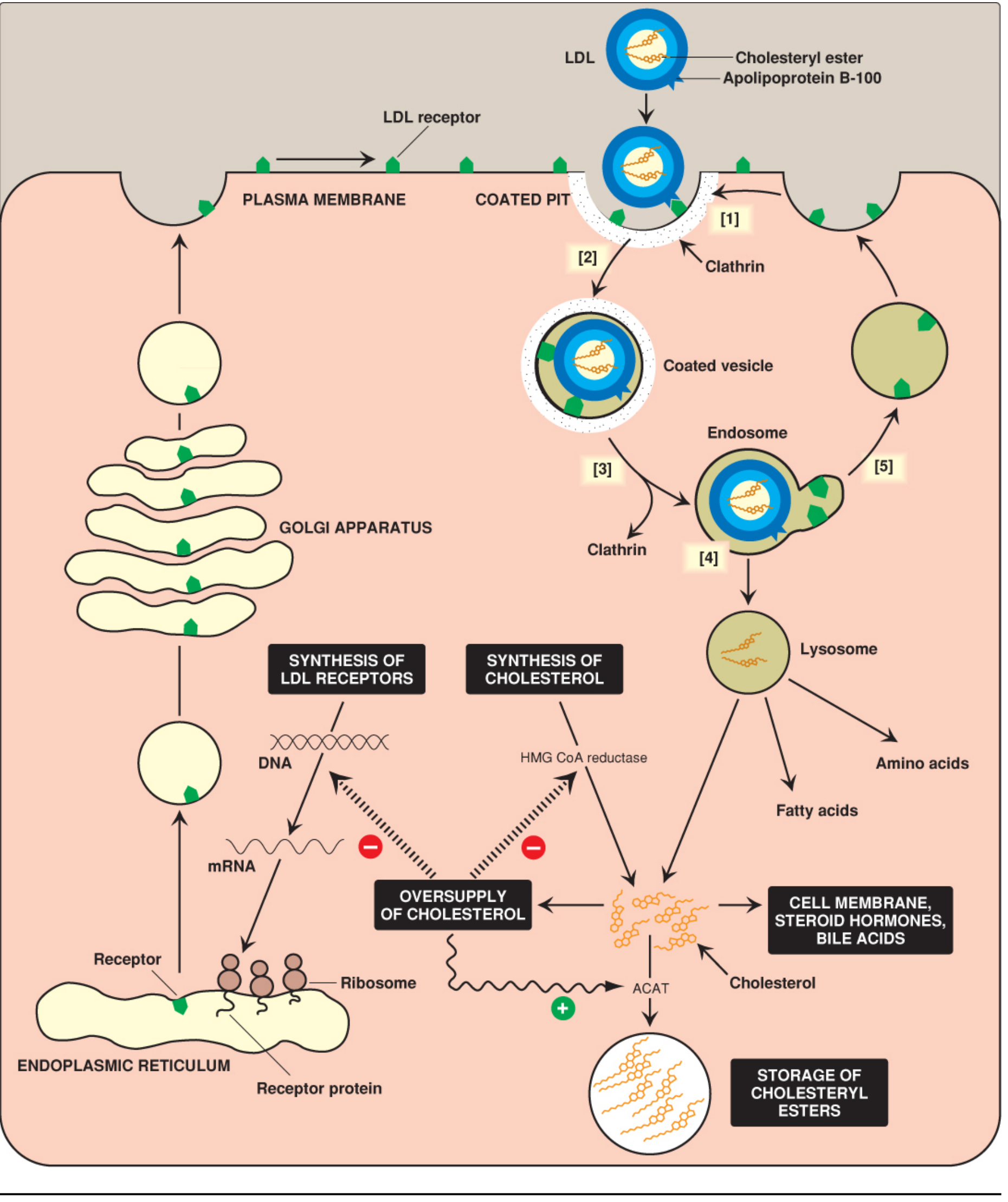
Figure: Cellular uptake and degradation of LDL particles (Lippincott Illustrated Reviews: Biochemistry, 8e, Fig. 18.21)
Cholesterol homeostasis consequences of LDL uptake:
- Suppression of HMG-CoA reductase — excess intracellular cholesterol inhibits transcription of the gene for HMG-CoA reductase (via the SREBP-2 pathway), decreasing de novo cholesterol synthesis; the enzyme is also more rapidly degraded.
- Downregulation of LDL receptors — reduced LDL receptor gene expression via the SRE/SREBP-2 system limits further cholesterol entry, preventing overaccumulation.
- ACAT activation — excess cholesterol is esterified by acyl-CoA:cholesterol acyltransferase (ACAT) and stored as cholesteryl esters in the cytoplasm.
PCSK9 regulation: The protein PCSK9 binds the LDL receptor and targets it for lysosomal degradation rather than recycling, thereby reducing the number of active receptors on the cell surface. (Harper's Illustrated Biochemistry, 32e, p. 275)
Scavenger receptor pathway: Macrophages possess scavenger receptors (SR-A) that bind and internalize oxidised LDL without feedback regulation. This leads to uncontrolled cholesterol ester accumulation, transforming macrophages into foam cells — the hallmark of early atherosclerotic plaque. (Lippincott Illustrated Reviews: Biochemistry, 8e, p. 652)
4. Reverse Cholesterol Transport — HDL Metabolism
HDL acts as a repository of apo C and apo E needed in VLDL/chylomicron metabolism, and performs reverse cholesterol transport (RCT): removing excess cholesterol from peripheral cells → esterification by LCAT → transport to the liver for bile acid synthesis or biliary secretion. This is the basis of HDL's protective role against atherosclerosis.
(B) Biochemical Defect and Clinical Consequences (2 Marks)
Diagnosis: Familial Hypercholesterolemia (FH)
This 25-year-old male with tendinous xanthomas, markedly elevated LDL-C, and a family history of premature cardiovascular disease has classic Familial Hypercholesterolemia (FH) — an autosomal dominant disorder.
Molecular Defect
The most common cause is loss-of-function mutations in the LDL receptor gene (LDLR) on chromosome 19. Other causes include:
- Mutations in apo B-100 (familial defective apoB) — reduce LDL binding affinity to its receptor
- Gain-of-function mutations in PCSK9 — increase receptor degradation, reducing LDL clearance
Heterozygotes (frequency ~1 in 250–500): produce ~50% of normal LDL receptors → plasma LDL-C typically >190 mg/dL.
Homozygotes (~1 in 1 million): virtually no functional LDL receptors → plasma LDL-C >500 mg/dL. (Basic Medical Biochemistry, 6e, p. 1209)
Homozygotes (~1 in 1 million): virtually no functional LDL receptors → plasma LDL-C >500 mg/dL. (Basic Medical Biochemistry, 6e, p. 1209)
Clinical Consequences
| Feature | Mechanism |
|---|---|
| Tendinous xanthomas | Cholesterol deposition in tendons (Achilles, extensor tendons of hands) |
| Xanthelasma | Cholesterol deposits in periorbital skin |
| Corneal arcus | Lipid deposition in corneal stroma (before age 45 = pathological) |
| Premature atherosclerosis | Elevated circulating LDL → oxidative modification → foam cell formation → plaque → coronary artery disease |
| Ischemic heart disease | Significantly increased risk; homozygotes may have MI in childhood |
FH is the most common single-gene cause of premature ASCVD. Heterozygotes are at greatly increased risk of coronary artery disease; homozygotes have severe, often fatal, ischemic heart disease in childhood or early adulthood. (Robbins & Kumar Basic Pathology, p. 151; Harrison's Principles of Internal Medicine, 22e, p. 3286)
(C) Diagnostic and Therapeutic Approaches in Hyperlipoproteinemia (2 Marks)
Diagnosis
1. Lipid Profile (Fasting):
- Total cholesterol, LDL-C, HDL-C, triglycerides
- LDL-C >190 mg/dL in adults should prompt FH evaluation
2. Classification — Fredrickson/WHO Classification:
| Type | Lipoprotein Elevated | Lipid | Example |
|---|---|---|---|
| I | Chylomicrons | ↑↑ TAG | LPL deficiency |
| IIa | LDL | ↑ Cholesterol | Familial hypercholesterolemia |
| IIb | LDL + VLDL | ↑ Cholesterol + TAG | Familial combined hyperlipidemia |
| III | IDL/chylomicron remnants | ↑ Both | Familial dysbetalipoproteinemia (apo E2) |
| IV | VLDL | ↑ TAG | Familial hypertriglyceridemia |
| V | VLDL + Chylomicrons | ↑↑ TAG | Combined |
3. Clinical Criteria (Dutch Lipid Clinic / Simon Broome criteria):
- Family history of premature CVD
- Tendinous xanthomas
- LDL-C levels above age-/sex-specific thresholds
- Genetic testing for LDLR, APOB, PCSK9 mutations confirms diagnosis
4. Additional Tests:
- Coronary artery calcium (CAC) score for risk stratification
- Thyroid function (to exclude secondary causes)
- Liver function, renal function (to exclude secondary hyperlipidemia)
Therapeutic Approaches
Lifestyle Modification (First-line):
- Low saturated fat, high fibre diet
- Weight reduction, aerobic exercise
- Smoking cessation
Pharmacotherapy (Step-wise approach):
| Drug Class | Mechanism | Example |
|---|---|---|
| Statins (first-line) | Inhibit HMG-CoA reductase → ↑ LDL receptor expression → ↑ LDL clearance | Atorvastatin, rosuvastatin |
| Ezetimibe | Inhibits intestinal cholesterol absorption (NPC1L1 transporter) | Used as add-on if statin alone insufficient |
| PCSK9 inhibitors | Monoclonal antibodies that block PCSK9 → prevent LDL receptor degradation → ↑ receptor recycling | Evolocumab, alirocumab |
| Bile acid sequestrants | Bind bile acids in gut → ↑ bile acid synthesis from cholesterol → ↓ hepatic cholesterol | Cholestyramine, colesevelam |
| Bempedoic acid | Inhibits ATP-citrate lyase (upstream of HMG-CoA reductase) | Used in statin-intolerant patients |
Many FH patients require combination therapy — high-intensity statin + ezetimibe ± PCSK9 inhibitor — to achieve LDL-C targets (<70 mg/dL in high-risk patients). Homozygous FH may require LDL apheresis (extracorporeal removal of LDL). (Harrison's Principles of Internal Medicine, 22e, p. 3287)
Summary Table
| Question Part | Key Concept |
|---|---|
| (A) LDL metabolism | Chylomicrons → remnants; VLDL → IDL → LDL; LDL receptor-mediated endocytosis; cholesterol homeostasis via SREBP-2; scavenger receptor/foam cells |
| (B) FH defect | LDLR mutation → impaired LDL uptake → markedly elevated LDL-C → xanthomas + premature atherosclerosis |
| (C) Diagnosis/Rx | Lipid profile, Fredrickson classification, genetic testing; statins → ezetimibe → PCSK9 inhibitors → apheresis |
Sources: Lippincott Illustrated Reviews: Biochemistry, 8e; Harper's Illustrated Biochemistry, 32e; Basic Medical Biochemistry: A Clinical Approach, 6e; Robbins & Kumar Basic Pathology; Harrison's Principles of Internal Medicine, 22e
This is a shared conversation. Sign in to Orris to start your own chat.