Management of parkinson disease
Reading File
Finding Sources
Searching PubMed
"Parkinson disease"[MeSH Terms] AND "management"[MeSH Terms]
Finding Sources
Searching PubMed
"Parkinson disease" AND treatment
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have everything needed for a comprehensive answer. Here is the full management guide for Parkinson's disease.
Management of Parkinson's Disease
Overview
Parkinson's disease (PD) is the second most common progressive neurodegenerative disorder after Alzheimer's disease, affecting ~1% of people over 60 and 4–5% over 85. It results from loss of dopaminergic neurons in the substantia nigra and formation of Lewy bodies. The cardinal features are resting tremor, rigidity, and bradykinesia of asymmetric onset, plus postural instability.
Key principle: Drug therapy should be initiated only when symptoms are significant enough to cause functional impairment. Current therapies are symptomatic — none has been clearly shown to slow disease progression.
Pharmacological Management
The diagram below shows the sites of action of anti-Parkinson drugs:
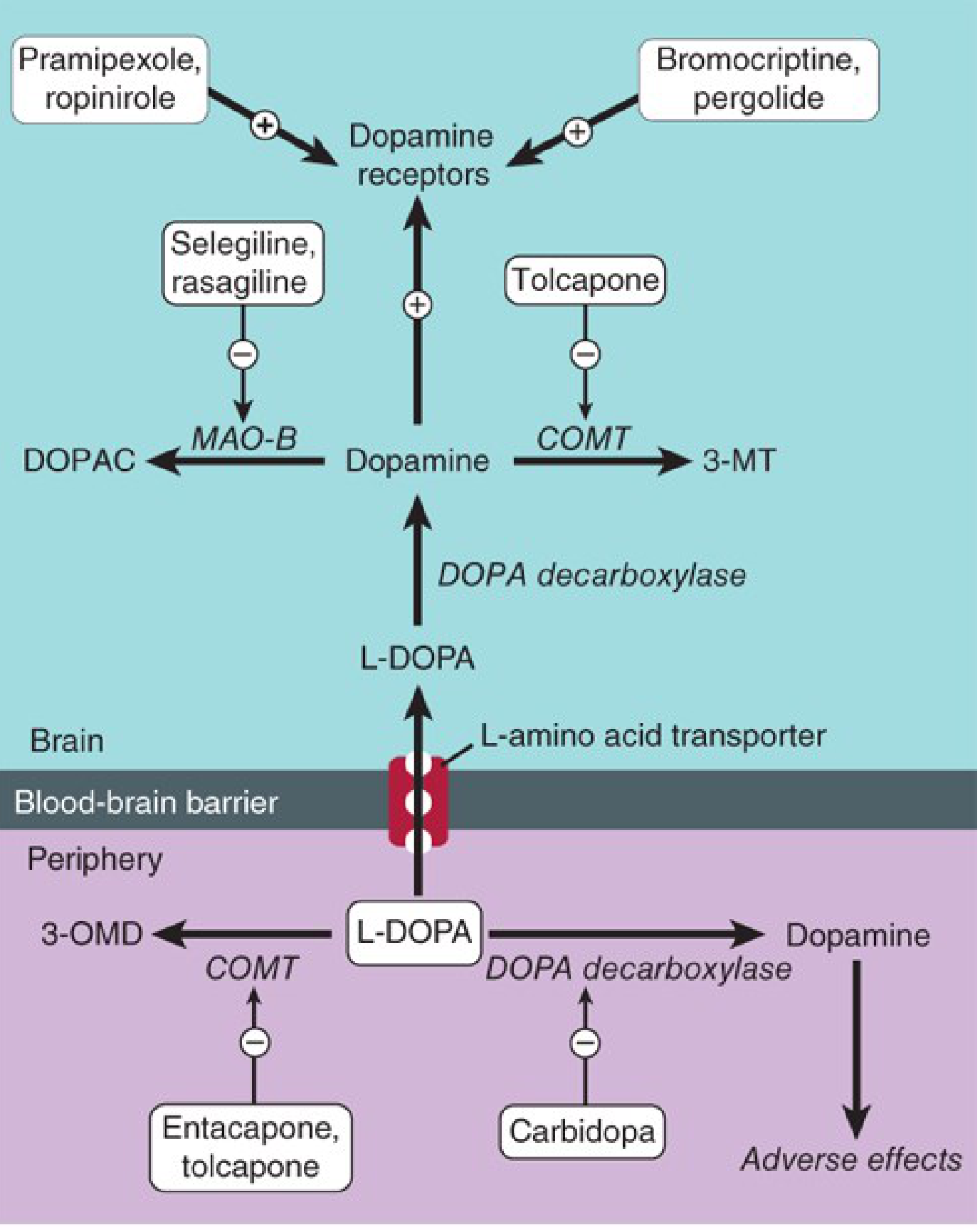
1. Levodopa + Carbidopa (Gold Standard)
Levodopa is the most effective agent, particularly for bradykinesia and rigidity. It is a dopamine precursor that crosses the blood-brain barrier.
- Carbidopa is a peripheral DOPA decarboxylase inhibitor that cannot cross the BBB. It prevents peripheral conversion of levodopa → dopamine, reducing side effects (nausea, vomiting, hypotension) and increasing CNS availability.
- Carbidopa saturates at 70–100 mg/day.
- Starting dose: Carbidopa/levodopa 25 mg/100 mg TID, increasing by ½ tablet every 4–7 days.
- Available as:
- Immediate release (Sinemet)
- Controlled release (Sinemet-CR) — 1 tablet BID
- Combined with entacapone (Stalevo)
Long-term complications:
| Complication | Description |
|---|---|
| Motor fluctuations | "Wearing-off" — benefit wanes before next dose |
| On-off phenomenon | Unpredictable swings between mobile ("on") and rigid/akinetic ("off") states |
| Dyskinesias | Involuntary choreiform movements at peak dose |
| Neuropsychiatric | Confusion, hallucinations (most common dose-limiting side effect) |
Side effects of levodopa include nausea, vomiting, dystonias, hallucinations, dyskinesias, and motor fluctuations. Hallucinations become the most common side effect limiting titration.
Contraindications: Psychosis; angle-closure glaucoma; active peptic ulcer (use caution); history of melanoma (monitor regularly). Do NOT combine with MAO-A inhibitors.
2. Dopamine Receptor Agonists
Act directly on postsynaptic dopamine receptors. Advantages over levodopa:
- No enzymatic conversion needed
- No toxic metabolites
- Do not compete with amino acids for transport
- Associated with lower incidence of motor fluctuations and dyskinesias with long-term use
Used as first-line monotherapy in early/mild disease OR as adjuncts to levodopa in advanced disease to smooth response fluctuations.
| Drug | Type | Dosing | Notes |
|---|---|---|---|
| Pramipexole | Non-ergot, D2-preferential | Start 0.125 mg TID; titrate to 0.5–1.5 mg TID | Extended-release available (once daily); may help affective symptoms; renally cleared |
| Ropinirole | Non-ergot, D2 agonist | Start 0.25 mg TID; titrate to 8–24 mg/day | Extended-release available; generic available |
| Rotigotine | Non-ergot | Transdermal patch | Useful for patients with swallowing issues |
| Apomorphine | Potent D1/D2 agonist | SC injection (1–6 mg PRN) | Rescue therapy for disabling "off" periods; requires antiemetic pretreatment |
| Bromocriptine | Ergot, D2 | 7.5–30 mg/day | Rarely used now due to side effect profile |
| Pergolide | Ergot, D1+D2 | — | No longer available in US (valvular heart disease risk) |
Common side effects: Nausea, somnolence, hallucinations, impulse control disorders (pathological gambling, hypersexuality), peripheral edema, sudden sleep attacks (pramipexole, ropinirole — must warn patients re: driving).
Contraindications: History of psychosis, recent MI, active peptic ulcer. Ergot agonists — avoid in peripheral vascular disease.
3. MAO-B Inhibitors
Selectively inhibit monoamine oxidase B → reduce dopamine breakdown → enhance and prolong levodopa effect.
| Drug | Dose | Use |
|---|---|---|
| Selegiline (deprenyl) | 5 mg with breakfast + 5 mg with lunch | Adjunctive; may reduce wearing-off; avoid later in day (insomnia) |
| Rasagiline | 1 mg/day (monotherapy) or 0.5–1 mg/day (adjunct) | First-line monotherapy for mild disease; adjunct for advanced disease |
| Safinamide | 50 mg/day → 100 mg/day after 2 weeks | Adjunct only (NOT effective as monotherapy); reduces off-periods |
⚠ Drug interactions (serious): Do NOT combine with meperidine, tramadol, methadone, propoxyphene, cyclobenzaprine, dextromethorphan, St. John's wort. Caution with TCAs and SSRIs (serotonin syndrome risk).
4. COMT Inhibitors
Inhibit catechol-O-methyltransferase → prevent peripheral breakdown of levodopa → prolong its effect. Used as adjuncts to levodopa for motor fluctuations (wearing-off).
| Drug | Dose | Notes |
|---|---|---|
| Entacapone (Comtan) | 200 mg with each levodopa dose | Preferred; no hepatotoxicity; peripheral COMT only |
| Tolcapone (Tasmar) | 100–200 mg TID | Central + peripheral COMT; monitor LFTs (hepatotoxicity risk) |
Side effects: Diarrhea, orange urine discoloration, dyskinesias (reduce levodopa dose accordingly).
5. Amantadine
- Originally an antiviral; has dopaminergic, anticholinergic, and NMDA antagonist properties.
- Main use: treatment of levodopa-induced dyskinesias (most useful in this regard).
- Also provides mild symptomatic benefit early in disease.
- Dose: 100 mg BID–TID.
- Side effects: Livedo reticularis, peripheral edema, confusion, insomnia, urinary retention, heart failure.
- Do NOT stop abruptly — may precipitate acute confusion, hyperpyrexia, and severe parkinsonism rebound.
6. Adenosine A2A Antagonist
- Istradefylline (20–40 mg daily) — caffeine analog; reduces off-periods in patients on carbidopa-levodopa.
- Side effects: Dyskinesias, dizziness, constipation, nausea, hallucinations.
7. Anticholinergics
Helpful primarily for tremor and drooling; minimal effect on bradykinesia.
| Drug | Dose |
|---|---|
| Trihexyphenidyl (Artane) | 1–2 mg TID |
| Benztropine (Cogentin) | 0.5 mg at bedtime → up to 6 mg/day |
Poorly tolerated in elderly — side effects include impaired memory, blurred vision, urinary retention, constipation, dry mouth. Avoid in patients with cognitive impairment.
Staged Pharmacotherapy Approach
Minor symptoms (no functional impairment)
→ No pharmacologic treatment; observation
Mild-moderate disease
→ Start dopamine agonist (pramipexole/ropinirole) OR
→ MAO-B inhibitor (rasagiline) as monotherapy OR
→ Low-dose carbidopa-levodopa (25/100 TID)
(Rule: "start low and go slow")
Tremor-predominant
→ Add anticholinergic (trihexyphenidyl)
Motor fluctuations / wearing-off
→ Add dopamine agonist OR COMT inhibitor OR MAO-B inhibitor
→ Adjust levodopa dosing intervals
Dyskinesias
→ Reduce levodopa dose
→ Add amantadine
Advanced / refractory motor symptoms
→ Consider surgical intervention (DBS)
Non-Pharmacological Management
Exercise
Exercise is a critical component — recent evidence designates it "exercise as medicine" in PD. Aerobic, resistance, balance, and tango/dance-based programs all show benefit for motor function, gait, and quality of life. — J Neurol Neurosurg Psychiatry, 2024
Physiotherapy
- Gait training, balance exercises
- Cueing strategies (visual/auditory) to overcome freezing of gait
- Referral for falls prevention
Speech Therapy
- For hypophonia and dysarthria (Lee Silverman Voice Treatment — LSVT LOUD)
- For dysphagia: videofluoroscopic swallowing study; swallowing exercises; dietary modification
Occupational Therapy
- Adaptive equipment for ADLs
- Environmental modification for safety
Nutrition
- High-protein meals may compete with levodopa absorption — space protein intake away from medication doses
- Adequate hydration; fiber for constipation
Psychological Support
- Depression occurs commonly and worsens motor symptoms in a vicious cycle; treat with SSRIs/SNRIs
- Cognitive behavioral therapy for anxiety and depression
- Caregiver support and education
Surgical Management
Deep Brain Stimulation (DBS)
- Targets: Subthalamic nucleus (STN) or globus pallidus internus (GPi)
- Indications: Disabling motor fluctuations or dyskinesias despite optimized medical therapy; good levodopa response (predicts DBS benefit); no dementia
- Contraindications: Secondary/atypical parkinsonism, dementia, failure to respond to dopaminergic therapy
- Mechanism: High-frequency stimulation modulates dysfunctional cortico-striato-thalamo-cortical circuitry
- Has largely replaced older ablative procedures (pallidotomy, thalamotomy) due to lower morbidity and reversibility
rTMS (Investigational)
- Repetitive transcranial magnetic stimulation targeting motor cortex to increase excitability — pilot studies promising but large RCTs remain ambiguous; not currently standard of care.
Management of Non-Motor Symptoms
| Symptom | Management |
|---|---|
| Depression | SSRIs, SNRIs; ECT can be considered (carbidopa-levodopa can continue with monitoring) |
| Psychosis/hallucinations | Reduce antiparkinsonian medications; quetiapine or clozapine (avoid risperidone/haloperidol) |
| Dementia | Rivastigmine (approved for PD dementia); donepezil |
| Orthostatic hypotension | Fludrocortisone, midodrine, compression stockings, adequate fluid/salt intake |
| Constipation | Fiber, hydration, polyethylene glycol, macrogol |
| Drooling | Anticholinergics, botulinum toxin to parotid glands |
| Insomnia/REM sleep behavior disorder | Melatonin, clonazepam for RBD |
| Urinary urgency | Oxybutynin (with caution re: cognition) |
Drug-Induced Parkinsonism (Important Differential)
This is reversible — identify and remove the causative agent:
- Neuroleptics, metoclopramide, prochlorperazine
- Amiodarone, valproic acid, lithium
- Methyldopa (dopamine synthesis blockade)
- Reserpine (dopamine depletion)
Recent Evidence Updates
- Motor fluctuation treatment review (MDS Evidence-Based Medicine review, Mov Disord 2025, PMID 40055961): Updated evidence on treatments for wearing-off phenomena
- Motor symptom treatment review (Neurol Clin 2025, PMID 40185525): Comprehensive review of current pharmacological approaches
- Exercise as medicine in PD (JNNP 2024, PMID 38418216): Strong evidence for structured exercise programs across disease stages
Sources: Katzung's Basic and Clinical Pharmacology 16e; Textbook of Family Medicine 9e; Swanson's Family Medicine Review; Bradley and Daroff's Neurology in Clinical Practice
This is a shared conversation. Sign in to Orris to start your own chat.