22 year old male, has patchy beard, complaints of low libido, no morning erections, low energy, masculine form but poorly built, what is the physiology of testosterone and its release, what is the diagnosis? What is the treatment considering his fertility?
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching PubMed
"hypogonadotropic hypogonadism" AND "fertility" AND treatment
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have all the information needed for a comprehensive, well-structured answer. Let me compile it.
Hypogonadotropic Hypogonadism in a 22-Year-Old Male
The Clinical Picture
This presentation — patchy beard, low libido, absent morning erections, low energy, masculine habitus but poorly developed musculature — is the classic picture of hypogonadotropic hypogonadism (HH), also called secondary hypogonadism. The onset before/during puberty accounts for incomplete secondary sexual development (sparse beard); the persistence into adulthood explains the low libido, absent nocturnal/morning erections, and reduced muscle mass.
Part 1: Physiology of Testosterone and Its Release
The hypothalamic-pituitary-gonadal (HPG) axis governs testosterone secretion through a hierarchical, feedback-regulated cascade.
The HPG Axis (Goldman-Cecil Medicine, Fig. 216-1)
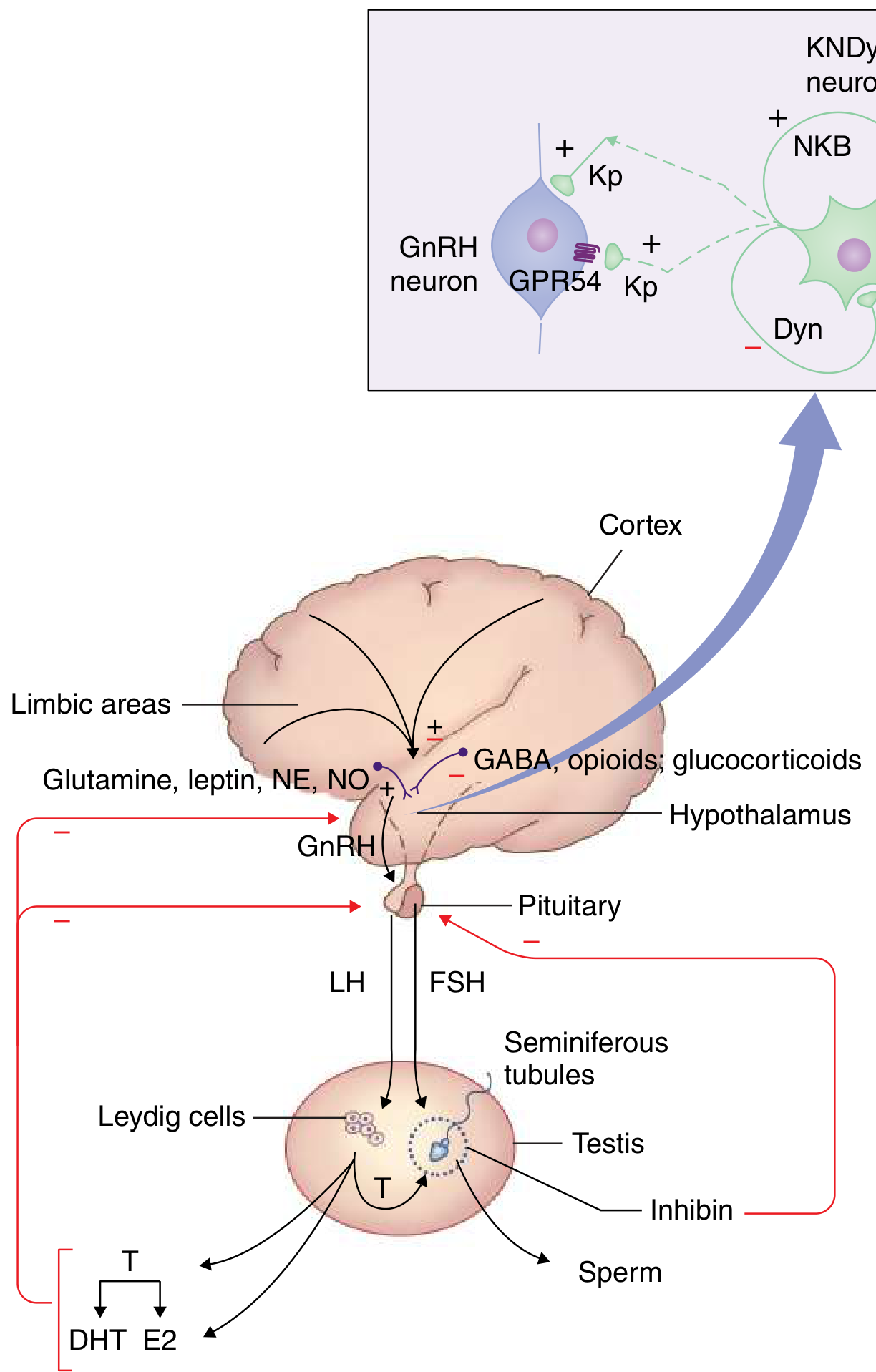
Step 1 — Hypothalamus (KNDy neurons → GnRH)
The arcuate nucleus contains kisspeptin-neurokinin B-dynorphin (KNDy) neurons that generate pulsatile GnRH secretion. Kisspeptin (acting via GPR54) and neurokinin B stimulate GnRH release; dynorphin inhibits it. GnRH is released in pulses every 60–90 minutes into the portal circulation — this pulsatility is essential; continuous GnRH desensitizes the pituitary. Higher inputs include glutamate, leptin, norepinephrine, and nitric oxide (stimulatory) vs. GABA, opioids, and glucocorticoids (inhibitory). — Goldman-Cecil Medicine, p. 2533
Step 2 — Anterior Pituitary (GnRH → LH & FSH)
Pulsatile GnRH binds Gq-coupled receptors on gonadotrophs, triggering secretion of:
- LH — acts on Leydig cells (interstitial) → stimulates testosterone synthesis via cholesterol side-chain cleavage (StAR protein, P450scc → pregnenolone → DHEA → androstenedione → testosterone)
- FSH — acts on Sertoli cells → stimulates spermatogenesis and inhibin B secretion
Step 3 — Testis
- Leydig cells synthesize testosterone from cholesterol. Normal serum testosterone: 264–916 ng/dL in men aged 19–39.
- Testosterone undergoes peripheral conversion: to dihydrotestosterone (DHT) via 5α-reductase (responsible for prostate growth, male pattern baldness, external genital virilization), and to estradiol (E2) via aromatase (responsible for bone closure, libido modulation).
- Sertoli cells, under FSH and high intratesticular testosterone, produce inhibin B and support spermatogenesis. Inhibin B feeds back to suppress FSH.
Feedback:
- Testosterone and estradiol exert long-loop negative feedback on both hypothalamus and pituitary — suppressing GnRH pulse frequency and LH/FSH amplitude.
- A strong diurnal rhythm exists: testosterone peaks in the morning (before 10 AM), hence the clinical importance of morning blood draws. — Goldman-Cecil Medicine, p. 2539
Actions of testosterone:
- Androgenic: secondary sexual characteristics (beard, body hair, phallus, scrotum), voice deepening, libido, sexual function
- Anabolic: skeletal muscle growth, erythropoiesis (EPO stimulation), bone density
- Via DHT: external genitalia development, prostate, sebaceous glands, androgenic alopecia
- Via E2 (aromatization): bone maturation, epiphyseal fusion, some aspects of libido and sexual function
Part 2: Diagnosis
Most Likely Diagnosis: Hypogonadotropic Hypogonadism (Secondary Hypogonadism)
Why?
| Feature | Significance |
|---|---|
| Patchy beard at 22 | Incomplete androgenization → low testosterone during puberty |
| Low libido, no morning erections | Classic testosterone deficiency symptoms |
| Low energy, poor muscle mass | Anabolic effects of testosterone absent |
| Masculine body form | Enough androgen to male-differentiate in utero, but insufficient for full pubertal maturation |
In HH, the defect is at the hypothalamus or pituitary — the testis is intact but receives insufficient LH/FSH stimulation. This contrasts with primary (hypergonadotropic) hypogonadism where the testis itself fails.
Differential within HH:
- Congenital HH (Kallmann syndrome or normosmic idiopathic HH) — most likely here given young age, no systemic disease, incomplete puberty. Kallmann syndrome adds anosmia (absent GnRH neurons fail to migrate from olfactory placode). FGFR1, GNRH1, PROKR2 gene mutations are the most common.
- Functional HH — from chronic illness, starvation, anorexia, excessive exercise, opioid use
- Pituitary tumor (e.g., prolactinoma — check for visual field defects, galactorrhea, and headache)
- Infiltrative disease (hemochromatosis, sarcoidosis)
Diagnostic workup:
- Morning serum total testosterone (×2, before 10 AM) — confirm low level (<264 ng/dL; treatment indicated if consistently <230 ng/dL with symptoms)
- Serum LH and FSH — will be low/inappropriately normal (not elevated as in primary hypogonadism) — this is the key distinguishing lab
- Serum prolactin — mandatory in all HH to exclude prolactinoma
- MRI pituitary — to exclude structural lesion
- Serum FSH/inhibin B/AMH — assess spermatogenic potential
- Olfactory testing — to distinguish Kallmann syndrome
- Genetic testing — if congenital HH suspected (mutations in GNRH1, FGFR1, PROKR2, KISS1R, etc.)
- Free testosterone or bioavailable testosterone if SHBG abnormality suspected
Goldman-Cecil Medicine, pp. 2538–2540
Part 3: Treatment — Considering Fertility
This is the critical fork: testosterone replacement therapy (TRT) suppresses spermatogenesis via negative feedback on FSH/LH, making it inappropriate when fertility is desired.
If Fertility Is Desired (Current Priority in a 22-Year-Old):
First-line: Gonadotropin Replacement Therapy
Directly stimulates the testis since the HPG axis below the testis is intact.
| Agent | Mechanism | Dosing |
|---|---|---|
| hCG (human chorionic gonadotropin) | LH analog → stimulates Leydig cells → testosterone production | 1500–2000 IU SC 3×/week |
| FSH (recombinant FSH or hMG) | Stimulates Sertoli cells → spermatogenesis | Added after 3–6 months of hCG if azoospermic |
- hCG alone often raises testosterone into the normal range and normalizes libido, energy, erections, and muscle mass
- Adding FSH (as recombinant FSH or hMG) is necessary to initiate or restore spermatogenesis
- Spermatogenesis may take 12–24 months to appear; previous testosterone use delays this further
- This is the only reliable pharmaceutical treatment for infertility in HH — Goldman-Cecil Medicine notes that specific gonadotropin treatment is available for the 1–2% of infertile men with gonadotropin insufficiency, while most other causes lack equivalent medical treatment. — Goldman-Cecil Medicine, p. 2736
Second-line alternative (if compliance is an issue): Pulsatile GnRH pump
- Mimics physiological hypothalamic pulsatility (60-min pulses via portable subcutaneous pump)
- Effective when the defect is hypothalamic (Kallmann/idiopathic HH) with an intact pituitary
- Less practical but highly effective for fertility; not widely available
Adjunct: Clomiphene citrate (off-label)
- Selective estrogen receptor modulator (SERM) — blocks estrogen negative feedback at hypothalamus/pituitary → increases endogenous LH/FSH → stimulates testicular testosterone and spermatogenesis
- Useful in functional HH or mild secondary hypogonadism
- Evidence from a 2024 meta-analysis (PMID 37306109) supports estrogen modulation in improving semen parameters in secondary hypogonadism
If Fertility Is NOT a Current Concern:
Testosterone Replacement Therapy (TRT) is appropriate:
- Testosterone enanthate or cypionate IM 100–200 mg every 1–2 weeks
- Testosterone gel (transdermal) — daily, mimics diurnal variation
- Goals: normalize testosterone levels, restore libido, morning erections, energy, and muscle mass
- Monitor serum testosterone midway between injections (goal: mid-normal range)
- TRT suppresses LH → testicular atrophy and azoospermia — this must be explained to the patient before initiating
Goodman & Gilman's Pharmacological Basis of Therapeutics, p. 995–996
Summary Algorithm
22M, hypogonadal symptoms
↓
Morning testosterone × 2 → LOW
LH / FSH → LOW/normal → Secondary (hypogonadotropic) hypogonadism
↓
Prolactin → check (exclude prolactinoma)
MRI pituitary → check for structural lesion
Olfactory testing → Kallmann?
↓
FERTILITY DESIRED?
YES → hCG ± FSH (gonadotropin therapy)
— Raises testosterone AND enables spermatogenesis
— Add FSH after 3–6 months if azoospermia persists
NO → Testosterone replacement (gel, IM injection)
— Counsel: causes azoospermia; reversible with switch to gonadotropins
Recent Evidence
Three recent systematic reviews are directly relevant:
- PMID 41206002 (Dwyer & Stamou, J Clin Endocrinol Metab 2026) — Reviews reversal of congenital HH; some patients achieve normal reproductive function with treatment
- PMID 39673783 (J Clin Endocrinol Metab 2025) — Gonadotropin therapy in HH
- PMID 37306109 (Andrology 2024, meta-analysis) — Estrogen modulation (clomiphene/tamoxifen) improves semen parameters in secondary hypogonadism
Key clinical pearl: In a 22-year-old with HH who wants future children, never start TRT without counseling about its contraceptive effect on spermatogenesis. Gonadotropin therapy both treats symptoms and preserves/induces fertility — making it the preferred first approach in this age group.
Sources: Goldman-Cecil Medicine (International Ed.), pp. 2530–2544; Goodman & Gilman's Pharmacological Basis of Therapeutics, pp. 995–996; Guyton & Hall Medical Physiology
He gives additional history of have small breast bud beneath nipple
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Finding Sources
I now have everything needed. Here is the updated, focused analysis.
The New Finding Changes the Diagnosis
Gynecomastia — a palpable breast bud beneath the nipple in a male — combined with the previous findings now creates a complete, recognizable syndrome. This is no longer just hypogonadotropic hypogonadism. This is Klinefelter Syndrome until proven otherwise.
Why Gynecomastia Shifts the Diagnosis
In the previous scenario, the working diagnosis was secondary (hypogonadotropic) hypogonadism — low LH/FSH driving low testosterone. Gynecomastia doesn't fit cleanly there.
Klinefelter syndrome is primary (hypergonadotropic) hypogonadism — the testes fail directly. The mechanism of gynecomastia is:
"Testosterone is decreased and estradiol is increased, leading to clinical features of undervirilization and gynecomastia. The ratio of estrogens to testosterone determines the degree of feminization in individual cases." — Harrison's Principles of Internal Medicine, 22E
The pathophysiology is:
- Damaged Leydig cells → ↓ testosterone
- Despite damage, residual Sertoli cells and Leydig cells continue to aromatize androgens → disproportionately elevated estradiol
- ↑ Estradiol/testosterone ratio → stimulates ductal proliferation in breast tissue = gynecomastia
- Low testosterone also fails to suppress breast growth as it normally would
- Elevated LH/FSH (pituitary sensing low T) → further drives estrogen production peripherally
Revised Diagnosis: Klinefelter Syndrome (47,XXY)
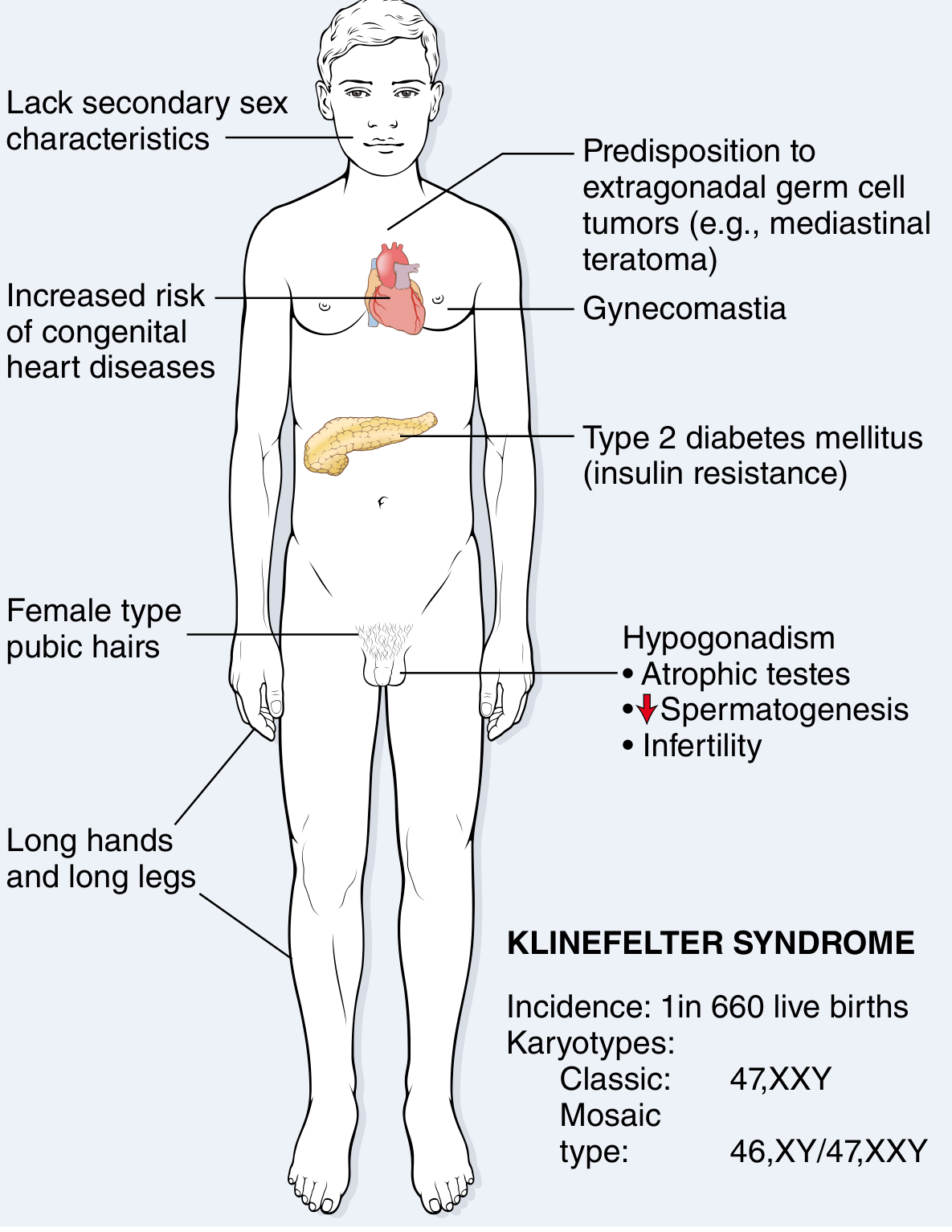
The complete syndrome in this patient:
| Feature | Classic Klinefelter Finding | This Patient |
|---|---|---|
| Age 22, incomplete virilization | Manifestations appear in puberty/young adulthood | ✓ Patchy beard |
| Low libido, no morning erections | Testosterone deficiency | ✓ |
| Low energy, poor muscle bulk | Anabolic effects of testosterone absent | ✓ |
| Masculine form | Male sex determination (Y chromosome present) | ✓ |
| Gynecomastia | ↑ Estradiol/testosterone ratio | ✓ NEW |
Additional features to look for on examination:
- Small, firm atrophic testes (< 5 mL; the hallmark)
- Long legs relative to trunk (eunuchoid proportions — span > height; pubis-to-floor > pubis-to-crown)
- Female-pattern pubic hair distribution
- Decreased facial, axillary, and body hair
Revised Hormone Profile
| Hormone | Expected Finding |
|---|---|
| Testosterone | Low (↓ Leydig cell function) |
| LH | High (primary gonadal failure → lost negative feedback) |
| FSH | Very high (seminiferous tubule failure → lost inhibin B feedback) |
| Estradiol | Elevated (excess aromatization) |
| Inhibin B | Very low or undetectable |
This is the opposite of hypogonadotropic hypogonadism — here LH and FSH are elevated because the pituitary is working hard to stimulate failing testes. — Robbins & Kumar Pathologic Basis of Disease, p. 167
Confirming the Diagnosis
- Karyotype (gold standard) — Classic: 47,XXY (90% of cases); Mosaic: 46,XY/47,XXY (10%)
- Higher polysomic forms exist: 48,XXXY / 49,XXXXY → more severe phenotype
- Hormones: ↓ total testosterone, ↑↑ FSH, ↑ LH, ↑ estradiol
- Semen analysis — azoospermia is the rule in 47,XXY; some sperm may be found in mosaic forms
- Testicular ultrasound — small, hypoechoic testes; assess for microlithiasis or tumor
- Bone density (DEXA) — osteoporosis risk from chronic hypogonadism
Pathology of the Testis
"In some patients the testicular tubules are totally atrophied and replaced by pink, hyaline, collagenous 'ghosts.' In others, apparently normal tubules are interspersed with atrophic tubules... Leydig cells appear prominent due to atrophy in the germ cell compartment." — Robbins & Kumar Pathologic Basis of Disease, p. 167
Spermatogonial stem cells with the 47,XXY complement are progressively lost after puberty. However, isolated foci of spermatogenesis can persist, which has important implications for fertility.
Treatment — Revised for Klinefelter Syndrome
1. Testosterone Replacement Therapy (TRT) — Core Treatment
- Indicated to correct hypogonadism symptoms: libido, erections, energy, muscle mass, mood, bone density
- Formulations: testosterone enanthate/cypionate IM, or testosterone gel (transdermal)
- Goal: mid-normal serum testosterone range
- Benefit: Within months → improved libido, energy, haematocrit; within 6 months → increased muscle mass, decreased fat; bone density improves over 2 years
- Important: TRT will suppress residual spermatogenesis via LH/FSH suppression — fertility must be addressed before starting TRT
2. Fertility — The Critical Point in a 22-Year-Old
Unlike idiopathic HH (where gonadotropins stimulate a normal testis), Klinefelter syndrome has intrinsic testicular failure. Gonadotropin therapy cannot create new sperm from destroyed tubules. The approach is:
Microsurgical Testicular Sperm Extraction (micro-TESE)
"Fertility can be achieved by intracytoplasmic injection of sperm retrieved surgically from the testes of men with Klinefelter syndrome, including some men with the nonmosaic form." — Harrison's 22E, p. 102 (block 44)
- A urologist performs microdissection TESE (mTESE) to locate and extract residual sperm from isolated spermatogenic foci
- Sperm retrieval success rates: ~50% in non-mosaic 47,XXY; higher in mosaic forms
- At age 22, sperm may still be present — retrieval success decreases with age as progressive tubular atrophy continues
- Retrieved sperm are used for intracytoplasmic sperm injection (ICSI) with partner's oocytes
Timing is Critical:
"Although sperm retrieval in adolescence for fertility preservation offers no benefit over harvesting in adulthood, fertility counseling, including the potential for sperm retrieval, should be offered prior to starting testosterone replacement therapy." — Harrison's 22E
Therefore: Refer for fertility counseling and potential mTESE BEFORE initiating TRT.
3. Gynecomastia Management
- Grade I (small bud, as in this patient): Often improves with TRT as testosterone rises and estradiol/testosterone ratio normalizes
- If persistent or cosmetically distressing after testosterone normalization: subcutaneous mastectomy (the definitive treatment)
- Aromatase inhibitors (anastrozole, letrozole) may reduce estradiol and gynecomastia, but evidence for long-term use in Klinefelter is limited
4. Surveillance (Klinefelter-specific Risks)
| Complication | Action |
|---|---|
| Breast cancer risk (20-30× increased) | Annual breast self-exam; consider periodic mammography |
| Osteoporosis | DEXA scan baseline; ensure TRT is started; calcium + vitamin D |
| Metabolic syndrome / T2DM | Annual fasting glucose, lipid panel |
| Congenital heart disease (MVP ~50%) | Echocardiography |
| Extragonadal germ cell tumors | Mediastinal imaging if chest symptoms |
| Autoimmune disease (SLE, Sjögren's) | Clinical vigilance |
Summary
Patchy beard + low libido + no morning erections +
low energy + poor muscle bulk + GYNECOMASTIA
↓
KLINEFELTER SYNDROME (47,XXY)
↓
Labs: ↓ Testosterone, ↑↑ LH, ↑↑ FSH, ↑ Estradiol
Confirm: Karyotype
↓
MANAGEMENT SEQUENCE:
1. Fertility counseling NOW (age 22 — sperm still retrievable)
2. Offer micro-TESE + sperm cryopreservation BEFORE TRT
3. Start TRT (testosterone enanthate/gel) after fertility addressed
4. Monitor and treat gynecomastia
5. Lifelong surveillance for comorbidities
The gynecomastia is the diagnostic pivot: it reflects elevated estradiol relative to testosterone from primary testicular failure — not what you see in simple hypogonadotropic hypogonadism where the testis is intact. Combined with the clinical picture, Klinefelter syndrome is the unifying diagnosis.
Sources: Harrison's Principles of Internal Medicine 22E (2025), block 44; Robbins & Kumar Pathologic Basis of Disease, p. 167; Schwartz's Principles of Surgery 11E, p. 3535–3539; Goldman-Cecil Medicine
How do ICSI and sperm retrieval improve fertility outcomes in Klinefelter?
Finding Sources
Finding Sources
Searching PubMed
"Klinefelter syndrome" AND "sperm retrieval" AND ICSI
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.