Stages of sleep and sleep disorders full details for md exam answer 15 marks
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive MD exam answer on Stages of Sleep and Sleep Disorders (15 marks):
STAGES OF SLEEP AND SLEEP DISORDERS
INTRODUCTION
Sleep is not a passive state but an active, organized neurobiological process characterized by distinct electrophysiological and behavioral stages. Sleep is studied using polysomnography (PSG), which simultaneously records the EEG, electrooculogram (EOG), and electromyogram (EMG). Sleep is scored in 30-second epochs according to criteria originally defined by Rechtschaffen and Kales (1968) and updated in 2007/2020 by the American Academy of Sleep Medicine (AASM).
PART I: STAGES OF SLEEP
Classification
Sleep is broadly divided into two major types:
- NREM sleep (Non-Rapid Eye Movement) — Stages N1, N2, N3
- REM sleep (Rapid Eye Movement) — Stage R
The AASM current nomenclature:
| AASM Stage | Former Name | EEG | EOG | EMG | Behavioral State |
|---|---|---|---|---|---|
| W (Wake) | — | Alpha (8–13 Hz, eyes closed); low-voltage fast (eyes open) | Voluntary eye movements, blinks | High tonic activity | Awake |
| N1 | Stage 1 | Low-voltage, mixed-frequency; theta (4–7 Hz); vertex sharp waves | Slow, rolling eye movements | Slightly decreased tone | Drowsy; may not perceive as sleep |
| N2 | Stage 2 | Sleep spindles (11–16 Hz, ≥0.5 s) + K-complexes (negative sharp wave followed by positive slow wave) | None | Low tonic activity | Light sleep |
| N3 | Stages 3 & 4 | High-amplitude slow waves ≥75 μV at ≤2 Hz, occupying ≥20% of epoch | None | Low tonic activity | Deep/slow-wave sleep |
| R | REM | Low-voltage mixed frequency; sawtooth waves (2–6 Hz) | Rapid conjugate eye movements | Tonic atonia with phasic twitches | Dreaming predominates |
EEG Patterns at Each Stage
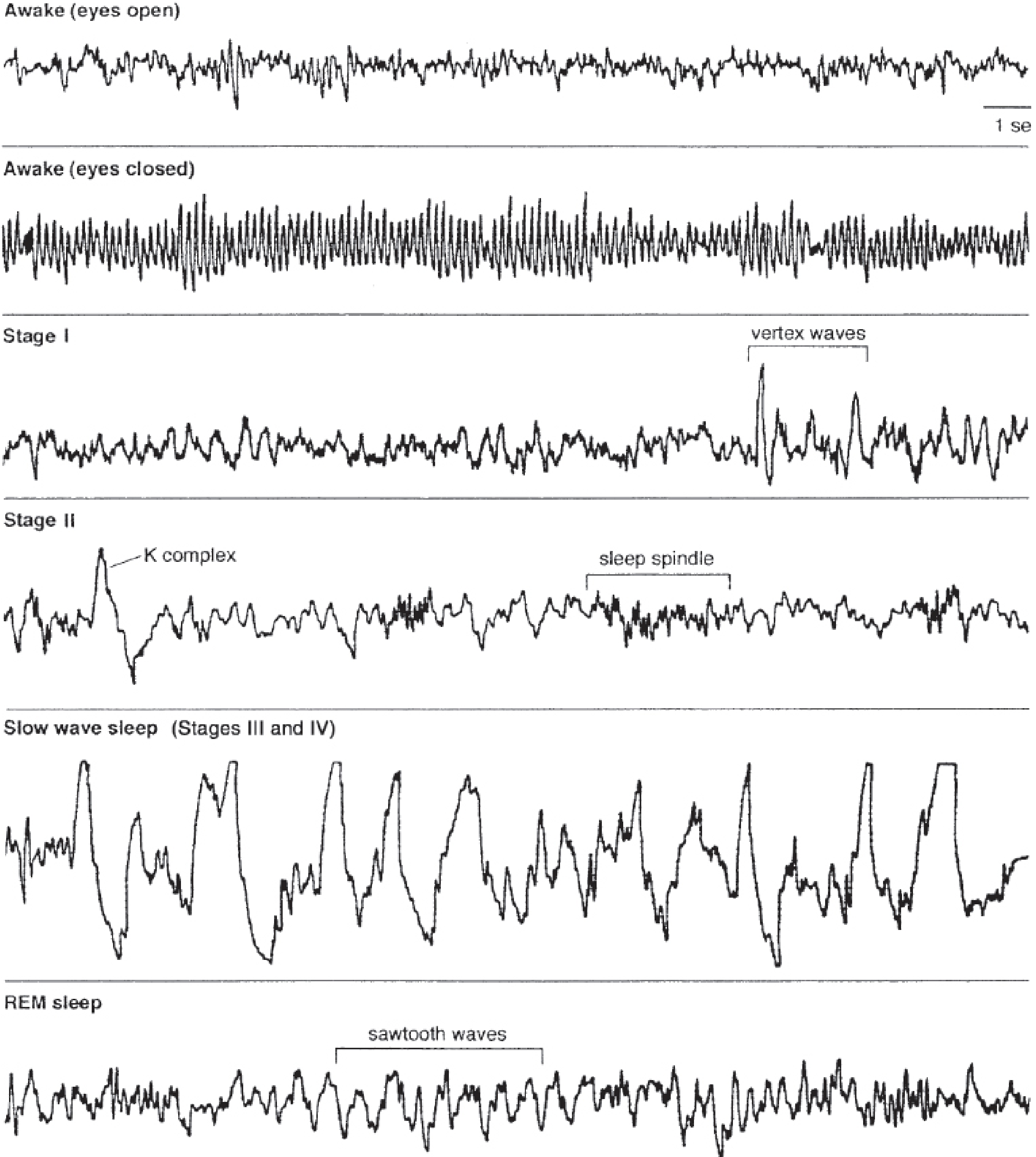
EEG patterns across stages of sleep — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
Stage N1 (Light NREM Sleep)
- Transition from wakefulness to sleep
- EEG: Alpha waves disappear; replaced by low-voltage, mixed-frequency theta (4–7 Hz); vertex sharp waves (V waves) may appear over central regions
- EOG: Slow, rolling eye movements
- EMG: Muscle tone mildly reduced
- Patient may not subjectively recognize this as sleep; decreased awareness of sensory stimuli, especially visual; mental activity becomes dreamlike
- Comprises ~3–5% of total sleep time (TST)
Stage N2 (Light-to-Intermediate NREM Sleep)
- Heralded by two cardinal EEG features:
- Sleep spindles: 11–16 Hz sinusoidal bursts lasting ≥0.5 seconds; generated by thalamo-cortical circuits
- K-complexes: High-amplitude negative sharp wave followed by positive slow wave, lasting ≥0.5 seconds; thought to reflect cortical response to environmental stimuli
- EOG: No eye movements
- EMG: Further decreased muscle tone
- Comprises ~50% of TST in healthy young adults
Stage N3 (Slow-Wave Sleep / Deep Sleep)
- Also called delta sleep or slow-wave sleep (SWS)
- EEG: High-amplitude (≥75 μV) slow delta waves at 0.5–2 Hz occupying ≥20% of epoch; most prominent over frontal regions
- Previously subdivided into Stage 3 (20–50% slow waves) and Stage 4 (>50% slow waves) — now combined into N3
- Arousal threshold is highest; person is most difficult to wake
- Comprises ~10–20% of TST; decreases markedly with age (virtually absent in persons >70 years)
- Functions: physical restoration, growth hormone secretion, memory consolidation
Stage R (REM Sleep)
- Tonic features (persistent throughout REM):
- Desynchronized (activated) low-voltage mixed-frequency EEG
- Generalized skeletal muscle atonia (except extraocular muscles and diaphragm) — mediated by glycinergic/GABAergic inhibition of spinal motor neurons from the pons
- Sawtooth waves (triangular, 2–6 Hz trains)
- Phasic features (episodic):
- Rapid conjugate eye movements (horizontal > vertical)
- Muscle twitches in face, hands, feet
- Fluctuations in heart rate, blood pressure, respiration (sympathetic activation)
- Penile/clitoral tumescence
- Dreaming: Most vivid, narrative dreams occur in REM; patients awoken from REM recall dreams most consistently
- Comprises ~20–25% of TST
Sleep Architecture and Cycling
A normal night's sleep follows a predictable pattern:
- A young adult enters sleep through N1 → N2 → N3 → back to N2 → then R (REM)
- First REM period occurs after ~70–100 minutes, is brief (5–10 min)
- NREM-REM cycle repeats 4–6 times per night, approximately every 90–100 minutes
- Early night: dominated by N3 (slow-wave) sleep
- Late night: cycles consist mainly of N2 and progressively longer REM periods
Proportion of TST by stage (young adult):
- N1: 3–5%
- N2: 50–60%
- N3: 10–20%
- REM: 20–25%
Age-related changes:
- Neonates: ~50% REM; 60-min cycles
- REM remains relatively constant across adult life
- N3 (slow-wave sleep) progressively decreases with age and is virtually absent after age 70
Neurobiological Control of Sleep
Sleep-wake switching is governed by a "flip-flop" mechanism (Saper et al.):
- VLPO (ventrolateral preoptic nucleus): GABAergic/galaninergic sleep-promoting neurons; inhibit arousal centers during sleep
- Arousal centers: Locus coeruleus (NE), dorsal raphe (5-HT), tuberomammillary nucleus (histamine) — all inhibit VLPO during wakefulness
- Orexin (hypocretin) neurons (lateral hypothalamus): stabilize the flip-flop switch; loss of these neurons → narcolepsy
REM/NREM switching similarly involves a secondary flip-flop between REM-on cholinergic neurons (pedunculopontine/laterodorsal tegmental nuclei) and REM-off GABAergic neurons.
PART II: SLEEP DISORDERS
Classification (ICSD-3, AASM 2014)
- Insomnia disorders
- Sleep-related breathing disorders
- Central disorders of hypersomnolence (narcolepsy, hypersomnia)
- Circadian rhythm sleep-wake disorders
- Parasomnias
- Sleep-related movement disorders
1. INSOMNIA
Definition: Inability to initiate sleep, maintain sleep, or early morning awakening despite adequate opportunity for sleep, causing daytime impairment or dissatisfaction.
Types (ICSD-3):
- Short-term insomnia disorder (<3 months): Precipitated by identifiable stressor; resolves when stressor is removed
- Chronic insomnia disorder (≥3 months, ≥3 nights/week): Usually comorbid with psychiatric (depression, anxiety), medical, neurological disorders, or substance abuse
Epidemiology: ~35% of adults report insomnia complaints; 10% have persistent/chronic insomnia; more common in women, elderly, divorced/widowed, low socioeconomic status
Consequences: Impaired work performance, reduced quality of life; 2–5× increased risk of developing depression; increased risk of hypertension, CAD, diabetes mellitus, and increased mortality in men with objectively measured short sleep
Management:
- First-line: Cognitive behavioral therapy for insomnia (CBT-I) — sleep restriction, stimulus control, relaxation techniques
- Pharmacological: benzodiazepines (temazepam), non-BZD hypnotics (zolpidem, eszopiclone), melatonin receptor agonists (ramelteon), dual orexin receptor antagonists (suvorexant)
2. OBSTRUCTIVE SLEEP APNEA (OSA)
Definition: Recurrent episodes of partial (hypopnea) or complete (apnea) upper airway obstruction during sleep, despite respiratory effort.
Pathophysiology: Collapse of the pharyngeal airway during sleep → hypoxia → arousal → cycle repeats. Associated with obesity, male sex, retrognathia, large neck circumference, tonsillar hypertrophy.
Diagnosis: Polysomnography (PSG) — gold standard; Apnea-Hypopnea Index (AHI):
- Mild: 5–14 events/hour
- Moderate: 15–30 events/hour
- Severe: >30 events/hour
Clinical features: Loud snoring, witnessed apneas, non-restorative sleep, excessive daytime sleepiness, morning headache, nocturia, impaired concentration
Complications: Systemic hypertension, pulmonary hypertension, arrhythmias (AF), right heart failure, increased risk of myocardial infarction and stroke
Treatment:
- CPAP (continuous positive airway pressure) — first-line for moderate-severe OSA
- Weight loss (strongly recommended)
- Oral appliances (mandibular advancement device) — for mild-moderate OSA
- Positional therapy
- Surgical options: uvulopalatopharyngoplasty (UPPP), maxillomandibular advancement, hypoglossal nerve stimulation
3. NARCOLEPSY
Definition: A neurological disorder of excessive daytime sleepiness characterized by dysregulation of REM sleep intrusion into wakefulness.
Pathophysiology: Loss of orexin (hypocretin)-producing neurons in the lateral hypothalamus — autoimmune destruction. Deficiency of HCRT destabilizes the sleep-wake flip-flop switch → REM components intrude inappropriately.
Tetrad of Narcolepsy:
- Excessive daytime sleepiness (EDS) — irresistible sleep attacks; most prominent feature
- Cataplexy — sudden bilateral loss of muscle tone triggered by strong emotion (laughter, surprise), without loss of consciousness; pathognomonic
- Sleep paralysis — temporary inability to move at sleep onset or awakening
- Hypnagogic/hypnopompic hallucinations — vivid hallucinations at sleep onset (hypnagogic) or on awakening (hypnopompic)
Types:
- Narcolepsy type 1: With cataplexy; low/absent CSF orexin-A (<110 pg/mL)
- Narcolepsy type 2: Without cataplexy; CSF orexin usually normal
Diagnosis: Multiple Sleep Latency Test (MSLT) — mean sleep latency ≤8 min with ≥2 sleep-onset REM periods (SOREMPs)
Treatment:
- EDS: Modafinil (first-line), amphetamines, methylphenidate, sodium oxybate (GHB)
- Cataplexy: Sodium oxybate, TCAs (clomipramine), SSRIs, venlafaxine, selegiline
- Behavioral: Regular sleep schedules, scheduled daytime naps
4. PARASOMNIAS
Parasomnias are undesirable behavioral or physiological events during sleep or sleep-wake transitions.
NREM Parasomnias (disorders of arousal from N3 — mostly in children):
- Sleepwalking (somnambulism): Complex motor behaviors; amnesia for event; may be triggered by alcohol, sleep deprivation
- Sleep terrors: Sudden arousal from N3; intense autonomic arousal; screaming, tachycardia, sweating; no recall
- Confusional arousals: Disorientation on awakening from N3
- All share: occur in first third of night, from N3 sleep; no dream recall; HLA associations
REM Parasomnias:
- REM Sleep Behavior Disorder (RBD): Loss of normal REM atonia → acting out of vivid/violent dreams; potential self/bed-partner injury; strong association with synucleinopathies (Parkinson's disease, Lewy body dementia, MSA — may precede neurodegeneration by decades); diagnosed by PSG showing REM without atonia; treatment: clonazepam, melatonin
- Nightmare disorder: Recurrent distressing dreams recalled on awakening; differ from sleep terrors (occur from REM; full recall; minimal autonomic features)
- Sleep paralysis (isolated): Transient inability to move at sleep-wake transition; fully conscious
Drugs causing/worsening parasomnias: MAOIs, SSRIs, TCAs, venlafaxine, alcohol, caffeine
5. RESTLESS LEGS SYNDROME (RLS) / PERIODIC LIMB MOVEMENT DISORDER (PLMD)
RLS (Ekbom syndrome):
- Uncomfortable sensory urge to move legs, worse at rest, worse in evening/night, relieved by movement
- May be primary (idiopathic) or secondary to iron deficiency, uremia, pregnancy, medications (dopamine antagonists, SSRIs, antihistamines)
- Treatment (AASM first-line): Dopamine agonists (ropinirole, pramipexole) in the evening; gabapentin/pregabalin
PLMD: Stereotyped periodic leg movements during sleep; associated with sleep disruption and EDS; diagnosed by PSG
6. CIRCADIAN RHYTHM SLEEP-WAKE DISORDERS
Misalignment between the internal circadian clock and the desired/required sleep-wake schedule:
- Delayed sleep phase disorder: Unable to sleep until late; cannot wake early; common in adolescents
- Advanced sleep phase disorder: Sleepy in early evening; wake in early morning; common in elderly
- Shift-work disorder / Jet lag
- Treatment: Light therapy, melatonin, chronotherapy
SUMMARY TABLE
| Disorder | Key Feature | Stage Affected | Investigation | Treatment |
|---|---|---|---|---|
| Insomnia | Difficulty initiating/maintaining sleep | All stages | Sleep diary, PSG | CBT-I, hypnotics |
| OSA | Apneas + snoring + EDS | N3, REM worse | PSG (AHI) | CPAP, weight loss |
| Narcolepsy | EDS + cataplexy | REM dysregulation | MSLT (SOREMP) | Modafinil, Na-oxybate |
| RBD | Acting out dreams | REM (loss of atonia) | PSG (REM without atonia) | Clonazepam, melatonin |
| Sleepwalking | Complex motor acts during sleep | N3 (NREM) | PSG | Safety, treat precipitants |
| RLS | Urge to move legs at rest | Sleep onset/N1 | Clinical, serum iron | Dopamine agonists |
Sources: Adams and Victor's Principles of Neurology 12e; Kaplan & Sadock's Comprehensive Textbook of Psychiatry; Ganong's Review of Medical Physiology 26e; Miller's Anesthesia 10e; Kaplan and Sadock's Synopsis of Psychiatry; Bradley and Daroff's Neurology in Clinical Practice; Murray & Nadel's Textbook of Respiratory Medicine
This is a shared conversation. Sign in to Orris to start your own chat.