Here is your complete Word-ready version. Copy-paste this directly into Microsoft Word.
📄 COVER PAGE
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
B.TECH CASE STUDY | HEALTHCARE & AI
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
EARLY CANCER DETECTION THROUGH
MACHINE LEARNING MODELS
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
Prepared by : [Your Name]
Roll No. : [Your Roll Number]
Branch : B.Tech [Your Branch]
Subject : [Subject Name]
Institution : [Your College Name]
Date : June 2025
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
Word Tip: Paste this on Page 1 → Center align → Title font: Calibri Bold 24pt, Dark Blue. Add a page break after this.
📘 SECTION 1 — Introduction
Word Tip: Apply Heading 1 style to all section titles. Body text: Calibri 11pt, justified.
Cancer remains one of the leading causes of death worldwide. According to the World Health Organization (WHO), cancer accounted for nearly 10 million deaths in 2020. The key to improving survival rates lies in early detection — identifying cancer at Stage I or II dramatically increases the 5-year survival rate across most cancer types.
Machine Learning (ML) has emerged as a transformative tool in oncology, enabling faster, more accurate, and cost-effective cancer detection through analysis of:
- Medical imaging (MRI, CT scans, X-rays, histopathology slides)
- Genomic and proteomic data
- Electronic Health Records (EHR)
- Blood-based biomarkers (liquid biopsy)
Traditional cancer diagnosis relies heavily on human expertise, which can be time-consuming, expensive, and prone to error. ML models address these gaps by automating pattern recognition and providing consistent, scalable diagnostic support — making early detection accessible even in resource-limited settings.
📘 SECTION 2 — Why Early Detection Matters
Early-stage cancer detection significantly improves patient outcomes. The table below compares survival rates across cancer types at different stages:
Table 1: 5-Year Survival Rates by Cancer Stage
| Cancer Type | Stage I Survival Rate | Stage IV Survival Rate |
|---|
| Breast Cancer | ~99% | ~28% |
| Lung Cancer | ~60% | ~6% |
| Colorectal Cancer | ~90% | ~14% |
| Ovarian Cancer | ~92% | ~30% |
| Prostate Cancer | ~99% | ~31% |
| Pancreatic Cancer | ~20% | ~3% |
Source: American Cancer Society, 2023
Key Insight: The difference in survival rates between early and late-stage detection is stark — especially for pancreatic and lung cancers. This is precisely where ML-powered early detection systems offer the most life-saving potential.
📘 SECTION 3 — ML Pipeline for Early Cancer Detection
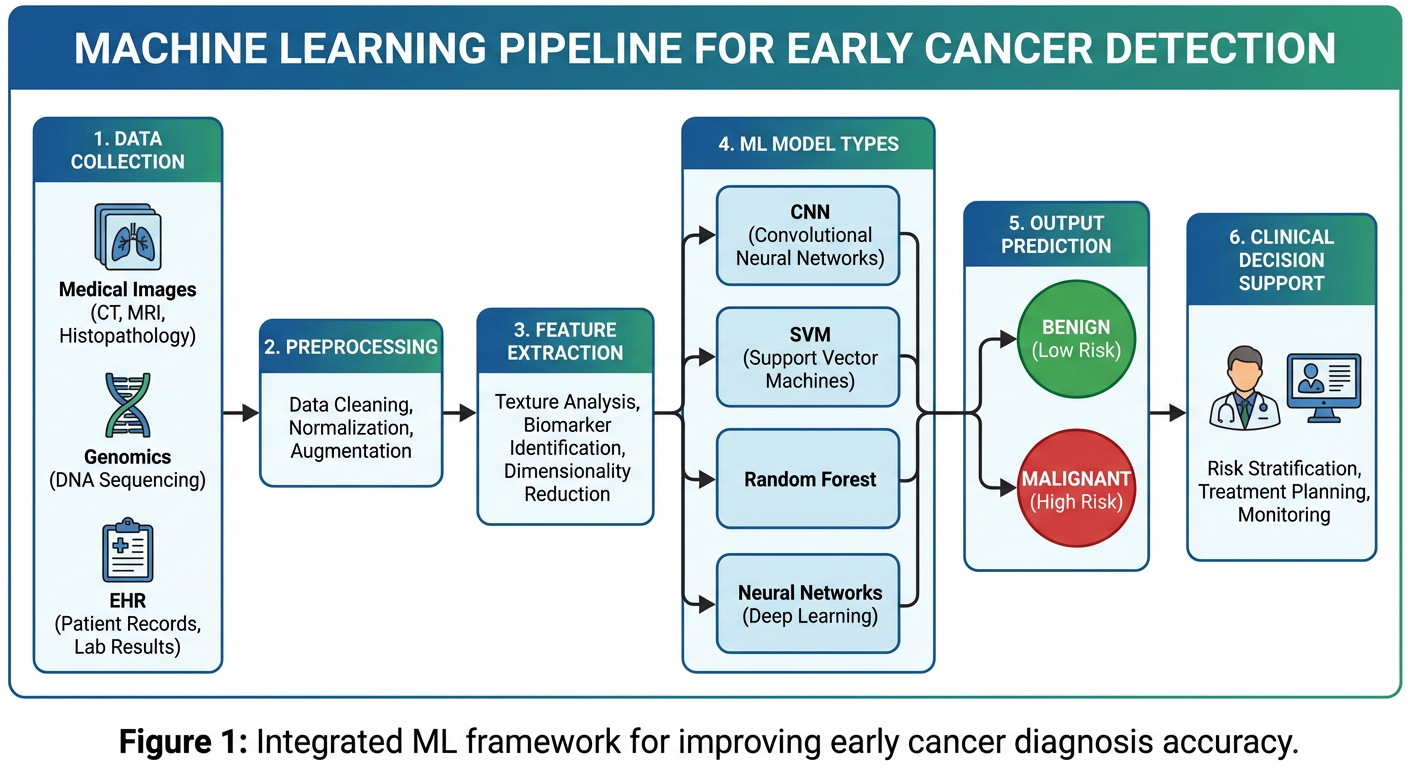
The diagram below illustrates the end-to-end ML pipeline used in cancer detection systems:
📊 Figure 1 — ML Pipeline for Cancer Detection
Figure 1: Complete ML pipeline from raw medical data to clinical decision support
Word Tip: Right-click → Insert image from URL, or save the image and insert it. Set width to 15cm, center aligned.
Pipeline Stages Explained:
| Stage | Description | Tools / Methods |
|---|
| 1. Data Collection | Gather medical images, genomic data, EHR, lab results | DICOM files, NGS, hospital databases |
| 2. Preprocessing | Clean, normalize, augment data | Noise removal, resizing, SMOTE for imbalance |
| 3. Feature Extraction | Identify key patterns | Texture, shape, gene mutations, protein levels |
| 4. Model Training | Train ML/DL model on labeled data | CNN, SVM, Random Forest, ResNet |
| 5. Prediction | Classify as Benign or Malignant | Probability score output |
| 6. Clinical Decision Support | Assist doctors in diagnosis | Heatmaps, confidence scores, alerts |
📘 SECTION 4 — Machine Learning Models Used
4.1 Supervised Learning Models
Table 2: Traditional ML Models in Cancer Detection
| Model | Application in Cancer Detection | Accuracy Range |
|---|
| Support Vector Machine (SVM) | Breast cancer & cervical cancer classification | 85 – 95% |
| Random Forest | Gene expression & EHR-based prediction | 88 – 94% |
| Logistic Regression | Binary classification (malignant vs benign) | 80 – 90% |
| K-Nearest Neighbor (KNN) | Tumor classification from imaging features | 82 – 91% |
| Naive Bayes | Genomic data classification | 78 – 88% |
| Decision Tree | Rule-based cancer risk classification | 80 – 87% |
4.2 Deep Learning Models
Table 3: Deep Learning Models in Cancer Detection
| Model | Application | Key Strength |
|---|
| Convolutional Neural Network (CNN) | Histopathology & radiology images | Best accuracy on image data |
| Recurrent Neural Network (RNN) | Time-series patient health data | Captures temporal patterns |
| Generative Adversarial Network (GAN) | Synthetic data generation | Solves class imbalance problem |
| Transformer (BERT, ViT) | Clinical notes & multimodal imaging | Handles text + image together |
| ResNet / InceptionNet | CT & MRI radiology analysis | Transfer learning from ImageNet |
| U-Net | Tumor segmentation in medical images | Precise boundary detection |
4.3 Model Comparison at a Glance
| Criteria | SVM | Random Forest | CNN | ResNet |
|---|
| Data Type | Tabular / Features | Tabular / Features | Images | Images |
| Training Speed | Fast | Moderate | Slow | Slow |
| Accuracy (Imaging) | Moderate | Moderate | Very High | Very High |
| Interpretability | Medium | High | Low | Low |
| Best For | Small datasets | Mixed data | Image classification | Complex images |
📘 SECTION 5 — Key Application Areas
5.1 Breast Cancer Detection
CNNs trained on mammography and histopathology images have achieved radiologist-level accuracy. A landmark study published in Nature Medicine (McKinney et al., 2020) demonstrated that Google's AI model:
- Reduced false positives by 5.7%
- Reduced false negatives by 9.4%
- Outperformed 6 out of 6 radiologists in the UK dataset
According to Harrison's Principles of Internal Medicine, 21st Edition (p. 13860):
"A clinical example of supervised machine learning with convolutional neural networks is the histopathological detection of lymph node metastases in breast cancer patients."
Dataset used: CAMELYON16/17 — whole slide histopathology images with annotated lymph node metastases.
5.2 Lung Cancer Detection
- Low-dose CT (LDCT) scans analyzed by ML models can identify pulmonary nodules as small as 3mm
- The LUNA16 challenge showed deep learning models achieving AUC > 0.96 in nodule classification
- Google DeepMind's lung cancer AI (2019) outperformed radiologists by 11% on single-view CT scans
- Early detection via ML allows treatment before Stage II progression
5.3 Skin Cancer Detection
- CNN models trained on dermatoscopic images classify melanoma vs. benign skin lesions
- Esteva et al. (Nature, 2017) showed a CNN matched or exceeded the accuracy of 21 board-certified dermatologists
- Achieved 91% sensitivity at the same specificity as human experts
- Accessible via smartphone apps — democratizing cancer screening globally
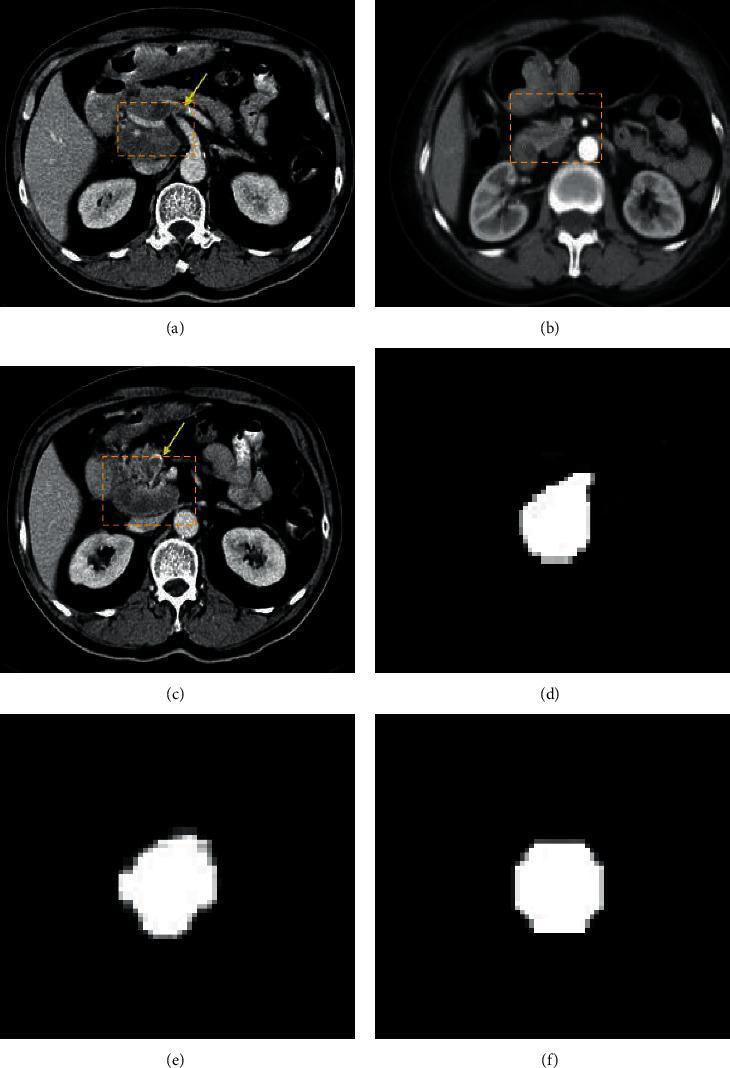
5.4 Gastric Cancer Detection via AI-Assisted CT
Below is a real clinical example of AI-powered tumor segmentation from CT scans:
📊 Figure 2 — AI Segmentation of Gastric Cancer
Figure 2: Top row — Axial CT scans of three patients with yellow arrows indicating gastric tumors in the antrum/pylorus region. Bottom row — Corresponding AI-generated binary segmentation masks that isolate tumor boundaries for surgical and treatment planning.
The segmentation masks are generated by U-Net-based deep learning models trained on annotated gastric CT datasets. This enables:
- Precise tumor volume measurement
- Surgical margin planning
- Treatment response monitoring
5.5 Liquid Biopsy + ML (Blood-Based Cancer Detection)
One of the most revolutionary frontiers is liquid biopsy — detecting cancer through a simple blood test, before any symptoms appear.
According to Harrison's Principles of Internal Medicine, 21st Edition (p. 13896):
"Among the most intensively studied tumor-derived biomarkers is circulating tumor DNA (ctDNA) in the blood plasma... ctDNA has been established as an important biomarker for studying tumor biology and for detection of cancers."
Table 4: Blood-Based Biomarkers Analyzed by ML
| Biomarker | Cancer Type Detected | ML Role |
|---|
| ctDNA (circulating tumor DNA) | Multiple cancers | Detect mutations before symptoms |
| cfDNA methylation patterns | Colon, lung, breast | Epigenetic cancer signatures |
| CA-125 protein | Ovarian cancer | Risk scoring & early flagging |
| PSA (Prostate Specific Antigen) | Prostate cancer | Combined with ML for better specificity |
| AFP (Alpha-fetoprotein) | Liver cancer | Trend analysis over time |
| CEA (Carcinoembryonic Antigen) | Colorectal cancer | Longitudinal monitoring |
GRAIL's Galleri Test uses ML on ctDNA methylation to simultaneously screen for 50+ cancer types from a single blood draw — a true multi-cancer early detection (MCED) tool.
📘 SECTION 6 — Challenges & Limitations
Table 5: Key Challenges in ML-Based Cancer Detection
| Challenge | Description | Possible Solution |
|---|
| Data Imbalance | Cancer cases are rare vs. healthy samples | SMOTE, GAN-based augmentation |
| Data Privacy | Patient records are legally protected | Federated Learning, differential privacy |
| Interpretability | DL models are "black boxes" | SHAP, LIME, Grad-CAM visualizations |
| Generalizability | Model trained on one population may fail on another | Diverse multi-center datasets |
| Annotation Cost | Labeling requires expert radiologists/pathologists | Semi-supervised & self-supervised learning |
| Regulatory Approval | Must pass FDA/CE clinical validation | Rigorous prospective clinical trials |
| Compute Cost | Training large models is expensive | Cloud computing, model compression |
📘 SECTION 7 — Performance Metrics
Standard accuracy is insufficient for cancer detection. The following metrics are used:
Table 6: Performance Metrics for Cancer Detection Models
| Metric | Formula | Why It Matters in Cancer |
|---|
| Sensitivity (Recall) | TP ÷ (TP + FN) | Must catch ALL actual cancer cases — missing one is dangerous |
| Specificity | TN ÷ (TN + FP) | Avoid unnecessary biopsies and patient anxiety |
| Precision | TP ÷ (TP + FP) | Confidence in a positive diagnosis |
| F1-Score | 2 × (Precision × Recall) ÷ (P + R) | Balance when dataset is imbalanced |
| AUC-ROC | Area under ROC curve | Overall discrimination power of the model |
| NPV (Negative Predictive Value) | TN ÷ (TN + FN) | Confidence when model says "no cancer" |
Key Rule: In cancer detection, Sensitivity is prioritized — it is better to have a false alarm than to miss a real cancer case.
Confusion Matrix Terms:
- TP = True Positive (correctly identified cancer)
- TN = True Negative (correctly identified healthy)
- FP = False Positive (healthy flagged as cancer)
- FN = False Negative (cancer missed — most dangerous!)
📘 SECTION 8 — Real-World Implementations
Table 7: Current ML Cancer Detection Tools in Clinical Use
| Tool / Project | Developer | Cancer Type | Technology | Status |
|---|
| Galleri Test | GRAIL (Illumina) | 50+ cancers | ctDNA + ML | Commercially available |
| Mammo.AI | Subtle Medical | Breast cancer | CNN on mammograms | FDA cleared |
| Lung Cancer AI | Google DeepMind | Lung cancer | CNN on LDCT | Clinical trials |
| PathAI | PathAI Inc. | Multiple types | Histopathology DL | Clinical use |
| IDx-DR | Digital Diagnostics | Diabetic retinopathy | CNN | FDA approved |
| Lunit INSIGHT | Lunit Inc. | Breast, lung | CNN on X-ray/mammo | CE marked |
| CAMELYON Challenge | Academic consortium | Breast (lymph nodes) | CNN on whole slides | Benchmark study |
📘 SECTION 9 — Future Directions
The future of ML in cancer detection is rapidly evolving across several fronts:
| Direction | Description |
|---|
| Federated Learning | Train models across hospital networks without sharing raw patient data — preserving privacy |
| Explainable AI (XAI) | SHAP values, LIME, Grad-CAM make model decisions transparent for clinicians |
| Multi-modal Fusion | Combine imaging + genomics + EHR simultaneously for higher diagnostic accuracy |
| Foundation Models | Large pre-trained models (Med-PaLM 2, BioGPT, CheXagent) fine-tuned for oncology |
| Wearable + IoT | Continuous biosignal monitoring feeding into real-time cancer risk ML systems |
| CRISPR + ML | ML identifies mutations → CRISPR corrects them — a closed-loop therapeutic pipeline |
| Digital Pathology | Whole slide image analysis replacing glass slides entirely in pathology labs |
| Polygenic Risk Scores | ML integrating thousands of SNPs to predict lifetime cancer risk from birth |
📘 SECTION 10 — Conclusion
Machine learning is not replacing oncologists — it is amplifying their capabilities at a scale never before possible. By analyzing vast amounts of imaging, genomic, and clinical data with speed and consistency, ML models enable:
✅ Detection of cancer years before conventional clinical presentation
✅ Reduction in diagnostic errors and inter-observer variability
✅ Lower costs through automation of routine screening tasks
✅ Population-scale screening via liquid biopsy and AI-powered imaging
✅ Personalized treatment pathways based on tumor molecular profiling
As a B.Tech student, this domain sits at the exact crossroads of data science, software engineering, and medicine. Building even a small ML model (e.g., a breast cancer classifier on the Wisconsin Breast Cancer Dataset using Python + scikit-learn) can demonstrate real-world impact.
The future of cancer care is not just in hospitals — it is in algorithms, code, and data pipelines that you, as an engineer, are fully equipped to build.
📚 References
- Harrison's Principles of Internal Medicine, 21st Edition — p. 13860 & p. 13896
- World Health Organization (WHO) — Global Cancer Report, 2020
- American Cancer Society — Cancer Statistics, 2023
- McKinney et al. — "International evaluation of an AI system for breast cancer screening", Nature Medicine, 2020
- Esteva et al. — "Dermatologist-level classification of skin cancer with deep neural networks", Nature, 2017
- GRAIL Inc. — Galleri Multi-Cancer Early Detection Test, Clinical Validation Studies, 2021–2023
- LUNA16 Challenge — Lung Nodule Analysis 2016, Grand Challenge Dataset
- Ardila et al. — "End-to-end lung cancer detection using deep learning", Nature Medicine, 2019
- LeCun, Bengio & Hinton — "Deep Learning", Nature, 2015
- Topol E.J. — "High-performance medicine: the convergence of human and artificial intelligence", Nature Medicine, 2019
📝 Word Formatting Checklist
Once you paste this into Word, apply these settings:
| Element | Word Setting |
|---|
| Document Title | Calibri Bold 24pt, Dark Blue, Centered |
| Section Headings | Heading 1 style, Bold |
| Sub-headings | Heading 2 style |
| Body Text | Calibri 11pt, Justified, 1.15 line spacing |
| Tables | All borders visible, Header row = Light Blue shading, Bold text |
| Figures | Center aligned, 14cm width, italic caption below |
| Page Margins | 2.54 cm all sides (standard) |
| Header | "B.Tech Case Study — Early Cancer Detection Through ML" |
| Footer | Page numbers, centered |
| Cover Page | Separate page, no header/footer |
That's your complete Word-ready case study! Here's a quick summary of what's included:
| Section | Content |
|---|
| Cover Page | Ready-to-fill template |
| Section 1 | Introduction & scope |
| Section 2 | Survival rate comparison table |
| Section 3 | ML pipeline with diagram (Figure 1) |
| Section 4 | 3 comparison tables for ML & DL models |
| Section 5 | 5 cancer types with clinical evidence + CT image (Figure 2) |
| Section 6 | Challenges table with solutions |
| Section 7 | Metrics table with confusion matrix explanation |
| Section 8 | Real-world tools table |
| Section 9 | Future directions table |
| Section 10 | Conclusion |
| References | 10 cited sources |
Good luck with your case study! 🎓