All the physiological changes in different trimesters on pregnancy
physiological changes in pregnancy trimesters cardiovascular respiratory hematological
pregnancy physiological changes renal gastrointestinal endocrine musculoskeletal skin
Physiological Changes in Pregnancy by Trimester
🫀 Cardiovascular System
| Parameter | 1st Trimester | 2nd Trimester | 3rd Trimester |
|---|---|---|---|
| Cardiac Output (CO) | ↑ 10–20% | ↑ 30–50% (peak ~20–24 wks) | ↑ 40–50% |
| Heart Rate | ↑ 10–15 bpm | ↑ 15–20 bpm | ↑ 15–20 bpm |
| Stroke Volume | Begins ↑ | Peak ↑ | Maintained |
| Blood Pressure (BP) | Slight ↓ | Lowest (~mid-trimester) | Returns to near-normal |
| Systemic Vascular Resistance | ↓ | ↓ (lowest mid-pregnancy) | Slight ↑ |
| Blood Volume | ↑ begins | ↑ 40–50% | ↑ 40–50% (peak 32–34 wks) |
- CO increases starting at 6 weeks due to progesterone-mediated vasodilation and increased estrogen driving plasma volume expansion.
- Uterine blood flow increases from ~50 mL/min at 10 weeks to ~500–750 mL/min at term.
- Aortocaval compression by the gravid uterus after 20 weeks can reduce CO in the supine position (relieved by left lateral decubitus).
🩸 Hematological Changes
| Parameter | Change | Timing |
|---|---|---|
| Plasma volume | ↑ 40–50% | Peaks at 32–34 weeks |
| Red cell mass | ↑ 20–30% | Progressive |
| Hemoglobin | ↓ (dilutional) ~11–12 g/dL | Mid-pregnancy nadir |
| Hematocrit | ↓ | Lowest 2nd trimester |
| WBC | ↑ (up to 12,000/μL) | All trimesters |
| Platelets | Slight ↓ (gestational thrombocytopenia) | 3rd trimester |
| Fibrinogen | ↑ (up to 600 mg/dL) | Progressive |
| Clotting factors (VII, VIII, X, XII) | ↑ | Progressive |
| Protein S | ↓ | Progressive |
- The disproportionate rise in plasma vs. red cell mass creates physiological anemia of pregnancy.
- The hypercoagulable state (↑ procoagulants, ↓ anticoagulants) protects against hemorrhage at delivery but increases VTE risk 4–5x.
🫁 Respiratory System
| Parameter | Change | Trimester |
|---|---|---|
| Tidal Volume | ↑ 30–40% | Progressive (especially 3rd) |
| Respiratory Rate | Minimal change or slight ↑ | — |
| Minute Ventilation | ↑ 40–50% | Progressive |
| Functional Residual Capacity (FRC) | ↓ 10–25% | 3rd trimester (diaphragm elevation) |
| Residual Volume | ↓ | 3rd trimester |
| Oxygen Consumption | ↑ 15–20% | Progressive |
| PaCO₂ | ↓ to ~30 mmHg | Progressive |
| PaO₂ | ↑ slightly | Progressive |
| Serum HCO₃⁻ | ↓ to ~18–22 mEq/L (compensated) | Progressive |
- Progesterone directly stimulates the respiratory center → chronic respiratory alkalosis with renal compensation.
- Dyspnea is reported in ~70% of pregnancies even without pathology (due to increased ventilatory drive).
- Capillary engorgement of upper airway mucosa → nasal congestion, epistaxis, difficult intubation.
🫘 Renal System
| Parameter | Change |
|---|---|
| GFR | ↑ 40–60% by 1st trimester, maintained |
| Renal plasma flow | ↑ 50–80% |
| Serum creatinine | ↓ to ~0.4–0.5 mg/dL (upper normal = 0.8) |
| BUN | ↓ |
| Serum uric acid | ↓ early, ↑ late in 3rd trimester |
| Glycosuria | Present (not pathological) |
| Proteinuria | Up to 300 mg/day (normal) |
| Ureteral dilation | ↑ (especially right side) |
- Renal sodium reabsorption increases to maintain ECF expansion despite high GFR.
- Ureteral dilation (hydroureteronephrosis) is due to progesterone-mediated smooth muscle relaxation + uterine compression → increased UTI risk.
- Creatinine and BUN are lower; values considered "normal" outside pregnancy may indicate renal impairment in pregnancy.
🦠 Gastrointestinal System
| Parameter | Change | Clinical Implication |
|---|---|---|
| Gastric emptying | Delayed (especially 2nd/3rd trimester) | ↑ Aspiration risk under anesthesia |
| GI motility/transit time | ↓ (progesterone effect) | Constipation, bloating |
| Lower esophageal sphincter tone | ↓ | GERD, heartburn |
| Gastric pH | ↑ (less acidic) | Affects oral drug absorption |
| Liver: alkaline phosphatase | ↑ (placental isoform) | Not a marker of liver disease |
| Serum albumin | ↓ | ↓ Drug protein binding |
| Gallbladder motility | ↓ | ↑ Risk of cholelithiasis |
- Nausea and vomiting (1st trimester) are mediated by hCG peaking at 8–10 weeks.
- Increased gastric pressure from the gravid uterus worsens GERD in the 3rd trimester.
⚗️ Endocrine System
Thyroid
- Thyroid-binding globulin (TBG) ↑ → total T3 and T4 ↑, but free T4 and T3 remain normal.
- hCG (structural similarity to TSH) → mild thyroid stimulation in 1st trimester → TSH transiently ↓.
- Iodine requirements ↑; iodine clearance ↑.
Adrenal
- Cortisol ↑ (CBG increases; free cortisol also slightly ↑ — physiological hypercortisolism).
- Aldosterone ↑ significantly to maintain sodium balance.
Pancreas / Glucose Metabolism
| Trimester | Insulin Sensitivity | Glucose Effect |
|---|---|---|
| 1st | Normal to slightly ↑ | Normal or lower fasting glucose |
| 2nd | ↓ begins (HPL effect) | Begins insulin resistance |
| 3rd | Marked ↓ insulin sensitivity | Risk of gestational diabetes |
- Human placental lactogen (hPL/HPL), progesterone, cortisol, and prolactin all antagonize insulin → progressive insulin resistance favoring fetal glucose supply.
- Fasting hypoglycemia can occur in 1st trimester (glucose diverted to fetus).
Pituitary
- Pituitary enlarges up to 135% (mainly prolactin-secreting cells).
- Prolactin ↑ progressively (up to 200 ng/mL at term).
- GH secretion from pituitary is suppressed; placental GH takes over.
🦴 Musculoskeletal System
- Relaxin (peaks at 12 weeks, then again at term) → ligamentous laxity throughout the body.
- Pubic symphysis widens (~3–4 mm normally, pathological if >10 mm).
- Sacroiliac joint mobility ↑ → lower back pain.
- Center of gravity shifts forward → lumbar lordosis increases → lordotic posture.
- Diastasis recti abdominis (separation of rectus muscles) develops in 3rd trimester.
🧴 Skin & Integumentary
| Change | Mechanism | Trimester |
|---|---|---|
| Hyperpigmentation (melasma, linea nigra) | ↑ MSH, estrogen, progesterone | 2nd–3rd |
| Striae gravidarum | Skin stretching + altered collagen | 3rd |
| Spider angiomata, palmar erythema | ↑ Estrogen → vasodilation | 2nd–3rd |
| Increased sweating | ↑ Metabolic rate | All |
| Hair: anagen phase prolonged | ↑ Estrogen | All (telogen effluvium postpartum) |
🧠 Neurological & Posture
- Mild cognitive changes (forgetfulness — "pregnancy brain") due to hormonal shifts and sleep disruption.
- Carpal tunnel syndrome in 3rd trimester (fluid retention compresses median nerve).
- Meralgia paresthetica (lateral femoral cutaneous nerve compression).
📊 Summary: Key Lab Value Changes in Pregnancy
| Lab Value | Non-Pregnant | Pregnant |
|---|---|---|
| Hemoglobin | 12–16 g/dL | ≥10.5 g/dL (acceptable) |
| Creatinine | 0.6–1.1 mg/dL | 0.4–0.8 mg/dL |
| BUN | 10–20 mg/dL | 5–12 mg/dL |
| Albumin | 3.5–5.0 g/dL | 2.8–4.0 g/dL |
| Alk Phos | 30–130 U/L | Up to 2–4× normal |
| WBC | 4.5–11 × 10³ | Up to 15–16 × 10³ |
| Fibrinogen | 200–400 mg/dL | 400–600 mg/dL |
| TSH | 0.4–4.0 mIU/L | Lower in 1st trimester |
| PaCO₂ | 35–45 mmHg | ~30 mmHg |
| HCO₃⁻ | 22–26 mEq/L | 18–22 mEq/L |
Conditions where fsh is low
low FSH causes hypogonadotropic hypogonadism conditions
FSH suppression hyperprolactinemia hypothalamic pituitary disorders secondary hypogonadism
Conditions Where FSH is Low
🧠 Mechanism of FSH Regulation
Hypothalamus → GnRH (pulsatile)
↓
Anterior Pituitary → FSH + LH
↓
Gonads → Sex steroids (Estrogen/Testosterone) + Inhibin B
↓ (negative feedback)
Hypothalamus + Pituitary ← suppressed
📋 Classification of Conditions Causing Low FSH
1. 🧠 Hypothalamic Causes (↓ GnRH → ↓ FSH + LH)
A. Functional Hypothalamic Amenorrhea / Anovulation
- Most common cause of low FSH in reproductive-age women
- Triggered by:
- Excessive exercise (female athlete triad)
- Caloric restriction / anorexia nervosa / malnutrition
- Psychological stress
- FSH is low-normal or low; LH pulsatility is disrupted
- Reversible with restoration of body weight/stress reduction
B. Kallmann Syndrome
- Congenital GnRH deficiency + anosmia (absent sense of smell)
- X-linked (KAL1/ANOS1 gene) or autosomal (FGFR1, PROKR2 mutations)
- Presents as complete absence of puberty, sexual infantilism
- FSH and LH both very low/undetectable (Harrison's, p. 10973)
C. Idiopathic Hypogonadotropic Hypogonadism (IHH)
- Same as Kallmann but without anosmia
- GnRH neuron migration or secretion defect
- FSH + LH low; testosterone/estrogen low
D. Hypothalamic Tumors / Infiltrative Disease
- Craniopharyngioma — most common hypothalamic tumor causing HH
- Germinoma, glioma, hamartoma
- Infiltrative: sarcoidosis, histiocytosis X (Langerhans cell histiocytosis), tuberculosis, hemochromatosis
- Radiation to hypothalamus
2. 🫐 Pituitary Causes (Direct gonadotrope failure → ↓ FSH)
A. Hyperprolactinemia
- PRL inhibits hypothalamic GnRH secretion via dopaminergic pathways; may also destroy gonadotropes via tumor mass effect
- Causes:
- Prolactinoma (most common pituitary tumor)
- Drugs (dopamine antagonists: antipsychotics, metoclopramide, domperidone)
- Hypothyroidism (↑ TRH → ↑ PRL)
- Renal failure, chest wall lesions
- FSH and LH both suppressed; treatment with dopamine agonists reverses deficiency (Harrison's, p. 10982)
B. Pituitary Tumors (Non-functioning or Functioning)
- Mass effect compresses gonadotropes
- GH-secreting (acromegaly), ACTH-secreting (Cushing's disease), or non-functioning adenomas
- Pituitary carcinoma (rare)
C. Sheehan's Syndrome
- Postpartum pituitary necrosis due to ischemia (massive obstetric hemorrhage)
- Panhypopituitarism; FSH/LH among first to be lost
- Presents with failure of lactation, amenorrhea, hypothyroidism, adrenal insufficiency
D. Pituitary Apoplexy
- Acute hemorrhage or infarction into a pituitary adenoma
- Sudden-onset headache, visual loss, panhypopituitarism
- Emergency
E. Empty Sella Syndrome
- Herniation of subarachnoid space into sella → pituitary compression
- Primary or secondary (post-surgery/radiation)
- Variable hormone deficiencies including low FSH
F. Pituitary Infiltrative/Inflammatory Disease
- Lymphocytic hypophysitis (autoimmune; common peripartum)
- Sarcoidosis, tuberculosis, hemochromatosis involving pituitary
- Radiation-induced hypopituitarism (delayed; dose-dependent)
G. Isolated FSH Deficiency
- Very rare; mutations in FSH-β subunit gene
- Women: amenorrhea, infertility; Men: azoospermia
3. 📉 Negative Feedback / Suppression States
A. Pregnancy
- High estrogen + progesterone + hCG → strong negative feedback → FSH suppressed throughout pregnancy
- Physiological; FSH normalizes postpartum
B. Exogenous Sex Steroids / Hormonal Contraceptives
- Combined oral contraceptive pills (COCPs), hormone replacement therapy, anabolic steroids
- Testosterone therapy in men → suppress FSH via negative feedback
- Anabolic steroid abuse — major cause of low FSH + LH + testosterone in men (despite normal/high androgens)
C. Ovarian / Testicular Hypersecretion
- Estrogen-secreting tumors (granulosa cell tumor) → suppress FSH
- Testosterone-secreting tumors (Leydig cell tumor, adrenal tumor)
- Congenital adrenal hyperplasia (CAH) — excess androgens suppress HPG axis
D. High Inhibin B / Inhibin A States
- Granulosa cell tumors secrete inhibin → selectively suppress FSH (LH may be normal)
- Used as tumor marker
4. 🔁 Systemic / Chronic Illness
| Condition | Mechanism |
|---|---|
| Obesity | ↑ Estrogen (aromatization in adipose), leptin resistance, insulin resistance → HPG suppression |
| Type 2 diabetes / metabolic syndrome | Insulin resistance affects GnRH pulsatility |
| Chronic kidney disease | Hyperprolactinemia + direct HPG disruption |
| Liver cirrhosis | ↑ Estrogen metabolism impairment → feedback suppression |
| Hemochromatosis | Iron deposition in pituitary → selective gonadotrope destruction |
| Chronic illness / critical illness | Functional suppression (cytokine-mediated) |
| HIV/AIDS | Multifactorial HPG suppression |
| Opioid use | Opioids suppress GnRH pulsatility → opioid-induced hypogonadism |
| Glucocorticoid excess (Cushing's) | Suppresses GnRH + LH/FSH secretion |
5. 🧒 Special Populations
Prepubertal Children (Normal)
- FSH is physiologically low before puberty — not pathological.
Constitutional Delay of Growth and Puberty (CDGP)
- Delayed but ultimately normal puberty
- Temporarily low FSH/LH; bone age delayed
- Distinguished from true HH by GnRH stimulation test
🔬 Diagnostic Approach to Low FSH
Low FSH confirmed
│
├─ Check LH, testosterone/estradiol, prolactin, TSH
│
├─ PRL elevated? → Prolactinoma / drug-induced / hypothyroidism
│
├─ MRI pituitary/hypothalamus
│ → Tumor, empty sella, infiltration
│
├─ Anosmia? → Kallmann syndrome
│
├─ BMI low / extreme exercise / stress?
│ → Functional hypothalamic HH
│
├─ Exogenous steroids / opioids?
│ → Drug-induced suppression
│
└─ GnRH stimulation test
→ Differentiates hypothalamic vs. pituitary cause
📊 Quick Reference Summary
| Category | Examples | FSH | LH | Testosterone/Estradiol |
|---|---|---|---|---|
| Functional HH | Anorexia, exercise | ↓ | ↓ | ↓ |
| Kallmann / IHH | Genetic GnRH deficiency | ↓↓ | ↓↓ | ↓↓ |
| Hyperprolactinemia | Prolactinoma, drugs | ↓ | ↓ | ↓ |
| Pituitary tumor/damage | Adenoma, Sheehan's | ↓ | ↓ | ↓ |
| Exogenous steroids | COCP, anabolic steroids, TRT | ↓ | ↓ | Variable |
| Granulosa cell tumor | Inhibin excess | ↓ | Normal | ↑ Estrogen |
| Pregnancy | hCG + estrogen feedback | ↓↓ | ↓↓ | ↑↑ |
| Cushing's disease | Cortisol excess | ↓ | ↓ | ↓ |
| Opioid use | Endogenous opioid excess | ↓ | ↓ | ↓ |
Tranverse vaginal septum
transverse vaginal septum embryology classification clinical features management
transverse vaginal septum anatomy diagram
Transverse Vaginal Septum (TVS)
🧬 Embryology & Pathogenesis
- Upper 2/3 — from the Müllerian (paramesonephric) ducts, which fuse to form the uterovaginal canal.
- Lower 1/3 — from the urogenital sinus (sinovaginal bulbs), which proliferate upward as the vaginal plate.
- Failure of complete canalization of the vaginal plate, OR
- Failure of fusion between the Müllerian duct-derived upper vagina and the urogenital sinus-derived lower vagina at their junction
📍 Location & Classification
By Location
| Level | Frequency | Clinical Significance |
|---|---|---|
| Upper 1/3 (near cervix) | ~46% | Most common; thicker, harder to resect |
| Middle 1/3 | ~35% | Intermediate difficulty |
| Lower 1/3 | ~19% | Least common; easiest surgical access |
Higher septum = thicker septum = more difficult surgery and worse prognosis for fertility.
By Morphology
| Type | Description |
|---|---|
| Complete (Imperforate) | Total obstruction; menstrual blood cannot drain → hematocolpos/hematometra |
| Incomplete (Microperforate) | Small opening allows some drainage; may present later or be asymptomatic until coitus |
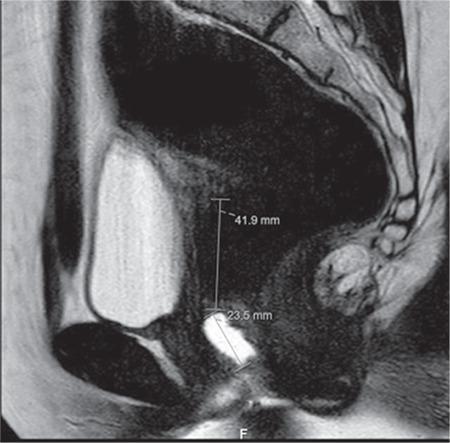
🔬 MRI Appearance
🏥 Clinical Presentation
Neonatal Period
- Usually identified incidentally or if mucocolpos (mucus accumulation) causes a palpable pelvic mass or urinary obstruction.
Adolescence (Most Common Presentation)
- Primary amenorrhea with cyclic pelvic pain (cryptomenorrhea) — classic
- Hematocolpos — blood accumulates below septum → lower abdominal mass
- Hematometra — blood accumulates in uterus if long-standing
- Hematosalpinx — retrograde filling of fallopian tubes
- Possible urinary symptoms (retention, frequency) from mass compression
- Endometriosis (from retrograde menstruation) is a long-term complication
Reproductive Age (Incomplete Septum)
- Dyspareunia — painful intercourse
- Infertility or recurrent pregnancy loss
- Difficulty with tampon insertion
- Abnormal vaginal bleeding patterns
- May only be diagnosed during gynecological examination or infertility workup
🆚 Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Imperforate hymen | Bulging bluish membrane at introitus; hymen is at the vaginal opening, not inside |
| Müllerian agenesis (MRKH) | No vagina or rudimentary vagina; normal karyotype (46,XX); no uterus on imaging |
| Androgen Insensitivity Syndrome (AIS) | 46,XY; absent pubic/axillary hair; testosterone in male range; absent uterus |
| Cervical agenesis/stenosis | Blind upper vagina with uterus present; cervix absent on imaging |
| Longitudinal vaginal septum | Vertical septum; causes double vaginal canal rather than obstruction |
🔍 Diagnosis
Clinical Examination
- Inspection: Normal external genitalia and hymen (unlike imperforate hymen)
- Speculum/digital exam: Blind-ending vaginal pouch; cervix not visualized
- In complete TVS: vault appears smooth with no cervix visible
Imaging
| Modality | Role |
|---|---|
| Pelvic MRI (gold standard) | Defines septum location, thickness, distance from introitus, hematocolpos/metra extent; essential for surgical planning |
| Transabdominal/transperineal USS | Initial investigation; identifies hematocolpos, uterus, ovaries |
| Transvaginal USS | Limited if septum is low; helpful for upper anatomy |
| Vaginoscopy | Direct visualization; useful in children |
MRI is critical — it measures:
- Distance from introitus to inferior septum (determines surgical approach)
- Septum thickness (predicts surgical difficulty)
- Upper vaginal length (predicts functional outcome)
🔧 Management
Surgical Correction — Excision of the Septum
Surgical Principles
- Excision of the septal tissue (not just incision — to prevent re-stenosis)
- Re-anastomosis of upper and lower vaginal segments
- Primary closure with interrupted absorbable sutures
- Vaginal stenting/dilation postoperatively to prevent re-stenosis
Approach Based on Location
| Septum Level | Surgical Approach |
|---|---|
| Low (lower 1/3) | Transvaginal excision; straightforward |
| Mid (middle 1/3) | Transvaginal with possible intraoperative USS or laparoscopic guidance |
| High (upper 1/3) | Most challenging; may need combined abdominal-vaginal approach or laparoscopic assistance; pull-through vaginoplasty |
Key Surgical Steps (Transvaginal Approach)
- Identify the septum
- Aspirate hematocolpos (if present) to decompress and identify the upper vagina
- Cruciate incision in the septum → excise septal ring completely
- Mobilize upper and lower vaginal walls
- End-to-end anastomosis
- Insert vaginal stent/mold postoperatively (worn for weeks to months)
Postoperative Care
- Vaginal dilators — used regularly to maintain patency and prevent restenosis (critical especially for high septa)
- Follow-up examinations to confirm patency
- Psychological support and counseling
⚕️ Complications
If Untreated
- Progressive hematocolpos → hematometra → hematosalpinx
- Endometriosis (retrograde menstruation) — significantly impairs fertility
- Pelvic adhesions
- Pyocolpos (infected collection)
- Renal anomalies (associated in some cases)
Post-Surgical
- Restenosis / re-obstruction — most important complication, especially with high septa
- Dyspareunia
- Infertility (reduced fertility with high septa despite surgery)
- Wound dehiscence
🤰 Fertility Outcomes
| Septum Location | Fertility Prognosis |
|---|---|
| Low | Generally good with timely surgery |
| Mid | Moderate |
| High | Reduced — associated with worse outcomes due to endometriosis, cervical proximity, scarring |
- Early diagnosis and surgery before endometriosis develops significantly improves fertility.
- Pregnancy rates after surgical correction range from 25–50% depending on septum level and extent of associated disease.
🔗 Associated Anomalies
- Renal anomalies (horseshoe kidney, renal agenesis) — due to shared embryological origin with mesonephric system
- Longitudinal vaginal septum
- Uterine anomalies (septate/bicornuate uterus)
- Ovarian dysgenesis (rare)
📝 Summary
| Feature | Details |
|---|---|
| Incidence | ~1 in 30,000–80,000 females |
| Embryological defect | Failed canalization of vaginal plate / fusion failure at sinovaginal junction |
| Most common site | Upper 1/3 of vagina |
| Classic presentation | Primary amenorrhea + cyclic pain in adolescence |
| Gold standard imaging | Pelvic MRI |
| Treatment | Surgical excision + anastomosis + postop dilation |
| Key complication | Restenosis; endometriosis if delayed |
| Differentiator from imperforate hymen | Normal hymen; septum is intravaginal |
What is the steps of investigation for primary amenorrhoea
primary amenorrhea investigation workup steps algorithm
primary amenorrhea causes differential diagnosis karyotype FSH LH
Investigation of Primary Amenorrhea — Step-by-Step Approach
📌 Definition & When to Investigate
| Situation | Age to Investigate |
|---|---|
| Normal growth + normal secondary sexual characteristics | Age 15–16 |
| No secondary sexual characteristics | Age 13 |
| Height < 3rd percentile | Age 13 |
| Breast development + cyclic pelvic pain (suggests outflow obstruction) | Age 12–13 |
| Breast development present but no menarche | Within 2 years of breast development |
🧭 Overview of the Approach
- Are secondary sexual characteristics (breasts, pubic hair) present? → Reflects estrogen/androgen action
- Is the uterus present? → Reflects Müllerian development
🔢 Step-by-Step Investigation
✅ STEP 1 — History & Physical Examination
- Age of onset of any pubertal signs
- Cyclic pelvic pain (suggests outflow obstruction with functional uterus)
- Anosmia (Kallmann syndrome)
- Headaches, visual field defects (pituitary tumor)
- Weight changes, exercise habits, stress (functional hypothalamic amenorrhea)
- Drug history (antipsychotics, chemotherapy, steroids)
- Family history of delayed puberty or amenorrhea
- Symptoms of hypothyroidism, Cushing's, chronic illness
- Height, weight, BMI
- Turner stigmata: short stature, webbed neck, wide-carrying angle, shield chest, low hairline
- Breast development (Tanner staging) — reflects estrogen
- Pubic/axillary hair (Tanner staging) — reflects androgens
- External genitalia: clitoromegaly, labial fusion, hymenal appearance
- Vaginal examination: depth, presence of cervix, blind vaginal pouch
- Thyroid palpation
- Signs of hyperandrogenism (acne, hirsutism)
- Signs of galactorrhea (→ hyperprolactinemia)
✅ STEP 2 — Pregnancy Test
- β-hCG (urine or serum) — must be excluded first in any woman of reproductive age regardless of history.
✅ STEP 3 — Assess Secondary Sexual Characteristics
🔵 BRANCH A: No Breast Development (No Estrogen Effect)
| Test | Purpose |
|---|---|
| FSH + LH | Key differentiator |
| Karyotype | Identify gonadal dysgenesis |
| Estradiol | Confirms hypoestrogen state |
| Testosterone | Elevated in some XY disorders |
| Prolactin | Rule out hyperprolactinemia |
| TSH | Rule out hypothyroidism |
| MRI brain/pituitary | If FSH/LH low → hypothalamic or pituitary pathology |
| Pelvic USS | Assess uterus and ovaries/streak gonads |
| Bone age X-ray | If constitutional delay suspected |
| FSH/LH | Likely Cause |
|---|---|
| ↑ FSH + ↑ LH (Hypergonadotropic) | Primary gonadal failure → check karyotype |
| ↓ FSH + ↓ LH (Hypogonadotropic) | Hypothalamic or pituitary cause → MRI pituitary |
- 45,X → Turner syndrome (most common cause of gonadal dysgenesis)
- 46,XX → Pure gonadal dysgenesis (Swyer if XY)
- 46,XY → Swyer syndrome (46,XY gonadal dysgenesis)
- Mosaic patterns (45,X/46,XX etc.)
- Kallmann syndrome (+ anosmia)
- Idiopathic hypogonadotropic hypogonadism (IHH)
- Pituitary tumors (craniopharyngioma, prolactinoma)
- Hypothalamic infiltrative disease
- Constitutional delay (diagnosis of exclusion)
🔴 BRANCH B: Breast Development Present (Estrogen Present)
🔴 Branch B1: Uterus Absent on USS
| Condition | Karyotype | Key Features |
|---|---|---|
| MRKH syndrome (Müllerian agenesis) | 46,XX | Normal female; absent uterus + upper vagina; normal ovaries; normal pubic/axillary hair; testosterone normal (female range) |
| Complete Androgen Insensitivity Syndrome (AIS) | 46,XY | Female phenotype; absent/sparse pubic + axillary hair; blind vaginal pouch; testosterone in male range; testes (often inguinal) |
- Karyotype (46,XX vs. 46,XY) — essential
- Testosterone (male range in AIS, female range in MRKH)
- Pelvic MRI — confirm absence of uterus, locate gonads
- Renal USS in MRKH (30–40% have renal anomalies)
🔴 Branch B2: Uterus Present on USS
- Uterus is present and functional → menstrual blood cannot exit
- Cyclic pelvic pain ± hematocolpos is characteristic
| Test | Findings |
|---|---|
| Pelvic USS | Hematocolpos (blood-filled vagina), hematometra, hematosalpinx |
| MRI pelvis (gold standard) | Precise location + thickness of septum; surgical planning |
| Clinical examination | Bulging bluish hymen (imperforate hymen) vs. intravaginal septum (TVS) |
- Imperforate hymen — membrane at vaginal introitus; bulging, bluish with Valsalva
- Transverse vaginal septum — intravaginal; normal hymen visible
- Cervical agenesis/stenosis — rare; uterus present, no cervix
- Vaginal agenesis with functional uterus — rare variant
✅ STEP 4 — Additional Hormonal Workup (All Branches)
| Hormone | Why |
|---|---|
| Prolactin | Hyperprolactinemia → suppresses GnRH → low FSH/LH |
| TSH | Hypothyroidism → ↑ TRH → ↑ PRL → amenorrhea |
| Cortisol / DHEAS | If Cushing's or adrenal disease suspected |
| AMH (Anti-Müllerian Hormone) | Reflects ovarian reserve; very low in gonadal dysgenesis |
| Inhibin B | Low in primary ovarian insufficiency |
✅ STEP 5 — Imaging
| Modality | When to Use |
|---|---|
| Pelvic USS (transabdominal ± transperineal) | All cases — assess uterus, ovaries, streak gonads, hematocolpos |
| MRI pelvis | Outflow obstruction (TVS, cervical agenesis); anatomy before surgery |
| MRI brain/pituitary | Low FSH/LH; symptoms of pituitary lesion (headache, visual changes, galactorrhea) |
| Bone age (wrist X-ray) | Constitutional delay vs. pathological hypogonadism |
| Renal USS | MRKH, TVS (associated renal anomalies) |
| Echocardiogram | Turner syndrome (bicuspid aortic valve, coarctation) |
✅ STEP 6 — Karyotype
- No breast development
- Absent uterus
- Any suspicion of gonadal dysgenesis
- Short stature with Turner features
- Male-range testosterone
🗺️ Investigation Algorithm at a Glance
Primary Amenorrhea
│
├─ β-hCG → Exclude pregnancy
│
├─ SECONDARY SEXUAL CHARACTERISTICS?
│
├─ NO BREASTS (No estrogen)
│ │
│ ├─ FSH/LH HIGH → Gonadal failure
│ │ └─ Karyotype:
│ │ 45,X → Turner syndrome
│ │ 46,XX → Pure gonadal dysgenesis
│ │ 46,XY → Swyer syndrome
│ │
│ └─ FSH/LH LOW → Hypothalamic/Pituitary
│ └─ MRI brain → Kallmann / IHH / Tumor
│
└─ BREASTS PRESENT (Estrogen present)
│
├─ UTERUS ABSENT (USS)
│ └─ Karyotype:
│ 46,XX → MRKH syndrome
│ 46,XY → Complete AIS
│ (testosterone to differentiate)
│
└─ UTERUS PRESENT (USS)
└─ Outflow tract obstruction
├─ Imperforate hymen (bulging introitus)
├─ Transverse vaginal septum (MRI)
└─ Cervical agenesis (MRI)
📊 Summary Table of Causes by Branch
| Breast | Uterus | Karyotype | Diagnosis |
|---|---|---|---|
| Absent | Present | 46,XX | Hypogonadotropic HH (Kallmann, IHH, hypothalamic) |
| Absent | Present | 46,XX | Primary ovarian insufficiency (↑FSH) |
| Absent | Absent/streak | 45,X | Turner syndrome |
| Absent | Absent | 46,XY | Swyer syndrome |
| Present | Absent | 46,XX | MRKH syndrome |
| Present | Absent | 46,XY | Complete AIS |
| Present | Present | 46,XX | Outflow obstruction (imperforate hymen, TVS) |
| Present | Present | 46,XX | Hypothalamic amenorrhea (low FSH) |
| Present | Present | 46,XX | Hyperprolactinemia |
Scenrios where fsh levels are high
high FSH elevated levels causes hypergonadotropic hypogonadism conditions
elevated FSH menopause premature ovarian insufficiency gonadal dysgenesis Turner syndrome
Scenarios Where FSH is High
🔬 Why FSH Rises
- Estradiol + Inhibin B (from granulosa cells / Sertoli cells) suppress FSH at the pituitary
- Testosterone suppresses LH predominantly
FSH is a better marker of gonadal failure than LH because it reflects loss of both estradiol AND inhibin B feedback, and it is less variable. (Harrison's, p. 11055)
📋 All Scenarios Where FSH is Elevated
1. 🔴 Natural Menopause
- Most common physiological cause of high FSH
- Ovarian follicular reserve depleted → ↓ estradiol + ↓ inhibin B → FSH rises markedly
- FSH >25–40 IU/L on two occasions confirms menopause
- Average age: 51 years
- LH also elevated; estradiol low
2. 🟠 Premature Ovarian Insufficiency (POI)
- Ovarian failure before age 40; accounts for ~10% of secondary amenorrhea
- FSH >25 IU/L on two measurements ≥4 weeks apart (in the presence of amenorrhea ≥4 months)
- AMH very low; estradiol low
- May wax and wane — serial measurements needed to confirm
- Causes include:
| Category | Examples |
|---|---|
| Genetic | Turner syndrome (45,X), FMR1 premutation (Fragile X), 46,XX gonadal dysgenesis, BMP15 mutations |
| Autoimmune | Autoimmune oophoritis (often with Addison's disease, thyroid disease — polyglandular syndrome) |
| Iatrogenic | Chemotherapy, pelvic radiotherapy, bilateral oophorectomy |
| Infectious | Mumps oophoritis (rare) |
| Idiopathic | ~50–75% of cases |
3. 🟡 Gonadal Dysgenesis
Turner Syndrome (45,X)
- Streak gonads with no functional follicles → no estrogen/inhibin → markedly elevated FSH from birth
- FSH very high in infancy, falls slightly in childhood, rises again at expected puberty
- Primary amenorrhea, absent breast development, short stature, typical stigmata
Pure Gonadal Dysgenesis
- 46,XX gonadal dysgenesis — normal female phenotype, streak gonads, high FSH
- 46,XY gonadal dysgenesis (Swyer syndrome) — female phenotype, XY karyotype, streak gonads → high FSH; gonads must be removed (gonadoblastoma risk)
Mosaic Turner (45,X/46,XX)
- Variable gonadal function; FSH may be elevated or normal depending on residual ovarian tissue
4. 🟢 Post-Oophorectomy / Surgical Castration
- Bilateral oophorectomy (surgical menopause) → abrupt loss of ovarian feedback
- FSH rises rapidly within days; can reach very high levels (>100 IU/L)
- Sudden onset of menopausal symptoms
5. 🔵 Iatrogenic Gonadal Damage
| Cause | Mechanism |
|---|---|
| Chemotherapy (alkylating agents: cyclophosphamide, busulfan, chlorambucil) | Direct follicular toxicity |
| Pelvic/gonadal radiation | Dose-dependent follicular destruction; >6 Gy often causes permanent POI |
| Galactosemia | Galactose metabolites toxic to ovarian follicles → POI + high FSH |
6. ⚫ Primary Testicular Failure (Males)
| Condition | Notes |
|---|---|
| Klinefelter syndrome (47,XXY) | Most common cause; small firm testes, azoospermia, gynecomastia; FSH markedly elevated |
| Bilateral cryptorchidism | If untreated; tubular atrophy |
| Mumps orchitis | Post-inflammatory tubular damage |
| Testicular torsion (bilateral) | Ischemic damage |
| Chemotherapy/radiotherapy | As in females |
| Sertoli cell-only syndrome (Del Castillo) | Absent germinal epithelium; FSH high, LH + testosterone often normal |
| Y chromosome microdeletions (AZF regions) | FSH high; azoospermia or severe oligospermia |
| Post-orchidectomy | FSH rises after removal |
In men, FSH reflects tubular/Sertoli cell integrity (inhibin B). LH and testosterone reflect Leydig cell function. FSH can be elevated while LH + testosterone remain normal (isolated tubular failure).
7. 🟣 Enzyme Deficiencies Affecting Steroidogenesis
| Deficiency | FSH Status | Mechanism |
|---|---|---|
| Aromatase deficiency | ↑ FSH | Cannot convert androgens to estrogen → ↓ estradiol → loss of feedback; hyperandrogenism |
| 17α-hydroxylase deficiency | ↑ FSH | ↓ estradiol + ↓ cortisol; mineralocorticoid excess → hypertension; sexual infantilism |
| FSH receptor mutation | ↑ FSH | FSH produced normally but cannot act; ovarian resistance |
| LH receptor mutation (female) | FSH normal/↑ | LH resistance; amenorrhea despite gonadotropins |
8. 🔶 Perimenopause (Menopausal Transition)
- FSH begins rising years before the final menstrual period
- Irregular cycles; FSH may fluctuate between normal and elevated
- Inhibin B falls first → FSH rises selectively (LH often still normal initially)
- FSH >10–12 IU/L on day 3 suggests diminished ovarian reserve in reproductive-age women
9. 🔷 Diminished Ovarian Reserve (DOR)
- Not complete ovarian failure, but reduced follicular pool
- Day 3 FSH >10 IU/L = marker of poor ovarian reserve
- Used in infertility assessment alongside AMH and antral follicle count (AFC)
- Associated with poor IVF response
10. 🟤 Gonadotropin-Secreting Pituitary Tumors
- FSH-secreting adenomas (rare) → autonomous FSH secretion independent of GnRH feedback
- May cause ovarian hyperstimulation, large multicystic ovaries, elevated estradiol
- Paradoxically: high FSH + high estradiol (not low) — distinguishes from gonadal failure
- Diagnosed on MRI pituitary + elevated α-subunit
11. 🩶 Physiological States
| State | FSH Level | Notes |
|---|---|---|
| Early follicular phase | 3–10 IU/L | Mild FSH rise drives follicle recruitment |
| Mid-cycle LH surge | FSH also surges (smaller) | Triggers ovulation alongside LH |
| Neonatal period | Temporarily elevated | "Mini-puberty" — HPG axis transiently active in first 3–6 months |
| Post-term pregnancy | Rises after delivery | Returns as gonadotropin suppression is removed |
📊 Quick Reference Summary
| Scenario | FSH | LH | Estradiol/Testosterone | Key Feature |
|---|---|---|---|---|
| Natural menopause | ↑↑ | ↑ | ↓↓ | Age >45, no menses |
| POI | ↑↑ | ↑ | ↓ | Age <40, amenorrhea |
| Turner syndrome | ↑↑↑ | ↑↑ | ↓↓ | 45,X, short stature |
| Swyer syndrome | ↑↑ | ↑↑ | ↓↓ | 46,XY, female phenotype |
| Klinefelter syndrome | ↑↑ | ↑ | Low-normal T | 47,XXY, azoospermia |
| Sertoli-cell-only | ↑↑ | Normal | Normal T | Azoospermia only |
| Post-oophorectomy | ↑↑↑ | ↑↑↑ | ↓↓↓ | Surgical history |
| Aromatase deficiency | ↑ | ↑ | ↓ (↑ androgens) | Virilization |
| 17α-hydroxylase def. | ↑ | ↑ | ↓ | Hypertension |
| FSH-secreting tumor | ↑↑ | Variable | ↑ or normal | Ovarian hyperstimulation |
| Diminished ovarian reserve | Mildly ↑ | Normal | Normal | Infertility workup |
| Perimenopause | Fluctuating ↑ | Normal-↑ | Variable | Irregular cycles |
🔑 Key Clinical Pearl
In gonadal failure (the most common cause): FSH is high + sex steroids are LOW. In FSH-secreting tumor: FSH is high + sex steroids are HIGH. This distinction is critical and prevents misdiagnosis.
Causes for premature ovarian failure
premature ovarian insufficiency failure causes etiology genetic autoimmune iatrogenic
primary ovarian insufficiency POI pathophysiology diagnosis management
Causes of Premature Ovarian Failure (Primary Ovarian Insufficiency)
📌 Definition & Terminology
- Premature Ovarian Insufficiency (POI) — preferred modern term — refers to loss of ovarian function before age 40 (Harrison's, p. 11055)
- Premature menopause = permanent cessation of menses before 40
- Early menopause = cessation between ages 40–45
- POI differs from premature menopause in that ovarian function may wax and wane — intermittent ovulation and even spontaneous pregnancy can occur in ~5–10% of cases
- Accounts for ~10% of secondary amenorrhea
- Causes span genetic, autoimmune, toxic, metabolic, and iatrogenic categories (Hormone Therapy Position Statement 2022, p. 7)
🔬 Pathophysiology
| Mechanism | Description |
|---|---|
| Follicle depletion | Accelerated atresia or inadequate initial follicle endowment → pool exhausted prematurely |
| Follicle dysfunction | Adequate follicles present but fail to respond to gonadotropins (FSH/LH resistance) |
📋 Causes of Premature Ovarian Failure
1. 🧬 Genetic Causes (~25–30% of POI)
A. Chromosomal Abnormalities
| Condition | Karyotype | Mechanism |
|---|---|---|
| Turner Syndrome | 45,X (or mosaic 45,X/46,XX) | Most common genetic cause; streak gonads; accelerated follicle atresia in utero; absent puberty (complete) or early menopause (mosaic) |
| Turner mosaicism | 45,X/46,XX; 45,X/47,XXX | Variable residual ovarian function; may have spontaneous puberty then early POI |
| Triple X syndrome | 47,XXX | Associated with early menopause/POI |
| Trisomy 21 (Down syndrome) | 47,XX+21 | Earlier menopause than general population |
| Deletions of X chromosome | del(Xq) | Critical region Xq13–q26 required for folliculogenesis |
B. Single Gene Mutations
| Gene | Mechanism | Features |
|---|---|---|
| FMR1 premutation (55–200 CGG repeats) | Most common single gene cause (~6% of sporadic POI, ~12% of familial); FMR1 premutation carriers at high risk | Associated with Fragile X syndrome in offspring; screen all POI patients |
| BMP15 (X-linked) | Bone morphogenetic protein 15; regulates folliculogenesis | POI in heterozygous females |
| GDF9 | Growth differentiation factor 9; follicle development | |
| FOXL2 | Transcription factor for granulosa cell development | FOXL2 mutations → blepharophimosis-ptosis-epicanthus inversus syndrome (BPES) + POI |
| NR5A1 (SF-1) | Steroidogenesis regulation | POI + adrenal insufficiency in some |
| NOBOX, FIGLA, SOHLH1/2 | Oocyte-specific transcription factors | Folliculogenesis failure |
| MCM8, MCM9 | DNA repair; meiotic progression | POI + chromosomal instability |
| STAG3, SYCE1, HFM1 | Synaptonemal complex / meiosis genes | Primary amenorrhea phenotype |
| EIF2B (Vanishing White Matter disease) | Ovarioleukodystrophy | POI + neurological disease |
| POLG (polymerase gamma) | Mitochondrial DNA replication | POI + progressive external ophthalmoplegia |
| ATM, BRCA1/2 | DNA damage repair | ↑ POI risk; BRCA1 particularly associated |
2. 🛡️ Autoimmune Causes (~5–30% of POI)
- Most common identifiable cause of POI in reproductive-age women
- Two subtypes:
A. Autoimmune Oophoritis
- Lymphocytic infiltration of theca cells (not granulosa initially)
- Anti-steroidogenic cell antibodies (anti-21-hydroxylase, anti-17α-hydroxylase, anti-side-chain cleavage enzyme)
- Anti-ovarian antibodies (less specific)
- Often part of Autoimmune Polyglandular Syndrome (APS):
| APS Type | Components |
|---|---|
| APS Type 1 (APS-1/APECED) | Hypoparathyroidism + Mucocutaneous candidiasis + Addison's disease; AIRE gene mutation; POI in ~60% |
| APS Type 2 (Schmidt syndrome) | Addison's disease + autoimmune thyroid disease ± type 1 diabetes; POI in ~10% |
- Adrenal antibodies (anti-21-hydroxylase) are the best marker — positive in ~4% of POI but predict autoimmune oophoritis
B. Associated Autoimmune Conditions
| Condition | Notes |
|---|---|
| Autoimmune thyroid disease (Hashimoto's, Graves') | Most common autoimmune association; screen all POI patients |
| Addison's disease | Critical to diagnose — life-threatening; 50% of Addison's patients with adrenal antibodies develop POI |
| Type 1 Diabetes Mellitus | Shared autoimmune predisposition |
| Myasthenia gravis | |
| Systemic lupus erythematosus (SLE) | Direct autoimmune damage + iatrogenic (cyclophosphamide) |
| Rheumatoid arthritis | |
| Crohn's disease |
3. ☢️ Iatrogenic Causes
A. Chemotherapy
- Alkylating agents — most gonadotoxic:
- Cyclophosphamide, busulfan, chlorambucil, melphalan, procarbazine
- Destroy primordial follicles directly
- Risk factors: older age, higher cumulative dose, combined regimens
- Platinum compounds (cisplatin), taxanes — intermediate risk
- Methotrexate, vincristine — low risk
- Fertility preservation (oocyte/embryo cryopreservation, ovarian tissue cryopreservation) recommended before chemotherapy
B. Radiotherapy
- Dose-dependent follicular destruction:
| Dose to Ovary | Effect |
|---|---|
| <2 Gy | Minimal risk |
| 2–6 Gy | Significant risk, especially in older women |
| >6 Gy | High risk of permanent POI |
| >20 Gy | Near-certain permanent ovarian failure |
- Whole-body irradiation (bone marrow transplant conditioning) → very high POI risk
- Cranial irradiation → hypothalamic-pituitary damage (central, not primary ovarian)
- Ovarian transposition (oophoropexy) can reduce radiation exposure
C. Surgery
- Bilateral oophorectomy → immediate surgical menopause
- Unilateral oophorectomy → accelerates decline in ovarian reserve; earlier menopause
- Ovarian cystectomy (especially for endometrioma) → removes healthy ovarian cortex → reduces reserve
- Hysterectomy without oophorectomy → may impair ovarian blood supply → earlier POI
4. ⚗️ Metabolic / Enzyme Deficiency Causes
| Condition | Mechanism |
|---|---|
| Galactosemia (GALT gene) | Galactose-1-phosphate accumulates → direct follicular toxicity; POI in ~80% of affected females; diagnosed on newborn screening |
| 17α-hydroxylase deficiency | Block in steroidogenesis → no estrogen/androgens; ↑ FSH; hypertension + sexual infantilism |
| Aromatase deficiency (CYP19A1) | Cannot convert androgens to estrogen → ↑ FSH + virilization |
| Cholesterol desmolase (StAR) deficiency | Lipoid congenital adrenal hyperplasia; no steroid production; ↑ FSH |
5. 🦠 Infectious Causes (Rare)
| Infection | Notes |
|---|---|
| Mumps oophoritis | Most recognized; analogous to mumps orchitis in males; rare since MMR vaccination |
| Tuberculosis | Granulomatous oophoritis |
| Cytomegalovirus (CMV) | Immunocompromised patients |
| Varicella-Zoster | Rare case reports |
| HIV | Multifactorial — direct viral effect + treatment toxicity + immune dysregulation |
6. 🔌 FSH Receptor / Gonadotropin Resistance
- Inactivating FSH receptor mutations (FSHR gene) — follicles present but cannot respond to FSH → arrested folliculogenesis
- "Savage syndrome" or ovarian resistance syndrome
- High FSH, normal-sized ovaries with arrested follicles on biopsy
- More common in Finnish population (Ala189Val mutation)
- LH receptor mutations — LH resistance; secondary amenorrhea
- Antibodies against FSH receptor — postulated in some autoimmune POI cases
7. 🧠 Syndromic / Systemic Associations
| Syndrome | POI Association |
|---|---|
| Blepharophimosis-Ptosis-Epicanthus Inversus Syndrome (BPES) Type I | FOXL2 mutation; eyelid anomaly + POI |
| Ataxia Telangiectasia | ATM gene; DNA repair defect; cerebellar ataxia + POI |
| Fanconi Anemia | DNA repair defect; pancytopenia + POI |
| Perrault Syndrome | POI + sensorineural hearing loss; HSD17B4, LARS2, CLPP mutations |
| Bloom Syndrome | DNA helicase defect; short stature, photosensitivity + POI |
| Werner Syndrome | Premature aging + early menopause |
| Nijmegen Breakage Syndrome | NBN gene; immunodeficiency + POI |
8. ❓ Idiopathic (~50–75% of Cases)
- Despite thorough investigation, no identifiable cause is found in the majority
- Likely represents undiscovered genetic variants, oligogenic inheritance, or gene-environment interactions
- Research ongoing with whole-exome/genome sequencing identifying new candidate genes
📊 Summary by Category
| Category | Proportion | Key Examples |
|---|---|---|
| Idiopathic | 50–75% | Unknown |
| Genetic | 25–30% | Turner syndrome, FMR1 premutation, FOXL2, BMP15 |
| Autoimmune | 5–30% | Autoimmune oophoritis, APS-1, APS-2, thyroid disease |
| Iatrogenic | Variable | Chemotherapy, radiotherapy, surgery |
| Metabolic/Enzymatic | Rare | Galactosemia, 17α-hydroxylase deficiency |
| Infectious | Very rare | Mumps, TB |
| Gonadotropin resistance | Rare | FSHR mutation (Savage syndrome) |
🔍 Investigations Recommended in All POI Patients
| Investigation | Purpose |
|---|---|
| FSH, LH, estradiol (×2, ≥4 weeks apart) | Confirm diagnosis (FSH >25 IU/L) |
| Karyotype | Exclude Turner syndrome, X chromosome abnormalities |
| FMR1 premutation screen | Most common single gene cause |
| Anti-21-hydroxylase antibodies | Screen for autoimmune oophoritis / Addison's risk |
| Thyroid function + anti-TPO antibodies | Most common autoimmune association |
| Fasting glucose / HbA1c | Screen for T1DM |
| AMH, antral follicle count (USS) | Assess residual ovarian reserve |
| DEXA scan | Baseline bone density (estrogen deficiency risk) |
| Adrenal function (if adrenal antibodies positive) | Rule out Addison's disease |
| Echocardiogram | If Turner syndrome confirmed (cardiac anomalies) |
| Renal USS | If Turner syndrome (renal anomalies) |
⚠️ Key Clinical Points
- FMR1 premutation must always be screened — has implications for genetic counseling (risk of Fragile X in offspring) and for sisters/daughters of the proband
- Adrenal antibodies positivity warrants adrenal function testing — Addison's disease can be life-threatening if missed
- POI is not the same as menopause — spontaneous pregnancy occurs in ~5–10%; contraception still needed if pregnancy is not desired
- Hormone replacement therapy (HRT) is recommended until the natural age of menopause (~51 years) to protect bone, cardiovascular, and neurological health
Can multiparity be a cause
🔬 Evidence & Mechanisms
1. Accelerated Follicle Recruitment During Each Pregnancy
- Each pregnancy involves suspension of ovulation but not suspension of follicular atresia
- Primordial follicles continue to undergo atresia even during pregnancy
- However, during pregnancy, FSH is suppressed (by placental estrogen/progesterone/inhibin) → theoretically follicle recruitment is reduced → this is actually somewhat protective
- The net effect is debated
2. Postpartum Hormonal Disruption
- Each postpartum period involves a surge in FSH (as pituitary suppression is lifted) → accelerated follicle recruitment in the postpartum window
- Breastfeeding-associated hyperprolactinemia suppresses GnRH → intermittent FSH fluctuations during lactation may affect follicular dynamics
3. Repeated Physiological Stress on Ovarian Vasculature
- Multiple pregnancies may cause cumulative microangiopathic changes in ovarian blood supply
- Reduced ovarian perfusion → accelerated follicular atresia
4. Nutritional Depletion
- Grand multiparity (≥5 pregnancies) in resource-limited settings → chronic nutritional deficiency, micronutrient depletion (zinc, folate, iron) → may impair follicular health and mitochondrial function in oocytes
📊 What the Data Shows
| Finding | Detail |
|---|---|
| Higher parity → earlier menopause | Some epidemiological studies show a modest association between grand multiparity and earlier age at menopause |
| Low parity / nulliparity → also associated with earlier menopause | Confounds the relationship — may reflect underlying subfertility rather than a protective effect of pregnancy |
| Effect size is small | Any effect of parity on menopausal age is modest (~1–2 years at most) |
| Confounders | Socioeconomic status, nutrition, breastfeeding duration, inter-pregnancy interval all modify the relationship |
🧭 Conclusion
- Grand multiparity (≥5) in the context of poor nutrition, short inter-pregnancy intervals, and prolonged lactation may contribute to earlier ovarian reserve depletion
- It is best viewed as a modifying/contributing factor that accelerates an already ongoing biological process, rather than an independent etiology
- In clinical practice, when evaluating POI in a multiparous woman, other causes (genetic, autoimmune, iatrogenic) should still be systematically excluded before attributing it to parity alone
Fibroid management with different indications
uterine fibroid leiomyoma management treatment options indications surgery medical
fibroid myomectomy hysterectomy uterine artery embolization MRI guided focused ultrasound fertility
uterine fibroids classification FIGO symptoms heavy menstrual bleeding pelvic pressure
GnRH agonist ulipristal acetate tranexamic acid fibroid medical treatment
Fibroid (Uterine Leiomyoma) Management by Indication
📌 Background
- Symptoms (heavy bleeding, pressure, pain, infertility)
- Fibroid characteristics (size, number, location — FIGO classification)
- Patient's reproductive wishes
- Age and proximity to menopause
📍 FIGO Classification of Fibroids (Location)
| Type | Location | Clinical Relevance |
|---|---|---|
| Type 0 | Pedunculated submucosal | Easily resected hysteroscopically |
| Type 1 | Submucosal <50% intramural | Hysteroscopic resection possible |
| Type 2 | Submucosal ≥50% intramural | Hysteroscopic ± open/laparoscopic |
| Type 3 | Intramural, contacts endometrium | Medical/surgical |
| Type 4 | Intramural entirely | Medical/surgical |
| Type 5 | Subserosal ≥50% intramural | Surgical |
| Type 6 | Subserosal <50% intramural | Surgical |
| Type 7 | Pedunculated subserosal | Surgical |
| Type 8 | Other (cervical, parasitic) | Individualized |
🗂️ Management by Indication
1. 🟢 Asymptomatic Fibroids
- No treatment required in most cases
- Annual surveillance with pelvic USS
- Reassurance — fibroids regress spontaneously after menopause
- Indications to intervene despite no symptoms:
- Rapid growth (raises concern for leiomyosarcoma — rare, ~0.5%)
- Size >10–12 cm causing silent ureteric compression
- Significant distortion of uterine cavity before planned IVF
2. 🔴 Heavy Menstrual Bleeding (HMB)
🔹 Medical Management (First-line, especially if fertility desired or surgery declined)
| Drug | Mechanism | Notes |
|---|---|---|
| Tranexamic acid | Antifibrinolytic; reduces menstrual blood loss ~50% | Non-hormonal; taken during menses only; does not shrink fibroid |
| NSAIDs (mefenamic acid, ibuprofen) | ↓ Prostaglandin-mediated vasodilation; reduces blood loss ~25% | Also helps dysmenorrhea |
| Levonorgestrel IUS (Mirena) | Progestogen-induced endometrial atrophy | Effective for HMB; may be expelled if cavity distorted; not suitable for large submucosal fibroids |
| Combined oral contraceptive pill (COCP) | Reduces endometrial proliferation | Moderate effect; does not shrink fibroids |
| Progestogens (norethisterone, medroxyprogesterone) | Endometrial suppression | Short-term use; cyclical or continuous |
| GnRH agonists (leuprolide, goserelin, buserelin) | Hypoestrogen state → fibroid shrinkage 30–50%; amenorrhea | Maximum 3–6 months (bone loss); used as preoperative downsizing; add-back HRT if >3 months |
| GnRH antagonists (elagolix, relugolix, linzagolix) | Rapid onset hypoestrogen; oral; dose-titratable | Approved for fibroid-associated HMB; fewer flare effects than agonists |
| Selective Progesterone Receptor Modulators (SPRMs) — ulipristal acetate | Progesterone receptor modulation → fibroid shrinkage + amenorrhea | Restricted use due to rare but serious hepatotoxicity (liver monitoring required); previously widely used |
🔹 Surgical / Interventional Management
| Procedure | Indication | Notes |
|---|---|---|
| Hysteroscopic myomectomy | Submucosal fibroids (FIGO 0, 1, 2); HMB | Gold standard for submucosal; day case; preserves fertility; can be repeated |
| Endometrial ablation | HMB without significant submucosal fibroid; family complete | Destroys endometrium; not suitable if fertility desired; less effective with large/multiple fibroids |
| Laparoscopic/open myomectomy | Intramural or subserosal fibroids; fertility desired | Removes fibroid, preserves uterus; risk of recurrence |
| Hysterectomy | Definitive; family complete; failed other treatments | Curative; total vs. subtotal; abdominal/laparoscopic/vaginal route |
| UAE (Uterine Artery Embolisation) | Symptomatic fibroids; uterus preservation desired; fertility not primary concern | Occludes uterine arteries → fibroid ischaemia + shrinkage; effective for HMB; less invasive than surgery |
| MRI-guided Focused Ultrasound (MRgFUS/HIFU) | Focal fibroids; uterus preservation; avoid surgery | Non-invasive; thermal ablation; suitable for limited fibroid burden (Bailey & Love's, p. 1607) |
3. 🟡 Pelvic Pressure / Bulk Symptoms
Management:
| Option | Notes |
|---|---|
| GnRH agonist (preoperative) | Shrinks fibroid bulk; improves haematological status before surgery |
| Myomectomy (laparoscopic or open) | If fertility desired; removes offending fibroid(s) |
| Hysterectomy | Definitive; preferred if family complete and uterus no longer desired |
| UAE | Reduces fibroid volume 40–60%; improves bulk symptoms; useful if surgery high-risk |
| MRgFUS | Effective for localised large fibroids; limited by fibroid number/location |
- Surgical approach chosen based on fibroid size, number, location, and patient preference
- Abdominal myomectomy preferred for very large (>10 cm) or multiple (>3–4) fibroids
- Laparoscopic myomectomy for ≤3–4 fibroids of moderate size (<8–10 cm)
4. 🟠 Dysmenorrhoea / Pelvic Pain
| Situation | Management |
|---|---|
| Dysmenorrhoea | NSAIDs; hormonal suppression; myomectomy if medical fails |
| Acute red degeneration (pregnancy) | Conservative: analgesia (paracetamol, opioids), hydration, NSAIDs (caution in pregnancy); usually self-limiting |
| Pedunculated fibroid torsion | Surgical emergency; laparoscopic/open myomectomy |
| Fibroid with degeneration (non-pregnant) | Analgesia; plan elective myomectomy |
5. 🔵 Fibroids and Infertility / Recurrent Miscarriage
Classification-Based Approach:
| Fibroid Type | Impact on Fertility | Management |
|---|---|---|
| Submucosal (FIGO 0, 1, 2) | Significantly reduces implantation rates | Hysteroscopic myomectomy — improves fertility outcomes; recommended before IVF |
| Intramural (FIGO 3, 4) distorting cavity | Moderately reduces fertility | Myomectomy (laparoscopic or open) |
| Intramural not distorting cavity | Controversial; mild effect | Consider myomectomy if >4–5 cm or recurrent implantation failure |
| Subserosal (FIGO 5, 6, 7) | Minimal impact | Usually no intervention for fertility alone |
Key Points:
- Hysteroscopic myomectomy is gold standard for submucosal fibroids before IVF
- After abdominal/laparoscopic myomectomy, recommend 3–6 months delay before attempting conception (uterine scar healing)
- UAE and MRgFUS — generally not recommended when fertility is desired (reduced endometrial blood flow, risk of premature ovarian failure with UAE)
- GnRH agonists preoperatively reduce fibroid size but do not improve fertility outcomes independently
6. 🟣 Fibroids in Pregnancy
| Complication | Management |
|---|---|
| Red degeneration (most common complication in pregnancy) | Conservative: analgesia, hydration, rest; NSAIDs caution (avoid after 32 weeks) |
| Preterm labour | Tocolysis; standard obstetric management |
| Malpresentation | Obstetric management; consider mode of delivery |
| Placenta praevia (if fibroid near lower segment) | Obstetric management |
| Obstructed labour (cervical/lower segment fibroid) | Caesarean section |
| Caesarean myomectomy | Generally avoided (risk of haemorrhage); only for pedunculated or easily accessible fibroids by experienced surgeons |
| Postpartum haemorrhage | Uterotonic agents; surgical haemostasis |
7. ⚫ Fibroid with Anaemia
- Often secondary to chronic HMB
- Preoperative optimisation:
- Iron supplementation (oral or IV ferric carboxymaltose)
- GnRH agonist for 3 months → induces amenorrhea → allows haemoglobin recovery + fibroid shrinkage → safer surgery
- Blood transfusion if Hb critically low before urgent surgery
- Cell salvage during myomectomy/hysterectomy
📊 Management Summary by Indication
| Indication | First-line | Second-line | Definitive |
|---|---|---|---|
| Asymptomatic | Watchful waiting | — | — |
| HMB (fertility desired) | Tranexamic acid / LNG-IUS / COCP | GnRH agonist → hysteroscopic myomectomy | Myomectomy |
| HMB (family complete) | Medical (tranexamic, LNG-IUS) | UAE / endometrial ablation | Hysterectomy |
| Bulk symptoms | GnRH agonist | UAE / myomectomy | Hysterectomy |
| Infertility / submucosal | Hysteroscopic myomectomy | — | — |
| Infertility / intramural | Laparoscopic/open myomectomy | — | — |
| Dysmenorrhoea | NSAIDs + hormonal | Myomectomy | Hysterectomy |
| Pregnancy complications | Conservative / analgesia | Obstetric management | CS if obstructed |
| Anaemia preoperative | Iron + GnRH agonist | IV iron | Surgery after optimisation |
🔑 Decision-Making Principles
Symptomatic fibroid
│
├─ Fertility desired?
│ YES → Medical suppression → Myomectomy (route by fibroid type)
│ NO → Medical → UAE / Ablation → Hysterectomy
│
├─ Submucosal?
│ → Hysteroscopic myomectomy (regardless of fertility wish)
│
├─ Near menopause (<5 years)?
│ → Medical management / GnRH agonist to bridge to natural regression
│
└─ High surgical risk?
→ UAE / MRgFUS / GnRH agonist long-term
- Hysterectomy is the only truly curative option — recurrence impossible
- Myomectomy carries a ~30% recurrence rate at 10 years; higher with multiple fibroids
- UAE — ~20% require further intervention within 5 years
- MRgFUS — emerging; best for single, accessible, T2-hypointense fibroids (Bailey & Love's, p. 1607)
47 year old patient with acuþe urinary retention with retained volume of 1500ml and on uss fibroid mass 8cm×8cm in the posterior wall. What is the best management option
Clinical Analysis: 47-Year-Old with Acute Urinary Retention + 8×8cm Posterior Wall Fibroid
🔍 Understanding the Scenario
| Parameter | Detail |
|---|---|
| Age | 47 years (perimenopausal) |
| Presentation | Acute urinary retention (AUR) |
| Retained volume | 1500 mL (massive retention) |
| Fibroid | 8×8 cm, posterior wall (intramural/subserosal) |
Why is a Posterior Wall Fibroid Causing AUR?
- Uterine retroversion/retroflexion — posterior fibroid tips the uterus forward and downward → cervix displaced anteriorly → urethral kinking/compression at the bladder neck
- Pelvic mass effect — 8×8 cm fibroid occupies significant pelvic space → compresses bladder trigone and urethra indirectly
- Uterine incarceration — gravid/enlarged uterus becomes incarcerated in the pelvis → urethral obstruction (same mechanism as incarcerated retroverted uterus in pregnancy)
🚨 Step 1 — Immediate Emergency Management
Urethral Catheterisation (Immediate)
- Insert indwelling Foley catheter (14–16 Fr)
- Drain slowly — avoid rapid decompression of >500 mL at a time
- Risk of decompression haematuria and post-obstructive diuresis if drained too rapidly
- Clamp catheter every 30 minutes or use controlled drainage
- Monitor urine output hourly after catheterisation
- Send urine for MC&S (exclude concurrent UTI)
Monitor for Post-Obstructive Diuresis
- Common after prolonged high-pressure retention
- Monitor fluid balance, electrolytes (U&E), creatinine
- IV fluid replacement if diuresis >200 mL/hour
🔬 Step 2 — Investigations
| Investigation | Purpose |
|---|---|
| FBC | Anaemia from associated HMB; baseline pre-surgery |
| U&E, creatinine | Assess renal function (bilateral ureteric obstruction possible with large fibroid) |
| LFTs, coagulation | Pre-surgical baseline |
| Serum hCG | Exclude pregnancy (47 years — still possible) |
| FSH, LH, estradiol | Assess menopausal status (guides long-term management) |
| Pelvic MRI | Definitive fibroid characterisation — location, relationship to bladder/urethra, uterine vessels, bowel; exclude sarcoma features |
| Renal USS / IVU | Exclude bilateral hydronephrosis/ureteric compression from mass |
| Cervical smear | If not up to date |
| Endometrial biopsy | If HMB present; exclude endometrial pathology |
🎯 Step 3 — Definitive Management
The Best Management Option: Total Hysterectomy
| Factor | Supports Hysterectomy |
|---|---|
| Age 47 | Perimenopausal; reproductive function likely no longer desired |
| AUR caused by fibroid | Fibroid is causing significant obstructive complications — not just symptomatic |
| 8×8 cm single posterior fibroid | Large; causing structural displacement |
| Obstructive uropathy | Retained 1500 mL = significant complication requiring definitive treatment |
| Proximity to menopause | Even if treated conservatively, fibroid unlikely to regress for several more years |
| Myomectomy risk | For a single large posterior intramural fibroid, myomectomy carries significant haemorrhage risk and ~30% recurrence risk — not justified when fertility is not a concern |
Why Not Other Options?
| Option | Reason Not Preferred Here |
|---|---|
| Medical management (GnRH agonist) | Will shrink fibroid 30–50% but takes 3 months; will NOT relieve AUR immediately; not definitive |
| Myomectomy | Appropriate only if fertility desired; significant haemorrhage risk for 8 cm posterior fibroid; 30% recurrence risk; not justified at 47 |
| UAE | Reduces fibroid volume but slowly; not suitable as emergency; does not guarantee resolution of urethral obstruction; 20% require further intervention |
| MRgFUS | Elective procedure; too slow for this presentation; not suitable for acute complication |
| Watchful waiting | Completely inappropriate — patient presented with a complication (AUR) |
📋 Surgical Plan
Preoperative Optimisation (While Catheterised)
| Step | Detail |
|---|---|
| Catheter drainage | Leave catheter in situ until surgery |
| Correct anaemia | Oral/IV iron; transfuse if Hb <8 g/dL |
| GnRH agonist (optional) | 1–2 injections (4–8 weeks) preoperatively to reduce fibroid size and vascularity → reduces intraoperative blood loss; only if surgery can be delayed |
| Renal function | Ensure normalisation of creatinine post-decompression |
| Thromboprophylaxis planning | VTE risk high (pelvic surgery + large mass) |
| Bowel prep | If posterior fibroid adherent to bowel |
| Consent | Include risk of bladder/ureteric injury, haemorrhage, bowel injury |
Surgical Approach
| Decision | Reasoning |
|---|---|
| Abdominal route preferred | Fibroid 8×8 cm — too large for straightforward vaginal/laparoscopic approach; abdominal gives best access and control |
| Laparoscopic-assisted | Possible in skilled hands if uterine size manageable; converts to open if needed |
| BSO vs. ovarian conservation | At 47 years — discuss with patient: ovarian conservation avoids surgical menopause but increases adnexal disease risk; most guidelines recommend ovarian conservation before natural menopause unless other indications |
| Ureteric identification | Critical — posterior fibroid may displace ureters; consider intraoperative ureteric stents if MRI shows proximity |
| Posterior fibroid dissection | Care needed — fibroid may be adherent to posterior broad ligament, rectum, or uterosacral ligaments |
🗓️ Management Timeline
Day 0 (Presentation)
│
├─ Immediate: Urethral catheterisation → controlled bladder drainage
├─ Monitor post-obstructive diuresis
├─ Blood tests, MRI pelvis, renal function
│
Weeks 1–4
├─ Correct anaemia (iron ± transfusion)
├─ Consider GnRH agonist depot injection (if surgery to be delayed)
├─ Urology review if renal function impaired
├─ Counsel patient regarding hysterectomy
│
Weeks 4–8
└─ Total Abdominal Hysterectomy
└─ Intraoperative ureteric identification
└─ Ovarian conservation decision
└─ Remove catheter post-op once voiding confirmed
⚠️ Special Considerations
- Leiomyosarcoma — rare (~0.5%) but must be considered for rapidly growing large fibroid; MRI features (heterogeneous signal, irregular margins, central necrosis) raise suspicion; final diagnosis on histology post-hysterectomy
- Ureteric injury risk — posterior fibroid displaces ureters medially; always identify ureters before clamping uterine vessels
- Post-op voiding trial — remove catheter 24–48 hours post-op; perform voiding trial; ensure residual volume <150 mL before discharge
✅ Summary Answer
The best management for this 47-year-old with AUR (1500 mL) caused by an 8×8 cm posterior uterine fibroid is:
- Immediate: Urethral catheterisation for bladder decompression
- Short-term: Investigate, correct anaemia, MRI pelvis, optimise for surgery
- Definitive: Total Abdominal Hysterectomy — curative, appropriate for her age, eliminates the obstructing mass permanently, and avoids the limitations and recurrence risk of all conservative options
Indications for myotomy and historectomy in fibroids
myomectomy indications fibroids fertility uterine preservation criteria
hysterectomy indications uterine fibroids leiomyoma definitive surgical treatment
Indications for Myomectomy vs Hysterectomy in Fibroids
🔑 Core Principle
The single most important differentiating factor is whether the patient desires future fertility.
- Fertility desired → Myomectomy
- Family complete / uterus not desired → Hysterectomy
✂️ MYOMECTOMY
Indications
1. 🤰 Fertility Preservation (Primary Indication)
- Woman desires future pregnancy
- Fibroids causing infertility — particularly:
- Submucosal fibroids distorting uterine cavity (FIGO 0, 1, 2)
- Large intramural fibroids (>4–5 cm) distorting cavity
- Fibroids associated with recurrent implantation failure in IVF
- Fibroids associated with recurrent miscarriage
- Hysteroscopic myomectomy before IVF significantly improves implantation rates
2. 🩸 Heavy Menstrual Bleeding — Uterus to be Preserved
- Significant HMB unresponsive to medical management
- Patient declines hysterectomy or is not yet ready
- Younger patient who wishes to retain uterus even if fertility not immediately desired
- Submucosal fibroid causing HMB → hysteroscopic myomectomy
3. 💊 Failed or Unsuitable Medical Management
- Symptoms persist despite adequate medical therapy (tranexamic acid, LNG-IUS, GnRH agonists)
- Patient intolerant of medical treatment
- Medical therapy contraindicated
4. 📏 Symptomatic Fibroids with Uterine Preservation Desired
- Pelvic pressure/bulk symptoms from large fibroid(s)
- Urinary symptoms (frequency, retention) caused by fibroid
- Pelvic pain / dysmenorrhoea unresponsive to analgesia
- Patient requests uterus preservation on personal, cultural, or religious grounds
5. 🚑 Pedunculated Fibroid Torsion
- Acute surgical emergency
- Pedunculated subserosal fibroid with torsion → laparoscopic/open myomectomy
6. 📐 Specific Fibroid Characteristics Suitable for Myomectomy
- Solitary or limited number of fibroids (ideally ≤3–4)
- Well-defined, accessible location
- Size amenable to chosen surgical route
7. 🔲 Pre-IVF / Pre-Conception Optimisation
- Submucosal or cavity-distorting fibroids identified during infertility workup
- Hysteroscopic myomectomy recommended before embryo transfer
Routes of Myomectomy
| Route | Indication |
|---|---|
| Hysteroscopic | FIGO 0, 1, 2 submucosal fibroids; HMB; infertility with cavity distortion; day case |
| Laparoscopic | Subserosal / intramural fibroids; ≤3–4 fibroids; size ≤8–10 cm; skilled surgeon |
| Open (Abdominal) | Large fibroids (>10 cm); multiple fibroids (>4); deep intramural; failed laparoscopic |
| Robotic-assisted | Centres with robotic capability; complex cases |
Contraindications to Myomectomy
- Suspected malignancy (leiomyosarcoma) — hysterectomy preferred
- Very large uterus (>20 weeks size) — hysterectomy often safer
- Desire for permanent solution — hysterectomy more appropriate
- Coexisting uterine/endometrial pathology requiring hysterectomy
Post-Myomectomy Considerations
- Delay conception 3–6 months after open/laparoscopic myomectomy (scar healing)
- Caesarean section may be recommended if deep myometrial incision made (risk of uterine rupture in labour)
- Recurrence rate: ~30% at 10 years; higher with multiple fibroids, younger age, Black ethnicity
🏥 HYSTERECTOMY
Indications
1. ✅ Family Complete / No Desire for Future Fertility (Primary Indication)
- Symptomatic fibroids in a woman who has completed her family
- No desire for uterine preservation
- Most common indication globally for hysterectomy
2. 🩸 Severe / Refractory Heavy Menstrual Bleeding
- HMB causing significant iron-deficiency anaemia unresponsive to:
- Medical therapy
- Conservative surgical procedures (hysteroscopic myomectomy, endometrial ablation)
- Quality of life severely impaired
- Recurrent HMB after previous myomectomy
3. 📏 Severe Bulk / Pressure Symptoms
- Large uterus (equivalent to ≥14–16 weeks size) causing:
- Urinary retention or obstructive uropathy (as in the previous case)
- Bilateral hydronephrosis / ureteric compression
- Severe urinary frequency/urgency unresponsive to other treatment
- Significant bowel compression → constipation/tenesmus
- Venous obstruction → lower limb oedema, varicosities
4. 🔁 Recurrence After Myomectomy
- Symptomatic fibroid recurrence after previous myomectomy
- Multiple recurrences
- Patient no longer desires further uterus-preserving attempts
5. ❌ Failed Conservative / Interventional Treatments
- Failure of:
- Medical management
- UAE (uterine artery embolisation)
- MRgFUS
- Endometrial ablation
- Previous myomectomy
- Persistent or worsening symptoms
6. 🔴 Suspected or Confirmed Leiomyosarcoma
- Rapid fibroid growth (especially post-menopausal)
- MRI features suspicious for sarcoma (heterogeneous signal, irregular borders, central necrosis)
- Elevated LDH
- Total hysterectomy mandatory — morcellation contraindicated
7. 🧩 Coexisting Uterine Pathology
- Fibroids + endometrial hyperplasia/carcinoma
- Fibroids + adenomyosis causing refractory dysmenorrhoea/HMB
- Fibroids + cervical dysplasia/carcinoma
- Fibroids + endometriosis requiring definitive treatment
- Fibroids + uterine prolapse (vaginal hysterectomy preferred)
8. ⚕️ High Surgical Risk Favouring Single Definitive Procedure
- Multiple comorbidities making repeat surgeries hazardous
- Patient preference for one definitive operation
- Obesity, previous multiple pelvic surgeries, anticipated difficult adhesions
9. 🔢 Multiple / Diffuse Fibroids
- Too numerous for myomectomy to be practical
- Diffuse leiomyomatosis (entire myometrium replaced by fibroids)
- Would leave an unacceptably thin or structurally compromised uterus after removal
10. 👵 Perimenopausal / Postmenopausal Women
- Symptomatic fibroids in women approaching or past menopause
- Avoids multiple procedures; fibroids will not regress fast enough to relieve current symptoms
- Postmenopausal fibroid growth → always suspect sarcoma → hysterectomy
Types of Hysterectomy in Fibroid Management
| Type | Definition | When Used |
|---|---|---|
| Total Hysterectomy (TH) | Remove uterus + cervix | Standard; eliminates cervical stump complications |
| Subtotal / Supracervical Hysterectomy | Remove uterus, preserve cervix | Technically easier; faster; less bladder risk; cervical screening still needed |
| Hysterectomy + BSO | Remove uterus, cervix, both tubes and ovaries | If adnexal pathology; postmenopausal; high ovarian cancer risk (BRCA) |
| Hysterectomy + ovarian conservation | Preserve ovaries | Premenopausal — avoid surgical menopause; ovaries left if healthy |
Routes of Hysterectomy
| Route | Indication |
|---|---|
| Vaginal | Small uterus; uterine prolapse; no previous surgery |
| Laparoscopic / LAVH | Moderate uterine size; skilled surgeon; less morbidity |
| Robotic-assisted | Complex cases; centres with robotic facility |
| Open (Abdominal) | Large uterus (>14–16 weeks); multiple fibroids; dense adhesions; suspected malignancy; previous multiple surgeries |
📊 Side-by-Side Comparison
| Feature | Myomectomy | Hysterectomy |
|---|---|---|
| Fertility | Preserved ✅ | Lost ❌ |
| Cure | Not curative (30% recurrence) | Curative ✅ |
| Uterus | Preserved | Removed |
| Menstruation | Continues | Ceases |
| Blood loss (intraop) | Potentially high (fibroid bed) | Controlled |
| Hospital stay | Shorter (hysteroscopic) | Slightly longer |
| Recovery | Variable by route | 4–6 weeks (open) |
| Future pregnancies | Possible | Impossible |
| Repeat procedure | Possible (recurrence) | Not needed |
| Scar/adhesion risk | Yes (future fertility risk) | Minimal concern |
| Best age group | Reproductive age (<40) | Perimenopausal / family complete |
🧭 Decision Algorithm
Symptomatic Fibroid — Surgical Management Needed
│
├─ Fertility desired?
│ │
│ YES → MYOMECTOMY
│ ├─ Submucosal → Hysteroscopic
│ ├─ Intramural/subserosal → Laparoscopic / Open
│ └─ Counsel re: recurrence, CS in future pregnancy
│
└─ NO (Family complete / uterus not desired)
│
├─ Coexisting pathology? → Hysterectomy + address pathology
├─ Failed previous myomectomy? → Hysterectomy
├─ Suspected sarcoma? → Hysterectomy (open, no morcellation)
├─ Multiple/diffuse fibroids? → Hysterectomy
├─ Large uterus with obstructive complications? → Hysterectomy
└─ HYSTERECTOMY
├─ Route: Abdominal / Laparoscopic / Vaginal
└─ BSO vs. ovarian conservation based on age/risk
⚠️ Key Points to Remember
- Myomectomy ≠ cure — recurrence rate 30% at 10 years; counsel patients accordingly
- Hysterectomy = only cure — but irreversible; must confirm no future fertility desired
- Suspected sarcoma → open hysterectomy; never morcellate (risk of disseminating malignancy)
- GnRH agonist preoperatively (3 months) for both procedures — reduces fibroid size, vascularity, intraoperative blood loss, and corrects anaemia
- Post-myomectomy mode of delivery depends on depth of myometrial incision — deep entry into cavity → elective CS recommended
Heavy menstrual bleeding and severe migraine with normal TVUSS findings
heavy menstrual bleeding treatment management normal uterus non-hormonal hormonal options
migraine with aura combined oral contraceptive contraindication progestogen only management
UKMEC WHO MEC migraine contraception stroke risk estrogen progestogen
HMB + Severe Migraine with Normal TVUSS — Management
🔍 Clinical Problem Analysis
| Feature | Implication |
|---|---|
| Heavy Menstrual Bleeding | Needs effective treatment to reduce blood loss |
| Severe Migraine | Critically limits hormonal options — especially estrogen-containing preparations |
| Normal TVUSS | No structural pathology (no fibroid, polyp, adenomyosis) → dysfunctional uterine bleeding (DUB) / AUB-O or AUB-E (FIGO PALM-COEIN classification) |
⚠️ The Critical Issue: Migraine + Estrogen = Contraindicated
WHO / UKMEC Classification for Migraine
| Migraine Type | Combined Hormonal Contraceptives (CHC) / Estrogen | Progestogen-Only Methods |
|---|---|---|
| Migraine WITHOUT aura | UKMEC 3 (relative contraindication; risks outweigh benefits) | UKMEC 2 (generally usable) |
| Migraine WITH aura | UKMEC 4 (ABSOLUTE contraindication) | UKMEC 2 (generally usable) |
Why Estrogen is Dangerous in Migraine
- Estrogen promotes a prothrombotic state — increases coagulation factors, decreases antithrombin III
- Migraine with aura is an independent risk factor for ischaemic stroke (RR ~2–4×)
- Combining estrogen + migraine with aura → multiplicative stroke risk (~8× above baseline)
- Estrogen fluctuations also trigger migraines (menstrual migraine — estrogen withdrawal)
- Therefore:
- COCP, patch, vaginal ring, combined injectable → all contraindicated
- Even HRT with estrogen should be used with extreme caution
✅ Treatment Options — Safe and Effective
- Non-hormonal options — safe regardless of migraine
- Progestogen-only options — UKMEC 2 (benefits outweigh risks)
🥇 FIRST LINE — Levonorgestrel Intrauterine System (LNG-IUS / Mirena)
| Feature | Detail |
|---|---|
| Effect on HMB | Reduces menstrual blood loss by 90–95%; most effective medical treatment for HMB |
| Migraine safety | Progestogen-only — UKMEC 2; safe to use; minimal systemic absorption |
| Mechanism | Local endometrial atrophy → markedly reduced shedding |
| Additional benefits | Amenorrhoea in ~50% at 1 year; contraception; lasts 5–8 years |
| Normal TVUSS | Ideal candidate — no cavity distortion; easy insertion |
| Migraine impact | May actually improve menstrual migraine by eliminating estrogen withdrawal trigger |
| NICE guidance | First-line recommended for HMB in women with normal uterus |
LNG-IUS (Mirena) is the single most appropriate treatment — addresses HMB effectively, is safe in migraine, and avoids estrogen entirely.
🥈 SECOND LINE — Non-Hormonal Medical Management
A. Tranexamic Acid
| Feature | Detail |
|---|---|
| Mechanism | Antifibrinolytic — inhibits plasminogen activation → prevents clot breakdown |
| Efficacy | Reduces blood loss ~50% |
| Dosing | 1g TDS–QDS during menstruation (max 4–5 days) |
| Migraine safety | ✅ Completely safe — non-hormonal |
| Limitation | Taken only during menses; does not provide contraception |
B. NSAIDs (Mefenamic Acid / Ibuprofen)
| Feature | Detail |
|---|---|
| Mechanism | ↓ Prostaglandin synthesis → reduced uterine vasodilation + vasoconstriction |
| Efficacy | Reduces blood loss ~25–30% |
| Migraine safety | ✅ Safe — also useful for menstrual migraine (dual benefit) |
| Dosing | Mefenamic acid 500 mg TDS during menstruation |
| Bonus | Reduces dysmenorrhoea simultaneously |
C. Tranexamic Acid + NSAIDs Combined
- Complementary mechanisms
- Can be used together for additive effect on HMB
- Both safe in migraine
🥉 THIRD LINE — Progestogen-Only Hormonal Options
A. Oral Norethisterone (High Dose)
| Feature | Detail |
|---|---|
| Dose for HMB | 5 mg TDS (days 5–26 of cycle) |
| Efficacy | Moderate — reduces HMB ~80% |
| Migraine safety | ✅ UKMEC 2 |
| Limitation | Bloating, mood changes; not as effective as LNG-IUS; must remember daily dosing |
B. Depo-Provera (DMPA — Depot Medroxyprogesterone Acetate)
| Feature | Detail |
|---|---|
| Effect on HMB | Induces amenorrhoea in ~50% after 1 year |
| Migraine safety | ✅ UKMEC 2 |
| Limitation | Irregular bleeding initially; irreversible for 3 months; bone density loss with long-term use |
C. Progestogen-Only Pill (POP — Desogestrel 75 mcg)
| Feature | Detail |
|---|---|
| Effect on HMB | Modest reduction; causes amenorrhoea in ~50% |
| Migraine safety | ✅ UKMEC 2 |
| Limitation | Less effective for HMB than LNG-IUS; daily pill |
D. Etonogestrel Implant (Nexplanon)
| Feature | Detail |
|---|---|
| Effect on HMB | Variable — may cause irregular bleeding; amenorrhoea in some |
| Migraine safety | ✅ UKMEC 2 |
| Limitation | Unpredictable bleeding pattern — not ideal for HMB specifically |
🔷 GnRH Agonists (Adjunct / Bridge)
| Feature | Detail |
|---|---|
| Mechanism | Pituitary downregulation → hypoestrogen → amenorrhoea |
| Efficacy for HMB | Complete amenorrhoea; rapidly effective |
| Migraine | ⚠️ Causes hypoestrogenism — may worsen migraine initially (estrogen withdrawal); may improve menstrual migraine long-term |
| Add-back HRT | Low-dose estrogen add-back complicates migraine safety |
| Duration | Max 6 months (bone loss); used as bridge to surgery |
| Role here | Short-term only; not for long-term management |
🔴 What to AVOID
| Treatment | Reason to Avoid |
|---|---|
| Combined Oral Contraceptive Pill (COCP) | Contains estrogen → UKMEC 4 if migraine with aura; UKMEC 3 if without aura → contraindicated |
| Combined patch / ring / injection | Same estrogen-related stroke risk |
| Cyclical low-dose norethisterone (day 19–26) | Inadequate dose for HMB; not recommended by NICE for HMB treatment |
| HRT with estrogen | Caution; avoid if migraine with aura |
📋 Management Algorithm for This Patient
HMB + Severe Migraine + Normal TVUSS
│
├─ Is migraine WITH AURA?
│ YES → Estrogen ABSOLUTELY contraindicated (UKMEC 4)
│ NO → Estrogen relatively contraindicated (UKMEC 3)
│
├─ FIRST LINE:
│ LNG-IUS (Mirena) ← BEST OPTION
│ Progestogen-only; 90-95% ↓ blood loss; safe in migraine
│
├─ If LNG-IUS declined / awaiting insertion:
│ Tranexamic acid (during menses) +/- Mefenamic acid
│ → Non-hormonal; dual benefit (HMB + menstrual migraine)
│
├─ If hormonal supplementation needed:
│ Progestogen-only options (norethisterone, DMPA)
│ Avoid all estrogen-containing preparations
│
├─ Persistent / failed medical management:
│ Endometrial ablation (if family complete)
│ Hysterectomy (definitive; last resort)
│
└─ Address migraine separately:
Triptans for acute attacks
Propranolol / topiramate / amitriptyline for prophylaxis
(Topiramate caution if pregnancy possible)
🔗 Additional Considerations
Menstrual Migraine
- Many women with severe migraine have menstrual migraine — attacks triggered by estrogen withdrawal at menstruation
- LNG-IUS can help by eliminating menstruation → removes estrogen-withdrawal trigger → may reduce migraine frequency
- Continuous progestogen (DMPA, implant) achieving amenorrhoea → eliminates hormonal fluctuation trigger
Iron Deficiency Anaemia
- Check FBC, ferritin, iron studies
- If anaemic → oral iron supplementation alongside HMB treatment
- LNG-IUS will correct anaemia over time as bleeding reduces
Investigate Migraine Properly
- Confirm whether migraine is with or without aura — changes UKMEC category and urgency of avoiding estrogen
- Neurology referral if poorly controlled
- Ensure adequate migraine prophylaxis in place
✅ Summary
| Priority | Treatment | Rationale |
|---|---|---|
| 1st choice | LNG-IUS (Mirena) | Most effective for HMB; progestogen-only; safe in migraine; may reduce menstrual migraine |
| 2nd choice | Tranexamic acid + Mefenamic acid | Non-hormonal; safe; effective for moderate HMB; mefenamic acid also helps migraine |
| 3rd choice | Oral norethisterone / DMPA | Progestogen-only; moderate HMB control; safe in migraine |
| Avoid | All estrogen-containing preparations | Stroke risk — especially if migraine with aura |
| Surgical (refractory) | Endometrial ablation / Hysterectomy | If all medical options fail; family complete |
Postmenopausal bleeding investigation steps
postmenopausal bleeding investigation steps endometrial cancer workup transvaginal ultrasound biopsy
postmenopausal bleeding causes differential diagnosis endometrial thickness pipelle hysteroscopy
Postmenopausal Bleeding (PMB) — Step-by-Step Investigation
📌 Definition
⚠️ Key Principle
PMB = Endometrial cancer until proven otherwise. ~10% of women with PMB have endometrial cancer. The majority (~90%) have benign causes — but malignancy must be excluded first in every case.
🔢 Causes of PMB (Differential Diagnosis)
| Cause | Frequency |
|---|---|
| Atrophic vaginitis / endometrial atrophy | Most common (~60–80%) |
| Endometrial polyp | ~10% |
| Endometrial hyperplasia | ~5–10% |
| Endometrial carcinoma | ~10% |
| Cervical pathology (polyp, carcinoma) | ~5% |
| HRT-related bleeding | Common if on HRT |
| Ovarian tumours (estrogen-secreting) | Rare |
| Vulval / vaginal carcinoma | Rare |
| Coagulation disorders | Rare |
| Urethral caruncle | Rare |
| Rectal bleeding misidentified | Rare |
🔢 Step-by-Step Investigation
✅ STEP 1 — History
- Duration, amount, frequency, colour (fresh red vs. brown/dark)
- Single episode vs. recurrent
- Associated symptoms: discharge (offensive → pyometra), pelvic pain, weight loss, bloating
| Risk Factor | Notes |
|---|---|
| Obesity (BMI >30) | ↑ peripheral estrogen aromatisation |
| Nulliparity | ↑ unopposed estrogen exposure |
| Late menopause (>55 years) | Prolonged estrogen exposure |
| Diabetes mellitus | Associated with endometrial cancer |
| Hypertension | Associated |
| Tamoxifen use | Endometrial proliferant; ↑ polyp + cancer risk |
| Unopposed estrogen HRT | Without progestogen |
| PCOS | Chronic anovulation → unopposed estrogen |
| Lynch syndrome (HNPCC) | 40–60% lifetime endometrial cancer risk |
| Family history | Endometrial/colorectal cancer |
| Breast cancer history | Tamoxifen; shared risk factors |
- Combined OCP use
- Multiparity
- Progestogen-containing HRT
- HRT type (combined vs. unopposed estrogen)
- Tamoxifen
- Anticoagulants
- Antipsychotics (↑ prolactin)
✅ STEP 2 — Physical Examination
| Examination | Purpose |
|---|---|
| General | BMI, pallor (anaemia), lymphadenopathy |
| Abdominal examination | Uterine/adnexal mass, ascites, hepatomegaly |
| Speculum examination | Inspect vulva, vagina, cervix: atrophy, ulceration, polyps, cervical mass, discharge, source of bleeding |
| Bimanual examination | Uterine size, mobility, tenderness; adnexal masses; parametrial involvement |
| Rectal examination | If rectal bleeding confused; posterior uterine extension |
- Atrophic vaginitis: pale, thin, dry vaginal walls, loss of rugae, petechiae — reassuring
- Cervical polyp/mass: visible on speculum — may explain bleeding
- Uterine enlargement + tenderness: suggests pyometra or carcinoma
✅ STEP 3 — Transvaginal Ultrasound (TVUSS)
Endometrial Thickness (ET) Measurement:
| ET on TVUSS | Interpretation | Action |
|---|---|---|
| ≤4 mm | Low risk of endometrial cancer (~1%) | Reassure; no biopsy needed if single episode of PMB and no other risk factors |
| >4 mm | Abnormal; requires further investigation | Endometrial biopsy mandatory |
| >10 mm | High suspicion of pathology | Biopsy + hysteroscopy |
| Cannot visualise | Technically inadequate | Proceed to hysteroscopy |
Additional TVUSS Assessment:
- Endometrial appearance: homogeneous vs. heterogeneous, polypoidal lesions, echogenic foci (calcification), fluid in cavity (haematometra/pyometra)
- Myometrium: invasion depth if mass visible; adenomyosis
- Ovaries: size, morphology, masses (estrogen-secreting tumour)
- Adnexa: free fluid, masses
- Cervical canal: stenosis, mass
TVUSS Findings and Their Implications:
| Finding | Implication |
|---|---|
| Thin ET ≤4 mm, homogeneous | Atrophy — benign; low cancer risk |
| ET >4 mm, uniform | Hyperplasia vs. carcinoma — biopsy |
| Polypoidal intracavitary lesion | Polyp vs. carcinoma — hysteroscopy + biopsy |
| Heterogeneous endometrium | High suspicion carcinoma |
| Fluid in cavity (hydrometra) | Cervical stenosis; pyometra; carcinoma obstructing outflow |
| Adnexal mass | Estrogen-secreting ovarian tumour |
✅ STEP 4 — Endometrial Biopsy (Histological Diagnosis)
- ET >4 mm on TVUSS
- Persistent PMB even if ET ≤4 mm (recurrent bleeding always warrants biopsy)
- Abnormal endometrial appearance on TVUSS regardless of thickness
- Unable to measure ET adequately on TVUSS
- High-risk patient (Lynch syndrome, tamoxifen, obesity)
Methods:
| Method | Detail |
|---|---|
| Pipelle endometrial biopsy | Gold standard outpatient biopsy; thin suction curette; performed in clinic without anaesthesia; sensitivity ~90% for endometrial cancer; may miss focal lesions |
| Hysteroscopy + directed biopsy | Gold standard overall; visualises entire cavity; directed biopsy of suspicious areas; detects polyps, focal lesions missed by blind biopsy |
| Dilatation & Curettage (D&C) | Traditional; now largely replaced by outpatient pipelle + hysteroscopy; still used if office biopsy fails or inconclusive |
| Hysteroscopy + LLETZ/resection | If polyp identified — polypectomy at same time |
When Pipelle is Insufficient → Proceed to Hysteroscopy:
- Inadequate sample on pipelle
- Cervical stenosis preventing pipelle entry
- Focal lesion on TVUSS (pipelle may miss)
- Persistent PMB despite negative pipelle
- Patient anxiety / office procedure not tolerated
✅ STEP 5 — Hysteroscopy
| Setting | Notes |
|---|---|
| Outpatient hysteroscopy | Preferred; "see and treat"; no anaesthesia required; vaginoscopic technique |
| Inpatient / GA hysteroscopy | If outpatient not tolerated; cervical stenosis; significant pathology requiring resection |
| Finding | Significance |
|---|---|
| Normal atrophic endometrium | Reassuring |
| Endometrial polyp | Polypectomy + histology |
| Irregular, friable, necrotic tissue | Highly suspicious carcinoma → directed biopsy |
| Submucosal fibroid | Myomectomy if symptomatic |
| Haematometra / pyometra | Drain + send fluid for cytology/culture |
| Uterine synechiae | Asherman's syndrome (rare in postmenopausal) |
✅ STEP 6 — Laboratory Investigations
| Test | Purpose |
|---|---|
| FBC | Anaemia from chronic bleeding |
| Coagulation screen (PT, APTT) | Coagulopathy as rare cause |
| U&E, LFTs | Pre-operative baseline; renal/hepatic comorbidities |
| Ca-125 | If adnexal mass / concern for ovarian pathology or advanced endometrial cancer |
| HbA1c / fasting glucose | Risk factor assessment (diabetes) |
| TFTs | Hypothyroidism (rare indirect cause) |
| Cervical smear | If not up to date; exclude cervical pathology |
✅ STEP 7 — Further Imaging (If Endometrial Cancer Confirmed)
| Investigation | Purpose |
|---|---|
| MRI pelvis | Gold standard for local staging — myometrial invasion depth, cervical extension, parametrial involvement |
| CT chest/abdomen/pelvis | Lymph node involvement, distant metastases |
| PET-CT | Selected cases; high-grade tumours; suspected distant spread |
| MRI abdomen | Para-aortic lymph node assessment |
| Chest X-ray | Pulmonary metastases (if CT not available) |
| Cystoscopy / sigmoidoscopy | If bladder/bowel involvement suspected (advanced disease) |
🗺️ Investigation Algorithm
Postmenopausal Bleeding
│
├─ History + Examination
│ ├─ Speculum: cervical polyp/mass → refer colposcopy/gynaecology
│ └─ Atrophic vaginitis → note but still investigate
│
├─ TVUSS (First-line)
│ │
│ ├─ ET ≤4 mm + normal appearance
│ │ └─ Single episode → Reassure + watchful waiting
│ │ Recurrent PMB → Pipelle biopsy anyway
│ │
│ ├─ ET >4 mm OR abnormal appearance
│ │ └─ ENDOMETRIAL BIOPSY (Pipelle)
│ │ ├─ Adequate sample → Histology report
│ │ └─ Inadequate / focal lesion → HYSTEROSCOPY
│ │
│ └─ Cannot visualise / technically inadequate
│ └─ Proceed directly to HYSTEROSCOPY
│
├─ Hysteroscopy + Directed Biopsy
│ ├─ Polyp → Polypectomy + histology
│ ├─ Suspicious endometrium → Directed biopsy
│ └─ Normal → Reassure
│
└─ Histology Result
├─ Benign (atrophy, polyp, simple hyperplasia) → Treat accordingly
├─ Hyperplasia without atypia → Progestogen / LNG-IUS + surveillance
├─ Atypical hyperplasia → Hysterectomy (high malignant potential)
└─ Endometrial carcinoma → Staging (MRI + CT) → MDT → Surgery
📊 Histological Outcomes and Management
| Histology | Management |
|---|---|
| Atrophy | Topical oestrogen for atrophic vaginitis; reassure |
| Endometrial polyp | Hysteroscopic polypectomy; annual surveillance |
| Simple hyperplasia without atypia | LNG-IUS or oral progestogen; 6-monthly biopsy surveillance |
| Complex hyperplasia without atypia | LNG-IUS / progestogen; close surveillance; consider hysterectomy |
| Atypical hyperplasia (EIN) | Hysterectomy — 40–50% progression to carcinoma if untreated |
| Endometrial carcinoma | Surgical staging → Total hysterectomy + BSO + sentinel lymph node biopsy / lymphadenectomy ± adjuvant therapy |
🔑 Key Points
- PMB = endometrial cancer until excluded — never attribute to benign cause without investigation
- TVUSS ET ≤4 mm + single episode of PMB + no risk factors = reassure, but recurrent PMB always requires biopsy regardless of ET
- Pipelle biopsy is the workhorse outpatient test — but hysteroscopy is needed for focal lesions and failed/inadequate pipelle
- Atypical hyperplasia (EIN) → hysterectomy without delay (40–50% harbour concurrent carcinoma)
- Lynch syndrome patients → lifelong annual surveillance even without symptoms
Cyclical progestrone or medroxy progesterone better to treat HMB on 16 year old girl
heavy menstrual bleeding adolescent treatment progestogen norethisterone medroxyprogesterone management
HMB teenager anovulatory bleeding hormonal treatment options COCP tranexamic acid
Cyclical Progesterone vs. MPA for HMB in a 16-Year-Old
🔍 Understanding the Clinical Context First
| Feature | Detail |
|---|---|
| Age | 16 years — likely within 2–3 years of menarche |
| Most likely cause | Anovulatory DUB — hypothalamic-pituitary-ovarian (HPO) axis immaturity → no corpus luteum → no endogenous progesterone → unopposed estrogen → endometrial proliferation → heavy, irregular shedding |
| Structural pathology | Unlikely at this age but must exclude |
| Coagulation disorder | Must be excluded — Von Willebrand disease (vWD) presents in up to 20% of adolescents with HMB; also platelet disorders, factor deficiencies |
Mandatory Initial Workup Before Starting Treatment
| Investigation | Purpose |
|---|---|
| FBC + ferritin | Anaemia assessment; iron deficiency |
| Coagulation screen (PT, APTT, bleeding time) | Screen for coagulopathy |
| vWF antigen + activity, Factor VIII | Exclude vWD (highest yield in adolescent HMB) |
| TFTs | Hypothyroidism → HMB |
| Prolactin | Hyperprolactinemia → anovulation → HMB |
| TVUSS / transabdominal USS | Exclude structural pathology (polyp, fibroid — rare at 16) |
| Pregnancy test (β-hCG) | Always in any adolescent with abnormal bleeding |
| LH, FSH, testosterone | If PCOS suspected |
⚖️ The Direct Answer: Cyclical Progesterone vs. MPA
Both are progestogens — but they differ significantly in:
| Feature | Cyclical Norethisterone (Synthetic 19-nortestosterone) | Medroxyprogesterone Acetate (MPA) (Synthetic 17-hydroxyprogesterone) |
|---|---|---|
| Structural class | 19-nortestosterone derivative | 17α-hydroxyprogesterone derivative |
| Androgenic activity | Mild androgenic effect | Minimal androgenic effect |
| Dose for HMB | 5 mg TDS (days 5–26 of cycle) | 10–20 mg/day (days 5–26 or 16–26) |
| Efficacy for HMB | ~80% reduction in blood loss | Moderate reduction |
| Cycle regularity | Regulates cycle | Regulates cycle |
| Effect on bone density | Neutral | ↓ Bone density with DMPA injectable form (not oral MPA) |
| Side effects | Bloating, mood changes, acne, weight gain, headache | Bloating, mood changes, irregular spotting, depression |
| Metabolic effects | Mild adverse lipid profile | Moderate adverse lipid profile |
| Adolescent bone health | Safe | Oral MPA safe; DMPA injectable — concern |
🎯 Which is Better for a 16-Year-Old?
Neither is the ideal first-line treatment in a 16-year-old with anovulatory HMB.
❌ Why Cyclical Progestogen (Norethisterone or MPA) is NOT the Best Choice Here
-
Mechanism mismatch — Cyclical progestogen works by opposing estrogen-driven endometrial proliferation. In anovulatory DUB, the problem IS lack of progesterone. Cyclical progestogen helps but:
- Requires precise cycle-day timing (difficult in irregular cycles)
- Does not address the underlying HPO axis immaturity
- High dose norethisterone has androgen side effects problematic in a teenager (acne, weight gain)
-
DMPA (Depo-Provera injectable MPA) — not recommended as first-line in adolescents:
- Causes significant bone mineral density (BMD) loss — critical during peak bone mass acquisition (ages 12–18)
- WHO, ACOG, and FSRH advise caution with DMPA in adolescents under 18
- If used, duration should be limited and calcium/vitamin D supplementation given
-
Oral MPA cyclically — acceptable but evidence for norethisterone in HMB is stronger; norethisterone 5 mg TDS (days 5–26) is better studied for HMB reduction
✅ Preferred Evidence-Based Options for HMB in a 16-Year-Old
🥇 FIRST LINE: Combined Oral Contraceptive Pill (COCP)
| Feature | Detail |
|---|---|
| Efficacy | Reduces menstrual blood loss ~50%; regularises cycles |
| Mechanism | Suppresses ovulation + endometrial proliferation; withdrawal bleed is predictable and lighter |
| Adolescent suitability | ✅ Safe; widely used; improves cycle predictability |
| Bone health | Neutral to protective (estrogen component maintains BMD) |
| Additional benefits | Dysmenorrhoea ↓; acne ↓; cycle regulation; contraception if needed |
| Duration | Can be used long-term |
| Contraindications | Migraine with aura, VTE risk, thrombophilia — check first |
For anovulatory DUB in adolescents, COCP is the most appropriate hormonal option — addresses both the HMB and the underlying anovulatory cycle dysregulation.
🥇 FIRST LINE (Non-hormonal): Tranexamic Acid
| Feature | Detail |
|---|---|
| Mechanism | Antifibrinolytic → reduces blood loss ~50% |
| Use | Taken only during menses |
| Adolescent suitability | ✅ Safest option; no hormonal side effects; first-line in vWD-associated HMB |
| Especially indicated | If coagulation disorder confirmed (vWD) |
| Dose | 1g (15–25 mg/kg) TDS–QDS during menses, max 5 days |
🥇 FIRST LINE (Non-hormonal): NSAIDs (Mefenamic acid / Ibuprofen)
| Feature | Detail |
|---|---|
| Mechanism | ↓ Prostaglandins → ↓ uterine vasodilation + vasoconstriction |
| Reduction in blood loss | ~25–30% |
| Additional benefit | Dysmenorrhoea relief |
| Use | During menses only; combined with tranexamic acid for additive effect |
🥈 SECOND LINE: Cyclical Norethisterone (if COCP contraindicated/declined)
- Norethisterone 5 mg TDS, days 5–26 of cycle
- Better evidence for HMB reduction than MPA
- Regulates cycle
- Monitor for androgenic side effects in teenager
- Not ideal long-term due to side effect profile
🥈 SECOND LINE: Oral MPA cyclically (if norethisterone not tolerated)
- MPA 10–20 mg/day, days 16–26 (luteal phase supplementation)
- Better tolerated than norethisterone for some patients
- Less androgenic
- Less data specifically for HMB than norethisterone
⚠️ LNG-IUS (Mirena) — Consider in Selected Adolescents
| Feature | Detail |
|---|---|
| Efficacy | Most effective (90–95% reduction) |
| Adolescent use | Increasing use; technically feasible; requires counselling |
| Advantage | Avoid systemic side effects; long-acting (5–8 years) |
| Barrier | Insertion discomfort; patient acceptability; cultural/social factors |
| Consider if | COCP contraindicated; severe refractory HMB; excellent long-term option |
📋 Direct Comparison Table: Cyclical Norethisterone vs. Oral MPA in a 16-Year-Old
| Criterion | Cyclical Norethisterone | Oral MPA |
|---|---|---|
| Efficacy for HMB | ✅ Higher (~80% reduction) | Moderate |
| Cycle regulation | ✅ Good | ✅ Good |
| Evidence base for HMB | ✅ Stronger | Moderate |
| Androgenic side effects | ⚠️ Mild (acne, weight gain) | ✅ Minimal |
| Bone density | ✅ Neutral | ✅ Oral — neutral (DMPA ❌) |
| Mood effects | ⚠️ Possible | ⚠️ Possible |
| Adolescent suitability | Acceptable (2nd line) | Acceptable (2nd line) |
| First-line status | ❌ Not first-line | ❌ Not first-line |
🧭 Recommended Approach for This Patient
16-year-old with HMB
│
├─ Exclude: Pregnancy, coagulopathy (vWD), thyroid disease
│
├─ Assess severity:
│ Mild-Moderate → Non-hormonal first
│ Severe (Hb <10, flooding, school disruption) → Hormonal
│
├─ FIRST-LINE OPTIONS:
│ Non-hormonal: Tranexamic acid + Mefenamic acid (during menses)
│ Hormonal: COCP (if no contraindication) → best for anovulatory DUB
│
├─ IF COCP CONTRAINDICATED:
│ Cyclical Norethisterone 5mg TDS (days 5–26) — better than MPA for HMB
│ OR Oral MPA 10mg BD (days 16–26) — if norethisterone poorly tolerated
│
├─ AVOID:
│ DMPA (Depo-Provera) injectable — bone density loss in adolescence
│
├─ IF REFRACTORY / SEVERE:
│ LNG-IUS (Mirena)
│ Haematology referral if coagulopathy confirmed
│
└─ Correct anaemia: Oral ferrous sulphate 200mg TDS
✅ Direct Answer to the Question
| Recommendation | |
|---|---|
| Cyclical norethisterone vs. MPA | Cyclical norethisterone has stronger evidence for HMB and achieves better blood loss reduction (~80%). MPA is an alternative if norethisterone is poorly tolerated (less androgenic). |
| But more importantly | Neither is the ideal first-line in a 16-year-old. COCP (for anovulatory HMB) or tranexamic acid + NSAIDs (non-hormonal) should be tried first. DMPA injectable MPA is specifically to be avoided in adolescents due to bone density concerns. |
What if it is a 40 year old person
HMB Management in a 40-Year-Old — Cyclical Progesterone vs. MPA
🔍 Key Differences from the 16-Year-Old
| Factor | 16-Year-Old | 40-Year-Old |
|---|---|---|
| Likely cause | Anovulatory DUB (HPO immaturity) | Anovulatory DUB (perimenopausal HPO decline) OR structural pathology |
| Structural pathology risk | Low | Significantly higher — fibroids, polyps, adenomyosis, endometrial hyperplasia |
| Endometrial cancer risk | Very low | ↑ Risk — must exclude |
| Coagulation disorder | Must exclude | Less likely as new diagnosis |
| Bone density concern | Critical | Less critical (peak bone mass achieved) |
| Fertility | Likely desired | May or may not be desired — key question |
| Contraception need | Possible | Yes — still fertile; must address |
| COCP use | Safe generally | More caution (VTE risk ↑ with age, smoking, BMI) |
| DMPA | Avoid | Acceptable; amenorrhoea beneficial |
| Proximity to menopause | Far | ~10 years away; symptoms may self-resolve |
⚠️ Step 1 — Mandatory Investigation BEFORE Treatment
| Investigation | Purpose |
|---|---|
| FBC + ferritin | Anaemia; iron deficiency |
| TFTs | Hypothyroidism → HMB |
| Coagulation screen | Less likely but exclude |
| β-hCG | Always exclude pregnancy |
| TVUSS | Fibroids, polyps, adenomyosis, endometrial thickness |
| Endometrial biopsy (Pipelle) | Mandatory at 40 if HMB + risk factors; ET >7 mm or abnormal TVUSS appearance |
| Hysteroscopy | If TVUSS abnormal or biopsy inconclusive |
| LH, FSH, estradiol | Assess perimenopausal status |
At 40, never start hormonal treatment for HMB without first excluding endometrial pathology (hyperplasia/carcinoma) — especially if irregular bleeding.
🎯 Direct Answer: Cyclical Progesterone vs. MPA at 40
Both are now more appropriate than in the 16-year-old — but still not the best first-line options.
⚖️ Head-to-Head Comparison at Age 40
| Feature | Cyclical Norethisterone | Oral MPA | DMPA (Injectable MPA) |
|---|---|---|---|
| Efficacy for HMB | ✅ ~80% reduction (days 5–26) | Moderate (days 16–26) | ✅ Amenorrhoea ~50% at 1 year |
| Cycle regulation | ✅ Good | ✅ Good | Irregular initially then amenorrhoea |
| Endometrial protection | ✅ Opposes estrogen-driven hyperplasia | ✅ | ✅ |
| Bone density at 40 | ✅ Neutral | ✅ Oral neutral | ⚠️ ↓ BMD — less critical at 40 than 16 |
| Androgenic effects | ⚠️ Mild acne/weight gain | ✅ Minimal | ✅ Minimal |
| Contraception | ❌ No contraceptive effect | ❌ No (oral) | ✅ Effective contraception |
| Perimenopausal symptoms | Not addressed | Not addressed | ✅ Reduces symptoms |
| Amenorrhoea induction | Sometimes | Sometimes | ✅ High rate |
| Reversibility | ✅ Immediate | ✅ Immediate | ⚠️ 3 months per injection |
| Fertility impact | ✅ Reversible | ✅ Reversible | Delayed return (~6–12 months) |
🥇 Best Options at Age 40 — Ranked
1. 🏆 LNG-IUS (Mirena) — BEST OVERALL OPTION
Still the single most effective and appropriate treatment for HMB at 40 — as at any age with a normal uterus.
| Feature | Detail |
|---|---|
| Efficacy | 90–95% reduction in blood loss; amenorrhoea in ~50% |
| Endometrial protection | ✅ Strongly protects against hyperplasia |
| Contraception | ✅ Highly effective — important at 40 |
| Duration | 5–8 years — bridges to menopause |
| Side effects | Minimal systemic absorption; local progestogen only |
| Perimenopausal | Can be used as the progestogen component of HRT if needed |
| Fertility | Rapidly reversible on removal |
| At 40 | Ideal — may last until menopause; eliminates need for further treatment |
2. COCP — Good Option if No Contraindications
| Feature | Detail |
|---|---|
| Efficacy | ~50% reduction; regular withdrawal bleeds |
| Contraception | ✅ Effective |
| Additional benefits | Perimenopausal symptom control; bone protection; acne; dysmenorrhoea |
| Caution at 40 | ↑ VTE risk with age; contraindicated if: smoker >35, migraine with aura, obesity + other VTE risk, hypertension, cardiovascular disease |
| UKMEC | Age 40 alone = UKMEC 2 (benefits outweigh risks); with additional risk factors → UKMEC 3–4 |
3. Non-hormonal Options — Still Valid First-line
| Drug | Role at 40 |
|---|---|
| Tranexamic acid | Effective; non-hormonal; no contraceptive effect; good if structural cause treated |
| NSAIDs (mefenamic acid) | Moderate HMB reduction + dysmenorrhoea; non-hormonal |
| Combination | Tranexamic acid + NSAID together — additive |
4. Cyclical Norethisterone (Days 5–26)
| Feature | Detail |
|---|---|
| At 40 | More appropriate than at 16 — bone density not a concern |
| Best use | When COCP contraindicated and LNG-IUS declined |
| Dose | 5 mg TDS, days 5–26 |
| Efficacy | ~80% blood loss reduction |
| Limitation | No contraceptive effect; androgenic side effects; requires daily compliance |
| Endometrial protection | ✅ Good — important at 40 with anovulatory cycles |
5. Oral MPA (Cyclical)
| Feature | Detail |
|---|---|
| At 40 | Acceptable alternative to norethisterone |
| Dose | 10 mg BD, days 16–26 (luteal phase) or days 5–26 for heavier bleeding |
| Vs Norethisterone | Less androgenic; similar efficacy for luteal phase supplementation; less data for full cycle use in HMB |
| Best indication | Anovulatory cycles where luteal phase support is the primary aim |
| Limitation | No contraception; daily pill |
6. DMPA (Depo-Provera) — Now Acceptable at 40
| Feature | Detail |
|---|---|
| Bone density | ⚠️ Still causes BMD loss but at 40, peak bone mass is achieved; WHO advises use with awareness but less restricted than in adolescents |
| HMB | Induces amenorrhoea in ~50% — excellent for HMB |
| Contraception | ✅ Effective |
| Perimenopausal | Useful — can bridge toward menopause |
| Limitation | Irregular bleeding initially; weight gain; delayed fertility return; cannot quickly reverse |
| Use | If fertility definitely not desired and LNG-IUS not acceptable |
7. GnRH Agonists — Bridge Only
| Feature | Detail |
|---|---|
| Role at 40 | Short-term (3–6 months) preoperative or bridge to menopause |
| Efficacy | Amenorrhoea; fibroid shrinkage |
| Limitation | Max 6 months (bone loss, menopausal symptoms); add-back HRT needed |
| Use | Preoperative optimisation; managing anaemia before surgery |
📋 Decision Based on Fertility Desire
If Fertility Still Desired:
Non-hormonal: Tranexamic acid + NSAIDs (during menses)
↓ (if insufficient)
Cyclical Norethisterone (days 5–26) OR Oral MPA
↓ (if insufficient)
LNG-IUS (still safe — rapid reversal on removal)
↓ (if structural cause found)
Hysteroscopic polypectomy / myomectomy as appropriate
If Fertility NOT Desired (Family Complete):
LNG-IUS ← BEST FIRST-LINE (also provides contraception)
OR
COCP (if no contraindications)
↓ (if declined/contraindicated)
Cyclical norethisterone / oral MPA
↓ (if failed)
DMPA / GnRH agonist (bridge)
↓ (if all medical options fail)
Endometrial ablation (family complete, normal cavity)
↓ (definitive)
Hysterectomy
📊 Summary Comparison — 16-Year-Old vs. 40-Year-Old
| Decision Point | 16-Year-Old | 40-Year-Old |
|---|---|---|
| First exclude | Coagulopathy, pregnancy | Structural pathology, endometrial pathology, pregnancy |
| Underlying mechanism | HPO immaturity (anovulation) | Perimenopause (anovulation) OR structural |
| Best first-line | Tranexamic acid + NSAID / COCP | LNG-IUS |
| COCP | ✅ Good first-line | ✅ If no contraindications (check VTE risk) |
| Cyclical norethisterone | 2nd line (androgenic SE concern) | 2nd–3rd line (acceptable) |
| Oral MPA | 2nd line (less data than NET) | 2nd–3rd line (less androgenic than NET) |
| DMPA | ❌ Avoid (bone density) | ⚠️ Acceptable with awareness |
| LNG-IUS | Consider (excellent but acceptability) | ✅ Best overall option |
| Endometrial ablation | ❌ Not appropriate | ✅ If family complete |
| Hysterectomy | ❌ Not appropriate | ✅ If all else fails |
✅ Direct Answer
At 40 years, both cyclical norethisterone and oral MPA are more appropriate than at 16 (bone density not a concern; higher risk of endometrial hyperplasia needing progestogen protection).
- Norethisterone (days 5–26) is superior to MPA for HMB reduction (~80% vs. moderate)
- Oral MPA is preferred if androgenic side effects of norethisterone are a concern
- However, LNG-IUS remains the single best treatment for HMB at 40 — more effective than either oral progestogen, provides contraception, and protects the endometrium until menopause
- Investigate first — endometrial biopsy + TVUSS mandatory before starting any treatment at 40
Fixed retroverted uterus and tender nodules at the pouch of douglas in a 35 year old women with a history of deep dyspareunia,dysmenorrhoea and pelvic pain what can be the diagnosis and the best management
endometriosis diagnosis fixed retroverted uterus pouch of douglas nodules deep dyspareunia dysmenorrhoea
endometriosis management treatment surgical medical options fertility deep infiltrating
endometriosis staging classification rASRM deep infiltrating bowel bladder uterosacral ligament surgery
endometriosis pouch of douglas deep infiltrating laparoscopy
Diagnosis & Management: Fixed Retroverted Uterus + POD Nodules + Deep Dyspareunia + Dysmenorrhoea
🎯 Diagnosis: Deep Infiltrating Endometriosis (DIE)
"Finding pelvic tenderness, a fixed retroverted uterus, tender uterosacral ligaments or enlarged ovaries on examination is suggestive of endometriosis. The diagnosis is more certain if deeply infiltrating nodules are found on the uterosacral ligaments or in the pouch of Douglas." (Bailey & Love's, p. 1603)
🔍 Clinching the Diagnosis — Feature by Feature
| Clinical Feature | Significance |
|---|---|
| Fixed retroverted uterus | Posterior adhesions from endometriotic implants tether the uterus posteriorly → cannot be anteverted on bimanual |
| Tender nodules in Pouch of Douglas | Deep infiltrating endometriotic deposits in rectovaginal septum / uterosacral ligaments — classic DIE finding |
| Deep dyspareunia | Endometriotic nodules in POD/uterosacral ligaments are compressed during deep penetration → severe pain |
| Dysmenorrhoea | Prostaglandin-mediated uterine contractions + inflamed peritoneal implants → progressive, severe, often debilitating |
| Chronic pelvic pain | Peritoneal sensitisation + nerve involvement by deeply infiltrating deposits |
| Age 35 | Peak reproductive age for symptomatic endometriosis |
📸 Laparoscopic Appearance of DIE
🧬 Pathophysiology
- Uterosacral ligaments
- Pouch of Douglas / rectovaginal septum
- Ovaries (endometriomas)
- Bowel (rectosigmoid)
- Bladder
- Pelvic peritoneum
- Repeated cyclical bleeding from implants → inflammation → fibrosis → adhesion formation → uterus becomes fixed in retroversion
🔢 Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Deep Infiltrating Endometriosis ✅ | Fixed retroverted uterus, POD nodules, deep dyspareunia, dysmenorrhoea — complete clinical picture |
| Pelvic Inflammatory Disease (PID) | Acute onset, fever, cervical excitation, discharge, bilateral adnexal tenderness; no fixed uterus typically |
| Ovarian endometrioma | USS shows characteristic ground-glass cyst; may coexist with DIE |
| Pelvic adhesions (post-surgical/infective) | History of surgery/PID; no specific nodules; laparoscopy differentiates |
| Uterosacral ligament fibroma | Very rare; no cyclical symptoms |
| Rectovaginal abscess | Acute; fever; USS shows collection |
| Ovarian malignancy | Fixed mass; Ca-125 ↑; USS features; usually older |
🔬 Investigations
First-Line
| Investigation | Finding in DIE |
|---|---|
| Pelvic USS (TVUSS) | Endometriomas (ground-glass ovarian cysts); POD obliteration; uterosacral thickening; sliding sign negative (bowel adherent to uterus) |
| Rectovaginal examination | Palpable nodules in rectovaginal septum; tenderness of uterosacral ligaments |
| CA-125 | Elevated in ~80% of advanced endometriosis; not diagnostic but supportive; elevated in many conditions |
| FBC | Baseline; exclude anaemia |
Gold Standard
| Investigation | Detail |
|---|---|
| Laparoscopy + histological confirmation | Definitive diagnosis — direct visualisation of lesions; biopsy confirms endometrial glands and stroma histologically (Endometriosis: Diagnosis and Management, p. 30) |
Additional Imaging for DIE (Pre-surgical Mapping)
| Investigation | Purpose |
|---|---|
| MRI pelvis | Best non-invasive modality for DIE mapping; identifies rectovaginal, uterosacral, bladder, bowel involvement; depth of infiltration; surgical planning |
| Transvaginal USS (expert) | Bowel endometriosis (rectosigmoid nodules); sliding sign to assess POD obliteration |
| Transrectal USS | Bowel wall infiltration depth |
| Cystoscopy | If bladder involvement suspected (urinary symptoms) |
| Colonoscopy / sigmoidoscopy | If deep bowel involvement suspected (PR bleeding, altered bowel habit cyclically) |
| IVU / Renal USS | Ureteric involvement — hydroureter/hydronephrosis (silent in some cases) |
"A digital rectal examination should be conducted to assess for disease involving the rectosigmoid area, as well as lateral and dorsal extension suggesting involvement of the hypogastric vessels and/or nerves." (Bailey & Love's, p. 1603)
📊 Staging (rASRM Classification)
| Stage | Description | This Patient |
|---|---|---|
| Stage I (Minimal) | Isolated superficial implants | ✗ |
| Stage II (Mild) | Deeper implants, small adhesions | ✗ |
| Stage III (Moderate) | Endometriomas, more adhesions | Possible |
| Stage IV (Severe) | Large endometriomas, dense adhesions, obliterated POD | Most likely ✅ |
Fixed uterus + obliterated POD + DIE nodules = Stage III–IV endometriosis
💊 Management
"Management options include pharmacological, non-pharmacological and surgical treatments. Endometriosis is an oestrogen-dependent condition. Most drug treatments work by suppressing ovarian function. The choice depends on the woman's preferences and priorities in terms of pain management and/or fertility." (Endometriosis: Diagnosis and Management, p. 30)
- Is fertility desired?
- Is pain the primary concern or infertility or both?
🔷 A. If FERTILITY DESIRED + PAIN
Medical (Temporising — does not restore fertility)
| Drug | Mechanism | Role |
|---|---|---|
| NSAIDs | ↓ Prostaglandins → pain relief | Symptomatic only; does not treat disease |
| COCP (continuous) | Suppresses ovulation + endometrial growth | Reduces pain; NOT used if trying to conceive |
| Progestogens (norethisterone, dienogest, DMPA, LNG-IUS) | Decidualisation + atrophy of implants | Pain relief; contraceptive; not for active fertility |
| GnRH agonists (goserelin, leuprolide) | Medical menopause; shrinks implants | Pre-surgical; not for concurrent fertility attempts |
| GnRH antagonists (elagolix, linzagolix) | Same; oral; titratable dose | NICE approved; better tolerability |
⚠️ All hormonal treatments suppress ovulation — they are contraceptive by nature. They do NOT improve fertility and should not be used when actively trying to conceive.
Surgical (Definitive for Fertility + Pain)
| Procedure | Detail |
|---|---|
| Laparoscopic excision of DIE (preferred over ablation) | Complete excision of nodules — uterosacral ligaments, rectovaginal septum, POD; restores anatomy; improves fertility and pain |
| Adhesiolysis | Divide adhesions fixing uterus; restores mobility |
| Ovarian endometrioma cystectomy | Stripping technique preferred over drainage; preserves ovarian cortex |
| Rectovaginal nodule excision | If rectovaginal septum involved; may require colorectal surgeon assistance |
| Bowel resection (if bowel deeply infiltrated) | Segmental resection; multidisciplinary (gynaecologist + colorectal surgeon) |
| Ureteric stenting / ureterolysis | If ureteric involvement |
| POD reconstruction | Restore normal pelvic anatomy |
- Excision > ablation for DIE — ablation misses depth of infiltration
- Complete excision at one operation preferred — reduces recurrence
- Multidisciplinary team (gynaecology, colorectal, urology) for complex DIE
- Postoperative hormonal suppression reduces recurrence
- Spontaneous pregnancy rates improve after laparoscopic excision of Stage III–IV disease
- If natural conception not achieved within 6–12 months post-surgery → IVF
- IVF may be primary approach in severe DIE with poor ovarian reserve (AMH low)
🔷 B. If FERTILITY NOT DESIRED + PAIN MANAGEMENT
Medical (Long-term Suppression)
| Option | Detail |
|---|---|
| LNG-IUS (Mirena) | First-line long-term; reduces dysmenorrhoea + HMB; local progestogen |
| COCP (continuous) | Suppress menstruation; reduce pain; prevent recurrence post-surgery |
| Dienogest 2 mg daily | Progestogen specifically licensed for endometriosis; highly effective for pain; preserves BMD |
| GnRH agonist + add-back HRT | Powerful suppression; max 6 months without add-back; with add-back can extend |
| GnRH antagonists (elagolix) | Oral; titratable; approved for endometriosis pain |
| DMPA | Amenorrhoea; suppresses implants; long-term use with BMD monitoring |
Surgical
| Procedure | Indication |
|---|---|
| Laparoscopic excision of DIE | First-line surgical treatment; pain relief + prevent progression |
| Laparoscopic uterosacral nerve ablation (LUNA) | Adjunct; interrupts pain pathways; limited evidence alone |
| Presacral neurectomy | Centralised midline pain; specialist centres |
| Hysterectomy + BSO | Definitive — for severe refractory disease, family complete; BSO prevents recurrence from ovarian estrogen; HRT can be given post-BSO (add-back) |
📋 Specific Management Plan for THIS Patient (35-year-old, DIE)
35-year-old — Fixed retroverted uterus + POD nodules + DIE symptoms
│
├─ INVESTIGATIONS:
│ TVUSS → endometriomas, POD obliteration
│ MRI pelvis → DIE mapping (bowel, bladder, uterosacral)
│ CA-125
│ Assess ovarian reserve (AMH, AFC) — fertility planning
│ DRE → rectovaginal nodule assessment
│
├─ FERTILITY DESIRED?
│ │
│ YES → Laparoscopic excision of DIE
│ + adhesiolysis + restore anatomy
│ + cystectomy if endometrioma present
│ → Attempt natural conception 6–12 months
│ → IVF if not achieved
│ → Post-surgical: hormonal suppression
│ (COCP / progestogen) until ready to conceive
│
└─ FERTILITY NOT DESIRED / COMPLETE:
Pain management priority:
Dienogest / COCP / LNG-IUS (medical suppression)
↓ (if inadequate)
Laparoscopic excision of DIE
↓ (post-surgical)
Long-term hormonal suppression to prevent recurrence
↓ (refractory / severe)
Hysterectomy + BSO (definitive)
⚠️ Special Considerations in DIE
| Issue | Management |
|---|---|
| Bowel involvement (cyclical rectal bleeding, dyschezia) | Pre-op bowel prep; colorectal surgeon at laparoscopy; segmental resection if full-thickness |
| Ureteric involvement (hydronephrosis — often silent) | Renal USS; ureteric stents; ureterolysis at surgery |
| Bladder endometriosis (cyclical haematuria) | Cystoscopy; partial cystectomy |
| Obliterated POD | Most technically demanding dissection; risk of rectal injury; specialist centre |
| Recurrence | ~20–40% at 5 years; post-surgical hormonal suppression critical |
| Psychological impact | Chronic pain → anxiety, depression; multidisciplinary support |
| Pain sensitisation | Central sensitisation in long-standing disease; pain psychology + physiotherapy alongside surgical/medical treatment |
✅ Summary
| Detail | |
|---|---|
| Diagnosis | Deep Infiltrating Endometriosis (DIE) — Stage III–IV |
| Confirmatory test | Laparoscopy + histology (gold standard) |
| Best pre-surgical imaging | MRI pelvis + expert TVUSS |
| If fertility desired | Laparoscopic excision of DIE → attempt natural conception → IVF |
| If fertility not desired | Hormonal suppression (dienogest/COCP/LNG-IUS) → laparoscopic excision → hysterectomy + BSO if refractory |
| Key principle | Medical treatment suppresses but does not cure; complete laparoscopic excision gives best outcomes for both pain and fertility |
Difference between endometrioma and fibroids
endometrioma ovarian cyst diagnosis ultrasound features histology
endometrioma vs fibroid ultrasound comparison
endometrioma ovarian cyst ultrasound ground glass appearance
uterine fibroid leiomyoma ultrasound hypoechoic shadowing
Endometrioma vs. Uterine Fibroid — Complete Comparison
🔬 Basic Definition
| Feature | Endometrioma | Uterine Fibroid (Leiomyoma) |
|---|---|---|
| Nature | Ovarian cyst containing ectopic endometrial tissue + old blood ("chocolate cyst") | Benign smooth muscle tumour of the uterine myometrium |
| Tissue origin | Endometrial glands + stroma implanted on/in the ovary | Uterine smooth muscle cells (myometrium) |
| Hormone dependence | Estrogen-dependent — grows with estrogen, regresses with menopause | Estrogen + progesterone dependent — grows with both, regresses with menopause |
| Malignant potential | Very low (~0.3–1%) — risk of endometrioid/clear cell ovarian carcinoma | Very low (~0.5%) — risk of leiomyosarcoma |
| Location | Ovary (most common site of endometriosis) | Uterus — intramural, submucosal, subserosal, pedunculated |
🧬 Pathology
| Feature | Endometrioma | Fibroid |
|---|---|---|
| Gross appearance | Cystic; thick-walled; contains dark brown "chocolate" fluid (old haemolysed blood) | Solid; firm; whorled white/grey cut surface; well-circumscribed with pseudocapsule |
| Histology | Endometrial glands + stroma + haemosiderin-laden macrophages in cyst wall; may show fibrosis | Interlacing bundles of smooth muscle cells with variable fibrous tissue; mitoses rare; hyalinisation common |
| Consistency | Cystic (fluid-filled) | Solid |
| Capsule | Thick fibrous wall; adherent to surrounding structures | Well-defined pseudocapsule (compressed myometrium) |
| Contents | Dark brown viscous "chocolate" fluid | Solid (whorled muscle); may degenerate (hyaline, cystic, red, calcific) |
👩 Epidemiology & Risk Factors
| Feature | Endometrioma | Fibroid |
|---|---|---|
| Prevalence | ~17–44% of women with endometriosis; 1–5% general population | Most common pelvic tumour; ~25–50% of women of reproductive age |
| Peak age | 25–40 years (reproductive) | 30–50 years (reproductive/perimenopausal) |
| Race | No strong racial predisposition | Black women — 3× higher incidence, earlier onset, more severe |
| Risk factors | Retrograde menstruation; nulliparity; early menarche; short cycles; family history; müllerian anomalies | Nulliparity; obesity; Black race; early menarche; family history; no protective effect of smoking |
| Protective | Pregnancy; breastfeeding; OCP | Pregnancy; smoking (weak); OCP |
🩺 Clinical Presentation
| Symptom | Endometrioma | Fibroid |
|---|---|---|
| Pain | Dysmenorrhoea (severe, progressive); deep dyspareunia; chronic pelvic pain — cyclical | Dysmenorrhoea (usually less severe); pelvic heaviness/pressure; usually non-cyclical |
| Menstrual bleeding | Usually normal or slightly heavy; may have irregular bleeding | Heavy menstrual bleeding (HMB) — hallmark symptom, especially submucosal |
| Infertility | Common — ~50% of infertile women have endometriosis | Common — especially submucosal/intramural distorting cavity |
| Pelvic mass | Adnexal (ovarian) mass | Uterine enlargement; may feel irregular |
| Acute presentation | Rupture → acute peritonism (chocolate fluid spillage) | Torsion of pedunculated fibroid; red degeneration in pregnancy |
| Bladder/bowel | Cyclical symptoms (haematuria, dyschezia) if DIE coexists | Frequency, retention, constipation from mass effect (non-cyclical) |
| Examination | Adnexal tenderness; fixed uterus; POD nodules; ovarian mass | Enlarged irregular uterus; firm mass; mobile (unless adherent) |
🖥️ Ultrasound Appearances
Endometrioma on USS
| USS Feature | Endometrioma |
|---|---|
| Structure | Cystic — unilocular (1–4 locules) |
| Echogenicity | Ground-glass — homogeneous low-level internal echoes |
| Contents | Uniform; no solid nodules |
| Wall | Thick, regular, well-defined |
| Doppler | Peripheral flow only; avascular internally |
| Sensitivity/Specificity | Sensitivity 93%, Specificity 96% (Berek & Novak's Gynecology, p. 632) |
| Location | Ovary (adnexal) |
| Bilateral | ~30% bilateral |
Fibroid on USS
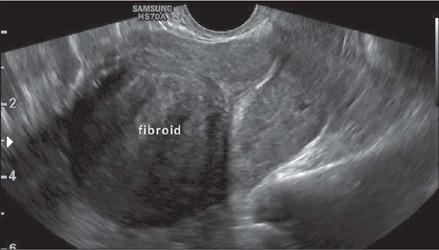
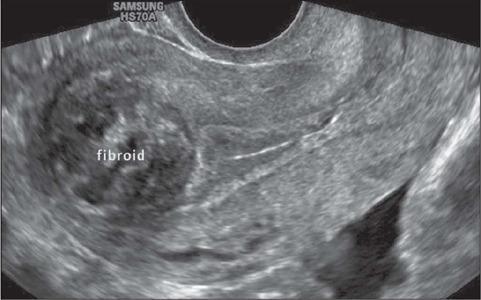
| USS Feature | Fibroid |
|---|---|
| Structure | Solid — not cystic |
| Echogenicity | Hypoechoic to isoechoic; heterogeneous |
| Contents | Solid whorled tissue; may have calcification (echogenic foci) |
| Acoustic shadow | ✅ Posterior acoustic shadowing — hallmark feature |
| Wall | Well-circumscribed pseudocapsule |
| Doppler | Peripheral + internal vascularity (unlike endometrioma) |
| Location | Uterine (intramural / submucosal / subserosal) |
| Uterus | Enlarged, lobulated, irregular |
📊 Side-by-Side Comparison Table
| Feature | Endometrioma | Fibroid |
|---|---|---|
| Tissue type | Cystic — endometrial tissue | Solid — smooth muscle |
| Location | Ovary | Uterus |
| USS structure | Cystic, ground-glass | Solid, heterogeneous |
| USS shadowing | ❌ None | ✅ Acoustic shadowing |
| Doppler | Peripheral only, avascular internally | Peripheral + internal flow |
| Contents | Chocolate fluid (old blood) | Solid muscle/fibrous tissue |
| Primary symptom | Dysmenorrhoea + dyspareunia | Heavy menstrual bleeding |
| Pain character | Cyclical, progressive | Pressure + dysmenorrhoea |
| Uterus on exam | May be fixed/retroverted | Enlarged, irregular, firm |
| Adnexa | Adnexal mass | Usually no adnexal mass |
| Fertility effect | ↓ Ovarian reserve; adhesions; tubal | Cavity distortion → implantation failure |
| CA-125 | Often elevated | Usually normal |
| AMH | ↓ (ovarian reserve reduced) | Usually normal |
| Hormone dependence | Estrogen only | Estrogen + progesterone |
| Behaviour in pregnancy | May enlarge/rupture; decidualisation | May cause red degeneration |
| Menopause | Regresses (estrogen-dependent) | Regresses (hormone-dependent) |
| Recurrence after surgery | ~30–40% at 5 years | ~30% at 10 years |
| Malignant transformation | Rare (~0.3–1%) → endometrioid/clear cell Ca | Rare (~0.5%) → leiomyosarcoma |
| Diagnosis | Laparoscopy + histology (gold standard) | USS; histology post-surgery |
| MRI | T1 bright, T2 dark (shading sign) | T2 hypointense whorled mass |
🧲 MRI Differences
| MRI Feature | Endometrioma | Fibroid |
|---|---|---|
| T1 signal | Bright (hyperintense) — blood products | Hypointense (dark) |
| T2 signal | Dark (T2 shading) — haemosiderin from repeated bleeding | Hypointense (dark) — fibrous/smooth muscle |
| Enhancement | Peripheral wall enhancement | Variable internal enhancement |
| Characteristic sign | T2 shading — diagnostic of endometrioma | Whorled hypointense mass |
| Differentiation | From dermoid, haemorrhagic cyst, corpus luteum | From adenomyoma, leiomyosarcoma |
🔬 Tumour Markers
| Marker | Endometrioma | Fibroid |
|---|---|---|
| CA-125 | Often ↑ (non-specific; raised in endometriosis) | Usually normal |
| CA-19.9 | May be mildly ↑ | Normal |
| AMH | ↓ — reduced ovarian reserve | Normal |
| LDH | Normal | ↑ in leiomyosarcoma (rapidly growing fibroid) |
💊 Medical Management Comparison
| Treatment | Endometrioma | Fibroid |
|---|---|---|
| COCP | ✅ Suppresses growth; pain relief | ✅ Mild HMB reduction |
| Progestogens | ✅ Decidualisation of implants | ✅ Endometrial atrophy; some shrinkage |
| GnRH agonists | ✅ Shrinks; controls pain; preoperative | ✅ Shrinks 30–50%; preoperative |
| LNG-IUS | ✅ Pain relief; reduces recurrence | ✅ HMB reduction |
| Tranexamic acid | Not applicable (not HMB-driven) | ✅ First-line for HMB |
| NSAIDs | ✅ Dysmenorrhoea | ✅ Dysmenorrhoea + HMB |
✂️ Surgical Management Comparison
| Procedure | Endometrioma | Fibroid |
|---|---|---|
| Cystectomy (stripping) | ✅ Gold standard — removes cyst wall | N/A |
| Myomectomy | N/A | ✅ Gold standard uterus-preserving |
| Hysteroscopic resection | N/A | ✅ Submucosal fibroids |
| Laparoscopic excision | ✅ Endometrioma + peritoneal disease | ✅ Laparoscopic myomectomy |
| Hysterectomy | ✅ Definitive (with BSO) | ✅ Definitive |
| UAE | ❌ Not appropriate | ✅ Effective option |
| MRgFUS | ❌ Not appropriate | ✅ Emerging option |
| Ovarian reserve | ⚠️ Cystectomy reduces reserve further — counsel carefully | Not affected |
⚠️ Key Clinical Pearls
| Endometrioma | Fibroid | |
|---|---|---|
| Never drain and leave | Drainage alone → near 100% recurrence; always excise cyst wall | Drainage not applicable |
| Fertility surgery | Bilateral endometriomas → check AMH first; risk of further reserve loss | Submucosal → must remove before IVF |
| Postmenopausal growth | → Suspect malignant transformation | → Suspect leiomyosarcoma |
| Coexistence | Endometrioma + fibroid can coexist in same patient | Same |
| Do not confuse on USS | Ground-glass cyst = endometrioma; avascular internally | Solid + acoustic shadow + internal flow = fibroid |
What are the differences on examination
Differences on Examination: Endometrioma vs. Fibroid
👁️ General / Abdominal Examination
| Finding | Endometrioma | Fibroid |
|---|---|---|
| Abdominal distension | Only if very large or with ascites | Present if uterus >14–16 weeks size |
| Palpable mass | Lower abdominal/pelvic adnexal mass if large (>8–10 cm) | Firm, irregular midline mass arising from pelvis; moves with uterus |
| Mass character | Soft-cystic, may be tender | Hard, irregular, nodular, firm |
| Mass origin | Lateral (adnexal) — may be bilateral | Central/midline (uterine) |
| Tenderness | ✅ Often tender (especially premenstrually) | Variable; tender if degenerating |
| Ascites | Rare (if ruptured or malignant transformation) | Very rare |
🤚 Speculum Examination
| Finding | Endometrioma | Fibroid |
|---|---|---|
| Cervix appearance | Usually normal; may see blue-black endometriotic deposits in posterior vaginal fornix / cervix if DIE coexists | Usually normal; cervical fibroid may be seen protruding through os |
| Vaginal wall | May show blue/purple nodules in posterior fornix (rectovaginal DIE) | Normal unless cervical/broad ligament fibroid distorts |
| Discharge | Usually normal | Normal; may have increased if submucosal causing irregular bleeding |
| Cervical excitation | ✅ Present — tenderness on moving cervix | ❌ Usually absent (unless degenerating) |
🖐️ Bimanual Examination
| Finding | Endometrioma | Fibroid |
|---|---|---|
| Uterine size | Usually normal size | Enlarged — uterus may feel equivalent to 8–20+ weeks size |
| Uterine shape | Normal | Irregular, lobulated, nodular |
| Uterine consistency | Normal | Firm to hard (like contracting uterus) |
| Uterine mobility | Restricted / fixed (if adhesions; retroversion) | Usually mobile (unless adherent) |
| Uterine position | Fixed retroversion — hallmark of DIE coexisting with endometrioma | Usually anteverted; can be retroverted if large posterior fibroid |
| Adnexal mass | ✅ Adnexal (ovarian) mass — cystic, tender, may be fixed to uterus/bowel | ❌ No adnexal mass (unless pedunculated subserosal mimics adnexal) |
| Mass character (adnexa) | Cystic, tender, tethered — does not move independently from uterus if adherent | Pedunculated fibroid feels solid, firm, moves with uterus |
| Tenderness | Marked — especially adnexal and uterosacral regions | Variable; mild unless degenerating or large |
| Uterosacral ligaments | Thickened, nodular, tender — highly specific for endometriosis/DIE | Smooth, non-tender |
| Pouch of Douglas | Obliterated / nodules palpable — pathognomonic of DIE | Clear; no nodules |
| Cervical excitation | ✅ Positive — tender on cervical movement | ❌ Usually negative |
| Bilateral adnexa | May be bilateral (~30%) | N/A — uterine mass |
🖐️ Rectal Examination (PR — Important in DIE)
| Finding | Endometrioma (with DIE) | Fibroid |
|---|---|---|
| Rectovaginal nodules | ✅ Palpable hard nodules in rectovaginal septum — diagnostic of DIE | ❌ Absent |
| Rectal wall | May feel indurated / tethered if bowel involved | Normal |
| Tenderness | ✅ Marked — especially premenstrually | Absent |
| Uterosacral thickening | ✅ Palpable from PR | ❌ Absent |
📋 Summary — Key Examination Differences at a Glance
| Examination Feature | Endometrioma | Fibroid |
|---|---|---|
| Uterine size | Normal | Enlarged |
| Uterine shape | Normal | Irregular/lobulated |
| Uterine consistency | Normal | Firm/hard |
| Uterine mobility | Restricted/fixed | Mobile (usually) |
| Uterine position | Fixed retroversion | Anteverted usually |
| Adnexal mass | ✅ Cystic, tender | ❌ (unless pedunculated) |
| Uterosacral ligaments | Nodular, thickened, tender | Normal, smooth |
| POD nodules | ✅ Palpable | ❌ |
| Cervical excitation | ✅ Positive | ❌ Negative |
| Rectovaginal nodules | ✅ (PR exam) | ❌ |
| Tenderness overall | Marked, cyclical | Mild/variable |
🔑 The Single Most Useful Examination Finding to Distinguish Them
| Finding | |
|---|---|
| Endometrioma | Fixed retroverted uterus + tender adnexal cystic mass + uterosacral nodularity + positive cervical excitation |
| Fibroid | Enlarged, firm, irregular, lobulated uterus + mobile + no adnexal tenderness + no POD nodules + no cervical excitation |
A cystic, tender, fixed adnexal mass with uterosacral nodularity and positive cervical excitation = endometrioma (± DIE)A firm, irregular, enlarged mobile uterus with no adnexal pathology and no POD findings = fibroid
2cm adenexal mass with fluid in Douglas pouch with 5w of gestation on a 30 year old primipara her pr is 100 and bp is 100/70 and beta hcg is 3000 what is recommended treatment option
Clinical Analysis
🚨 What Does This Picture Tell Us?
| Parameter | Finding | Interpretation |
|---|---|---|
| Age / parity | 30 years, primigravida | Fertility preservation critical |
| Gestational age | 5 weeks | Early pregnancy |
| Adnexal mass | 2 cm | Right/left adnexal — not intrauterine |
| Free fluid in POD | Present | Haemoperitoneum — blood in pelvis |
| β-hCG | 3000 mIU/mL | Positive pregnancy; no intrauterine sac visible at this level |
| Pulse | 100 bpm | Tachycardia — sign of haemodynamic compromise |
| BP | 100/70 mmHg | Hypotension — borderline shock |
🎯 Diagnosis: Ruptured Ectopic Pregnancy
Diagnostic Reasoning:
- At β-hCG 3000 mIU/mL, an intrauterine gestational sac should be visible on TVUSS
- The discriminatory zone for TVUSS = 1500–2000 mIU/mL (intrauterine sac visible above this level)
- β-hCG 3000 + no intrauterine sac = ectopic pregnancy until proven otherwise
- Represents the ectopic gestational sac / tubal pregnancy in the fallopian tube
- Most common site = ampullary portion of fallopian tube (~70%)
- In the context of ectopic pregnancy = haemoperitoneum
- Blood tracking into the pouch of Douglas from a ruptured or leaking ectopic
- PR 100 = tachycardia
- BP 100/70 = hypotension
- Together = Class II haemorrhagic shock → active intraperitoneal bleeding
- This rules out conservative (expectant) or medical (methotrexate) management
⚠️ Why Methotrexate is CONTRAINDICATED Here
| Methotrexate Criterion | This Patient | Eligible? |
|---|---|---|
| Haemodynamically stable | ❌ PR 100, BP 100/70 — unstable | ❌ |
| Unruptured ectopic | ❌ Free fluid = likely ruptured | ❌ |
| β-hCG <5000 mIU/mL | ✅ 3000 mIU/mL | ✅ |
| No fetal cardiac activity | Not stated | Possibly ✅ |
| Mass <3.5 cm | ✅ 2 cm | ✅ |
| No contraindications | Unknown | ? |
Haemodynamic instability + free fluid (haemoperitoneum) = absolute contraindication to methotrexate regardless of β-hCG level or mass size.
🚨 Recommended Treatment: Emergency Laparoscopic Surgery
Immediate Resuscitation (Before Theatre)
| Action | Detail |
|---|---|
| IV access | Two large-bore cannulae (14–16G) immediately |
| IV fluids | Crystalloid bolus (500 mL–1L stat) while preparing theatre |
| Blood group + crossmatch | 2–4 units packed red cells |
| FBC, coagulation, U&E | Urgent |
| Oxygen | High-flow via face mask |
| Catheterise | Monitor urine output |
| Consent | Emergency; explain salpingotomy vs. salpingectomy |
| Theatre | Activate immediately — do not delay for further investigations |
Definitive Treatment: Emergency Laparoscopy
- Experienced laparoscopic surgeon available
- Anaesthetic team ready
- If massive haemoperitoneum / extreme instability → convert to laparotomy
Surgical Decision: Salpingotomy vs. Salpingectomy
| Procedure | Definition | When to Choose |
|---|---|---|
| Salpingotomy (conservative) | Linear incision on tube → remove ectopic → leave tube intact | Contralateral tube damaged/absent; desires future natural conception; tube salvageable |
| Salpingectomy (radical) | Remove entire affected fallopian tube | Preferred in most cases — lower persistent ectopic rate; contralateral tube healthy |
For THIS Patient:
| Factor | Recommendation |
|---|---|
| Primigravida, 30 years | Fertility highly important — consider salpingotomy |
| Contralateral tube status | Assess at laparoscopy — if healthy contralateral tube → salpingectomy preferred (evidence-based) |
| Contralateral tube damaged/absent | → Salpingotomy to preserve fertility |
| NICE guidance | If contralateral tube is healthy → salpingectomy preferred (less risk of persistent trophoblast, lower ectopic recurrence in that tube) |
| IVF outcomes | Similar pregnancy rates after salpingectomy vs. salpingotomy when contralateral tube is intact |
In a primigravida with a healthy contralateral tube → Salpingectomy is the recommended procedure (NICE guidelines) — removes the affected tube, eliminates risk of persistent ectopic (which occurs in ~8% after salpingotomy), and does not significantly compromise future fertility if the other tube is normal.
If contralateral tube is absent, damaged, or pathological → Salpingotomy to maximise fertility preservation.
Post-Operative Management
| Action | Detail |
|---|---|
| β-hCG monitoring | Weekly until undetectable (<5 mIU/mL) — especially if salpingotomy performed (rule out persistent trophoblast) |
| Anti-D immunoglobulin | If patient is Rhesus negative — give within 72 hours |
| Iron supplementation | If significant blood loss + anaemia post-op |
| Counselling | Psychological support; risk of future ectopic (~10–15%); advice on early USS in future pregnancies |
| Contraception | Advise to wait 3 months before attempting next conception |
| Early scan next pregnancy | TVUSS at 6 weeks in all future pregnancies |
📋 Management Algorithm for This Patient
30-year-old — 5 weeks gestation
Adnexal mass 2 cm + POD fluid + PR 100 + BP 100/70 + β-hCG 3000
│
├─ DIAGNOSIS: RUPTURED ECTOPIC PREGNANCY
│ (Haemoperitoneum + haemodynamic compromise)
│
├─ IMMEDIATE:
│ IV access × 2 → Fluids → Crossmatch → O₂ → Catheter
│ Activate theatre URGENTLY
│
├─ METHOTREXATE? ❌ CONTRAINDICATED
│ (Haemodynamic instability + free fluid = rupture)
│
├─ EXPECTANT? ❌ CONTRAINDICATED
│ (Active bleeding + shock)
│
└─ SURGERY: Emergency Laparoscopy
│
├─ Evacuate haemoperitoneum
├─ Assess both tubes
│
├─ Contralateral tube HEALTHY?
│ YES → SALPINGECTOMY (preferred - NICE)
│ NO → SALPINGOTOMY (fertility preservation)
│
└─ Post-op:
Serial β-hCG (weekly until zero)
Anti-D if Rh negative
Counselling + early USS next pregnancy
✅ Summary Answer
The recommended treatment is emergency laparoscopic salpingectomy (or salpingotomy if the contralateral tube is compromised).This patient has a ruptured ectopic pregnancy with haemoperitoneum and haemodynamic compromise (tachycardia + hypotension) — this is a surgical emergency. Methotrexate and expectant management are both absolutely contraindicated. Resuscitation must begin immediately and she must be taken to theatre without delay.
When is laprotomy advised in this condition
Indications for Laparotomy in Ectopic Pregnancy
🔑 Core Principle
Laparoscopy is the gold standard for surgical management of ectopic pregnancy. Laparotomy is reserved for situations where laparoscopy is unsafe, impossible, or fails.
📋 Indications for Laparotomy
1. 🚨 Haemodynamic Instability / Cardiovascular Collapse
| Situation | Detail |
|---|---|
| Severe haemorrhagic shock | Systolic BP <90 mmHg unresponsive to fluid resuscitation |
| Massive haemoperitoneum | Large volume of blood causing abdominal distension and cardiovascular compromise |
| Cardiac arrest / peri-arrest | Immediate laparotomy to control bleeding |
| Rapidly deteriorating patient | No time to set up laparoscopic equipment safely |
The previous patient (PR 100, BP 100/70) is borderline — if she responds to initial resuscitation, laparoscopy remains appropriate. If she deteriorates further despite resuscitation → laparotomy.
- Key principle: Laparoscopy requires time to establish pneumoperitoneum and set up equipment — in extremis, a midline laparotomy gives fastest access to control haemorrhage
2. 🩺 Unavailability of Laparoscopic Expertise
| Situation | Detail |
|---|---|
| No experienced laparoscopic surgeon available | Laparotomy is safer than an inexperienced laparoscopic attempt |
| No laparoscopic equipment available | Resource-limited settings, rural hospitals, out-of-hours emergencies |
| Anaesthetic risk too high for prolonged laparoscopy | Laparotomy is faster |
3. 🔄 Failed / Converted Laparoscopy
| Situation | Detail |
|---|---|
| Inability to establish pneumoperitoneum | Dense adhesions from previous surgery preventing safe entry |
| Inadequate visualisation | Massive haemoperitoneum obscuring operative field |
| Uncontrollable bleeding laparoscopically | Haemostasis cannot be achieved; convert to open |
| Injury to surrounding structures | Bowel, bladder, major vessel injury during laparoscopy |
| Technically complex dissection | Anatomy too distorted to proceed safely laparoscopically |
4. 🏥 Significant Pelvic Adhesive Disease
| Situation | Detail |
|---|---|
| Previous multiple pelvic surgeries | Dense adhesions (previous salpingectomy, myomectomy, bowel surgery) → obliterated planes → laparoscopy hazardous |
| Previous pelvic inflammatory disease (PID) | Frozen pelvis; distorted anatomy |
| Previous laparotomy with midline scar | Relative indication; experienced surgeons can still attempt laparoscopy but conversion rate higher |
| Known frozen pelvis | Severe endometriosis + adhesions |
5. 📍 Unusual Location of Ectopic Pregnancy
| Ectopic Type | Why Laparotomy Needed |
|---|---|
| Cornual / Interstitial ectopic | Deep within uterine cornu; high vascularity; catastrophic haemorrhage risk; often requires cornual resection or hysterectomy |
| Cervical ectopic | May require hysterectomy if uncontrolled bleeding |
| Heterotopic pregnancy (ectopic + intrauterine) | Complex; requires careful dissection to preserve intrauterine pregnancy |
| Abdominal / peritoneal ectopic | Advanced; bowel/mesenteric involvement; complex vascular supply |
| Ovarian ectopic (large) | Large ovarian ectopics may require open approach |
| Broad ligament ectopic | Deep between leaves of broad ligament; ureteric risk |
6. 🔧 Technical / Equipment Factors
| Situation | Detail |
|---|---|
| Morbid obesity | Extreme BMI → inadequate pneumoperitoneum; poor visualisation; prolonged laparoscopy risky |
| Previous abdominal mesh | Adhesions to anterior wall preventing safe trocar insertion |
| Coagulopathy | Ongoing bleeding diathesis requiring fastest possible haemostasis |
7. ⚕️ Surgeon Preference / Training
| Situation | Detail |
|---|---|
| Surgeon not trained in advanced laparoscopy | Laparotomy is safer in inexperienced hands |
| Intraoperative complications beyond laparoscopic capability | Bowel/vessel injury requiring open repair |
📊 Laparoscopy vs. Laparotomy — Decision Guide
| Scenario | Approach |
|---|---|
| Haemodynamically stable + experienced surgeon | Laparoscopy |
| Mildly compromised but responding to resuscitation | Laparoscopy |
| Severe shock / cardiovascular collapse | Laparotomy |
| Massive haemoperitoneum + deteriorating | Laparotomy |
| No laparoscopic expertise available | Laparotomy |
| Dense pelvic adhesions | Laparotomy (or cautious laparoscopy with low threshold to convert) |
| Cornual / interstitial / abdominal ectopic | Laparotomy |
| Failed laparoscopy | Convert to laparotomy |
| Laparoscopic injury to bowel/vessel | Convert to laparotomy |
🔄 Conversion from Laparoscopy to Laparotomy
- Haemostasis cannot be achieved laparoscopically
- Visualisation is inadequate despite all manoeuvres
- Injury to bowel, bladder, ureter, or major vessel
- Pneumoperitoneum cannot be maintained
- Anaesthetic deterioration under laparoscopy
✅ Summary
| Indication | Category |
|---|---|
| Severe haemorrhagic shock / cardiovascular collapse | Emergency |
| Massive haemoperitoneum | Emergency |
| No laparoscopic expertise / equipment | Resource |
| Failed / converted laparoscopy | Intraoperative |
| Dense pelvic adhesions | Anatomical |
| Cornual, abdominal, broad ligament ectopic | Location |
| Morbid obesity / coagulopathy | Patient factors |
| Intraoperative complications | Intraoperative |
For the previous patient — if resuscitation stabilises her → proceed with laparoscopy. If she continues to deteriorate (BP falling further, HR >120, signs of Class III–IV shock) → immediate laparotomy without delay, as time to haemostasis is life-saving.
Explain cyclical HRT briefly and list the other HRT therapies by oestrogen
Cyclical HRT — Explanation + Full Classification by Oestrogen
🔵 What is Cyclical HRT?
Why Cyclical?
📅 How Cyclical HRT Works
Day 1 ──────────────────────────────────── Day 28
│ │
│ OESTROGEN ████████████████████████████████ │ (continuous, every day)
│ PROGESTOGEN ██████████████ │ (days 15–28 or days 17–28)
│ │
│ ↓ │
│ Withdrawal bleed │
│ (at end of progestogen) │
📋 Two Types of Cyclical HRT
| Type | Oestrogen | Progestogen | Bleed | Used In |
|---|---|---|---|---|
| Monthly cyclical (sequential) | Daily | 12–14 days/month | Monthly withdrawal bleed | Perimenopause; regular or irregular cycles |
| 3-monthly cyclical (long-cycle) | Daily | 14 days every 3 months | Bleed every 3 months | Perimenopausal; prefer less frequent bleeds |
👤 Who Gets Cyclical HRT?
- Perimenopausal women (still having periods or <1 year since last period)
- Women who want/need to know if they are still ovulating
- Women transitioning toward continuous combined HRT
- Not suitable for postmenopausal women (>1 year since last period) → they should use continuous combined HRT to avoid unnecessary bleeds
⚠️ Key Points About Cyclical HRT
- The withdrawal bleed is not a real period — it is pharmacological; does not indicate fertility
- Endometrial protection requires progestogen for at least 12 days per cycle — shorter duration increases hyperplasia risk
- Women should be counselled about the expected bleed — it should be light and predictable; heavy or irregular bleeding → investigate (endometrial biopsy)
- After 2 years of perimenopause (or confirmed postmenopause) → switch to continuous combined HRT to avoid bleeds
🗂️ Classification of HRT by Oestrogen Type
1. 🟠 Conjugated Equine Oestrogen (CEE)
- Derived from pregnant mare's urine (Premarin)
- Contains mixture of oestrone sulphate + equilin + multiple other conjugated oestrogens
- Standard dose: 0.625 mg/day (low dose 0.3 mg; higher 1.25 mg)
- Route: Oral tablet
- Oldest and most studied form of HRT (used in WHI trial)
- Higher first-pass liver metabolism → more hepatic effects (↑ SHBG, ↑ clotting factors, ↑ triglycerides) compared to transdermal
- Available as:
- Premarin (oestrogen alone — for hysterectomised women)
- Prempak-C (CEE + cyclical norgestrel)
- Premique (CEE + continuous MPA)
2. 🟡 Oestradiol (17β-oestradiol) — Bioidentical
- Identical to endogenous human oestradiol — most physiological form
- Preferred in modern HRT — especially transdermal (avoids first-pass liver metabolism)
- Available in multiple routes:
A. Oral Oestradiol
| Drug | Dose | Notes |
|---|---|---|
| Oestradiol valerate (Progynova) | 1–2 mg/day | Converted to oestradiol after absorption |
| Micronised oestradiol (Estrace) | 1–2 mg/day | Better absorbed than valerate |
| Oestradiol (Zumenon, Elleste Solo) | 1–2 mg/day | Standard oral preparations |
- Disadvantage: First-pass hepatic metabolism → stimulates liver protein synthesis → ↑ SHBG, ↑ clotting factors, ↑ CRP, ↑ triglycerides → higher VTE risk than transdermal
B. Transdermal Oestradiol — Patches
| Type | Examples | Dose | Notes |
|---|---|---|---|
| Matrix patch | Estradot, Evorel, FemSeven | 25–100 mcg/24h | Changed twice weekly or weekly |
| Reservoir patch | Estraderm | 25–100 mcg/24h | Older type; changed twice weekly |
- Advantage: Bypasses liver → no first-pass effect → lower VTE risk → preferred in women with VTE risk factors, obesity, migraines
- Available as oestrogen-only or combined patches:
- Evorel Sequi (oestradiol + sequential norethisterone) — cyclical
- Evorel Conti (oestradiol + continuous norethisterone) — continuous combined
- FemSeven Sequi — sequential patch
C. Transdermal Oestradiol — Gel
| Drug | Dose | Notes |
|---|---|---|
| Oestrogel (Besins) | 0.75 mg/actuation; 1–2 pumps/day | Applied to arm/thigh daily |
| Sandrena gel | 0.5–1.5 mg/day unit-dose sachets | Applied to thigh |
| Lenzetto | Transdermal spray (1.53 mg/spray) | 1–3 sprays/day on forearm |
- Same advantages as patches — avoids first-pass
- Patient controls dose more flexibly
- Must allow to dry; avoid skin contact with others
D. Subcutaneous Implants
| Drug | Notes |
|---|---|
| Oestradiol implants (25–100 mg pellets) | Inserted subcutaneously under LA; lasts 4–6 months; consistent levels; risk of tachyphylaxis (tolerance) with escalating doses; less commonly used now |
E. Vaginal Oestradiol (Local — Low Systemic Absorption)
| Drug | Type | Use |
|---|---|---|
| Vagifem / Vagirux (oestradiol 10 mcg tablet) | Vaginal tablet | Urogenital atrophy; minimal systemic absorption |
| Imvanesse (oestradiol 10 mcg) | Vaginal tablet | Same |
| Ovestin cream (oestriol) | Vaginal cream | Local use; oestriol (weakest oestrogen) |
| Estring | Vaginal ring (oestradiol 7.5 mcg/24h) | Changed every 3 months; local effect |
| Intrarosa (prasterone/DHEA) | Vaginal insert | Converted locally to androgens + oestrogens |
- Systemic absorption is very low at low doses → generally does not require progestogen cover
- Indicated for genitourinary syndrome of menopause (GSM): vaginal dryness, dyspareunia, recurrent UTIs
3. 🟢 Oestriol
- Weakest of the three natural oestrogens (oestrone, oestradiol, oestriol)
- Predominantly local/vaginal use
- Low systemic absorption → minimal endometrial stimulation
- Preparations: Ovestin cream/pessaries (oestriol 0.1%)
- Not suitable as systemic HRT — insufficient potency for menopausal symptom relief
4. 🔵 Oestrone
- Predominant oestrogen after menopause (produced by peripheral aromatisation of adrenal androgens in fat)
- Not commonly used as standalone HRT preparation
- Present in CEE as oestrone sulphate (main component)
- Oral oestrone sulphate (Harmogen) — used in some countries as systemic HRT
📊 Summary: HRT by Oestrogen Type
| Oestrogen | Route | Examples | VTE Risk | Best For |
|---|---|---|---|---|
| CEE | Oral | Premarin, Prempak-C | ↑ (first-pass) | Traditional; well-studied |
| Oestradiol | Oral | Progynova, Elleste, Zumenon | ↑ (first-pass) | Bioidentical; oral preferred |
| Oestradiol | Transdermal patch | Evorel, Estradot, FemSeven | ✅ Lowest | VTE risk; obesity; migraine |
| Oestradiol | Transdermal gel | Oestrogel, Sandrena, Lenzetto | ✅ Lowest | Flexible dosing; VTE risk |
| Oestradiol | Implant | Oestradiol pellet | Low | Compliance issues; surgical |
| Oestradiol | Vaginal (local) | Vagifem, Estring | Minimal | GSM only; no systemic effect |
| Oestriol | Vaginal (local) | Ovestin cream/pessary | Minimal | GSM; weakest oestrogen |
| Oestrone | Oral | Harmogen | ↑ | Limited use |
🔑 Key Clinical Points
| Point | Detail |
|---|---|
| Cyclical HRT = perimenopause | Still having periods → cyclical; postmenopause → continuous combined |
| Transdermal preferred | Lower VTE, stroke, liver effects vs. oral — especially in women with risk factors |
| Progestogen always needed | In women with a uterus — any oestrogen route requires progestogen cover (except low-dose local vaginal) |
| Hysterectomised women | Oestrogen alone — no progestogen needed |
| Bioidentical | Oestradiol (17β) is structurally identical to endogenous oestrogen — preferred modern choice |
| CEE | Most extensively studied (WHI trial); slightly higher risk profile vs. transdermal oestradiol |
Mechanism of action of insulin breifly
Mechanism of Action of Insulin
🔵 Step 1 — Insulin Secretion
- Insulin is a peptide hormone produced by β-cells of the islets of Langerhans in the pancreas
- Secreted in response to ↑ blood glucose (primary stimulus), amino acids, GLP-1, GIP, vagal stimulation
- Released as preproinsulin → cleaved to proinsulin → insulin + C-peptide (secreted in equimolar amounts)
🔵 Step 2 — Receptor Binding
-
Insulin binds to the insulin receptor (IR) — a transmembrane glycoprotein consisting of:
- 2 α-subunits (extracellular — contain insulin binding site)
- 2 β-subunits (transmembrane + intracellular — contain tyrosine kinase domain)
- Linked by disulphide bonds → α₂β₂ heterotetramer
-
Insulin binds to the α-subunits → conformational change → activates intrinsic tyrosine kinase activity on the β-subunits
🔵 Step 3 — Intracellular Signalling Cascade
Insulin binds α-subunit
↓
β-subunit tyrosine kinase activated
↓
Autophosphorylation of β-subunit tyrosine residues
↓
Phosphorylation of IRS-1/IRS-2
(Insulin Receptor Substrates)
↓
┌─────┴──────┐
│ │
PI3K MAPK pathway
pathway (Ras → Raf → MEK → ERK)
│ │
↓ ↓
Akt/PKB Gene transcription
activated Cell growth, proliferation
│
↓
Multiple metabolic effects
🔵 Step 4 — Key Metabolic Effects
A. Glucose Uptake (PI3K → Akt → GLUT4)
- Akt phosphorylation → GLUT4 vesicles translocate to cell membrane
- GLUT4 (Glucose Transporter 4) is the insulin-sensitive transporter in:
- Skeletal muscle (~80% of glucose disposal)
- Adipose tissue
- Glucose enters cells via facilitated diffusion down concentration gradient
- GLUT2 (liver, pancreas) and GLUT1 (brain, RBCs) are insulin-independent
B. Liver Effects (Hepatic)
| Action | Effect |
|---|---|
| ↑ Glycogenesis | Activates glycogen synthase → glucose → glycogen storage |
| ↓ Glycogenolysis | Inhibits glycogen phosphorylase → ↓ glycogen breakdown |
| ↓ Gluconeogenesis | Inhibits PEPCK and glucose-6-phosphatase → ↓ new glucose production |
| ↑ Glycolysis | ↑ glucokinase activity → ↑ glucose oxidation |
| ↑ Lipogenesis | ↑ acetyl-CoA carboxylase → ↑ fatty acid synthesis |
| ↓ Ketogenesis | Inhibits β-oxidation of fatty acids |
C. Muscle Effects
| Action | Effect |
|---|---|
| ↑ GLUT4 → ↑ glucose uptake | Primary glucose disposal site |
| ↑ Glycogenesis | Stores glucose as muscle glycogen |
| ↑ Protein synthesis | Stimulates ribosomal S6 kinase → anabolic |
| ↓ Proteolysis | Anti-catabolic |
D. Adipose Tissue Effects
| Action | Effect |
|---|---|
| ↑ GLUT4 → ↑ glucose uptake | |
| ↑ Lipogenesis | Promotes triglyceride storage |
| ↓ Lipolysis | Inhibits hormone-sensitive lipase → ↓ FFA release → ↓ substrate for gluconeogenesis and ketogenesis |
🔵 Step 5 — Net Effect
↑ Blood Glucose
│
↓ Insulin released
│
├─ Liver: ↓ glucose output (↓ gluconeogenesis, ↓ glycogenolysis)
├─ Muscle: ↑ glucose uptake + glycogen synthesis
├─ Fat: ↑ glucose uptake + lipogenesis; ↓ lipolysis
│
↓
↓ Blood Glucose → back to normal (5.0 mmol/L)
📊 Summary
| Level | Key Event |
|---|---|
| Receptor | Binds α-subunit → tyrosine kinase activation |
| Signal | IRS-1/2 → PI3K → Akt (metabolic) + MAPK (growth) |
| Glucose transport | GLUT4 translocation to membrane → ↑ glucose uptake |
| Liver | ↓ Gluconeogenesis, ↓ glycogenolysis, ↑ glycogenesis, ↑ lipogenesis |
| Muscle | ↑ Glucose uptake, ↑ glycogenesis, ↑ protein synthesis |
| Fat | ↑ Lipogenesis, ↓ lipolysis, ↓ FFA release |
| Net result | ↓ Blood glucose; anabolic state |
Insulin is the body's primary anabolic hormone — it drives glucose, amino acids, and fatty acids into cells for storage and use, while simultaneously suppressing all pathways that would raise blood glucose.
Assisted vaginal delivery indications and contra indications
Assisted Vaginal Delivery (Operative Vaginal Delivery) — Indications & Contraindications
📌 Definition
- Forceps — metal instruments applied to fetal head to provide traction ± rotation
- Ventouse (Vacuum extractor) — suction cup applied to fetal head to assist maternal pushing
✅ INDICATIONS
🔴 Fetal Indications
| Indication | Detail |
|---|---|
| Fetal distress / non-reassuring CTG | Most common indication; prolonged decelerations, bradycardia, pathological CTG in second stage → expedite delivery |
| Cord prolapse in second stage | Head at/near outlet → quickest to deliver vaginally |
| Prolonged second stage | Nullipara: >2 hours active pushing without epidural (>3 hours with epidural); Multipara: >1 hour active pushing (>2 hours with epidural) — descent arrested or inadequate |
🟡 Maternal Indications
| Indication | Detail |
|---|---|
| Maternal exhaustion | Unable to push effectively despite adequate contractions |
| Inadequate maternal effort | Epidural analgesia blunting urge to push; reduced expulsive effort |
| Maternal cardiac disease | Avoid prolonged expulsive effort (Valsalva ↑ cardiac workload) — e.g. severe valvular disease, aortic aneurysm, heart failure |
| Maternal respiratory disease | Severe asthma, pulmonary hypertension — limit active pushing |
| Hypertensive crisis | Pre-eclampsia/eclampsia — shorten second stage to prevent BP surges |
| Cerebrovascular disease | Avoid straining (risk of haemorrhage/re-bleed) — e.g. intracranial aneurysm, AVM, recent stroke |
| Neuromuscular disease | Myasthenia gravis, spinal cord injury — inadequate pushing ability |
| Maternal diabetes with macrosomia | Expedite delivery to reduce shoulder dystocia risk |
| Previous eye surgery / raised ICP | Avoid Valsalva |
📋 PREREQUISITES (Conditions That Must Be Met Before Attempting)
| Prerequisite | Detail |
|---|---|
| Cervix fully dilated (10 cm) | No cervix palpable |
| Membranes ruptured | Intact membranes → must rupture first |
| Head engaged | Head must be engaged; not high |
| Station | Head at ≥ +2 station (outlet) or at spines (0 station) for mid-cavity; head at outlet (+3 to +5) for straightforward cases |
| Position known | Exact position of occiput must be determined (OA, OL, OP, etc.) |
| Adequate pelvis | Clinical pelvimetry — no absolute cephalopelvic disproportion |
| Adequate analgesia | Epidural (ideal); pudendal block + local infiltration for outlet |
| Empty bladder | Catheterise before procedure — reduces trauma and improves access |
| Informed consent | Verbal consent obtained; risks explained |
| Operator competent | Trained operator; capable of recognising failure and converting to CS |
| Theatre available | In case of failed instrumental delivery → immediate CS |
| Neonatal team available | For resuscitation if needed |
| Episiotomy considered | Should be performed if needed to facilitate delivery |
❌ CONTRAINDICATIONS
Absolute Contraindications (Both Forceps and Ventouse)
| Contraindication | Reason |
|---|---|
| Unengaged head (above the brim) | High risk of failure, uterine rupture, trauma |
| Incomplete cervical dilation | Cervical laceration; failed delivery |
| Cephalopelvic disproportion (CPD) | Head too large for pelvis → CS only |
| Malpresentation (brow, face with mentoposterior) | Forceps unsafe; ventouse unsafe in face presentation |
| Unknown fetal position | Cannot apply instruments correctly → trauma |
| Fetal coagulation disorder / bleeding tendency | e.g. haemophilia, thrombocytopenia → ventouse especially increases intracranial haemorrhage risk |
| Dead fetus | Destructive operations used instead |
| Non-vertex presentation (except after-coming head in breech) | Forceps for after-coming head of breech is an exception |
Relative Contraindications / Cautions
| Contraindication | Relevant Instrument | Detail |
|---|---|---|
| Preterm < 34 weeks | Ventouse contraindicated | Immature skull → high risk of cephalhaematoma, subgaleal haemorrhage, intracranial haemorrhage |
| Preterm 34–36 weeks | Ventouse — caution | Increased risk vs. term; forceps preferable |
| Face presentation | Ventouse contraindicated | Cannot achieve correct cup placement; forceps (mentoanterior only) possible |
| Brow presentation | Both contraindicated | Cannot deliver vaginally — CS |
| After-coming head of breech | Forceps (Burns-Marshall / Lovset / Piper) | Ventouse not appropriate |
| Fetal macrosomia | Both — caution | Increased shoulder dystocia risk post-delivery |
| Suspected fetal coagulopathy | Ventouse — avoid | Forceps preferred if must assist |
| Failed ventouse | Proceed cautiously with forceps or CS | Sequential use ↑ neonatal trauma risk — use only if good prospects of success |
| Deflexed head / asynclitism | Ventouse — caution | Proper cup placement difficult |
| Previous uterine scar | Both — caution | Expulsive traction may risk scar dehiscence in obstructed cases |
⚖️ Forceps vs. Ventouse — Key Differences
| Feature | Forceps | Ventouse |
|---|---|---|
| Mechanism | Mechanical grip + traction ± rotation | Suction cup + maternal pushing |
| Rotation | Can actively rotate (Kielland's forceps) | Passive rotation with traction |
| Analgesia required | ✅ More — usually epidural/pudendal block | Less — pudendal block often sufficient |
| Maternal trauma | ↑ Perineal/vaginal tears; episiotomy often needed | Less maternal trauma |
| Neonatal scalp | Facial/scalp marks (temporary) | Chignon (caput); cephalhaematoma; subgaleal haemorrhage |
| Intracranial haemorrhage | Lower risk than ventouse | Slightly higher risk |
| Preterm | ✅ Preferred (<34 weeks) | ❌ Contraindicated |
| Face presentation | ✅ Possible (mentoanterior) | ❌ Contraindicated |
| Failed ventouse | May attempt forceps (with caution) | — |
| Operator skill | Higher skill required | Easier to learn |
| Success rate | Higher (especially for rotation) | Slightly lower; higher failure rate |
🗺️ Classification of Assisted Vaginal Delivery by Station
| Classification | Station | Position | Notes |
|---|---|---|---|
| Outlet | +3 to +5; visible at introitus | OA or slight rotation <45° | Lowest risk; straightforward |
| Low cavity | +2 and below | Any position | Common; moderate skill |
| Mid cavity | Head at spines (0) to +1 | Any position | Higher skill; Kielland's forceps for rotation; CS if doubt |
| High cavity | Above spines | — | Not attempted — proceed to CS |
⚠️ When to Abandon and Proceed to Caesarean Section
| Situation | Action |
|---|---|
| No descent with 3 pulls (ventouse) | Abandon → CS |
| Cup detaches twice (ventouse) | Abandon → CS |
| No rotation/descent with forceps | Abandon → CS |
| Persistent fetal bradycardia despite delivery attempt | Expedite → if not imminent → CS |
| Operator loses confidence | Abandon → CS |
| Total ventouse pull time >15–20 minutes | Abandon → CS |
📊 Summary
| Indications | Contraindications | |
|---|---|---|
| Fetal | Distress, cord prolapse, prolonged 2nd stage | Coagulopathy, preterm <34 wks (ventouse), unengaged head |
| Maternal | Exhaustion, cardiac/respiratory/neurological disease, hypertensive crisis | CPD, incomplete dilation, unknown position |
| Prerequisites | Fully dilated, ruptured membranes, engaged, position known, empty bladder, consent, theatre available | If any prerequisite not met → do not proceed |
| Ventouse-specific CI | — | Preterm <34 wks, face presentation, fetal coagulopathy |
| Forceps-specific advantage | — | Preterm, face presentation (mentoanterior), rotation needed |
Managment of miscarriage based on Gestational sac and foetal heart rate
Management of Miscarriage Based on Gestational Sac & Fetal Heart Rate
🔑 Diagnostic Framework
📐 USS Diagnostic Criteria (RCOG / NICE Guidelines)
| USS Finding | Diagnostic Threshold | Diagnosis |
|---|---|---|
| CRL ≥7 mm + no fetal heartbeat | Single scan | Embryonic/fetal demise confirmed |
| MSD ≥25 mm + no fetal pole | Single scan | Anembryonic pregnancy confirmed |
| CRL 5–6 mm + no FCA | Repeat scan in 7 days | Uncertain — rescan required |
| MSD 16–24 mm + no fetal pole | Repeat scan in 7 days | Uncertain — rescan required |
| CRL <5 mm + no FCA | Repeat scan in 7–14 days | Too early — rescan required |
| No gestational sac + β-hCG >1500–2000 | — | Suspect ectopic |
Key principle: When findings are below the diagnostic threshold, a single scan is insufficient — a repeat scan at 7–14 days is mandatory to avoid misdiagnosis and inadvertent termination of a viable pregnancy.
🗂️ Classification of Miscarriage + USS Findings + Management
1. 🟢 Threatened Miscarriage
| Feature | Detail |
|---|---|
| USS | Gestational sac present + fetal pole + FETAL HEARTBEAT PRESENT |
| Cervical os | Closed |
| Symptoms | Vaginal bleeding ± mild cramps; no tissue passed |
| Viability | Pregnancy potentially viable |
- Reassurance — majority (>85%) continue to viable pregnancy if FHR seen
- Pelvic rest — avoid intercourse, heavy activity
- Repeat USS in 1–2 weeks to confirm ongoing viability
- Progesterone — vaginal micronised progesterone (400 mg BD) if history of previous miscarriage AND bleeding in current pregnancy (PRISM trial evidence) — gives modest benefit in this subgroup
- Anti-D if Rh-negative (if gestation ≥12 weeks or heavy bleeding <12 weeks)
- No role for bedrest (no evidence)
2. 🟡 Missed Miscarriage (Silent / Delayed Miscarriage)
| Feature | Detail |
|---|---|
| USS | CRL ≥7 mm + NO fetal heartbeat (confirmed on single scan) OR MSD ≥25 mm + no fetal pole |
| Cervical os | Closed |
| Symptoms | No bleeding/pain; symptoms of pregnancy may have regressed; incidental finding |
| CRL 5–6 mm / MSD 16–24 mm | Repeat scan 7–14 days before confirming diagnosis |
A. Expectant Management
- Await natural passage of pregnancy
- Success rate: ~50–80% within 2 weeks (higher for incomplete, lower for missed)
- Monitor with serial β-hCG + USS
- Advise patient what to expect (bleeding, cramping, tissue passage)
- Suitable if: haemodynamically stable, no infection, patient accepts uncertainty and delay
- Follow-up USS at 2 weeks to confirm completion
B. Medical Management (Misoprostol)
- Misoprostol (prostaglandin E1 analogue) — 800 mcg vaginally (or sublingually)
- Causes uterine contractions → expulsion of products
- Success rate: ~80–85% within 2 weeks
- May repeat dose at 48 hours if incomplete
- Suitable if: first trimester, stable, no contraindications
- Side effects: cramping, bleeding, nausea, diarrhoea, fever
- Mifepristone 200 mg orally 24–48 hours before misoprostol improves efficacy (Swanson's, p. 434)
- Anti-D if Rh-negative
C. Surgical Management (Uterine Evacuation)
- Manual Vacuum Aspiration (MVA) — outpatient; local anaesthesia; 60 mL hand-held syringe; safe and effective; preferred in first trimester
- Electric Vacuum Aspiration (EVA) — theatre setting; general or local anaesthesia
- Surgical Evacuation of Retained Products of Conception (ERPC/ERPOC) — suction curettage under GA
- Success rate: >95%
- Preferred when:
- Failed expectant or medical management
- Patient preference for certainty
- Heavy bleeding / haemodynamic instability
- Infection (septic miscarriage)
- Gestational age >12 weeks (Swanson's, p. 434)
3. 🔴 Inevitable Miscarriage
| Feature | Detail |
|---|---|
| USS | Gestational sac may be present; fetal pole with no heartbeat OR sac low in uterus/in cervical canal |
| Cervical os | OPEN |
| Symptoms | Active bleeding + cramping; no tissue passed yet |
- Medical — misoprostol 800 mcg vaginally (higher success than in missed miscarriage) (Swanson's, p. 434)
- Surgical — MVA / EVA if bleeding heavy or patient preference
- Expectant — may complete naturally; closely monitor
- Anti-D if Rh-negative
4. 🟠 Incomplete Miscarriage
| Feature | Detail |
|---|---|
| USS | Heterogeneous intrauterine contents / echogenic material >15 mm in cavity (retained products); no fetal parts identifiable; no gestational sac |
| Cervical os | OPEN |
| Symptoms | Continued bleeding + cramping; some tissue already passed |
- Expectant — if clinically stable, bleeding not heavy, <50 mm of retained tissue → may complete naturally (48–72 hours)
- Medical — misoprostol 800 mcg vaginally — highest success rate of all types in this category (Swanson's, p. 434)
- Surgical — MVA/EVA preferred if:
- Heavy bleeding / haemodynamic instability
- Signs of infection
- Failed medical/expectant
- Retained tissue >50 mm on USS
5. 🟢 Complete Miscarriage
| Feature | Detail |
|---|---|
| USS | Empty uterus — endometrial thickness <15 mm; no retained products; no gestational sac |
| Cervical os | Closed (or recently closed) |
| Symptoms | Bleeding settling; cramping resolving; tissue passed |
| β-hCG | Falling to zero |
- No intervention required
- Confirm by USS — empty uterus
- Serial β-hCG to confirm falling to undetectable
- Reassurance + emotional support
- Anti-D if Rh-negative (if heavy bleeding or <12 weeks with significant haemorrhage)
- Advise regarding next conception planning
6. ⚫ Septic Miscarriage
| Feature | Detail |
|---|---|
| USS | Retained products ± gas in uterus |
| Cervical os | Usually open |
| Symptoms | Fever, offensive discharge, uterine tenderness, systemic sepsis signs |
- IV antibiotics immediately (broad spectrum — co-amoxiclav + metronidazole ± gentamicin)
- IV fluids + resuscitation
- Urgent surgical evacuation — do not delay surgery for antibiotics to work
- Blood cultures, FBC, CRP, U&E, coagulation before surgery
- HDU/ITU if septic shock
- Anti-D if Rh-negative
📊 USS Findings → Management Summary Table
| USS Finding | FHR | Diagnosis | Management |
|---|---|---|---|
| CRL ≥7 mm, no FCA (confirmed) | ❌ Absent | Missed miscarriage | Expectant / Medical (misoprostol ± mifepristone) / Surgical (MVA) |
| MSD ≥25 mm, no fetal pole | ❌ Absent | Anembryonic (blighted ovum) | Expectant / Medical / Surgical |
| CRL 5–6 mm, no FCA | ❌ Absent | Uncertain | Repeat USS in 7 days |
| MSD 16–24 mm, no fetal pole | ❌ Absent | Uncertain | Repeat USS in 7 days |
| CRL <5 mm, no FCA | ❌ Absent | Too early | Repeat USS in 7–14 days |
| Fetal pole + FCA present | ✅ Present | Threatened miscarriage | Expectant; progesterone if previous loss; rescan |
| Retained echogenic material >15 mm | N/A | Incomplete miscarriage | Misoprostol (best success) or MVA |
| Empty uterus, settling symptoms | N/A | Complete miscarriage | Confirm; no intervention; β-hCG follow-up |
| Retained products + fever/sepsis | N/A | Septic miscarriage | IV antibiotics + urgent surgical evacuation |
🗺️ Decision Algorithm
Suspected Miscarriage
│
├─ TVUSS
│ │
│ ├─ FHR PRESENT + sac/pole present
│ │ └─ THREATENED → Reassure; progesterone if prev loss; rescan
│ │
│ ├─ FHR ABSENT
│ │ │
│ │ ├─ CRL ≥7 mm OR MSD ≥25 mm → CONFIRMED non-viable
│ │ │ └─ Counsel re 3 options:
│ │ │ 1. Expectant (wait)
│ │ │ 2. Medical (misoprostol ± mifepristone)
│ │ │ 3. Surgical (MVA / EVA)
│ │ │
│ │ └─ Below threshold → UNCERTAIN → Repeat scan 7–14 days
│ │
│ ├─ OS OPEN + bleeding → INEVITABLE / INCOMPLETE
│ │ └─ Medical (misoprostol — best success) or Surgical
│ │
│ ├─ EMPTY UTERUS + settling → COMPLETE
│ │ └─ Confirm; no intervention; serial β-hCG
│ │
│ └─ RETAINED + FEVER → SEPTIC
│ └─ IV antibiotics URGENT + Surgical evacuation
│
└─ ALL: Anti-D if Rh-negative
Emotional support + counselling
Contraception / preconception planning
⚠️ Additional Management Points
| Issue | Action |
|---|---|
| Anti-D immunoglobulin | All Rh-negative women: 250 IU IM if <12 weeks (if surgical or medical management, or heavy bleeding); 500 IU if ≥12 weeks |
| β-hCG follow-up | Confirm return to zero — especially after medical/expectant management to exclude persistent trophoblastic disease |
| Failed medical management | Proceed to surgical evacuation |
| Recurrent miscarriage (≥3 losses) | Investigate: karyotype (both partners), antiphospholipid antibodies, thrombophilia screen, uterine anatomy (HSG/hysteroscopy), thyroid function |
| Emotional support | Miscarriage is a bereavement — acknowledge loss; refer to support groups; screen for depression/anxiety |
| Next pregnancy timing | Physically: can try after next period; emotionally: when ready |
Amniotic fluid index
Amniotic Fluid Index (AFI)
📌 Definition
"The AFI is an assessment of amniotic fluid that should include the cumulative measurement of 4 pockets of fluid. Normal AFI is 8–18; oligohydramnios is <5–6; and polyhydramnios is 20–24." (Campbell-Walsh Urology, p. 755)
🔬 How AFI is Measured
Technique:
- Patient lies supine
- Uterus divided into 4 quadrants by the umbilicus (horizontal) and linea nigra (vertical)
- Transducer held perpendicular to the floor (not to the uterine wall)
- Largest vertical pocket of fluid measured in each quadrant — free of umbilical cord and fetal parts
- Four measurements added together → AFI in centimetres (cm)
Umbilicus
┌────────┬────────┐
│ Q1 │ Q2 │ ← Upper quadrants
├────────┼────────┤
│ Q3 │ Q4 │ ← Lower quadrants
└────────┴────────┘
Linea Nigra
AFI = Q1 + Q2 + Q3 + Q4 (cm)
📊 Normal AFI Values
| Interpretation | AFI Value |
|---|---|
| Normal | 8–18 cm |
| Borderline | 5–8 cm |
| Oligohydramnios | < 5 cm (some use <6 cm) |
| Polyhydramnios | > 20–24 cm (some use >25 cm) |
AFI by Gestational Age (approximate normal ranges):
| Gestational Age | Mean AFI | 5th–95th Percentile |
|---|---|---|
| 16 weeks | 12.1 cm | 7.9–15.4 cm |
| 20 weeks | 14.1 cm | 8.6–18.6 cm |
| 24 weeks | 14.7 cm | 8.9–19.9 cm |
| 28 weeks | 14.6 cm | 7.7–21.6 cm |
| 32 weeks | 13.9 cm | 7.7–21.4 cm |
| 36 weeks | 13.2 cm | 6.8–20.7 cm |
| 40 weeks | 10.5 cm | 5.0–18.5 cm |
| 42 weeks | 8.1 cm | 3.7–14.8 cm |
AFI peaks at 32–34 weeks (~14–16 cm) then gradually declines toward term.
🔄 Alternative Method: Single Deepest Pocket (SDP)
| Method | Description | Normal | Oligohydramnios | Polyhydramnios |
|---|---|---|---|---|
| AFI | Sum of 4 quadrant measurements | 8–18 cm | <5 cm | >24 cm |
| Single Deepest Pocket (SDP) | Deepest single vertical pocket free of cord/fetal parts | 2–8 cm | <2 cm | >8 cm |
- RCOG/NICE now recommend SDP over AFI for surveillance in high-risk pregnancies (growth restriction, post-dates)
- SDP has lower false-positive rate for oligohydramnios → fewer unnecessary interventions
- AFI tends to overdiagnose oligohydramnios compared to SDP
💧 OLIGOHYDRAMNIOS (AFI < 5 cm / SDP < 2 cm)
Causes
Fetal Causes:
| Cause | Detail |
|---|---|
| Renal agenesis (Potter sequence) | No fetal urine production → severe oligohydramnios → pulmonary hypoplasia, limb deformities |
| Bilateral renal dysplasia/cystic kidneys | Reduced urine output |
| Posterior urethral valves (PUV) | Bladder outlet obstruction → back pressure → oligohydramnios |
| Urethral atresia | Complete obstruction |
| IUGR / placental insufficiency | Fetal hypoxia → ↓ renal perfusion → ↓ urine output |
| Post-term pregnancy (≥42 weeks) | Placental senescence → ↓ fluid |
| Chromosomal anomalies | Trisomy 18, triploidy |
Maternal / Placental Causes:
| Cause | Detail |
|---|---|
| PPROM (Preterm Prelabour Rupture of Membranes) | Most common cause of sudden oligohydramnios; fluid leaking |
| Pre-eclampsia / hypertension | Uteroplacental insufficiency → ↓ fetal renal perfusion |
| Diabetes (poorly controlled) | Vascular placental disease |
| NSAIDs | Cause ductus arteriosus constriction + ↓ fetal urine output (prostaglandin-mediated renal vasoconstriction) |
| ACE inhibitors / ARBs | Fetal renal tubular dysgenesis |
| Antiphospholipid syndrome | Placental insufficiency |
| Dehydration (maternal) | Mild, reversible |
Complications of Oligohydramnios
| Complication | Mechanism |
|---|---|
| Pulmonary hypoplasia | Inadequate fluid for lung development (fluid essential for airway distension) |
| Potter sequence | Oligohydramnios → characteristic facies, limb positioning defects, pulmonary hypoplasia |
| Cord compression | Reduced fluid cushion → cord accidents, variable decelerations |
| Fetal growth restriction | Often co-existing |
| Meconium aspiration | Concentrated meconium in reduced fluid |
| Fetal distress in labour | Cord compression → CTG abnormalities |
| Caesarean section rate | ↑ significantly |
Management of Oligohydramnios
| Severity | Gestation | Management |
|---|---|---|
| Mild (AFI 5–8 / borderline) | Any | Increase maternal hydration; repeat USS in 1 week; fetal monitoring (CTG, Doppler) |
| Moderate (AFI 3–5) | <37 weeks | Investigate cause; serial USS; CTG; umbilical artery Doppler; consider admission |
| Severe (AFI <2) | Any | Urgent assessment; consider delivery if ≥34 weeks |
| AFI <5 | ≥37 weeks | Induce labour / deliver |
| PPROM suspected | Any | Confirm (speculum, PAMG-1/IGFBP-1 swab); manage per PPROM protocol |
| Renal agenesis | Any gestation | Counsel — lethal condition; palliative care planning |
| Amnioinfusion (intrapartum) | Labour | Instil saline via intrauterine catheter → reduces cord compression, variable decelerations → ↓ CS rate |
🌊 POLYHYDRAMNIOS (AFI > 24–25 cm / SDP > 8 cm)
Grading:
| Grade | AFI | SDP |
|---|---|---|
| Mild | 25–29.9 cm | 8–9.9 cm |
| Moderate | 30–34.9 cm | 10–11.9 cm |
| Severe | ≥35 cm | ≥12 cm |
Causes
Fetal Causes (impaired swallowing / ↑ fluid production):
| Cause | Detail |
|---|---|
| Oesophageal atresia | Cannot swallow amniotic fluid → accumulates |
| Duodenal atresia ("double bubble") | Upper GI obstruction |
| Bowel obstruction (jejunal/ileal atresia) | Impaired gut absorption |
| CNS anomalies (anencephaly, holoprosencephaly) | Impaired swallowing reflex |
| Facial/cleft palate | Cannot swallow |
| Neuromuscular disorders | Floppy fetus → impaired swallowing |
| Hydrops fetalis (immune / non-immune) | ↑ fluid transudation |
| Twin-to-twin transfusion syndrome (TTTS) | Recipient twin — polyhydramnios |
| Sacrococcygeal teratoma | Large tumour → ↑ fluid transudation |
| Chromosomal anomalies | Trisomy 18, 21 |
Maternal Causes:
| Cause | Detail |
|---|---|
| Gestational Diabetes / Pre-existing DM | Most common identifiable cause; fetal hyperglycaemia → fetal polyuria |
| Idiopathic | ~50–60% of cases; no cause found |
Complications of Polyhydramnios
| Complication | Mechanism |
|---|---|
| Preterm labour | Uterine overdistension → contractions |
| Preterm PROM | Membrane stretch |
| Malpresentation | Excess fluid → unstable lie, breech |
| Cord prolapse | Unstable lie + SROM → cord first |
| Placental abruption | Rapid decompression of overdistended uterus at SROM |
| Postpartum haemorrhage | Uterine atony from overdistension |
| Maternal respiratory compromise | Diaphragmatic elevation |
| Maternal discomfort | Abdominal pain, dyspnoea |
Management of Polyhydramnios
| Step | Action |
|---|---|
| Investigate cause | Detailed anomaly scan; fetal echocardiogram; OGTT (gestational diabetes); TORCH serology; fetal karyotype if anomaly detected |
| Mild/asymptomatic | Serial USS monitoring; treat underlying cause (e.g. optimise diabetes control) |
| Moderate/severe + symptomatic | Amnioreduction (therapeutic amniocentesis) — drain fluid to relieve maternal symptoms and reduce preterm labour risk |
| Indomethacin | ↓ Fetal urine output (prostaglandin inhibition); effective but limited to <32 weeks (risk of premature ductus arteriosus closure) |
| TTTS | Fetoscopic laser photocoagulation of communicating vessels |
| Delivery timing | Individualised; severe polyhydramnios ≥37 weeks → consider delivery |
| At delivery | Controlled rupture of membranes to prevent cord prolapse; PPH precautions |
📊 Summary Table
| Parameter | Oligohydramnios | Normal | Polyhydramnios |
|---|---|---|---|
| AFI | < 5 cm | 8–18 cm | > 24–25 cm |
| SDP | < 2 cm | 2–8 cm | > 8 cm |
| Common causes | PPROM, renal anomalies, IUGR, post-term | — | Diabetes, GI obstruction, idiopathic |
| Fetal risk | Cord compression, pulmonary hypoplasia | — | Preterm, malpresentation, cord prolapse |
| Surveillance | Serial USS + CTG + Doppler | Routine | Serial USS + anomaly scan |
| Intervention | Amnioinfusion (intrapartum); deliver if ≥37 wks | — | Amnioreduction; indomethacin; laser (TTTS) |
🔑 Key Clinical Points
| Point | Detail |
|---|---|
| AFI peaks at 32–34 weeks then declines toward term | Normal physiological decline |
| SDP preferred over AFI in high-risk surveillance | Lower false-positive rate |
| Isolated oligohydramnios at term | Strong indication to deliver — high risk of cord compromise in labour |
| Sudden oligohydramnios at any gestation | → Suspect PPROM first; confirm with speculum + swab |
| NSAIDs cause reversible oligohydramnios | Stop if used >48 hours or after 32 weeks |
| Polyhydramnios + normal anomaly scan + normal OGTT | = Idiopathic (~50–60%); monitor with serial USS |
| Double bubble on scan + polyhydramnios | = Duodenal atresia until proven otherwise |
Breech extraction procedure in steps
Breech Extraction — Step-by-Step Procedure
📌 Definition & Distinction
| Term | Definition |
|---|---|
| Spontaneous breech delivery | Entire baby delivered by maternal effort alone without traction |
| Assisted breech delivery | Baby delivers spontaneously to umbilicus; operator assists delivery of shoulders, arms, and head |
| Total breech extraction | Operator actively extracts the entire baby from the beginning, including pulling the legs/buttocks down — NOT used in singleton vaginal breech except emergencies; primary use = second twin in transverse lie |
Assisted breech delivery is the standard for planned vaginal breech birth. Total breech extraction is reserved for emergencies (e.g. fetal distress in second stage, delivery of second twin).
✅ Prerequisites Before Breech Delivery
| Requirement | Detail |
|---|---|
| Fully dilated cervix | 10 cm — complete |
| Ruptured membranes | Intact membranes must be ruptured |
| Engaged breech | At or below ischial spines |
| Adequate pelvis | No CPD |
| Frank or complete breech | Footling breech — higher risk (cord prolapse) |
| EFW <3.8–4 kg | Macrosomia → CS preferred |
| Head flexed | Hyperextended head → CS |
| Experienced operator | Essential — trained accoucheur |
| Anaesthesia | Epidural preferred; general anaesthesia available |
| Theatre ready | Immediate CS capability |
| Neonatal team present | For resuscitation |
| IV access + consent | Ready |
🔢 Step-by-Step: Assisted Breech Delivery
🔵 PHASE 1 — Delivery of the Buttocks and Lower Body
Step 1 — Crowning of the Breech
- Patient in lithotomy position
- Allow breech to descend passively with maternal pushing — do not pull
- Buttocks appear at vulva and distend the perineum
- Perform generous episiotomy (mediolateral) as buttocks crown — prevents perineal obstruction to delivery of shoulders and head
- The sacrum should rotate anteriorly (sacroanterior position ideal)
Step 2 — Delivery of the Buttocks
- Continue allowing descent with maternal effort
- Grasp baby over iliac crests (not abdomen/flanks — risk of renal/splenic injury)
- Hold with thumbs on sacrum, fingers over iliac crests
- Apply gentle downward traction with contractions
- Do not pull on legs or abdomen
Step 3 — Delivery of the Legs (Frank Breech → Extended Legs)
- In frank breech (extended legs alongside trunk) — legs deliver spontaneously or:
- Pinard manoeuvre — insert two fingers along fetal thigh to the popliteal fossa → apply pressure to flex knee → foot brought down
- In complete/footling breech — legs already flexed; deliver feet first then legs
Step 4 — Loop the Cord
- Once umbilicus is delivered → loop a loose loop of umbilical cord downward to prevent traction on cord
- Check cord — pulsating (reassuring); pale/compressed → expedite delivery
- Delivery of umbilicus → 3–5 minutes maximum to deliver the head before hypoxia
🔵 PHASE 2 — Delivery of the Shoulders and Arms
Step 5 — Allow Trunk to Deliver to Scapulae
- Continue gentle downward traction as maternal pushing proceeds
- Allow baby to hang momentarily — weight helps flex the head and bring it into the pelvis
- Rotate trunk so back remains anterior (or lateral) — sacrum should stay anterior
Step 6 — Delivery of Arms (Normal Position — Arms Flexed)
- If arms flexed across chest → deliver spontaneously with gentle downward traction
- Sweep arms out one at a time by:
- Splint the humerus between two fingers
- Sweep arm downward and across the face (wash face manoeuvre)
- Deliver anterior arm first, then posterior
Step 7 — Extended/Nuchal Arms (Complication → Løvset's Manoeuvre)
- Hold baby at pelvis (over iliac crests / thighs)
- Rotate baby 180° (half turn) keeping back uppermost — the arm that was posterior is now brought under the pubic symphysis anteriorly where there is more space
- Deliver that arm from under the symphysis by sweeping across the face
- Rotate 180° back in opposite direction
- Deliver second arm the same way
- Principle: rotating 180° brings each arm to the anterior where it can be swept free
🔵 PHASE 3 — Delivery of the Aftercoming Head
Step 8 — Engage the Head
Option A: Mauriceau-Smellie-Veit (MSV) Manoeuvre ← Most Common
- Place baby astride your forearm (face down, body along your arm)
- Insert index and middle finger of the same hand into the baby's mouth / onto the malar prominences of the cheeks (NOT into the mouth — jaw fracture risk; place on cheekbones)
- With the other hand, place index and middle fingers on the occiput (to flex the head) — "forks" on either side of neck over shoulders
- Apply suprapubic pressure by an assistant — to maintain head flexion
- Traction: apply downward and outward traction following the curve of the pelvis (curve of Carus)
- As the occiput appears under the symphysis → sweep the baby upward in an arc → deliver the face, brow, vertex
- Control the speed — slow, controlled delivery to prevent sudden decompression and intracranial haemorrhage
Operator's left hand:
→ Baby's body straddles forearm
→ Index + middle finger on cheekbones/malar area
→ Flex head
Operator's right hand:
→ Index + middle finger on occiput/neck (fork-like)
→ Apply traction
Episiotomy indications and Contraindications
Episiotomy — Indications & Contraindications
📌 Definition
Modern consensus: Episiotomy should be selective (only when clinically indicated) — NOT routine. Evidence shows routine episiotomy does NOT reduce severe perineal trauma and increases morbidity.
✂️ Types of Episiotomy
| Type | Direction | Angle | Advantages | Disadvantages |
|---|---|---|---|---|
| Mediolateral (most common — UK/Europe) | From midpoint of fourchette toward ischiorectal fossa (right or left) | 45–60° from midline | Lower risk of extension to anal sphincter/rectum | More painful; more blood loss; harder to repair |
| Median / Midline (common — USA) | Directly toward anus along midline | 0° | Easy to repair; less painful; less blood loss | High risk of extension → 3rd/4th degree tear |
| J-shaped | Begins midline then curves lateral | — | Combines advantages of both | Rarely used |
| Lateral | Directly lateral | 90° | — | Bartholin gland injury; poor healing; rarely used |
Mediolateral episiotomy at 60° from midline is recommended by RCOG — angled cuts are less likely to extend into the anal sphincter.
✅ INDICATIONS
🔴 Fetal Indications
| Indication | Detail |
|---|---|
| Fetal distress in second stage | Pathological CTG, bradycardia, prolonged deceleration → need rapid delivery; episiotomy creates space to expedite |
| Preterm delivery (<34 weeks) | Premature perineum less distensible; episiotomy provides space; some debate but widely practised to protect fragile preterm head |
| Macrosomia / shoulder dystocia | Enlarging the outlet may facilitate manoeuvres (McRoberts, Rubin II, Woods screw) — though episiotomy itself doesn't resolve bony dystocia, it gives more room for internal manoeuvres |
| Breech delivery | Essential — allows safe delivery of shoulders and after-coming head; prevents sudden decompression |
| Face presentation (mentoanterior) | Perineum less able to accommodate face diameter |
| Multiple pregnancy (second twin) | Access for manipulation / internal version |
🟡 Maternal Indications
| Indication | Detail |
|---|---|
| Operative vaginal delivery (forceps or ventouse) | Most common indication — instruments need space; risk of uncontrolled perineal tearing is high without episiotomy; mandatory with rotational/mid-cavity forceps |
| Rigid / scarred perineum | Previous significant perineal surgery, scarring from FGM (female genital mutilation), previous 3rd/4th degree tear with repair → tissue inelastic → high risk of traumatic tearing |
| Imminent severe perineal tear | Perineum thinning and about to tear with jagged, irregular laceration → controlled incision preferable to uncontrolled tear |
| Previous 3rd or 4th degree tear (relative) | Controversial — RCOG recommends consideration of mediolateral episiotomy in subsequent delivery to divert away from previous sphincter repair |
| Maternal pushing difficulty | Prolonged second stage with inadequate progress + tight perineum |
🟠 Procedural / Obstetric Indications
| Indication | Detail |
|---|---|
| Internal manoeuvres | Shoulder dystocia internal rotational manoeuvres (Rubin, Woods, Zavanelli) require adequate access |
| Breech extraction (second twin) | Space needed for manipulation |
| Cord prolapse (in second stage) | Expedite delivery rapidly |
| Trial of instrumental delivery | Where failure necessitating CS is anticipated — episiotomy provides access |
❌ CONTRAINDICATIONS
Absolute Contraindications
| Contraindication | Reason |
|---|---|
| Unconsented patient | Medico-legal; patient must consent (in non-emergency) |
| Clotting disorders / coagulopathy | Active DIC, thrombocytopenia → episiotomy wound will bleed excessively and not heal well |
| Active vulval/vaginal infection (e.g. active HSV outbreak, Fournier's gangrene) | Spreading infection through incision |
| Avascular / ischaemic perineal tissue | Poor healing; breakdown |
Relative Contraindications
| Contraindication | Detail |
|---|---|
| Routine episiotomy without indication | Evidence-based practice: not recommended — does not prevent severe tears; increases morbidity |
| Normal spontaneous vaginal delivery progressing well | No clinical benefit shown for prophylactic episiotomy |
| Previous episiotomy with poor healing | Relative caution — may need to cut on opposite side or reconsider |
| FGM Type III (infibulation) | Requires defibulation (release of scar anteriorly) rather than standard episiotomy |
⚖️ Evidence: Selective vs. Routine Episiotomy
| Outcome | Routine Episiotomy | Selective Episiotomy |
|---|---|---|
| Severe perineal trauma (3rd/4th degree) | ↑ Risk (especially midline) | ↓ Risk |
| Anterior perineal tears | More common | Less common |
| Healing complications | More | Fewer |
| Dyspareunia | More | Fewer |
| Urinary incontinence | No benefit | — |
| Fecal incontinence | No benefit | — |
| Neonatal outcome | No difference | — |
WHO, RCOG, NICE — all recommend restrictive/selective episiotomy policy only.
🔪 Technique Briefly
| Step | Detail |
|---|---|
| Timing | Cut at crowning — when head distends perineum to ~3–4 cm diameter (maximum stretch = less blood loss) |
| Anaesthesia | Local infiltration (if no epidural) — 10–20 mL lidocaine 1% into perineum |
| Guard | Two fingers inserted between baby's head and perineum to protect fetus |
| Cut | Single decisive cut with straight scissors — 4–5 cm length at 60° from midline (mediolateral) |
| Direction | Away from anal sphincter toward right ischiorectal fossa |
| Repair | Continuous suture technique (Vicryl Rapide) — vaginal wall, then muscle, then skin (subcuticular preferred) |
⚠️ Complications of Episiotomy
| Complication | Notes |
|---|---|
| Haemorrhage | Especially if poorly timed or large cut |
| Extension → 3rd/4th degree tear | Most feared — especially midline episiotomy |
| Infection / wound breakdown | Particularly in perineal haematoma, poor hygiene |
| Haematoma | Especially if bleeding vessel not ligated |
| Dyspareunia | Post-healing scar tissue |
| Inadequate repair | Poor anatomical alignment → chronic pain, incontinence |
| Faecal/urinary incontinence | If sphincter involved unrecognised |
📊 Summary
| Indication | Contraindication | |
|---|---|---|
| Fetal | Distress, preterm, breech, macrosomia, shoulder dystocia | — |
| Maternal | Operative delivery (forceps/ventouse), rigid perineum, FGM, imminent severe tear, previous 3rd/4th degree | Coagulopathy, active infection, unconsented |
| Procedural | Internal manoeuvres, cord prolapse, breech extraction | Routine use without indication |
| Key rule | Selective, not routine — only cut when clinically necessary | Never as prophylaxis in normal delivery |