Describe the different stages of erythropoiesis. Explain the factors that control erythropoiesis 10 mark question
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the information needed for a comprehensive answer. Let me compose it.
Erythropoiesis: Stages and Controlling Factors
Definition
Erythropoiesis is the process by which red blood cells (erythrocytes) are produced from pluripotent stem cells in the bone marrow. It maintains the circulating red cell mass by replacing the ~1% of erythrocytes that reach senescence daily — roughly 250 billion cells, or 2.5–3 million per second under normal conditions.
Stages of Erythropoiesis
Phase 1: Committed Progenitor Stages (Morphologically Unrecognizable)
These cells cannot be identified on routine bone marrow smears but are defined by functional assays:
| Stage | Key Features |
|---|---|
| Pluripotent Stem Cell (HSC) | Self-renewing; gives rise to all blood lineages |
| CFU-GEMM | Mixed myeloid progenitor — committed to granulocyte, erythroid, monocyte, megakaryocyte lines |
| BFU-E (Burst-Forming Unit – Erythroid) | First cell exclusively committed to the erythroid line; only 10–20% are in cycle at any time; contains low levels of GATA-1 |
| CFU-E (Colony-Forming Unit – Erythroid) | Smaller colonies; expresses the highest density of EPO receptors; highly dependent on EPO for survival — without it, rapid apoptosis ensues; begins expressing blood group and Rh antigens |
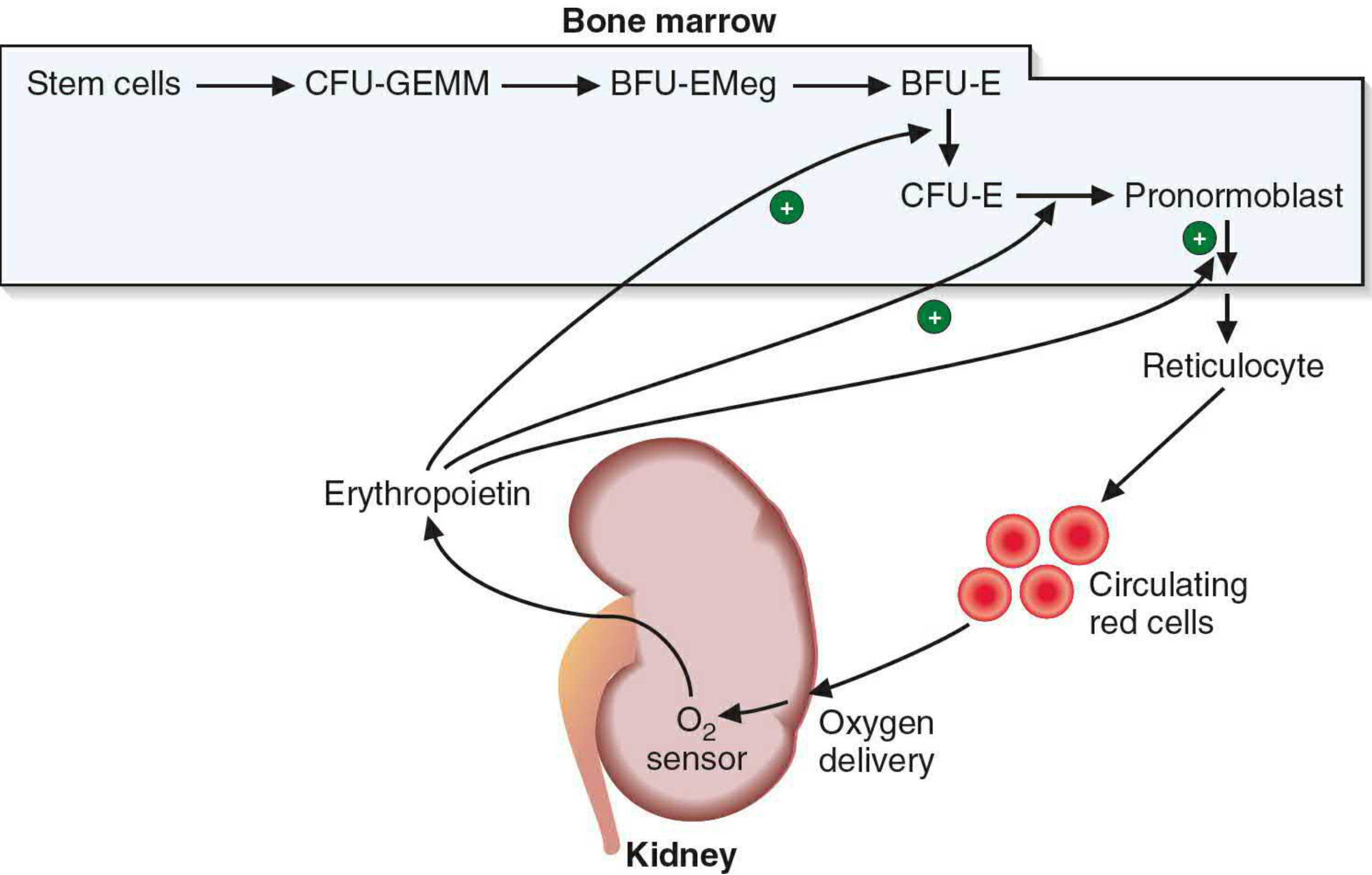
Erythropoietin stimulation of erythrocyte maturation — Basic Medical Biochemistry: A Clinical Approach, 6e
Phase 2: Morphologically Recognizable Stages (in Bone Marrow)
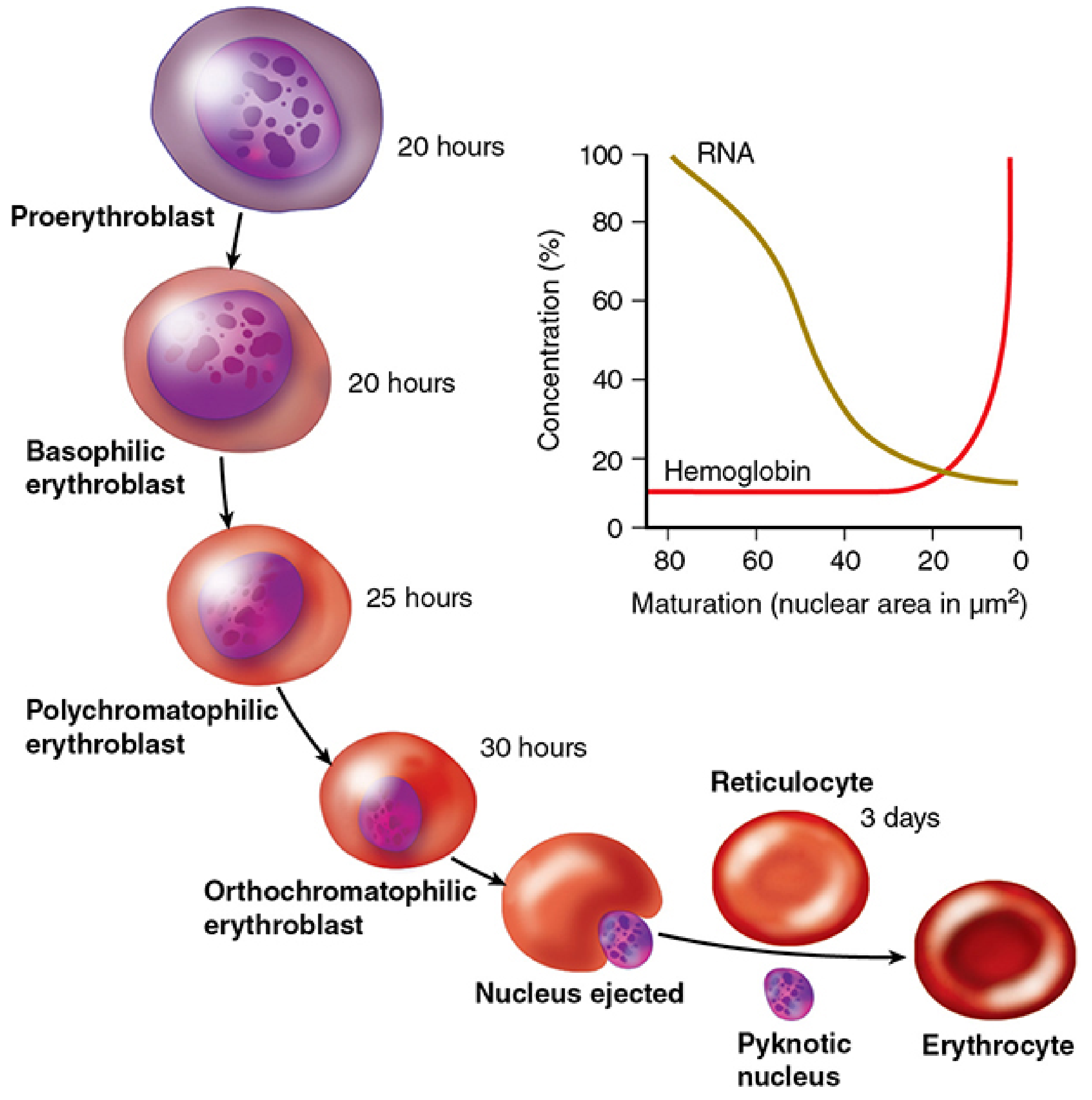
Erythropoiesis — major precursor stages with RNA/Hb curves — Junqueira's Basic Histology, 17e
1. Proerythroblast (Pronormoblast)
- Largest of the recognizable precursors (~20 µm); scarce in marrow
- Nucleus: Large, occupies most of the cell; loose, lacy (euchromatic) chromatin; multiple prominent nucleoli
- Cytoplasm: Deeply basophilic (reflects abundant ribosomes/polyribosomes; no hemoglobin yet)
- Golgi apparatus may appear as a pale perinuclear zone
- Capable of mitosis; duration ~20 hours
2. Basophilic Erythroblast (Early Normoblast)
- Smaller than the proerythroblast (arises by mitotic division)
- Nucleus: Smaller; chromatin begins to coarsen and condense; nucleoli disappear
- Cytoplasm: Strongly basophilic — reflects the increasing number of ribosomes and polyribosomes now actively synthesizing hemoglobin
- Capable of mitosis; duration ~20 hours
3. Polychromatophilic Erythroblast (Intermediate Normoblast)
- Further reduced cell size
- Nucleus: Smaller still; coarser, clumped heterochromatin (checkerboard pattern)
- Cytoplasm: Mixed blue-gray (polychromatic) — hemoglobin accumulation introduces eosinophilia, but residual RNA retains basophilia; the two staining qualities coexist
- Active hemoglobin synthesis
- Capable of mitosis (last stage that divides); duration ~25 hours
- This is the last EPO-dependent stage (along with earlier erythroblasts)
4. Orthochromatophilic Erythroblast (Late Normoblast / Normoblast)
- Cell is now smaller, slightly larger than a mature erythrocyte
- Nucleus: Extremely condensed (pyknotic); no visible chromatin pattern; occupies a small fraction of cell volume
- Cytoplasm: Nearly eosinophilic/pink — hemoglobin now predominates; very little RNA remains
- Cannot divide; duration ~30 hours
- Late in this stage: the pyknotic nucleus is ejected (extruded) and phagocytosed by macrophages at the erythroblastic island
5. Reticulocyte (Polychromatophilic Erythrocyte)
- Enucleate cell — nucleus has been expelled
- Still contains residual polyribosomes and mRNA, capable of some hemoglobin synthesis
- With brilliant cresyl blue stain: ribosomes precipitate into a faint blue network → "reticulum" (hence the name)
- Released from bone marrow into the peripheral bloodstream
- Circulates for 1–2 days, then matures in the spleen where ribosomes and mRNA are lost
- Normally constitutes ~1% of circulating red cells
6. Mature Erythrocyte
- Biconcave disc, ~7–8 µm
- No nucleus, no organelles; entirely eosinophilic cytoplasm filled with hemoglobin
- Life span: ~120 days
- Senescent cells are phagocytosed by macrophages in the spleen, bone marrow, and liver; iron is recycled
Key sequential changes summarized: cell size ↓ → nuclear size ↓ → nuclear chromatin condensation ↑ → nucleoli disappear → cytoplasm: basophilic → polychromatic → eosinophilic → nucleus ejected → reticulocyte → erythrocyte.
The total process from progenitor cell to circulating reticulocyte takes approximately 7 days, involving 3–5 mitotic divisions (occurring in the proerythroblast, basophilic, and polychromatophilic erythroblast stages).
Factors Controlling Erythropoiesis
1. Erythropoietin (EPO) — Primary Hormonal Regulator
- A 34 kDa glycoprotein (30.4 kDa protein core), produced mainly by peritubular interstitial cells of the kidney (and a minor amount by the liver)
- Regulated by a classic negative feedback loop: hypoxia → ↑ EPO → ↑ erythropoiesis → ↑ O₂ delivery → ↓ EPO release
- Mechanism: EPO binds the EPO receptor (EPO-R) on erythroid progenitors, activating the JAK2/STAT5 signaling pathway; this suppresses apoptosis and promotes proliferation/differentiation
- EPO receptors are most densely expressed on CFU-E cells
- Steady-state serum EPO: 10–30 U/L; in severe anemia/hypoxia: up to 10,000 U/L
- EPO is an essential survival factor from CFU-E through the basophilic erythroblast stage — without it, these cells rapidly undergo apoptosis
- Bone marrow is not a storage site for erythrocytes; erythrocytes are released as soon as formed
2. Oxygen Tension (Hypoxia-Sensing Mechanism)
- The kidney acts as an oxygen sensor via the HIF (Hypoxia-Inducible Factor) pathway
- In hypoxia: HIF-1α/HIF-2α are stabilized (normally degraded by prolyl hydroxylases under normoxia) → bind HRE (hypoxia-response elements) in the EPO gene promoter → ↑ EPO transcription
- HIF-2α is the principal isoform controlling EPO synthesis in renal peritubular cells
- Conditions stimulating EPO: high altitude, anemia, cardiopulmonary disease, hemorrhage, hemolysis
3. Iron
- Essential for hemoglobin (heme) synthesis; iron deficiency is the most common cause of anemia worldwide
- Iron supply must precisely match demand: ~1 mg/day absorbed from diet (5–10% of dietary intake); the majority comes from macrophage recycling of senescent RBCs
- HRI (Heme-Regulated Inhibitor / eIF-2α kinase): In iron-sufficient states, free heme binds HRI and inhibits eIF-2α phosphorylation → globin synthesis proceeds. In iron deficiency, HRI phosphorylates eIF-2α and suppresses mTORC1 → reduced globin synthesis → microcytic erythrocytes
- Iron deficiency also partially suppresses HIF-2α → reduces EPO production → inappropriately low reticulocyte count
4. Vitamin B₁₂ (Cobalamin) and Folic Acid
- Both are essential for DNA synthesis (purine and pyrimidine synthesis), required for the rapid cell divisions of erythropoiesis
- Deficiency → inability of erythroid progenitors to complete cell division → megaloblastic anemia (ineffective erythropoiesis with large abnormal cells)
- The stage most affected is at the transition from EPO-dependent effects to hemoglobin synthesis (around the CFU-E/proerythroblast boundary)
- Increased serum bilirubin, LDH, and accelerated iron turnover are hallmarks of the resulting ineffective erythropoiesis
5. Growth Factors and Cytokines
| Factor | Role |
|---|---|
| Stem Cell Factor (SCF/c-Kit ligand) | Early progenitor survival and proliferation |
| IL-3 | Stimulates early multipotent progenitors (CFU-GEMM) |
| GM-CSF | Acts on early myeloid progenitors |
| GATA-1 transcription factor | Master transcription factor for erythroid commitment; low in BFU-E, high in CFU-E and beyond |
| TNF-α, IFN-γ, TGF-β, IL-6 | Negative regulators produced by macrophages in chronic inflammation — suppress erythropoiesis (basis of anemia of chronic disease) |
6. The Erythroblastic Island Microenvironment
- Erythroblasts mature in close contact with central macrophages in erythroblastic islands within the bone marrow
- Macrophages supply ferritin and iron to developing erythroblasts, assist in enucleation, and support proliferation
- Osteoblasts in the hematopoietic niche can also locally produce EPO, modulating the response to systemic anemia
7. Negative Feedback
- Adequate oxygen delivery → reduced HIF-2α → ↓ EPO → ↓ erythropoiesis
- Mature erythrocytes themselves do not feed back directly, but their oxygen-carrying capacity determines the hypoxic stimulus
- Excess EPO → polycythemia → increased blood viscosity, which can paradoxically reduce tissue O₂ delivery
Summary Table
| Factor | Mechanism | Effect on Erythropoiesis |
|---|---|---|
| EPO (kidney) | JAK2/STAT5; inhibits apoptosis | Primary stimulator |
| Hypoxia/HIF-2α | ↑ EPO gene transcription | ↑ Erythropoiesis |
| Iron | Heme synthesis; HRI pathway | Deficiency → microcytic anemia |
| Vitamin B₁₂ / Folate | DNA synthesis for cell division | Deficiency → megaloblastic anemia |
| GATA-1 | Erythroid lineage commitment | Essential for differentiation |
| IL-3, SCF, GM-CSF | Progenitor proliferation | Supportive stimulation |
| TNF-α, IFN-γ, TGF-β | Macrophage-derived suppression | ↓ Erythropoiesis (chronic inflammation) |
| Macrophage niche | Iron delivery, enucleation support | Structural and nutritional support |
Sources:
- Junqueira's Basic Histology: Text and Atlas, 17e, pp. 644–645
- Histology: A Text and Atlas with Correlated Cell and Molecular Biology, Plate 10.3, pp. 776–777
- Basic Medical Biochemistry: A Clinical Approach, 6e, p. 1536
- Brenner and Rector's The Kidney, 2-Volume Set, pp. 2465–2469
This is a shared conversation. Sign in to Orris to start your own chat.