Cryptogenic organising pneumonia full details for md exams 15marks answer
Reading File
Finding Sources
Finding Sources
Searching PubMed
"cryptogenic organizing pneumonia" AND treatment
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
cryptogenic organising pneumonia CT scan chest histology Masson body
I now have comprehensive information from Harrison's 22E, Grainger & Allison's Radiology, Murray & Nadel's, and Fishman's. Let me compile the complete 15-mark MD exam answer.
Cryptogenic Organising Pneumonia (COP) — MD Examination Answer
Definition & Nomenclature
Cryptogenic Organising Pneumonia (COP) is an idiopathic interstitial pneumonia (IIP) characterised histologically by plugs of granulation tissue (Masson bodies / polypoid connective tissue buds) filling the lumens of alveolar ducts, alveoli, and small bronchioles, without destruction of the lung architecture.
- Historical term: Idiopathic bronchiolitis obliterans organising pneumonia (BOOP) — coined by Epler et al. (1985)
- Preferred term: COP, since "bronchiolitis obliterans" is a misnomer — the dominant process is in the air spaces, not the airways
- First described as a clinicoradiological entity by Davison et al. (1983) in eight patients with bilateral consolidation, polypoid fibrosis in alveoli, and striking corticosteroid response
- "Organising pneumonia" = histological pattern; "Cryptogenic" = only when no cause is identified
Classification
COP falls under idiopathic interstitial pneumonias per the 2002/2013 ATS/ERS classification. Organising pneumonia (OP) as a pattern can be:
| Type | Cause |
|---|---|
| Cryptogenic (COP) | Unknown; diagnosis of exclusion |
| Secondary OP | Infections, drugs, CTD, radiation, malignancy, transplantation |
Aetiology / Causes of OP Pattern
(Table 11.4 — Grainger & Allison)
- Unknown → COP
- Infections (bacterial, viral — including COVID-19)
- Toxic fume exposure
- Drugs — antibiotics, chemotherapeutics (bleomycin, amiodarone, nitrofurantoin), anti-inflammatory drugs
- Connective tissue diseases — especially polymyositis/dermatomyositis, RA, Sjögren's syndrome
- Vasculitis — Churg–Strauss, Wegener's granulomatosis
- Immunological disorders — common variable immunodeficiency, mixed cryoglobulinaemia
- Organ transplantation — bone marrow, lung, renal
- Radiation pneumonitis
- Malignancy — particularly lymphoma
Epidemiology
- Affects patients typically in their 50s–60s (range 40–70 years)
- Equal sex incidence; no association with smoking
- Incidence: ~2–7 per 100,000 population
Pathogenesis
- Initial injury (unknown trigger in COP) → damage to alveolar epithelial cells
- Exudation of fibrin into alveolar spaces
- Migration of fibroblasts and myofibroblasts into fibrin matrix
- Formation of polypoid granulation tissue buds (Masson bodies) in alveolar ducts and alveoli
- Inflammatory infiltrate of lymphocytes, plasma cells, macrophages in alveolar walls
- Architecture preserved — no honeycombing or significant fibrosis
- The process is potentially reversible with treatment (unlike UIP/IPF)
Pathology (Histology)
- Masson bodies (polypoid buds of loose connective tissue / fibroblastic plugs) in alveolar spaces, alveolar ducts, and respiratory bronchioles
- Inflammatory infiltrate in alveolar walls — lymphocytes, plasma cells, occasional eosinophils
- Alveolar architecture preserved (no destructive fibrosis)
- Patchy and temporally uniform (unlike UIP)
- Foamy macrophages may fill adjacent alveoli
- No hyaline membranes, no eosinophilic infiltrates, no granulomas (distinguishes from other IIPs)
Histopathologic findings include patchy regions of organizing pneumonia with granulation tissue commonly involving small airways, alveolar ducts, and alveoli with surrounding inflammation that can involve alveolar walls — Harrison's 22E, Fig. 304-3C
Clinical Features
| Feature | Detail |
|---|---|
| Onset | Subacute (weeks–months); flu-like prodrome |
| Common symptoms | Cough (dry/productive), progressive dyspnoea, fever, fatigue, malaise, weight loss, night sweats |
| Examination | Inspiratory crackles/rales; no clubbing |
| Often misdiagnosed as | Community-acquired pneumonia (fails antibiotic therapy — key clue) |
Key clinical pearls:
- Symptoms present for 4–10 weeks before diagnosis (average 3 months)
- Multiple failed courses of antibiotics → should raise suspicion
- Non-productive cough + dyspnoea + constitutional symptoms in middle-aged non-smoker
Investigations
Pulmonary Function Tests
- Restrictive pattern — reduced TLC, FVC, DLCO
- FEV1/FVC normal or increased
- Hypoxaemia at rest or on exertion
- Mild obstruction occasionally (due to bronchiolar involvement)
Blood Tests
- Elevated ESR and CRP (often markedly)
- Mild leukocytosis
- Autoantibody screen (ANA, anti-Jo-1, RF) — to exclude secondary causes (CTD)
Bronchoalveolar Lavage (BAL)
- Lymphocytosis (CD8 > CD4) — most characteristic
- Also: neutrophilia, eosinophilia (mixed cellularity)
- CD4:CD8 ratio decreased
- BAL pattern alone not diagnostic but supportive
Chest X-Ray Findings
- Bilateral patchy areas of consolidation — mid and lower zone predominance (though any zone can be affected)
- Subpleural and/or peribronchovascular distribution in ~2/3 of patients
- Migrating (fleeting) opacities — highly characteristic
- Nodules (up to 1 cm) — may be the sole manifestation
- Cavitation is NOT a feature
HRCT Findings (most important investigation)
| CT Pattern | Description |
|---|---|
| Consolidation (most common) | Bilateral, peripheral, subpleural; mid-lower zone; air bronchograms present |
| Ground-glass opacity (GGO) | Bilateral, often surrounding consolidation |
| Reversed Halo / Atoll sign | Central GGO surrounded by ring of consolidation — pathognomonic when present |
| Perilobular pattern | Consolidation outlining pulmonary lobules — regarded as pathognomonic |
| Nodules | 1 cm, no zonal predilection; rarely large solitary mass |
| Linear opacities | Type I: radial, bronchocentric; Type II: subpleural, curvilinear |
A perilobular distribution of consolidation, unlike a multitude of other radiological signs, may be regarded as pathognomonic of an organising pneumonia pattern — Grainger & Allison's Diagnostic Radiology
CT images illustrating COP patterns:
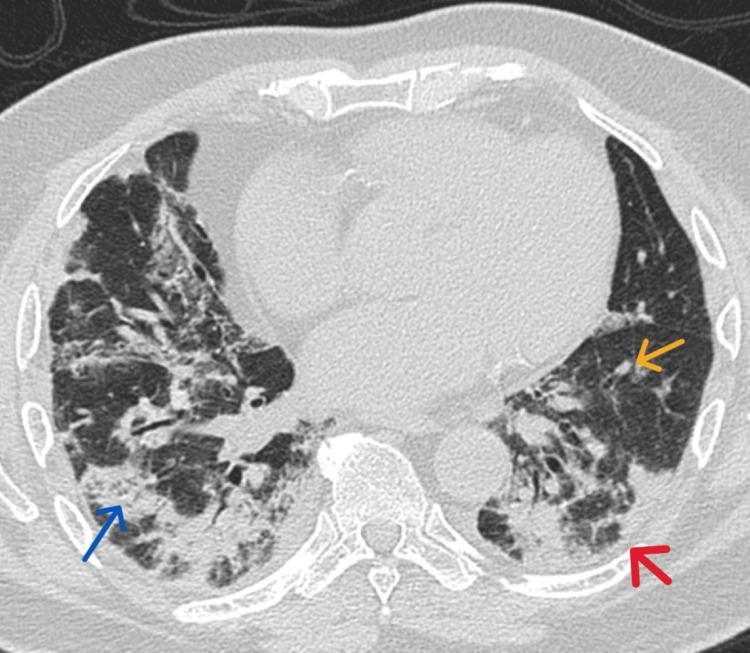
Axial HRCT showing bilateral patchy consolidation (blue arrow), small nodules (yellow arrow), and perilobular pattern (red arrow) — classic COP
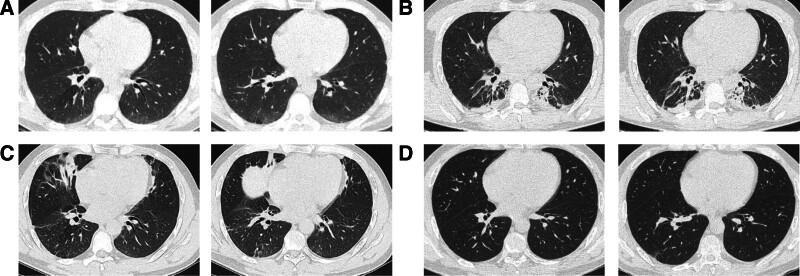
Serial HRCT: (A) bilateral lower lobe GGO; (B) progression; (C) new right middle lobe focus — migratory pattern; (D) near-complete resolution on steroids
Diagnosis
Diagnostic Criteria
Diagnosis of COP requires exclusion of:
- Infectious causes
- Drug-induced OP
- CTD-associated OP
- Post-radiation OP
Gold standard: Multidisciplinary discussion (MDT) — clinician + radiologist + pathologist
Biopsy:
- Transbronchial biopsy — sufficient in typical cases (shows Masson bodies); avoids surgical biopsy
- Surgical lung biopsy (VATS) — when diagnosis remains uncertain; gold standard for histology
- CT-guided — for peripheral nodular lesions
Diagnostic approach:
- Typical clinical + HRCT findings → MDT discussion
- BAL to support (lymphocytosis)
- Transbronchial biopsy if clinical picture not clear
- VATS/surgical biopsy if diagnosis still uncertain
Differential Diagnosis
| Condition | Distinguishing feature |
|---|---|
| CAP/pneumonia | Responds to antibiotics; no migration |
| Eosinophilic pneumonia | Peripheral opacities; eosinophilia; BAL eosinophilia |
| Lymphoma | PET-avid nodes; biopsy |
| IPF | Honeycombing; basal subpleural fibrosis; UIP pattern |
| NSIP | Diffuse ground glass; subpleural sparing; no consolidation |
| AIP (Hamman-Rich) | Acute onset; DAD on biopsy; hyaline membranes |
| Pulmonary alveolar proteinosis | "Crazy paving"; PAS+ material on BAL |
Treatment
Corticosteroids — First-line
| Regimen | Dose / Duration |
|---|---|
| Prednisone | 0.75–1 mg/kg/day (max 60 mg/day) for 4–8 weeks |
| Taper | Gradual taper over 6–12 months (total minimum 6 months) |
| Maintenance | Low-dose for 6–12 months reduces relapse |
- Response is typically dramatic and rapid — symptomatic improvement within days to weeks
- Radiological improvement within 4–8 weeks
Second-line / Steroid-sparing
| Agent | Indication |
|---|---|
| Macrolides (azithromycin/clarithromycin) | Mild disease; steroid-sparing; some studies show benefit |
| Azathioprine | Steroid-resistant / relapsing disease |
| Cyclophosphamide | Refractory cases |
| Mycophenolate mofetil | Increasing evidence for steroid-sparing |
| Rituximab | Biologic option; growing evidence |
Evidence is growing that alternate cytotoxic (e.g., mycophenolate, cyclophosphamide) or biologic (e.g., rituximab) therapies can be helpful in both treating the disease and reducing the need for steroids — Harrison's 22E
Spontaneous resolution
- Mild/asymptomatic cases may resolve spontaneously
- Watchful waiting acceptable only in very mild disease
Prognosis & Relapse
- Overall prognosis: Excellent — most patients respond well to corticosteroids
- Relapse rate: 30–58% — usually on steroid taper or after discontinuation
- Relapses respond to re-initiation of steroids
- Factors predicting relapse:
- Short duration of initial treatment
- Rapid steroid taper
- Secondary forms (CTD-associated)
- Progression to respiratory failure is rare
- Chronic fibrosing course seen in a minority (termed "fibrotic OP" or progression to NSIP/UIP pattern)
- Mortality: <5% in COP; higher in secondary forms
COP vs. Secondary OP — Key Distinction
| Feature | COP | Secondary OP |
|---|---|---|
| Cause | Unknown | Identified (drugs, CTD, infection) |
| Prognosis | Excellent | Depends on cause |
| Long-term therapy | Usually not needed | May be needed (esp. CTD) |
Summary / High-Yield Points for MD Exam
- COP = organising pneumonia without identifiable cause — diagnosis of exclusion
- Masson bodies = pathological hallmark (polypoid fibroblastic plugs in alveoli/alveolar ducts)
- Classic presentation: middle-aged non-smoker, subacute flu-like illness, bilateral consolidation failing antibiotic therapy
- HRCT: bilateral peripheral consolidation ± GGO ± reversed halo sign ± perilobular pattern
- Reversed halo/atoll sign and perilobular consolidation = near-pathognomonic CT signs
- BAL: mixed cellularity with lymphocytosis (CD4:CD8 ratio decreased)
- PFTs: restrictive pattern with reduced DLCO
- Treatment: Prednisolone 0.75–1 mg/kg/day for minimum 6–12 months
- Dramatic response to steroids is both diagnostic and therapeutic
- Relapse in 30–58% — especially on steroid taper; re-treat with steroids
Sources:
- Harrison's Principles of Internal Medicine, 22nd Edition (2025), Chapter 304
- Grainger & Allison's Diagnostic Radiology, Chapter 11
- Murray & Nadel's Textbook of Respiratory Medicine
- Fishman's Pulmonary Diseases and Disorders
This is a shared conversation. Sign in to Orris to start your own chat.