What is ursodiol and ursodeoxyclic acid in canine hepatitis
ursodiol ursodeoxycholic acid canine hepatitis liver disease dogs
ursodeoxycholic acid mechanism of action hepatoprotective bile acid
Ursodiol (Ursodeoxycholic Acid) in Canine Hepatitis
Mechanism of Action
| Mechanism | Detail |
|---|---|
| Bile acid pool modification | Replaces hydrophobic, cytotoxic bile acids (e.g., deoxycholate, lithocholate) with the hydrophilic, less toxic UDCA, reducing hepatocyte membrane damage |
| Cytoprotection | Stabilizes hepatocyte and cholangiocyte membranes against toxic bile acid-induced injury |
| Choleresis | Stimulates bile secretion and bile flow, promoting clearance of toxic metabolites |
| Immunomodulation | Reduces aberrant MHC class I and class II expression on hepatocytes and bile duct cells, downregulating immune-mediated liver injury |
| Anti-apoptotic effect | Inhibits mitochondrial membrane permeabilization and suppresses hepatocyte apoptosis triggered by hydrophobic bile acids |
| Antifibrotic | Indirectly reduces hepatic fibrosis by limiting ongoing inflammation and hepatocyte necrosis |
Role in Canine Hepatitis
Chronic Hepatitis (Canine Chronic Hepatitis / CCH)
- Chronic hepatitis in dogs is often immune-mediated or idiopathic, and toxic bile acid accumulation contributes to ongoing hepatocyte injury.
- UDCA reduces the intrahepatic concentration of hydrophobic bile acids, providing cytoprotection and reducing inflammation.
- It is typically used alongside primary treatments (immunosuppressives such as prednisolone, azathioprine) rather than as monotherapy.
- Dose: 10–15 mg/kg PO once daily (some sources use divided dosing; given with food as it is fat-soluble).
Cholestatic Liver Disease
- UDCA is particularly beneficial when cholestasis is present (elevated ALP, bilirubin, bile acids) as it actively promotes bile flow and reduces bile acid-induced hepatocyte toxicity.
Hepatic Copper Accumulation
- In copper-associated hepatitis (e.g., Bedlington Terriers, Labrador Retrievers), UDCA is used as supportive therapy alongside copper chelation (D-penicillamine or zinc) to protect hepatocytes from oxidative and bile acid-mediated damage.
Vacuolar Hepatopathy / Steroid Hepatopathy
- Used supportively to reduce hepatocyte stress and improve bile flow.
Biliary Disease / Mucocele
- Some use of UDCA in early or nonsurgical biliary mucocele cases has been reported to thin bile and reduce inspissation, though surgical management is often definitive.
Clinical Monitoring
- Liver enzymes (ALT, ALP, GGT, bilirubin) — to assess response; expect gradual improvement over weeks to months
- Serum bile acids (pre- and post-prandial) — to evaluate functional hepatic reserve
- Clinical signs — appetite, mentation, ascites, icterus
Key Considerations
- Safety: UDCA is well-tolerated in dogs. Side effects are rare but may include mild GI upset (vomiting, diarrhea) at higher doses.
- Contraindications: Avoid in complete biliary obstruction (can worsen bile accumulation if outflow is blocked).
- Not a cure: UDCA addresses bile acid toxicity and inflammation but does not reverse fibrosis or treat the primary underlying etiology.
- Evidence base: While robust randomized controlled trials in dogs are limited compared to humans, UDCA is widely recommended in veterinary internal medicine guidelines (e.g., WSAVA Liver Standardization Group) based on mechanism, safety profile, and clinical experience.
Summary
Why onset + metclopramide and Ranitidine +omeprazole are not used collectively in dog and cat from different books and ethnic sources
metoclopramide ondansetron drug interaction serotonin syndrome dogs cats
ranitidine omeprazole drug interaction antacid proton pump inhibitor H2 blocker veterinary
ondansetron metoclopramide combined antiemetic contraindication mechanism
omeprazole ranitidine combination gastric acid suppression pharmacodynamic antagonism redundancy
Why Ondansetron + Metoclopramide and Ranitidine + Omeprazole Are Avoided Together in Dogs and Cats
1. Ondansetron + Metoclopramide
(You wrote "onset" — this refers to ondansetron, a 5-HT₃ receptor antagonist)
Their Individual Mechanisms
| Drug | Class | Primary Mechanism |
|---|---|---|
| Metoclopramide | Dopamine (D₂) antagonist + prokinetic | Blocks D₂ receptors centrally (CRTZ) and peripherally; also has weak 5-HT₃ antagonist and 5-HT₄ agonist activity |
| Ondansetron | Selective 5-HT₃ antagonist | Blocks serotonin (5-HT₃) receptors in the gut and CRTZ — potent antiemetic |
Why They Should NOT Be Combined
A. Pharmacodynamic Antagonism at 5-HT₃ Receptors
- Ondansetron is a potent, selective 5-HT₃ blocker
- Metoclopramide's prokinetic effect via 5-HT₄ is opposed by ondansetron's blockade of 5-HT₃ receptors in a complex interplay that reduces metoclopramide's prokinetic efficacy
- Net result: the motility-promoting benefit of metoclopramide is blunted by concurrent ondansetron
B. Risk of Serotonin Syndrome (Serotonergic Toxicity)
- Metoclopramide inhibits serotonin reuptake and has serotonergic activity
- Ondansetron, while a 5-HT₃ antagonist, paradoxically contributes to elevated serotonin in certain receptor compartments
- Combined use can tip the balance toward serotonin syndrome, characterized by:
- Tremors, hyperthermia, mydriasis
- Agitation, disorientation
- Muscle rigidity or fasciculations
- Tachycardia
- Cats are particularly sensitive to serotonin toxicity due to differences in serotonin receptor density and metabolism compared to dogs
C. Redundancy Without Added Benefit
- Does not provide superior antiemetic control compared to adequate dosing of one agent
- Doubles CNS/autonomic side effect risk (extrapyramidal signs from metoclopramide, QT prolongation from ondansetron)
- Violates the principle of rational polypharmacy
D. QT Prolongation Risk
- Ondansetron is associated with dose-dependent QT interval prolongation
- Metoclopramide also has mild cardiac effects
- Combined use increases the risk of arrhythmias, particularly in cats with pre-existing cardiac disease
Veterinary references: Plumb's Veterinary Drug Handbook (10th ed.), Papich's Saunders Handbook of Veterinary Drugs, and the BSAVA Small Animal Formulary all flag the serotonergic interaction between metoclopramide and serotonergic drugs including 5-HT₃ antagonists.
2. Ranitidine + Omeprazole
Their Individual Mechanisms
| Drug | Class | Mechanism |
|---|---|---|
| Ranitidine | H₂-receptor antagonist | Competitively blocks histamine H₂ receptors on parietal cells → reduces cAMP → reduces acid secretion |
| Omeprazole | Proton pump inhibitor (PPI) | Irreversibly binds and inhibits the H⁺/K⁺-ATPase pump (the final common pathway for ALL acid secretion) |
Why They Should NOT Be Combined
A. Mechanistic Redundancy — Omeprazole Supersedes Ranitidine
- Ranitidine only blocks one upstream signal (histamine via H₂ receptors)
- Once omeprazole is active, ranitidine adds no meaningful additional acid suppression
- This is pharmacodynamic redundancy: combining them does not achieve greater gastric pH elevation than omeprazole alone at appropriate doses
B. Pharmacokinetic Antagonism — Ranitidine Blunts Omeprazole Activation
- Omeprazole is a prodrug — it requires an acidic environment in the secretory canaliculus of the parietal cell to be converted into its active sulfenamide form, which then binds the proton pump
- Ranitidine, by raising gastric (and parietal cell canalicular) pH, reduces the acidic microenvironment needed for omeprazole to activate
- Result: ranitidine partially prevents omeprazole from being activated → omeprazole's efficacy is reduced
- This is a true pharmacokinetic-pharmacodynamic interaction documented in human gastroenterology and applied in veterinary medicine
C. Ranitidine Has Additional Concerns in Cats
- Ranitidine contains a nitrosamine impurity (NDMA — N-nitrosodimethylamine), which led to its withdrawal from human markets (FDA 2020) and has influenced veterinary use
- In cats, ranitidine also has prokinetic properties (via acetylcholinesterase inhibition) which is a separate action from its H₂ blockade; this sometimes causes confusion about its role
D. Increased Side Effect Burden Without Benefit
- Ranitidine: headache-equivalent signs, rare hepatotoxicity in cats, drug interactions (alters absorption of other drugs requiring acidic stomach)
- Omeprazole: already alters GI microbiome, reduces B12/magnesium absorption with chronic use, may alter gut motility
- Combining both multiplies risks of altered nutrient absorption and GI dysbiosis without therapeutic gain
Veterinary references: Plumb's Veterinary Drug Handbook explicitly notes that H₂ blockers and PPIs should not be combined as the H₂ blocker may impair PPI activation. The BSAVA Small Animal Formulary, Merck Veterinary Manual, and Boothe's Small Animal Clinical Pharmacology and Therapeutics all support using either an H₂ blocker or a PPI, not both simultaneously.
Summary Table
| Drug Combination | Primary Reason to Avoid | Species Note |
|---|---|---|
| Ondansetron + Metoclopramide | Serotonin syndrome risk; ondansetron antagonizes metoclopramide's prokinetic effect; QT prolongation | Cats especially vulnerable to serotonin toxicity |
| Ranitidine + Omeprazole | Ranitidine raises gastric pH and impairs omeprazole activation (prodrug); pure redundancy; no added acid suppression | Ranitidine also withdrawn from human use (NDMA contamination) |
Clinical Bottom Line
- For antiemesis + prokinesis: choose metoclopramide (if prokinesis is needed) or ondansetron (for potent antiemesis) — not both. Maropitant (NK₁ antagonist) is now first-line in dogs/cats and avoids these interactions entirely.
- For acid suppression: omeprazole (or other PPIs like pantoprazole) is the preferred agent; H₂ blockers are a second-line alternative when PPIs are unavailable — never combine the two classes simultaneously.
How steroid enhance the corneal ulcers in cat and dog
corticosteroids corneal ulcer worsening dogs cats mechanism
steroid ophthalmic corneal healing collagenase matrix metalloproteinase ulcer progression
corneal ulcer stromal melting keratomalacia collagenase neutrophil protease veterinary ophthalmology
corneal ulcer melting stromal dog cat steroid effect
How Steroids Enhance (Worsen) Corneal Ulcers in Dogs and Cats
The Normal Corneal Healing Process (Baseline)
- Epithelial cells at the wound margin migrate and proliferate to re-cover the defect
- Inflammatory cells (neutrophils, macrophages) debride necrotic tissue
- Keratocytes (corneal stromal fibroblasts) produce new collagen to restore the stroma
- Matrix metalloproteinases (MMPs) are tightly regulated to remodel the stroma without over-digesting it
- Neovascularization may occur peripherally to deliver immune cells and nutrients
Mechanisms by Which Steroids Worsen Corneal Ulcers
1. Inhibition of Epithelial Healing
- Corticosteroids suppress epithelial cell mitosis and migration
- They downregulate growth factors including EGF (epidermal growth factor) and fibronectin essential for epithelial resurfacing
- Result: the surface defect fails to close, leaving the stroma exposed to environmental pathogens, proteases, and desiccation
- In cats especially, re-epithelialization is slow even without steroids; adding steroids causes persistent epithelial defects
2. Upregulation of Collagenases → Stromal Melting (Keratomalacia)
- Steroids paradoxically increase collagenase (MMP-1, MMP-2, MMP-9) activity in corneal tissue
- These enzymes digest stromal collagen fibrils — the structural backbone of the cornea
- Simultaneously, steroids suppress the natural inhibitors of MMPs (TIMPs — tissue inhibitors of metalloproteinases)
- The net result is uncontrolled enzymatic digestion of the corneal stroma → rapid thinning → descemetocele (only Descemet's membrane remains) → corneal perforation
This process is called keratomalacia or corneal melting and can progress from a simple ulcer to perforation within 24–48 hours in dogs, and even faster in cats.
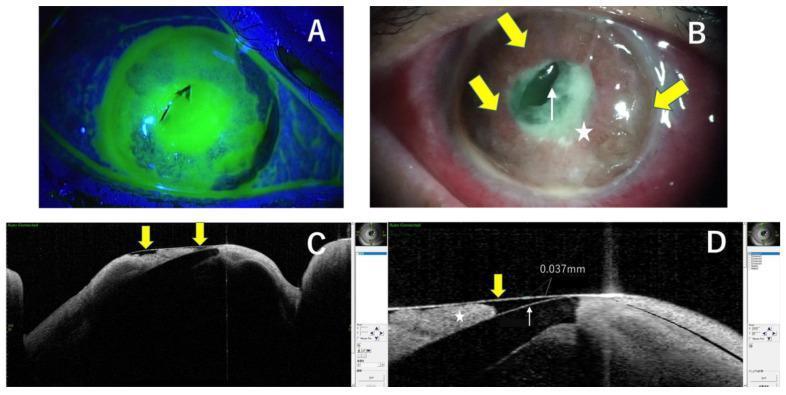
3. Immunosuppression → Potentiation of Infection
- Steroids suppress neutrophil and macrophage function, reducing phagocytosis of bacteria and fungi
- They impair T-cell–mediated immune responses at the ocular surface
- Pre-existing subclinical bacterial colonization (e.g., Pseudomonas aeruginosa, Staphylococcus, Streptococcus) rapidly becomes overt infectious keratitis
- Fungal keratitis (e.g., Aspergillus, Fusarium) is dramatically worsened — fungi thrive in the immunosuppressed corneal environment
- Pseudomonas is particularly dangerous as it secretes exogenous proteases that synergize with steroid-upregulated MMPs to cause explosive corneal melting
As noted in Bacterial Keratitis (p. 22–23): "Potential disadvantages [of corticosteroids] include recurrence of infection, local immunosuppression, inhibition of collagen synthesis predisposing to corneal melting."
4. Inhibition of Collagen Synthesis
- Steroids suppress keratocyte activity and reduce synthesis of new collagen (types I and III)
- Without collagen replacement, the thinned stroma cannot regenerate
- This creates a net catabolic state: collagen is being broken down faster than it is being made
- Clinically: the ulcer bed deepens rather than fills
5. Masking of Clinical Signs
- Steroids powerfully suppress pain, redness, and blepharospasm
- The animal (and owner) may appear to improve clinically even as the ulcer worsens structurally
- This false reassurance delays diagnosis and re-treatment
- By the time perforation is visible, it may be too late to save the globe
6. Increased Intraocular Pressure (Steroid-Induced Glaucoma)
- Topical and systemic steroids can reduce aqueous humor drainage → elevated IOP
- In an eye already compromised by corneal ulceration, raised IOP increases the risk of iris prolapse if perforation occurs
- Cats are particularly susceptible to steroid-induced IOP elevation
7. Species-Specific Considerations
| Feature | Dogs | Cats |
|---|---|---|
| Collagenase activity | High baseline; steroids dramatically amplify this | Moderate; melting less explosive but still significant |
| Common organisms | Pseudomonas, Staph, Strep — proteolytic | Herpesvirus (FHV-1) co-infection common |
| Herpesvirus | Rare concern | FHV-1 is a major complication — steroids cause viral reactivation and dramatic worsening of herpetic keratitis |
| Indolent ulcers | Common in boxers, corgis — steroids worsen epithelial adherence failure | Less common |
| Healing rate | Faster re-epithelialization | Slower; more vulnerable to steroid-induced persistent defects |
Special Note on Cats and FHV-1:
- Feline herpesvirus-1 establishes latency in the trigeminal ganglion
- Steroids cause reactivation of latent FHV-1, triggering dendritic/geographic ulcers, stromal keratitis, and corneal sequestrum formation
- This is a unique and critical reason why steroids are even more dangerous in cats with corneal disease than in dogs
Summary: Cascade of Steroid-Induced Corneal Damage
Topical/Systemic Corticosteroid Applied to Ulcerated Cornea
↓
┌───────────────┼────────────────┐
↓ ↓ ↓
Epithelial MMP upregulation Immunosuppression
healing blocked + TIMP suppression (bacterial/fungal
↓ overgrowth)
Collagen digestion ↓
exceeds synthesis Proteases amplify
↓ collagen destruction
Stromal thinning
↓
Descemetocele
↓
Corneal perforation
↓
Endophthalmitis / Globe loss
Clinical Rule in Veterinary Ophthalmology
"Never use steroids on an ulcerated eye."
- Topical NSAIDs (diclofenac, flurbiprofen) — with caution as they can also mildly impair healing
- Systemic NSAIDs for pain/inflammation
- Serum/plasma drops — contain natural TIMPs and anti-proteases to counteract melting
- N-acetylcysteine (NAC) drops — mucolytic and MMP inhibitor, used in melting ulcers
- Atropine — for reflex uveitis/mydriasis
- Treating the underlying cause (entropion, distichia, KCS, FHV-1 with antivirals)
Tell me the Mode of action of these drugs terbutaline sulfate, amoxicillin trihydrate,
terbutaline sulfate beta-2 agonist mechanism of action bronchodilation
amoxicillin trihydrate mechanism of action beta-lactam cell wall synthesis inhibition
beta-2 adrenergic receptor agonist cAMP adenylyl cyclase smooth muscle relaxation mechanism
amoxicillin penicillin binding protein peptidoglycan bacterial cell wall bactericidal spectrum
Mode of Action: Terbutaline Sulfate & Amoxicillin Trihydrate
1. Terbutaline Sulfate
Drug Class
Chemical Nature
- Synthetic catecholamine analogue
- Exists as a sulfate salt for stability and solubility
- Structurally related to epinephrine but designed for selective β₂ activity
Mechanism of Action — Step by Step
Terbutaline
↓
Binds β₂-adrenergic receptors (Gs-protein coupled)
↓
Activates adenylyl cyclase
↓
ATP → cyclic AMP (cAMP) ↑↑
↓
Activates Protein Kinase A (PKA)
↓
Phosphorylates myosin light-chain kinase (MLCK) → INACTIVATED
↓
Smooth muscle RELAXATION
At the Molecular Level:
| Step | Event |
|---|---|
| Receptor binding | Terbutaline binds β₂ receptors on bronchial smooth muscle, uterine muscle, and mast cells |
| G-protein activation | Gs protein is activated → stimulates adenylyl cyclase |
| cAMP production | Adenylyl cyclase converts ATP to cyclic AMP (cAMP) — intracellular second messenger |
| PKA activation | Elevated cAMP activates Protein Kinase A |
| MLCK inhibition | PKA phosphorylates and inactivates myosin light-chain kinase → prevents actin-myosin crossbridging |
| Ca²⁺ sequestration | PKA also promotes Ca²⁺ uptake into sarcoplasmic reticulum and efflux out of cell, further reducing intracellular Ca²⁺ |
| Net effect | Smooth muscle relaxation → bronchodilation, uterine relaxation, vasodilation |
Organ-Specific Effects
| Organ/System | Effect | Mechanism |
|---|---|---|
| Bronchi (primary target) | Bronchodilation | β₂-mediated smooth muscle relaxation |
| Mast cells | Reduced mediator release | Inhibits degranulation (histamine, leukotrienes) |
| Mucociliary clearance | Increased | Stimulates ciliary beat frequency |
| Uterus | Tocolysis (relaxation) | β₂ receptors on myometrium |
| Heart (minor) | Tachycardia | Weak β₁ activity (side effect) |
| Skeletal muscle/vasculature | Tremors, vasodilation | β₂ receptors in peripheral vessels |
| Metabolic | Hypokalemia, hyperglycemia | K⁺ shifts into cells; glycogenolysis |
Veterinary Applications (Dogs & Cats)
- Bronchospasm / feline asthma — first-line bronchodilator in cats
- Tracheal collapse in dogs — reduces bronchospasm
- Status asthmaticus — injectable terbutaline in acute severe bronchoconstriction
- Tocolysis — prevent premature uterine contractions
- Hyperkalemia (emergency) — drives K⁺ into cells via β₂ activation
Dosing (Veterinary):
- Cats (bronchospasm): 0.01 mg/kg SC/IM; or 0.1 mg/kg PO q4–8h
- Dogs: 0.01 mg/kg SC; or 1.25–5 mg PO q4–8h
Selectivity Note
2. Amoxicillin Trihydrate
Drug Class
Chemical Nature
- Semi-synthetic penicillin with an amino group on the side chain (improves oral absorption and spectrum)
- "Trihydrate" = the salt form with 3 water molecules — improves stability of the oral formulation
- Susceptible to β-lactamase enzymes unless combined with a β-lactamase inhibitor (e.g., clavulanic acid → Amoxicillin-clavulanate / co-amoxiclav)
Mechanism of Action — Step by Step
Amoxicillin enters bacterial periplasmic space
↓
Binds Penicillin-Binding Proteins (PBPs) — transpeptidase enzymes
↓
Irreversibly inhibits transpeptidase activity
↓
Cross-linking of peptidoglycan strands BLOCKED
↓
Peptidoglycan cell wall WEAKENS
↓
Bacterial autolysins (murein hydrolases) continue degrading wall
↓
Osmotic lysis of bacteria → BACTERICIDAL
Detailed Steps:
| Step | Detail |
|---|---|
| Target: Peptidoglycan | Bacterial cell walls are built from peptidoglycan (murein) — a rigid mesh of NAM-NAG sugar chains cross-linked by short peptide bridges |
| Transpeptidase (PBP) role | Transpeptidases catalyze the cross-linking reaction between peptide side chains of adjacent glycan strands — essential for structural wall integrity |
| β-Lactam binding | The β-lactam ring of amoxicillin mimics the D-Ala–D-Ala terminal dipeptide of the natural substrate → acts as a competitive/irreversible inhibitor of PBPs |
| Covalent inactivation | Amoxicillin forms a stable covalent acyl-enzyme complex with the active serine residue of PBPs → permanently inactivates the enzyme |
| Wall weakening | Existing peptidoglycan continues to be degraded by bacterial autolysins but no new cross-links form → wall becomes structurally deficient |
| Bactericidal lysis | Osmotic pressure inside the bacterium causes cell swelling and rupture — bacteria die (bactericidal, not just static) |
As cited in Harrison's Principles of Internal Medicine (21st ed., p. 4364): "β-lactams inhibit bacterial cell-wall synthesis by binding to cell-wall transpeptidases, cross-linking enzymes also called penicillin-binding proteins (PBPs)... The most common mechanism of resistance is degradation by β-lactamases, which break down the core β-lactam ring."
Spectrum of Activity
| Organism Type | Coverage | Notes |
|---|---|---|
| Gram-positive cocci | Excellent | Staphylococcus (non-β-lactamase), Streptococcus, Enterococcus |
| Gram-negative rods | Moderate | E. coli, Pasteurella, Haemophilus — broader than narrow-spectrum penicillins |
| Anaerobes | Some coverage | Useful for mixed infections |
| β-Lactamase producers | No coverage | Staph aureus (MRSA), many E. coli strains — need amoxicillin-clavulanate |
| Pseudomonas | None | Intrinsically resistant |
Resistance Mechanisms to Amoxicillin
- β-Lactamase production — most common; enzymes cleave the β-lactam ring, destroying activity
- Modified PBPs — altered target with reduced binding affinity (e.g., MRSA has PBP2a encoded by mecA gene)
- Efflux pumps — pump drug out of bacteria before it can act
- Reduced permeability — porin loss in gram-negative bacteria limits entry
Veterinary Applications (Dogs & Cats)
- Skin and soft tissue infections — pyoderma, wounds, abscesses (Staphylococcus, Streptococcus)
- Urinary tract infections (UTI)
- Respiratory tract infections — bronchopneumonia, Pasteurella in cats
- Gastrointestinal infections
- Dental/oral infections — periodontal disease
- Post-surgical prophylaxis
Dosing (Veterinary):
- Dogs & Cats: 11–22 mg/kg PO q8–12h (trihydrate oral formulation)
- Often used as amoxicillin-clavulanate (co-amoxiclav) to extend coverage against β-lactamase producers
Comparative Summary
| Feature | Terbutaline Sulfate | Amoxicillin Trihydrate |
|---|---|---|
| Class | β₂-adrenergic agonist | Aminopenicillin (β-lactam) |
| Target | β₂-adrenergic G-protein coupled receptor | Penicillin-binding proteins (PBPs) / transpeptidases |
| Second messenger | cAMP via adenylyl cyclase | None (direct enzyme inhibition) |
| Effect type | Functional (smooth muscle relaxation) | Bactericidal (cell wall lysis) |
| Primary use (vet) | Bronchospasm, feline asthma, tocolysis | Bacterial infections (skin, respiratory, UTI) |
| Onset of action | Rapid (minutes SC/inhaled) | Slower (hours — requires bacterial cell division) |
| Resistance concern | Receptor downregulation with overuse | β-lactamase production |
| Key side effect | Tachycardia, hypokalemia, tremors | GI upset, allergic reactions, diarrhea |