Essay answer for Diabetes management with flow charts under 800-1000 words
Reading File
Checking Date & Time
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
I now have sufficient information to write a comprehensive essay. Let me compose it with embedded flowcharts.
Diabetes Mellitus: Management
Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by persistent hyperglycemia resulting from defects in insulin secretion, insulin action, or both. Management is multifaceted, combining lifestyle modification, pharmacotherapy, glycemic monitoring, and complication surveillance. The goals are to maintain fasting blood glucose within 90–130 mg/dL and hemoglobin A1c (HbA1c) below 7%, thereby preventing microvascular and macrovascular complications. — ROSEN's Emergency Medicine
1. Classification and Initial Assessment
Two major forms guide therapeutic decisions:
- Type 1 DM — absolute insulin deficiency (autoimmune β-cell destruction); insulin is mandatory from diagnosis.
- Type 2 DM — relative insulin deficiency with peripheral resistance; managed stepwise, beginning with lifestyle changes and oral agents.
Baseline workup includes HbA1c (gold standard for chronic glycemia; reflects average glucose over 2–3 months), fasting glucose, renal function, lipid panel, urine microalbumin, and fundoscopy. — Harrison's Principles of Internal Medicine 22E, Goldman-Cecil Medicine
2. Non-Pharmacological Management
Lifestyle intervention is the foundation for both types but is particularly central in Type 2:
- Diet: High-fiber, low-glycemic index foods; caloric restriction in obese patients.
- Exercise: Regular physical activity reduces insulin resistance; must be balanced with caloric intake and drug regimens.
- Weight management: Even modest weight loss (5–10%) substantially improves glycemic control in Type 2 DM.
- Bariatric surgery: Considered in severe obesity with inadequately controlled Type 2 DM when pharmacotherapy fails. — Guyton and Hall Textbook of Medical Physiology
3. Pharmacological Management
Type 1 DM — Insulin-Dependent
Insulin is non-negotiable. Regimens follow a basal-bolus strategy:
┌─────────────────────────────────────────────────────────┐
│ TYPE 1 DM MANAGEMENT FLOWCHART │
└─────────────────────────────────────────────────────────┘
Newly Diagnosed Type 1 DM
│
▼
Start Insulin (Basal-Bolus Regimen)
• Basal: Glargine / Detemir (once daily)
• Bolus: Aspart / Lispro (pre-meals)
│
▼
Monitor Blood Glucose + HbA1c
(Target HbA1c < 7%; FBG 90–130 mg/dL)
│
┌─────┴──────┐
Achieved? Not Achieved?
│ │
Continue + annual Adjust dose /
complication consider CSII
screening (insulin pump)
Type 2 DM — Stepwise Pharmacotherapy
┌─────────────────────────────────────────────────────────────┐
│ TYPE 2 DM MANAGEMENT FLOWCHART │
└─────────────────────────────────────────────────────────────┘
Newly Diagnosed Type 2 DM
│
▼
Lifestyle Modification (Diet + Exercise)
± Metformin 500 mg OD/BD (first-line)
│
▼
Reassess HbA1c at 3 months
│
┌──────┴───────┐
HbA1c < 7% HbA1c ≥ 7%
│ │
Continue Add Second Agent
monitoring based on comorbidity:
│
┌──────────┴───────────┐
Heart Failure / No major
Renal Disease comorbidity
│ │
SGLT-2 inhibitor Sulfonylurea
(empagliflozin / (glipizide 5 mg OD)
canagliflozin) OR GLP-1 agonist
│ │
└──────────┬───────────┘
▼
Still not at target?
│
▼
Add/Intensify Insulin
Key oral agent classes (ROSEN's Emergency Medicine):
| Class | Mechanism | Example | Notes |
|---|---|---|---|
| Biguanides | ↓ hepatic glucose output | Metformin | First-line; no weight gain; avoid in severe CKD |
| Sulfonylureas | ↑ insulin secretion | Glipizide, Gliburide | Risk of hypoglycemia |
| SGLT-2 inhibitors | ↑ urinary glucose excretion | Empagliflozin | CV and renal mortality benefit |
| GLP-1 agonists | ↑ glucose-dependent insulin | Liraglutide | Weight loss; useful in obesity |
| Thiazolidinediones | ↓ insulin resistance | Pioglitazone | Risk of fluid retention |
| α-Glucosidase inhibitors | ↓ glucose absorption | Acarbose | GI side effects |
4. Monitoring and Targets
HbA1c should be measured every 3 months until stable, then every 6 months. Self-monitored blood glucose (SMBG) guides daily dose titration. Continuous glucose monitoring (CGM) is preferred in Type 1 and insulin-dependent Type 2 patients. The ADA recommends:
- HbA1c < 7% (or < 7.5% in older adults with comorbidities)
- Fasting glucose: 80–130 mg/dL
- 2-hour postprandial: < 180 mg/dL
5. Complications and Organ-Specific Management
┌──────────────────────────────────────────────────┐
│ DIABETIC COMPLICATIONS FLOWCHART │
└──────────────────────────────────────────────────┘
Diabetes Diagnosis
│
┌───────────┼────────────┐
▼ ▼ ▼
Nephropathy Retinopathy Neuropathy
│ │ │
Microalbumi- Annual Peripheral:
nuria screen fundoscopy Pregabalin /
│ │ Duloxetine
ACEi/ARB + Refer to Autonomic:
BP control ophthalmol. Symptomatic Rx
│ │ │
└───────────┼────────────┘
▼
Macrovascular Disease
(CAD, Stroke, Peripheral
Arterial Disease)
│
Statin + Antiplatelet
+ BP control (<130/80)
- Nephropathy: ~50% of end-stage renal disease in the US is diabetic in origin. ACE inhibitors reduce microalbuminuria and slow progression. — ROSEN's Emergency Medicine
- Retinopathy: Leading cause of adult blindness; severity directly related to glycemic control. Background retinopathy → proliferative retinopathy → vitreous hemorrhage. Referral to ophthalmology is mandatory.
- Neuropathy: Peripheral symmetric neuropathy affects 15–60% of patients. First-line analgesics: duloxetine 60 mg/day or pregabalin up to 600 mg/day, both achieving meaningful pain control in >50% of patients.
6. Inpatient and Special Situations
- Inpatient hyperglycemia: Target blood glucose 140–180 mg/dL; use insulin infusion in critically ill patients.
- CKD patients: Avoid metformin; use insulin ± GLP-1 agonists.
- Heart failure: SGLT-2 inhibitors (empagliflozin, canagliflozin) show documented mortality benefit.
- Perioperative period: Hold oral agents; use insulin sliding scale.
Conclusion
Effective diabetes management requires an individualized, stepwise approach: lifestyle modification → metformin monotherapy → combination oral agents → insulin, guided by HbA1c and glucose targets. Complication screening must be systematic and lifelong. SGLT-2 inhibitors and GLP-1 agonists have transformed Type 2 DM management by offering cardiovascular and renal benefits beyond glucose control alone. Sustained glycemic control remains the most powerful tool to prevent nephropathy, retinopathy, and neuropathy.
References:
- ROSEN's Emergency Medicine: Concepts and Clinical Practice — Management of Diabetes Mellitus
- Goldman-Cecil Medicine, International Edition — Inpatient Management of Diabetes
- Harrison's Principles of Internal Medicine, 22E (2025) — Glycemic Control and Complications
- Guyton and Hall Textbook of Medical Physiology — Type 2 Diabetes Management
Essay answer for ENT PG exam question "IntraTympanic Therapy" with diagrams under 800 words
Reading File
Checking Date & Time
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Searching Images
intratympanic injection technique tympanic membrane middle ear
I now have comprehensive source material. Let me compose the full essay with diagrams.
Intratympanic Therapy
Introduction
Intratympanic (IT) therapy refers to the direct injection of pharmacological agents through the tympanic membrane into the middle ear space, from where the drug diffuses across the round window membrane (RWM) into the inner ear. This route bypasses the blood-labyrinthine barrier, achieving high local drug concentrations while minimizing systemic side effects. It has become a cornerstone in the management of inner ear disorders, particularly Ménière's disease and idiopathic sudden sensorineural hearing loss (ISSNHL).
Rationale and Pharmacokinetic Basis
The inner ear is an immunologically privileged site protected by the blood-labyrinthine barrier, analogous to the blood-brain barrier. Systemic administration of drugs results in poor cochlear penetration. The IT route exploits the round window membrane as the primary pathway for drug entry into the scala tympani, with the stapes footplate and bony otic capsule serving as minor secondary routes.
Table 158.1 — Major Communication Routes to and from the Inner Ear (Cummings Otolaryngology):
| Boundary | Internal Compartment | External Compartment |
|---|---|---|
| Round window membrane | Perilymph (scala tympani) | Middle ear |
| Stapes footplate | Perilymph (vestibule) | Middle ear |
| Blood-perilymph barrier | Perilymph | Blood |
| Cochlear aqueduct | Perilymph | CSF |
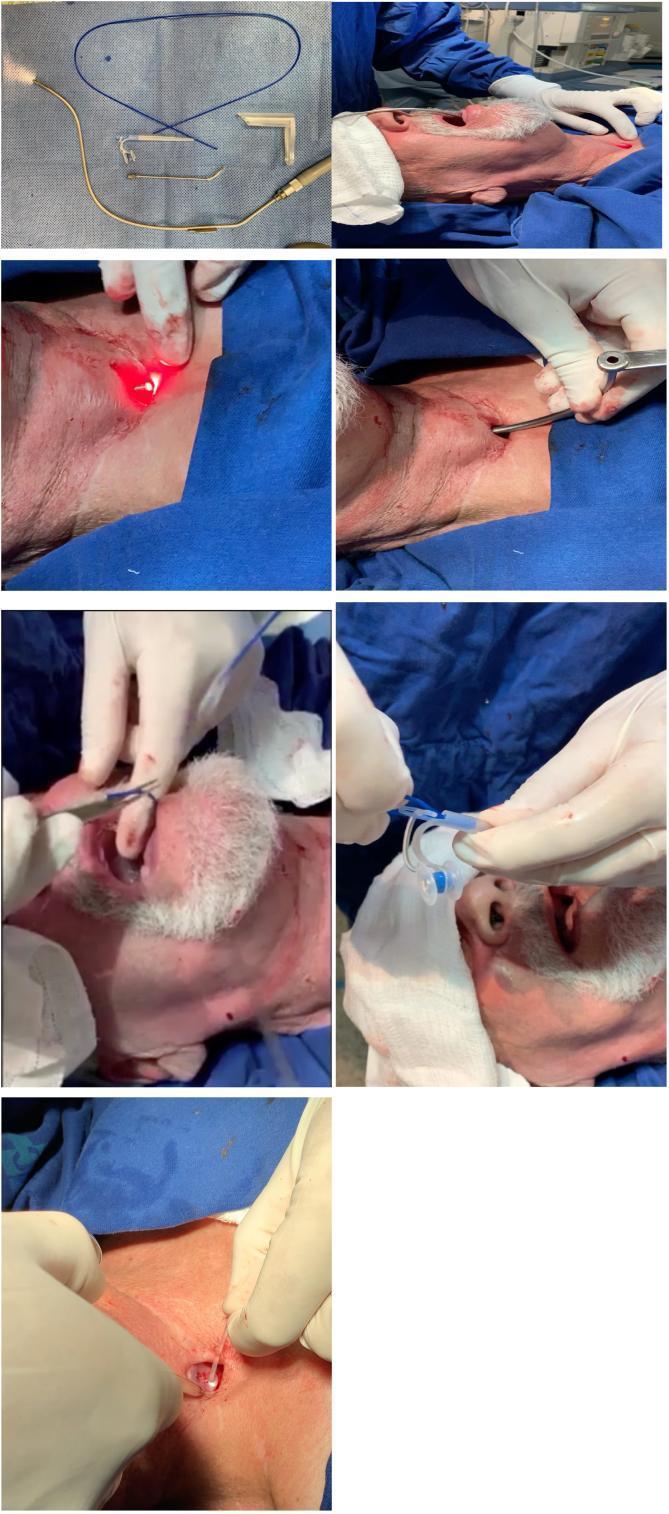
Technique of Intratympanic Injection
┌────────────────────────────────────────────────────────┐
│ INTRATYMPANIC INJECTION TECHNIQUE │
└────────────────────────────────────────────────────────┘
STEP 1: Patient positioned supine, head turned 45°
away from injected ear
STEP 2: Local anaesthetic (topical EMLA or
phenol) applied to tympanic membrane
STEP 3: Under microscopic or endoscopic guidance,
a 25–27 gauge needle inserted into the
POSTEROINFERIOR quadrant of TM
STEP 4: Drug injected slowly (0.3–0.5 mL)
filling the middle ear space
STEP 5: Patient instructed to remain still
with head tilted 45° for 20–30 mins
(promotes contact with round window)
STEP 6: Patient advised NOT to swallow or
yawn (prevents Eustachian tube
drainage of drug)
┌──────────────────┐
│ Tympanic │
│ Membrane │
│ │
│ [Posteroinferior│◄── Needle entry
│ quadrant] │
│ │
└────────┬─────────┘
│ Drug diffuses
▼
┌──────────────────┐
│ Middle Ear │
│ Space │
└────────┬─────────┘
│ Crosses
▼
┌──────────────────┐
│ Round Window │
│ Membrane │
└────────┬─────────┘
│
▼
┌──────────────────┐
│ Scala Tympani │
│ (Perilymph) │
└──────────────────┘
The image below shows actual needle placement through the tympanic membrane under endoscopic guidance:
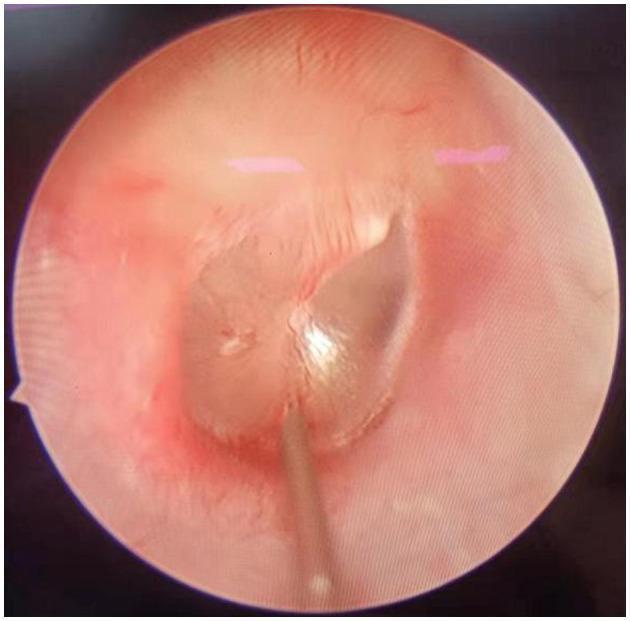
Agents Used and Their Indications
┌──────────────────────────────────────────────────────────────────┐
│ IT THERAPY — AGENTS & INDICATIONS │
└──────────────────────────────────────────────────────────────────┘
Intratympanic Therapy
│
┌────────────────┴────────────────┐
▼ ▼
CORTICOSTEROIDS AMINOGLYCOSIDES
(Dexamethasone / (Gentamicin)
Methylprednisolone)
│ │
┌─────┴──────┐ ┌─────┴──────┐
▼ ▼ ▼ ▼
ISSNHL Ménière's Ménière's Chemical
(primary Disease Disease labyrinthectomy
or salvage (vertigo (intractable (vestibular
therapy) control) vertigo) ablation)
1. Intratympanic Corticosteroids
Agents: Dexamethasone (4–24 mg/mL; typical 10 mg/mL) or methylprednisolone.
Mechanism: Anti-inflammatory; may modulate autoimmune inner ear disease; upregulates cochlear glucocorticoid receptors.
Indications:
- ISSNHL: Used as primary therapy or salvage treatment after failure of systemic steroids. Prospective RCTs confirm that IT methylprednisolone improves hearing in patients who fail oral steroids. — Cummings Otolaryngology
- Ménière's disease: Vertigo control achieved in 82% vs. 57% with saline placebo in a randomized trial. Effect on hearing and tinnitus is minimal. May require repeat injections every 3 months.
Protocol for Ménière's disease (Itoh and Sakata, first described 1987):
- 4–5 weekly injections of dexamethasone 2 mg/mL
- Vertigo relief in 80%; tinnitus reduction in 74%
2. Intratympanic Gentamicin
Mechanism: Selective vestibulotoxicity (hair cell destruction in cristae and maculae) with relative cochlear sparing — exploits the drug's higher affinity for vestibular vs. cochlear hair cells.
Indication: Intractable unilateral Ménière's disease with preserved useful hearing.
Outcomes (Cummings Otolaryngology):
- Wu & Minor: 90% complete vertigo control; profound SNHL in only 3%
- Nedzelski et al.: 83% vertigo control; 10% profound hearing loss
- Superior to dexamethasone for vertigo control (RCT, Casani et al.)
Current trend: Single-dose "titration therapy" — one injection with additional doses only if symptoms persist — to minimize cochleotoxicity risk.
Comparison of IT Agents
| Parameter | IT Steroid | IT Gentamicin |
|---|---|---|
| Mechanism | Anti-inflammatory / immunomodulatory | Vestibulotoxic (hair cell ablation) |
| Target symptom | SSNHL; vertigo (Ménière's) | Intractable vertigo (Ménière's) |
| Effect on hearing | Protective / restorative | Risk of SNHL (3–10%) |
| Effect on vertigo | 57–82% control | 83–90% control |
| Reversibility | Yes | Partially irreversible |
| Dosing | Repeat every 3 months | Single dose (titration) |
Complications
Complications of IT Therapy
├── Injection-related
│ ├── Tympanic membrane perforation (usually self-healing)
│ ├── Pain / discomfort at injection site
│ ├── Dizziness (transient, caloric effect of cold drug)
│ └── Otitis media (rare)
└── Drug-specific
├── Steroids: minimal; no documented ototoxicity
└── Gentamicin: sensorineural hearing loss (dose-dependent),
oscillopsia, imbalance (permanent in ablative cases)
Conclusion
Intratympanic therapy represents a precise, minimally invasive route to deliver therapeutic concentrations of drugs to the inner ear. Steroids (dexamethasone, methylprednisolone) are the cornerstone for ISSNHL and Ménière's disease with preserved hearing. Gentamicin offers chemical vestibular ablation for intractable Ménière's vertigo. The choice of agent, dosing protocol, and frequency must be individualized based on the clinical scenario, residual hearing status, and patient response. Advances in sustained-release formulations (e.g., OTO-104) promise more consistent drug delivery and reduced need for repeated injections.
References:
- Cummings Otolaryngology Head and Neck Surgery — Chapters 158, 167 (Intratympanic Drug Delivery; Ménière Disease)
- K.J. Lee's Essential Otolaryngology — Inner Ear, ISSNHL
- Harrison's Principles of Internal Medicine 22E — Sensorineural Hearing Loss
"Prosthetic Phonatory Devices" essay with images- diagrams under 800 words
Checking Date & Time
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching Images
tracheoesophageal voice prosthesis laryngectomy Provox diagram
Reading File
Reading File
Reading File
Searching Images
electrolarynx artificial larynx device neck placement phonation
Excellent — I now have rich textbook content and highly relevant images. Let me compose the full essay.
Prosthetic Phonatory Devices
Introduction
Total laryngectomy, performed for advanced laryngeal or hypopharyngeal carcinoma, results in permanent separation of the respiratory and digestive tracts and complete loss of the natural voice. Rehabilitation of voice is a critical component of post-laryngectomy care that profoundly affects quality of life. Three methods of alaryngeal voice rehabilitation are available: tracheoesophageal (TE) voice prostheses (gold standard), the artificial electrolarynx, and esophageal speech. Of these, prosthetic phonatory devices — particularly TE voice prostheses — represent the most effective and widely used approach. — Cummings Otolaryngology Head and Neck Surgery
Anatomy of Voice Rehabilitation After Laryngectomy
After total laryngectomy, the trachea is brought out as a permanent stoma in the anterior neck. The pharynx and upper esophagus form the neovibrator segment for sound production. A tracheoesophageal fistula (TEF) is surgically created between the posterior tracheal wall and the anterior esophagus to allow redirected airflow.
POST-LARYNGECTOMY ANATOMY
─────────────────────────
Oral cavity
│ (articulation)
Pharynx / PE segment ◄── Vibrates to produce voice
│
Oesophagus
│
┌────┴───────────────────┐
│ Tracheoesophageal │
│ Party Wall │
│ [TEP / Voice Prosth.] │◄── One-way valve
└────┬───────────────────┘
│ (exhaled lung air redirected)
Trachea
│
Tracheostoma
(permanent, anterior neck)
│
Lungs (air source)
1. Tracheoesophageal Voice Prosthesis (TEP / TEVP)
Mechanism
The tracheoesophageal puncture (TEP) technique, pioneered by Singer and Blom in 1980, involves creating a fistula between the posterior tracheal wall and the anterior esophagus. A one-way silicone valve prosthesis is placed in this tract. On digital occlusion of the stoma, exhaled pulmonary air is shunted through the prosthesis into the esophagus/hypopharynx, vibrating the pharyngoesophageal (PE) segment to produce voice. The valve prevents aspiration of food and fluid in the reverse direction. — Schwartz's Principles of Surgery; Cummings Otolaryngology
Types
| Type | Description | Example |
|---|---|---|
| Indwelling (clinician-changed) | Long-wearing; replaced by clinician in clinic every 3–6 months | Provox Vega, Blom-Singer Indwelling |
| Non-indwelling (patient-changed) | Patient-replaced; lower cost; shorter lifespan | Provox NID, Blom-Singer Classic |
TRACHEOESOPHAGEAL VOICE PROSTHESIS — KEY COMPONENTS
─────────────────────────────────────────────────────
Tracheal flange ──┐
├── Shaft (8 mm standard length)
Silicone valve ───┤ traverses party wall
└── Esophageal flange
← Trachea side │ Esophagus side →
(air IN) │ (air enters to vibrate PE segment)
│
One-way valve
(opens on exhalation,
closes at rest to prevent
liquid retrograde flow)
TEP Procedure — Primary vs. Secondary
- Primary TEP: Performed at the time of laryngectomy. Immediate VP fitting stabilizes the party wall and allows voicing by postoperative day 10.
- Secondary TEP: Performed ~4 weeks post-laryngectomy in the outpatient clinic under flexible esophagoscopy guidance, using a Seldinger-type trocar technique.
The puncture is placed ≤10 mm from the mucocutaneous border of the stoma to allow easy daily maintenance by the patient. — Cummings Otolaryngology, Box 111.1

Heat and Moisture Exchanger (HME)
Used in conjunction with the stoma, the HME filters, warms, and humidifies inhaled air — replacing the function of the nasal passages lost after laryngectomy. It can be housed within a peristomal hydrocolloid adhesive and retains ~60% of extra water loss through the stoma (≈500 mL/24 h more than normal nasal breathing). — Cummings Otolaryngology, Fig. 111.15
Outcomes
- TE voice achieves the gold standard for voice rehabilitation after laryngectomy.
- Voice production possible on the day of prosthesis insertion in many patients.
- Control of vertigo: >80% voice success rates in experienced centres.
- Cricopharyngeal spasm can impair fluency; treated with botulinum toxin injection percutaneously or endoscopically.
Complications
COMPLICATIONS OF TE VOICE PROSTHESIS
───────────────────────────────────────
Device complications
├── Candida biofilm → valve incompetence → leakage THROUGH prosthesis
├── Periprosthetic leakage (around the device, not through it)
├── TEP enlargement / fistula widening
└── Prosthesis dislodgement
Patient/anatomy complications
├── Cricopharyngeal spasm → poor voice fluency
├── Stomal stenosis
└── Pharyngeal stenosis (more common after fasciocutaneous reconstruction)
2. Artificial Electrolarynx
A battery-powered electromechanical device held externally against the neck or cheek. It generates a mechanical vibration that is transmitted transcutaneously into the pharynx and oral cavity, where it is articulated into speech.
Characteristics:
- Immediately usable postoperatively — no surgical procedure required
- Produces a monotone, robotic-sounding voice
- Used as a temporary bridge while awaiting TEP, or as long-term option in patients unable to undergo TEP
- Types: neck-type (most common) and intraoral (via tube into corner of mouth)
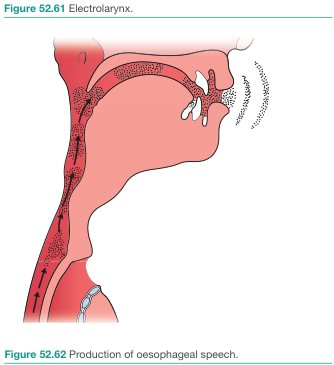
3. Esophageal Speech
Air is voluntarily swallowed or injected into the esophagus, then expelled in a controlled manner to vibrate the PE segment. No device or implant is needed. However, it requires up to one year of intensive training and produces a soft, low-pitched voice with limited phrase length. It is less commonly used today given the superiority of TE voice prostheses. — Cummings Otolaryngology
Comparison of Alaryngeal Voice Methods
| Feature | TE Voice Prosthesis | Electrolarynx | Esophageal Speech |
|---|---|---|---|
| Voice quality | Best — most natural | Robotic/mechanical | Soft, limited |
| Learning time | Same day | Immediate | Up to 1 year |
| Device required | Yes (implanted) | Yes (external) | No |
| Surgery needed | TEP required | No | No |
| Gold standard? | ✓ Yes | No | No |
| Longevity | 3–6 months per VP | Battery life | Lifelong once learned |
Conclusion
Prosthetic phonatory devices have transformed voice rehabilitation after total laryngectomy. The tracheoesophageal voice prosthesis (Provox, Blom-Singer) remains the gold standard, offering near-natural voice quality achievable on the day of insertion. The electrolarynx serves as an invaluable bridge device and long-term alternative where TEP is unsuitable. An experienced multidisciplinary team — surgeon, speech-language pathologist, and nurse specialist — is essential for optimal long-term outcomes. Ongoing innovations in sustained-release antifungal coatings and improved valve design aim to extend prosthesis lifespan and reduce leakage complications.
References:
- Cummings Otolaryngology Head and Neck Surgery — Chapters 111, 158 (Voice Rehabilitation; Prosthetic Voice)
- Schwartz's Principles of Surgery, 11th Ed. — Specific Considerations: Laryngectomy Speech Rehabilitation
- Bailey and Love's Short Practice of Surgery, 28th Ed. — Alaryngeal Speech Methods
This is a shared conversation. Sign in to Orris to start your own chat.