Clonazepam
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Here is a comprehensive overview of clonazepam drawn from multiple authoritative sources:
Clonazepam (Klonopin)
Class & Mechanism
Clonazepam is a high-potency, intermediate- to long-acting benzodiazepine. It acts as a positive allosteric modulator of GABA-A receptors, potentiating the inhibitory effect of GABA by increasing the frequency (not duration) of Cl⁻ channel openings. This enhances GABAergic synaptic inhibition throughout the CNS.
Unlike other 1,4-benzodiazepines, it has unusual potency against pentylenetetrazol-induced seizures while having minimal effect on maximal electroshock seizure patterns, suggesting particular efficacy against absence and myoclonic seizure types.
— Goodman & Gilman's Pharmacological Basis of Therapeutics
Pharmacokinetics
| Parameter | Value |
|---|---|
| Onset (peak plasma) | 1–4 hours after oral dose |
| Half-life | 20–60 hours (mean ~30 h; some sources cite 23–36 h) |
| Protein binding | ~85% |
| Metabolism | Hepatic — reduction of nitro group → inactive 7-amino derivatives; <1% excreted unchanged in urine |
| Steady state | Typically 5–8 days |
The manufacturer recommends divided dosing due to the intermediate half-life. Every-other-day dosing is not recommended for tapering because fluctuating plasma levels can precipitate inter-dose withdrawal.
Indications
Approved uses:
- Seizures: Absence seizures, myoclonic seizures (especially in children), Lennox-Gastaut syndrome
- Acute repetitive seizures (intranasal spray — orphan drug designation)
- Panic disorder (FDA-approved in adults)
Off-label / additional uses:
- Anxiety disorders (generalized anxiety, social phobia)
- Restless legs syndrome / REM sleep behavior disorder (first-line; if non-responsive, melatonin or pramipexole may be added)
- Acute mania (adjunct)
Dosing
Adults (seizures/panic):
- Initial: 1.5 mg/day PO in 3 divided doses
- Titrate: increase by 0.5–1 mg every 3–7 days
- Maximum: 20 mg/day
Pediatric (<10 years or <30 kg):
- Initial: 0.01–0.03 mg/kg/day PO ÷ BID–TID (max 0.05 mg/kg/day initially)
- Titrate: increase 0.25–0.5 mg/day every 3 days
- Maximum: 0.2 mg/kg/day ÷ TID
Therapeutic drug monitoring (not well established): proposed trough level 20–80 ng/mL, drawn within 30 min before oral dose.
— Harriet Lane Handbook, 23rd ed.
Adverse Effects
| System | Effects |
|---|---|
| CNS | Drowsiness, lethargy (~50% initially; tolerance often develops), ataxia, muscular incoordination, dysarthria, dizziness |
| Behavioral | Aggression, hyperactivity, irritability, poor concentration (especially in children) |
| Metabolic | Anorexia or hyperphagia |
| Respiratory | Increased bronchial/salivary secretions (problematic in children); respiratory depression with IV use |
| Cardiovascular | Cardiovascular depression (IV, especially when combined with other CNS depressants) |
| Hematologic | Thrombocytopenia, leukopenia (rare) |
| Psychiatric | Monitor for depression, suicidal ideation, unusual behavior changes |
Tolerability & Dependence
Tolerance to antiseizure effects typically develops within 1–6 months, after which some patients no longer respond at any dose.
Physical dependence can develop within days to weeks, even at prescribed doses (FDA boxed warning). Abrupt discontinuation can:
- Precipitate seizures (including status epilepticus)
- Cause a prolonged withdrawal syndrome lasting weeks to over 12 months (FDA warning)
Drug Interactions
- Carbamazepine, phenytoin, phenobarbital → decrease clonazepam levels (CYP3A4 inducers)
- CYP3A4 inhibitors (e.g., erythromycin) → increase clonazepam levels/toxicity
- CNS depressants (opioids, alcohol, other benzodiazepines) → additive respiratory/CNS depression
Contraindications
- Severe hepatic disease
- Acute narrow-angle glaucoma
- Caution: compromised respiratory function, porphyria, renal impairment
Tapering / Deprescribing
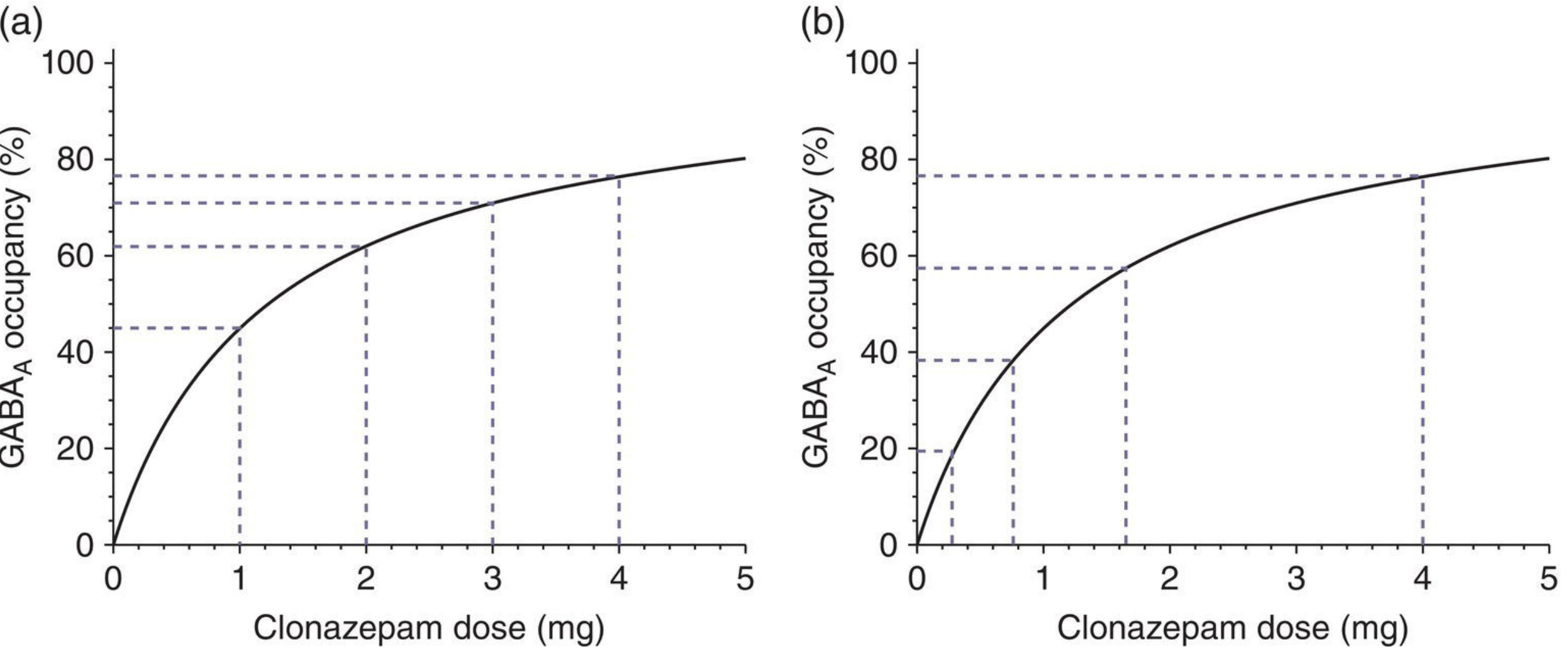
Due to the hyperbolic relationship between dose and receptor occupancy, linear dose reductions produce increasingly large reductions in effect at lower doses, risking more severe withdrawal. Guidelines recommend hyperbolic (exponentially decreasing) dose reductions to maintain equal-sized reductions in receptor occupancy at each step. Liquid formulations are often required for fine-grained tapering at low doses.
An alternative strategy is to switch to diazepam first (given its longer half-life and wide availability in liquid form), then taper from diazepam.
Key Comparisons Within Benzodiazepines
| Drug | Half-life | Protein Binding | Notes |
|---|---|---|---|
| Clonazepam | 20–60 h | 85% | High potency; nitro-reduction metabolism |
| Diazepam | 1–2 days (+ active metabolite ~60 h) | 99% | Active metabolite N-desmethyl-diazepam |
| Lorazepam | ~14 h | ~90% | Glucuronidation only; no active metabolites |
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics | The Maudsley Deprescribing Guidelines | Harriet Lane Handbook, 23rd ed. | Bradley & Daroff's Neurology in Clinical Practice | Kaplan & Sadock's Synopsis of Psychiatry
This is a shared conversation. Sign in to Orris to start your own chat.