Mechanisms of alpha and gamma interaction and how muscle spindle and muscle stretch reflex work
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching the Web
alpha gamma coactivation muscle spindle stretch reflex mechanism physiology
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Loading Image
Reading File
I now have comprehensive material from multiple authoritative textbooks with excellent diagrams. I can compose a thorough, well-illustrated answer.
Muscle Spindle, Stretch Reflex, and α–γ Coactivation
1. The Muscle Spindle: Structure and Innervation
The muscle spindle is a slender (3–10 mm) encapsulated proprioceptor embedded in parallel within the bulk of skeletal muscle. It contains 3–12 intrafusal muscle fibers — far smaller than the surrounding extrafusal (force-generating) fibers.
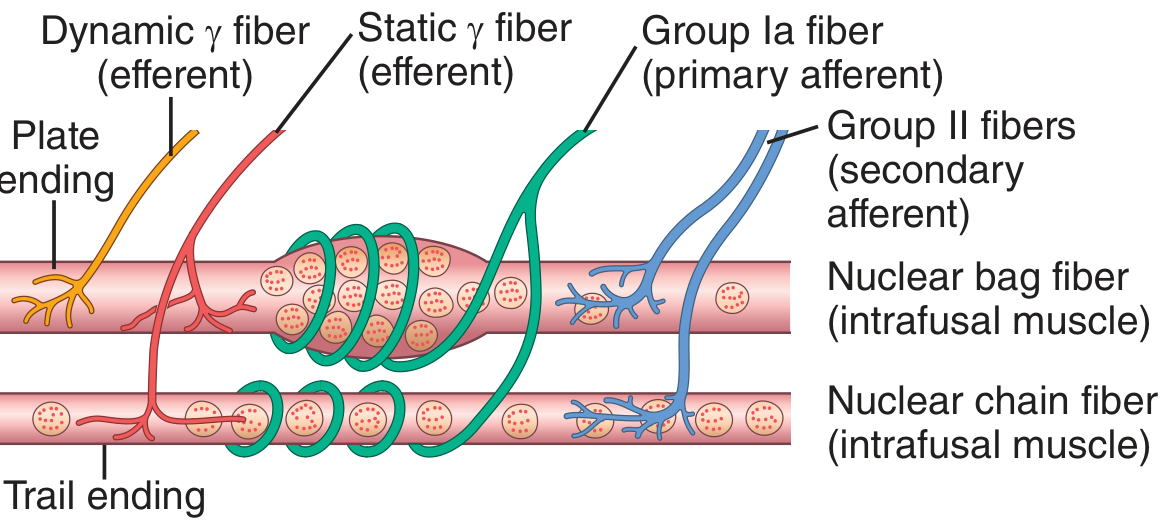
Fig. 55.4 — Guyton & Hall: nuclear bag and chain intrafusal fibers with their efferent (γ) and afferent (Ia, II) innervation.
Intrafusal fiber types
| Feature | Nuclear Bag Fibers | Nuclear Chain Fibers |
|---|---|---|
| Count per spindle | 1–3 | 3–9 |
| Nucleus arrangement | Clustered in a central "bag" | Single-file chain |
| Efferent input | γ-dynamic (γd) fibers → plate endings | γ-static (γs) fibers → trail endings |
| Sensory afferent | Primary (Ia) annulospiral wrapping | Primary (Ia) + secondary (II) flower-spray |
| Response speciality | Dynamic (velocity) response | Static (length) response |
Sensory endings
- Primary (annulospiral) ending — a Group Ia fiber (avg 17 µm, conduction 70–120 m/s) wraps around the central non-contractile equatorial region of both fiber types. It is highly sensitive to the rate of change of length (dynamic) and to sustained stretch (static).
- Secondary (flower-spray) ending — Group II fibers (avg 8 µm) flank the primary ending and respond mainly to static length. They project to interneurons in the cord rather than directly to α motor neurons.
Motor (efferent) innervation
The contractile polar ends of intrafusal fibers are controlled by small type Aγ (gamma) motor neurons in the anterior horn of the spinal cord — these are the γ-efferent fibers. About 31% of all motor fibers to a muscle are γ-efferents. They do not produce significant force in the whole muscle; their role is entirely to set spindle sensitivity. — Guyton & Hall, p. 679
2. How the Muscle Spindle Works as a Receptor
The spindle's central region has few/no contractile filaments — it is purely sensory. It is excited by two mechanisms:
- Passive stretch of the whole muscle → the equatorial region of the intrafusal fiber is elongated → Ia fibers fire faster.
- γ motor activation → the polar ends contract → the mid-region is stretched internally (even without any change in whole-muscle length) → Ia fibers fire faster.
Conversely, whole-muscle shortening (e.g., during α motor neuron activation) unloads the spindle → Ia firing falls or silences. This is the key problem that α–γ coactivation solves (see §4).
Under normal resting conditions, low-level tonic γ-efferent activity keeps the spindle under slight tension so it emits a continuous stream of Ia impulses — the spindle can signal both stretch (↑ firing) and unstretch (↓ firing). — Guyton & Hall, p. 678
3. The Muscle Stretch Reflex (Myotatic Reflex)
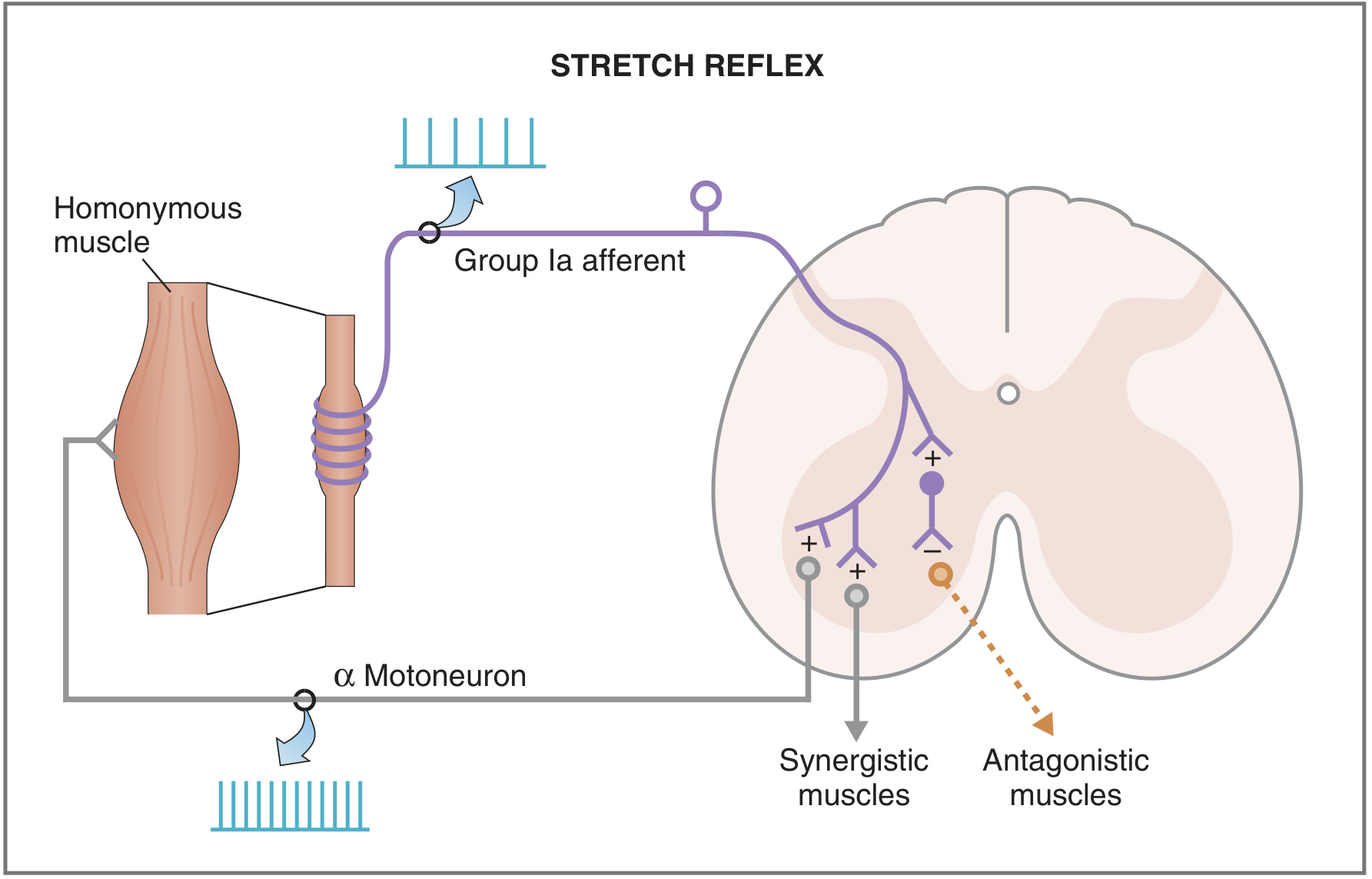
Fig. 3.33 — Costanzo Physiology: monosynaptic stretch reflex with reciprocal inhibition of antagonists.
Circuit
- Muscle is suddenly stretched (e.g., patellar tendon tap).
- Intrafusal fibers in the muscle spindle are stretched → Group Ia afferents fire at increased rate.
- Ia afferents enter via the dorsal root and make monosynaptic excitatory connections directly onto α motor neurons in the ventral horn that innervate the homonymous (same) muscle.
- α motor neurons fire → extrafusal fibers contract → muscle shortens → spindle unloads → Ia firing returns to baseline.
This is the only truly monosynaptic reflex in the body. — Costanzo Physiology, p. 111
Reciprocal innervation (Sherrington's law)
Simultaneously, Ia afferents synapse onto Ia inhibitory interneurons that project to α motor neurons of the antagonist muscle, causing it to relax. This prevents co-contraction from impeding the reflex movement.
Dynamic vs. static components
| Component | Mediator | Timing | Purpose |
|---|---|---|---|
| Dynamic stretch reflex | Primary (Ia) endings, nuclear bag fibers | Instantaneous, brief | Opposes sudden length changes; very strong during rapid stretch |
| Static stretch reflex | Primary + secondary (Ia + II) endings, nuclear chain fibers | Prolonged | Maintains constant degree of contraction against sustained load |
The dynamic reflex acts as a damper against sudden oscillations; the static reflex maintains postural tone. — Guyton & Hall, p. 679
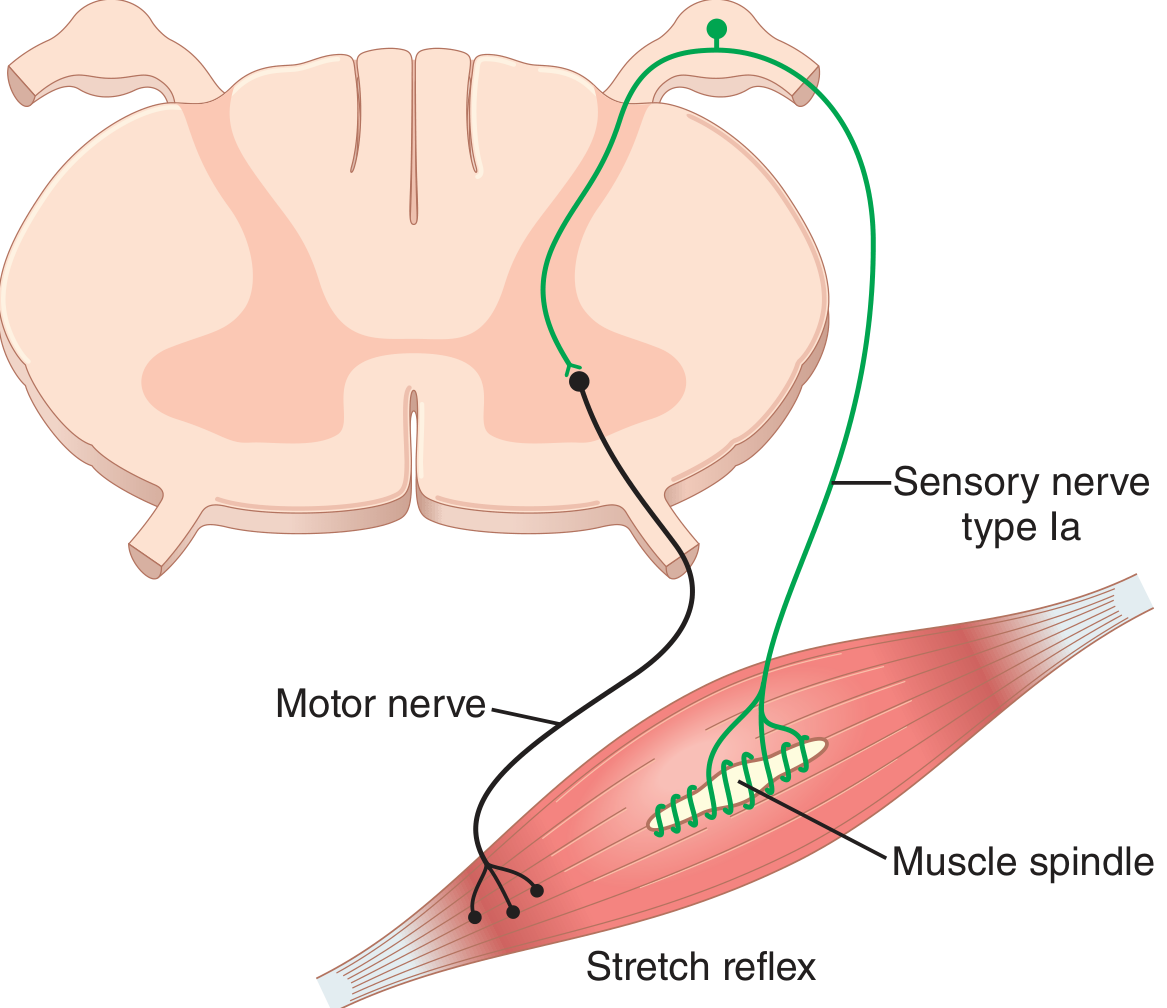
Fig. 55.5 — Guyton & Hall: monosynaptic Ia → α motor neuron loop.
4. α–γ Coactivation: The Core Mechanism
The problem it solves
When the brain commands a voluntary contraction, α motor neurons drive extrafusal fibers to shorten. As the muscle shortens, the spindle — arranged in parallel — is also physically shortened and unloaded. Without compensation, Ia firing would cease, and the spindle would lose all ability to signal any further stretch errors during the movement. The stretch reflex servomechanism would be blind during voluntary contraction.
The solution: simultaneous α and γ activation
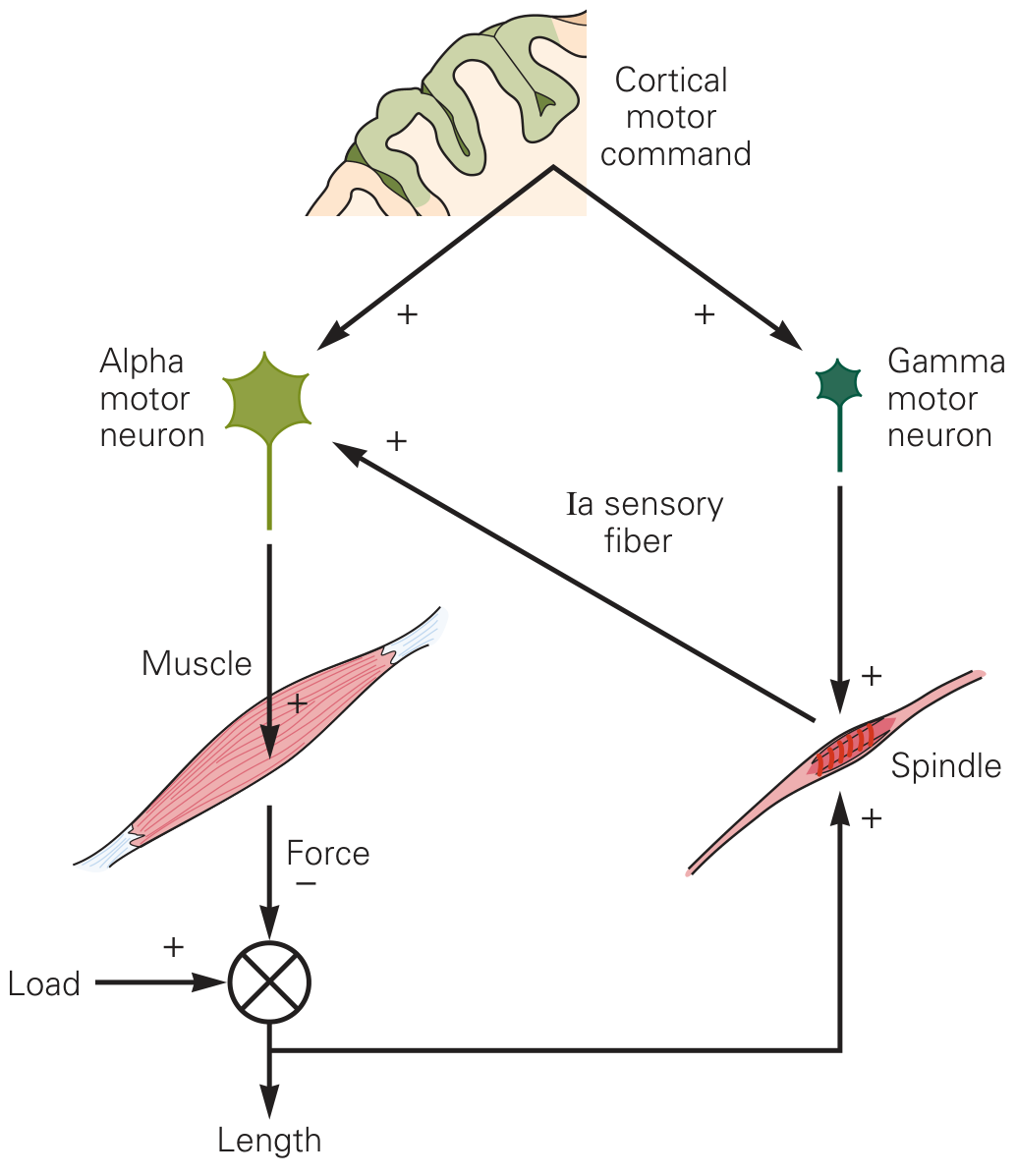
Fig. 32–10A — Kandel Principles of Neural Science: cortical motor command drives both α and γ motor neurons in parallel.
When the motor cortex (or any descending motor centre) issues a movement command, it simultaneously activates both α and γ motor neurons — a process called α–γ coactivation or α–γ coupling. The γ activation causes the polar ends of intrafusal fibers to contract at the same rate as the extrafusal muscle shortens. This keeps the equatorial region of the spindle taut at its correct resting length, maintaining continuous Ia discharge throughout the movement.
"Approximately 31% of all the motor nerve fibers to the muscle are the small type A gamma efferent fibers. Whenever signals are transmitted from the motor cortex or from any other area of the brain to the alpha motor neurons, in most instances the gamma motor neurons are stimulated simultaneously." — Guyton & Hall, p. 679
Functional consequences of α–γ coactivation
- Spindle sensitivity is maintained across all muscle lengths — the reflex loop stays operational throughout voluntary movement.
- Error detection during movement: if the actual muscle shortening is less than commanded (unexpected load), the intrafusal fibers (contracting via γ drive) will be stretched relative to the less-shortened extrafusal muscle → Ia firing increases → excites α motor neurons further → corrective force is added automatically.
- Error correction if overloading: if the muscle unexpectedly shortens too easily, intrafusal fibers slacken → Ia firing decreases → α motor neuron excitation falls → contraction is moderated.
- Damping of oscillations: prevents the limb from overshooting a desired position by providing continuous feedback correction.
Ragnar Granit's "servo-assist" hypothesis (1963)
Granit proposed that descending signals set a reference length by dialling both α and γ firing to the desired endpoint. The spindle then acts as a negative-feedback servomechanism, continuously correcting deviations from that reference. — Kandel Principles of Neural Science, p. 821
Microneurography evidence
In the late 1960s, Vallbo and Hagbarth developed microneurography (direct recording from peripheral nerve afferents in awake humans). Vallbo demonstrated that during slow voluntary finger flexion, Ia firing increased even as the muscle shortened — direct proof that γ activation maintains spindle sensitivity during α-driven contractions. — Kandel, p. 821
5. γ-Dynamic vs. γ-Static: Fine-Tuning the Response
| Parameter | γ-Dynamic (γd) | γ-Static (γs) |
|---|---|---|
| Intrafusal target | Nuclear bag fibers | Nuclear chain fibers |
| Effect | Dramatically enhances dynamic (velocity-sensitive) response of Ia | Enhances static (length-sensitive) response of Ia and II |
| Descending source | Primarily reticulospinal tract | Reticulospinal + corticospinal |
| Role | Velocity error detection; rapid reflex gain | Postural tone; sustained length maintenance |
The CNS can thus independently tune how "reactive" (dynamic gain) vs. how "tonic" (static gain) the spindle is for different motor tasks. — Guyton & Hall, p. 678
6. Brain Centers Controlling the γ System
- Bulboreticular facilitatory region (brain stem) — the primary driver; especially important for antigravity postural muscles, which have high spindle density.
- Cerebellum — modulates γ drive to smooth out movement trajectories.
- Basal ganglia — contributes to background tone.
- Cerebral cortex (motor cortex) — sends the descending α–γ co-activation signals during voluntary movement.
7. Clinical Relevance
| Finding | Mechanism |
|---|---|
| Hyperreflexia (upper motor neuron lesion) | Loss of descending inhibition → increased γ drive → over-sensitised spindles → exaggerated stretch reflexes |
| Hyporeflexia / areflexia (lower motor neuron / Ia afferent lesion) | Disruption of the Ia → α monosynaptic arc |
| Spasticity | Tonic enhancement of the static stretch reflex; velocity-dependent resistance to passive stretch |
| Clonus | Self-sustaining oscillation in the dynamic stretch reflex loop due to excessive gain |
| Rigidity (Parkinson's) | Abnormally elevated γ and α drive → lead-pipe or cogwheel resistance in all directions of passive movement |
| Deep tendon reflexes (DTR) | Clinical elicitation of the dynamic stretch reflex — graded 0–4+ as a measure of overall spinal cord excitability |
Summary
| Component | Role |
|---|---|
| Intrafusal fibers | Sensory organ; not significant force generators |
| Ia (primary) afferent | Reports both velocity and length; monosynaptically excites α motor neurons |
| II (secondary) afferent | Reports length; polysynaptic pathways; contributes to static reflex |
| α motor neuron | Drives extrafusal (force-generating) muscle fibers |
| γ motor neuron | Contracts intrafusal polar ends; sets spindle sensitivity |
| α–γ coactivation | Maintains spindle sensitivity during voluntary contraction; enables ongoing error correction |
| Stretch reflex arc | Monosynaptic negative-feedback loop resisting sudden length changes |
The key conceptual insight: the spindle is not just a passive stretch sensor — it is actively calibrated by the CNS through γ motor neurons, and the α–γ co-activation system transforms it into a real-time error-correction device for movement control.
Sources: Guyton & Hall Textbook of Medical Physiology (14th ed.), pp. 677–681; Kandel Principles of Neural Science (6th ed.), pp. 820–822; Costanzo Physiology (7th ed.), pp. 110–112.
This is a shared conversation. Sign in to Orris to start your own chat.