Regulation of insulin and diet
Reading File
Finding Sources
Finding Sources
Searching PubMed
"insulin secretion" AND "dietary regulation"
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have comprehensive information to provide a thorough answer. Here is a synthesis from multiple authoritative textbooks:
Regulation of Insulin and Diet
1. Basal Insulin Secretion
Fasting plasma insulin in normal humans is 0–70 μU/mL. The basal secretion rate is approximately 1 unit/hour, rising 5- to 10-fold after food ingestion — averaging ~40 units/day total. — Ganong's Review of Medical Physiology
2. Cellular Mechanism of Glucose-Stimulated Insulin Secretion
The β-cell is the primary glucose sensor of the body. The mechanism is a beautiful 8-step cascade:
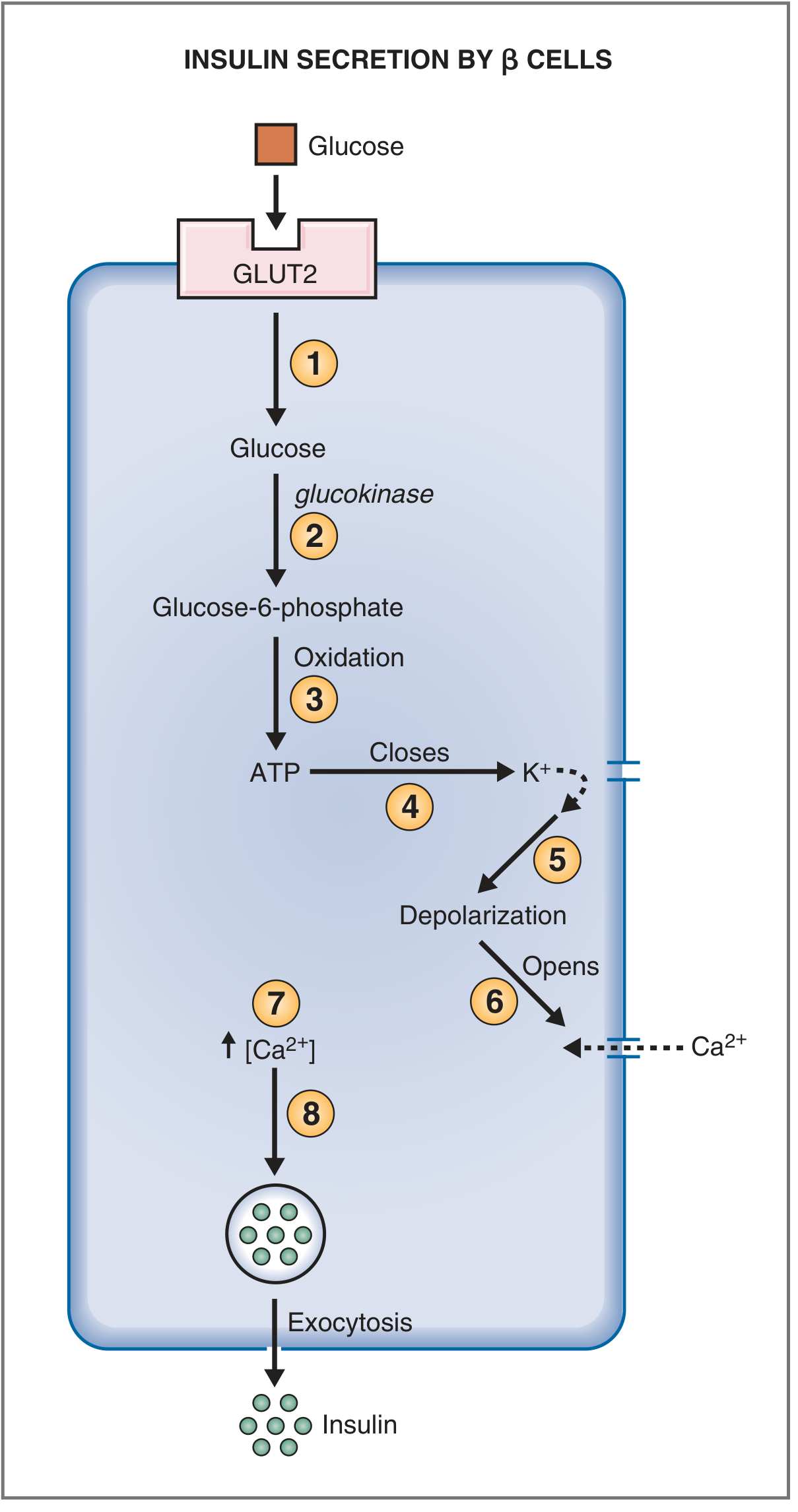
Fig. Mechanism of insulin secretion by pancreatic β cells stimulated by glucose. — Costanzo Physiology 7th Ed.
| Step | Event |
|---|---|
| 1 | Glucose enters via GLUT2 (facilitated diffusion; expressed only on β-cells and liver) |
| 2 | Glucokinase phosphorylates glucose → glucose-6-phosphate (rate-limiting sensor step) |
| 3 | Oxidation of G-6-P generates ATP |
| 4 | Rising ATP closes ATP-sensitive K⁺ channels |
| 5 | K⁺ efflux halts → membrane depolarizes |
| 6 | Depolarization opens voltage-gated Ca²⁺ channels |
| 7 | Ca²⁺ influx raises intracellular [Ca²⁺] |
| 8 | Ca²⁺ triggers exocytosis of insulin-containing granules |
Crucially, glucokinase acts as the "glucose sensor" — it has a low affinity for glucose (high Km), so its activity directly tracks blood glucose concentration.
The biphasic response to glucose is physiologically important:
- First phase (rapid, short-lived): release of pre-formed granules
- Second phase (slow, prolonged): newly synthesized and mobilized insulin
3. Dietary Macronutrients and Insulin
Carbohydrates (primary stimulus)
Blood glucose rising above ~100 mg/dL triggers insulin secretion that peaks at glucose levels of 400–600 mg/dL, reaching 10–25× basal levels. The feedback is rapid — secretion turns off within 3–5 minutes of glucose returning to fasting levels. — Guyton & Hall Textbook of Medical Physiology
Amino Acids (potentiating stimulus)
- Most potent: arginine and lysine
- Amino acids alone cause only modest insulin release
- But given alongside elevated glucose, they double glucose-induced insulin secretion
- This is physiologically important: dietary protein drives insulin secretion to promote amino acid uptake into muscle and protein synthesis
Fatty Acids and Ketoacids
- Elevated fatty acids and ketoacids also stimulate insulin secretion via metabolic pathways parallel to those used by glucose — Costanzo Physiology
4. Hormonal and Neural Regulators
Stimulatory Factors
| Factor | Mechanism |
|---|---|
| Glucose | ATP → K⁺ channel closure (primary) |
| Amino acids | Parallel metabolic pathways |
| GIP, GLP-1 (incretins) | Released by gut after a meal; amplify insulin response by ~2× |
| Glucagon | Gq protein → phospholipase C → IP₃ → ↑Ca²⁺ → exocytosis |
| Acetylcholine / vagus | Parasympathetic "cephalic phase" anticipatory response |
| Cortisol, GH, estrogen, progesterone | Potentiate glucose stimulus (chronic excess → β-cell exhaustion) |
| Sulfonylurea drugs | Close ATP-K⁺ channels directly, mimicking glucose depolarization |
| Obesity | Basal hyperinsulinemia due to insulin resistance |
Inhibitory Factors
| Factor | Significance |
|---|---|
| Fasting / hypoglycemia | Reduced glucose substrate |
| Somatostatin | Inhibits glucagon-stimulated exocytosis |
| α-Adrenergic agonists | Sympathetic stress response suppresses insulin (preserves glucose for fight-or-flight) |
| Exercise | Sympathetic activation reduces insulin; muscle uptake becomes insulin-independent |
| Diazoxide | Keeps ATP-K⁺ channels open; used clinically for insulinoma |
5. Incretins: The Gut-Brain-Pancreas Axis
GLP-1 and GIP are the key gut hormones released after carbohydrate/fat meals. They create an "anticipatory" insulin rise before glucose is even fully absorbed, enhancing insulin secretion and suppressing glucagon from α-cells. This incretin effect explains why oral glucose causes greater insulin release than intravenous glucose at the same blood glucose level. GLP-1 receptor agonists (semaglutide, liraglutide) and DPP-4 inhibitors exploit this pathway therapeutically. — Guyton & Hall
6. Insulin as the Master Anabolic Hormone: Fed vs. Fasting States
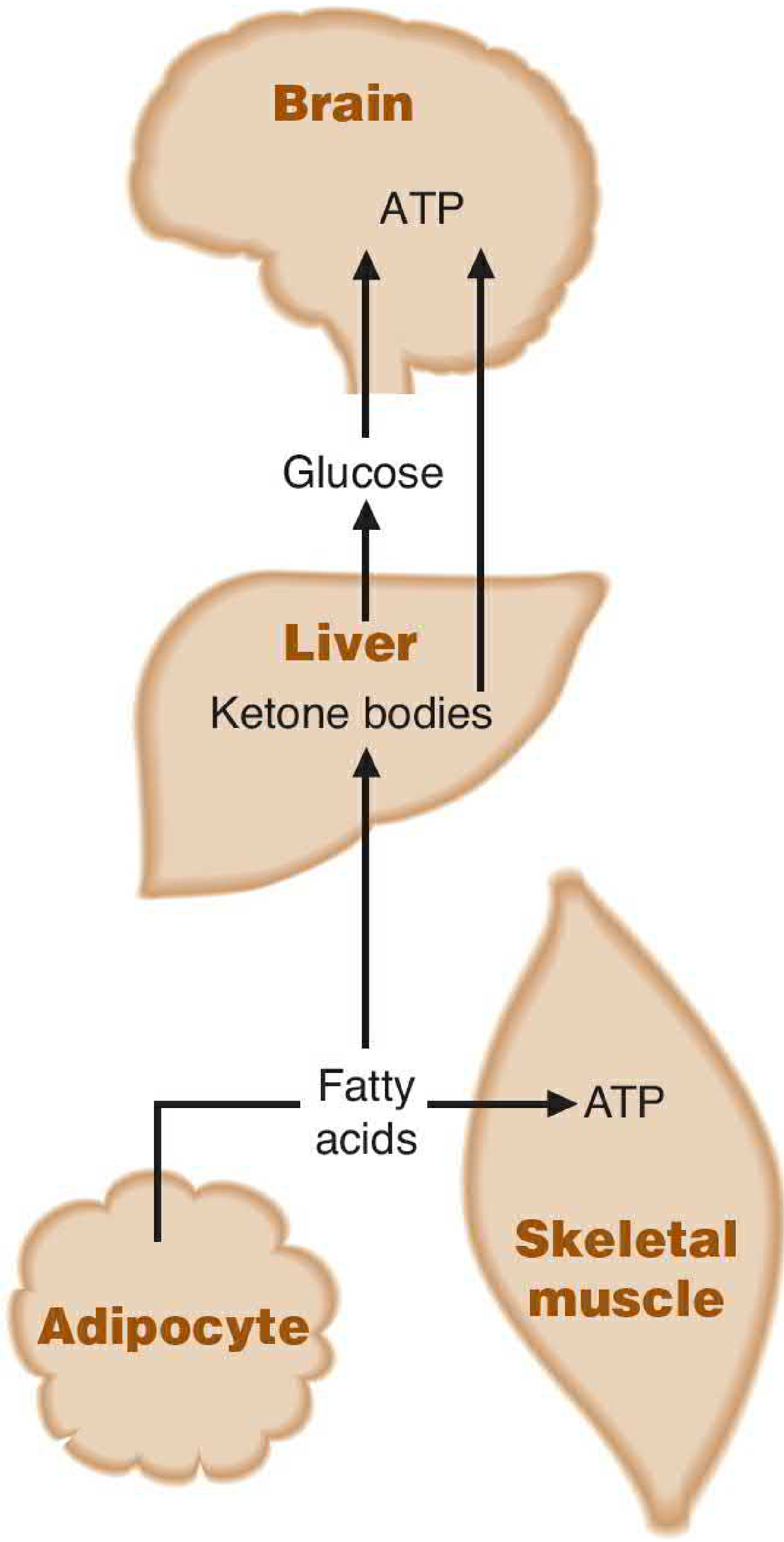
Fig. Fasting state: glucagon mobilizes fatty acids from adipose tissue; liver converts them to ketone bodies and releases glucose for the brain. — Basic Medical Biochemistry, 6th Ed.
| State | Insulin:Glucagon Ratio | Metabolic Priority |
|---|---|---|
| Fed (postprandial) | ↑↑ | Glucose uptake, glycogen synthesis, lipogenesis, protein synthesis |
| Fasting | ↓ | Glycogenolysis, gluconeogenesis, lipolysis, ketogenesis |
Insulin actions (fed state):
- Liver: promotes glycogen synthesis, active glycolysis, triglyceride synthesis
- Muscle: promotes glucose uptake (GLUT4 translocation), glycogen synthesis, protein synthesis
- Adipose: promotes glucose uptake, lipogenesis; inhibits hormone-sensitive lipase (stops lipolysis)
Glucagon actions (fasting state):
- Binds receptor → ↑cAMP → activates protein kinase A → phosphorylates key enzymes
- Opposes insulin's dephosphorylation effects on the same enzymes
- Mobilizes fatty acids from adipose → liver converts to ketone bodies for brain fuel during prolonged fasting
Blood glucose is defended tightly at 70–100 mg/dL despite widely varying carbohydrate intake. — Basic Medical Biochemistry – A Clinical Approach, 6th Ed.
7. Feedback Loop Summary
Dietary carbohydrate/protein/fat
↓
Blood glucose/amino acids ↑
↓
β-cells: GLUT2 uptake → glucokinase → ATP ↑
↓
K⁺ channels close → depolarization → Ca²⁺ influx
↓
Insulin secreted into portal blood
↓
Liver, muscle, adipose: glucose uptake & storage
↓
Blood glucose falls → insulin secretion turns off (3–5 min)
Key Clinical Correlates
- Type 2 diabetes: impaired β-cell response + peripheral insulin resistance → compensatory hyperinsulinemia → eventual β-cell exhaustion
- Acromegaly / Cushing's syndrome: chronic GH or cortisol excess → insulin antagonism → secondary diabetes risk
- Sulfonylureas: close ATP-K⁺ channels → insulin release independent of glucose (hypoglycemia risk)
- GLP-1 agonists / DPP-4 inhibitors: amplify the incretin effect, glucose-dependent (lower hypoglycemia risk)
- Low-carbohydrate diets: reduce postprandial glucose excursions → blunt insulin peaks → improved insulin sensitivity over time
Sources: Costanzo Physiology 7th Ed. | Guyton & Hall Medical Physiology | Ganong's Review of Medical Physiology 26th Ed. | Basic Medical Biochemistry – A Clinical Approach 6th Ed. | Robbins, Cotran & Kumar Pathologic Basis of Disease
My pediatrician told that we can’t give our 6 month old baby egg fish and non veg till the age of 1 year.
Reading File
Finding Sources
Finding Sources
Searching PubMed
"complementary feeding" AND "allergenic foods" AND infant
Searching the Web
WHO AAP guidelines egg fish introduction infant 6 months allergy prevention 2024
Finding Sources
Finding Sources
Reading File
Reading File
This is important — the evidence is clear and I have enough to give a thorough, well-cited answer. This advice from the pediatrician is outdated and potentially harmful. Here's the response:
This Advice Is Outdated — Current Evidence Says the Opposite
Your pediatrician's advice to delay eggs, fish, and non-vegetarian foods until 1 year is based on old guidelines from the 1980s–1990s that have since been reversed. Current evidence from multiple major medical organizations shows that early introduction of these foods actually reduces food allergy risk, while delaying them may increase it.
What the Old Thinking Was
In the 1980s and 1990s, experts believed delaying allergenic foods (eggs, fish, nuts) would prevent allergies. Following that advice, food allergy rates actually accelerated rather than declined. That alone prompted a major rethink. — Sleisenger & Fordtran's Gastrointestinal and Liver Disease
The Landmark Evidence That Changed Everything
The LEAP Trial (Learning Early About Peanut Allergy) — a large randomized controlled trial — showed:
- Infants 4–11 months who consumed peanut protein regularly had an 81% reduction in peanut allergy risk compared to those who avoided it
- This protection was maintained even after 12 months of stopping peanut
For eggs specifically, a meta-analysis found "moderate certainty" evidence that introducing egg between 4 and 6 months reduces egg allergy risk. For fish, findings were of "low certainty" but no evidence supports delaying it.
"There is no evidence that delayed introduction of allergenic solids is beneficial in prevention of food allergies." — Sleisenger & Fordtran's Gastrointestinal and Liver Disease
What Current Guidelines Actually Say
| Organization | Recommendation |
|---|---|
| AAP (American Academy of Pediatrics) | Withdrew the "delay allergenic foods" advice in 2008; now actively supports early introduction |
| NIAID (NIH) 2017 Guidelines | Introduce peanut and egg around 6 months, not before 4 months |
| USDA Dietary Guidelines 2020–2025 & 2025–2030 | Introduce eggs, fish, tree nuts, wheat, and shellfish alongside other complementary foods at ~6 months |
| AAAAI/ACAAI Consensus 2021 | Both peanut and egg should be introduced around 6 months for all infants |
| WHO | Start complementary foods at 6 months — no specific food group should be withheld |
A 2025 AAAAI study showed that when families received modern allergy prevention guidelines, egg allergy at age 1 dropped from 11.7% to 2.8% — by introducing eggs earlier rather than waiting.
The Biological Reason: Dual Allergen Hypothesis
Current science explains this through the dual allergen exposure hypothesis:
- Skin exposure to a food allergen (e.g., through eczema-damaged skin) before oral introduction → promotes allergic sensitization
- Oral introduction early in life → promotes immune tolerance through the gut mucosa
Delaying oral introduction while skin exposure happens passively is exactly what sets up allergies. — Sleisenger & Fordtran's GI & Liver Disease; Fitzpatrick's Dermatology
Practical Guidance for Your 6-Month-Old
At 6 months, when your baby is developmentally ready for solids (sitting with support, showing interest in food, loss of tongue-thrust reflex), you can introduce:
- Well-cooked egg (scrambled, mashed into puree) — start with a small amount
- Fish (well-cooked, boneless, flaked) — good source of iron, zinc, omega-3
- Well-cooked chicken, lentils, dal — excellent iron sources, especially important if breastfed
Important precautions:
- Introduce one new food at a time, 3–5 days apart, to identify any reaction
- If your baby has severe eczema or an existing food allergy, talk to a pediatric allergist before introducing peanuts specifically (other foods can still be introduced normally)
- Avoid honey (botulism risk) and cow's milk as a main drink until 1 year — these restrictions still stand
- Avoid whole nuts (choking hazard) — nut butters or powders are fine
- Avoid high-mercury fish (shark, swordfish, king mackerel) — opt for salmon, sardines, rohu, catla
What You Should Do
Have a direct conversation with your pediatrician and ask them to review the 2019 AAP Clinical Report on Allergenic Food Introduction and the NIAID 2017 Addendum Guidelines. It's possible they're following older training. If they remain firm without citing current evidence, a second opinion from a pediatric allergist or a more recently trained pediatrician is reasonable.
The goal at 6 months is a varied, nutrient-dense diet — eggs and fish are among the most valuable foods you can offer a growing infant, both nutritionally and immunologically.
This is a shared conversation. Sign in to Orris to start your own chat.