Difference between apoptosis and necrosis
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive comparison of apoptosis and necrosis, drawn from Robbins Pathologic Basis of Disease and Histology: A Text and Atlas.
Apoptosis vs. Necrosis
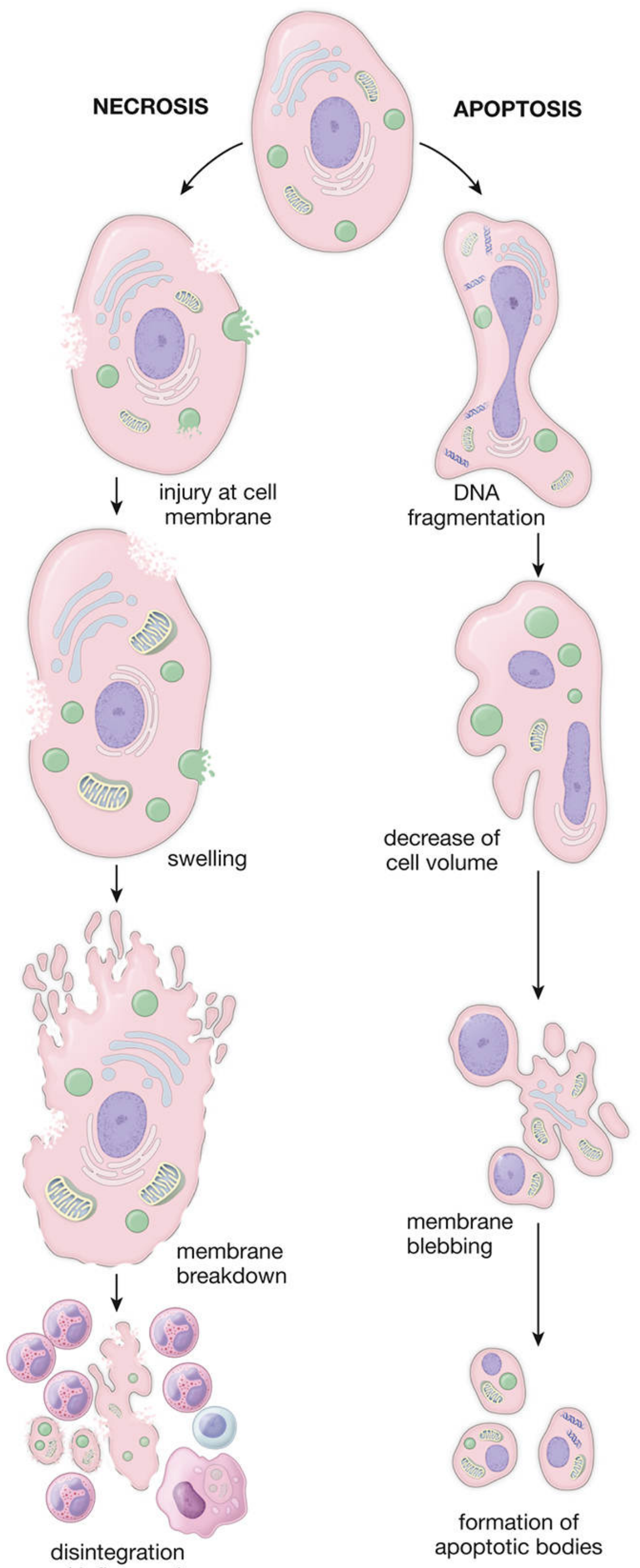
Fig. 3.18 — Histology: A Text and Atlas. Necrosis (left): cell swelling → membrane breakdown → disintegration. Apoptosis (right): DNA fragmentation → cell shrinkage → membrane blebbing → apoptotic bodies.
Overview
| Feature | Necrosis | Apoptosis |
|---|---|---|
| Nature | "Accidental," unregulated | "Programmed," regulated (suicide) |
| Cause | Severe, irreversible injury | Physiologic signals or DNA damage |
| Cell size | Enlarged (swelling) | Reduced (shrinkage) |
| Plasma membrane | Disrupted | Intact; altered lipid orientation |
| Nucleus | Pyknosis → karyorrhexis → karyolysis | Fragmented into nucleosome-sized pieces |
| Cellular contents | Enzymatic digestion; leak into surroundings | Intact; packaged into apoptotic bodies |
| DNA degradation | Random | Oligonucleosomal ("ladder" pattern) |
| Inflammation | Yes — prominent | No |
| End products | Cell debris phagocytosed by macrophages | Apoptotic bodies phagocytosed cleanly |
| Role | Usually pathologic | Often physiologic; sometimes pathologic |
Necrosis
Necrosis results when injury exceeds the cell's capacity for recovery. The sequence:
- Membrane injury → influx of water and extracellular ions
- Cell and organelle swelling
- Membrane breakdown → lysosomal enzymes spill into cytoplasm and extracellular space
- Disintegration → cell debris triggers an acute inflammatory response
Nuclear changes include:
- Pyknosis — nuclear shrinkage and condensation
- Karyorrhexis — nuclear fragmentation
- Karyolysis — nuclear dissolution
Patterns of tissue necrosis:
- Coagulative — ischemic infarcts (e.g., kidney, heart); preserved "ghost" outlines
- Liquefactive — brain infarcts and abscesses; tissue dissolves
- Caseous — tuberculosis; cheese-like, structureless debris
- Fat necrosis — pancreatic or traumatic fat injury; saponification
- Gangrenous — ischemia of a limb ± superimposed infection
Apoptosis
Apoptosis is caspase-dependent, genetically encoded cell death that eliminates unwanted or damaged cells without inflammation. The cell "dies with dignity."
Physiologic causes
- Embryologic development (tissue remodeling)
- Hormone-dependent involution (e.g., endometrial shedding, lactating breast regression)
- Cell turnover in epithelia (intestinal crypts)
- Elimination of autoreactive lymphocytes
- Neutrophil clearance after inflammation resolves
Pathologic causes
- DNA damage (radiation, chemotherapy)
- Viral infections (e.g., cytotoxic T-cell–mediated killing)
- Accumulation of misfolded proteins
Mechanisms
| Pathway | Trigger | Key mediators |
|---|---|---|
| Intrinsic (mitochondrial) | Growth factor withdrawal, DNA damage, ER stress | Cytochrome c release → caspase-9 → caspase-3 |
| Extrinsic (death receptor) | FasL/TRAIL binding Fas/DR | Caspase-8 → caspase-3 |
Both pathways converge on executioner caspases (caspase-3), which cleave structural proteins and activate DNases.
Morphologic sequence
- DNA fragmentation
- Cell shrinkage
- Membrane blebbing (intact membrane)
- Formation of apoptotic bodies (membrane-enclosed fragments)
- Phagocytosis by neighboring cells or macrophages — no inflammatory response
Key Conceptual Distinction
"Necrosis has historically been thought of as 'accidental' cell death… In contrast, apoptosis is 'regulated' cell death, mediated by defined molecular pathways that are activated under specific circumstances and kill cells with surgical precision, without inflammation or the associated collateral damage." — Robbins, Cotran & Kumar Pathologic Basis of Disease
Overlap
The distinction is not always absolute:
- Some regulated forms of necrosis exist (e.g., necroptosis, pyroptosis), mediated by defined genetic programs
- Apoptosis can progress to secondary necrosis if apoptotic bodies are not cleared
- Cytotoxic T-lymphocyte killing combines features of both (perforins + caspase-3 activation)
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease, Table 2.2; Histology: A Text and Atlas with Correlated Cell and Molecular Biology, Table 3.2 & Fig. 3.18
This is a shared conversation. Sign in to Orris to start your own chat.