Mitral Regurgitation
Reading File
Finding Sources
Searching PubMed
"mitral regurgitation"[MeSH Terms] AND management
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Now I have comprehensive information to provide a thorough overview of Mitral Regurgitation. Let me compile this into a well-structured answer.
Mitral Regurgitation (MR)
Definition & Overview
Mitral regurgitation is systolic backflow of blood from the left ventricle (LV) into the left atrium (LA) due to failure of the mitral valve to close competently. It is one of the most common valvular lesions and results from dysfunction of any component of the mitral valve apparatus: leaflets, chordae tendineae, annulus, or papillary muscles/LV wall.
Classification
Primary (Organic/Degenerative) MR
The valve leaflets themselves are structurally abnormal.
| Cause | Notes |
|---|---|
| Mitral valve prolapse (MVP) | ~90% of primary MR in the US; myxomatous degeneration |
| Ruptured chordae tendineae | Common cause of acute severe MR |
| Infective endocarditis | Leaflet destruction |
| Rheumatic heart disease | Leaflet thickening, fusion, scarring |
| Annular calcification | Common in elderly |
| Drug-induced | Pergolide, cabergoline, fenfluramine (high-dose, prolonged use) |
| Collagen vascular disease | e.g., Marfan, Ehlers-Danlos |
Secondary (Functional) MR
The valve leaflets are structurally normal — MR arises from LV or annular pathology causing leaflet malcoaptation.
- Dilated cardiomyopathy → papillary muscle displacement, apical tethering of leaflets
- Ischemic LV dysfunction → papillary muscle ischemia/infarction
- Atrial fibrillation → annular dilation causing malcoaptation
- ~75% of patients with heart failure with reduced ejection fraction have some degree of secondary MR; severe in ~20%
Pathophysiology — Three Phases
Phase A: Acute MR
The sudden regurgitant pathway "wastes" stroke volume into the LA. The LV is volume-overloaded, using the Frank-Starling mechanism maximally. Crucially, the regurgitant pathway unloads the LV in systole (ejection into the low-impedance LA), so EF appears supranormal — yet forward stroke volume is low.
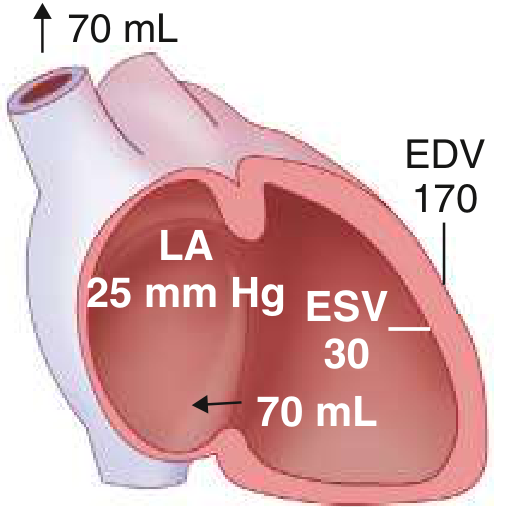
Phase B: Chronic Compensated MR
Eccentric LV hypertrophy develops; EDV increases substantially. Enlarged LA accommodates regurgitant volume at lower pressures. Forward stroke volume returns toward normal. Patient may remain asymptomatic even with severe MR.
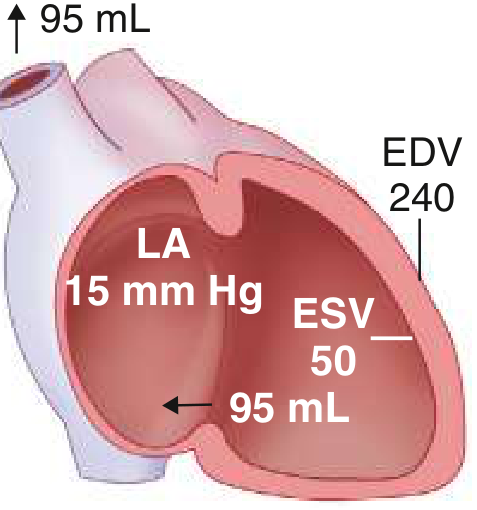
Phase C: Chronic Decompensated MR
Contractile dysfunction emerges. ESV rises markedly, forward SV falls. LV afterload increases as the enlarged LV radius raises wall stress. EF now falls — an EF of 55–60% in severe MR may actually represent significant myocardial dysfunction.
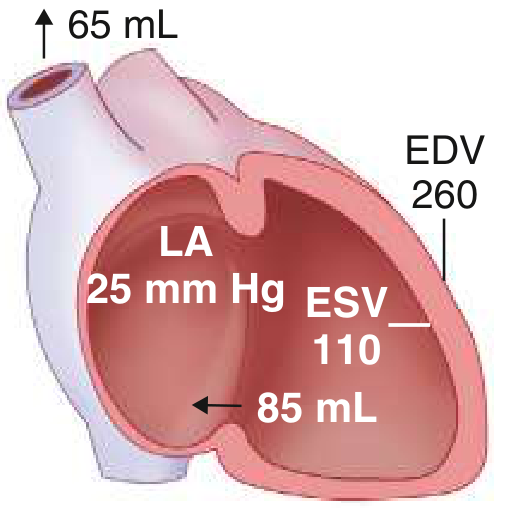
Key point: Reduced afterload occurs only in acute severe primary MR. In chronic MR, the increased LV radius returns wall stress to normal.
Clinical Features
Symptoms
- Often asymptomatic for years during compensation
- Exertional dyspnea and fatigue (earliest symptoms as LV function begins to decline)
- Orthopnea, PND, and pulmonary edema (advanced disease)
- Right-sided heart failure symptoms if pulmonary hypertension develops
- Acute severe MR: cardiogenic shock, flash pulmonary edema
Physical Examination
| Finding | Detail |
|---|---|
| Murmur | Holosystolic, best at apex, radiates to axilla; roughly correlates with severity but not reliable for grading |
| Apex beat | Displaced laterally and inferiorly (LV dilation) |
| S1 | Reduced intensity |
| S3 gallop | Present in severe MR — reflects rapid early filling of LV from enlarged LA; does not necessarily indicate HF |
| S2 splitting | Physiologically split (early A2 due to shortened LV systole) |
| Pulmonary HTN signs | Loud P2, RV lift, signs of right HF |
In acute MR: the large LA v-wave rapidly equilibrates LA and LV pressure, shortening/softening the murmur — the murmur can be deceptively soft despite severe regurgitation.
In secondary (functional) MR: the murmur may be unimpressive or even inaudible due to reduced LV contractile force.
Diagnosis
ECG
- Left atrial enlargement (notched P wave in II, biphasic P in V1)
- LV hypertrophy (voltage criteria)
- Atrial fibrillation (common complication)
Chest X-Ray
- Cardiomegaly (LV + LA enlargement)
- Pulmonary vascular congestion in decompensated states
- Absence of cardiomegaly suggests mild MR or acute onset
Echocardiography (Primary Investigation)
- TTE: LV and LA size/function, valve morphology, Doppler quantification of MR severity
- Color-flow Doppler: Identifies and semi-quantifies the regurgitant jet — but can over/underestimate severity, especially with eccentric jets
- Quantitative methods (preferred for clinical decisions):
- Vena contracta width
- PISA (Proximal Isovelocity Surface Area) — regurgitant volume and effective regurgitant orifice area (EROA)
- Pulmonary vein flow reversal (systolic flow reversal = severe MR)
- TEE: Superior valve anatomy, leaflet pathology, surgical planning; also used intraprocedurally
- 3D echocardiography: Pathoanatomic detail for surgical repair planning
Cardiac MRI
- Precise quantification of regurgitant fraction when echo is inconclusive
- Phase-contrast imaging: directly measures regurgitant volume
Cardiac Catheterization
- Reserved for cases where non-invasive assessment is inconclusive
- Coronary angiography if >40 years or ischemic symptoms suspected
Severity Grading (Echocardiographic)
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Vena contracta (cm) | <0.3 | 0.3–0.69 | ≥0.7 |
| EROA (cm²) | <0.2 | 0.2–0.39 | ≥0.4 |
| Regurgitant volume (mL) | <30 | 30–59 | ≥60 |
| Regurgitant fraction | <30% | 30–49% | ≥50% |
Management
Medical Therapy
Acute Severe MR:
- Goal: ↑ forward cardiac output, ↓ regurgitant volume
- Arterial vasodilators (nitroprusside): reduce SVR, preferentially increase forward flow
- Intra-aortic balloon counterpulsation: if hypotension precludes vasodilators (increases diastolic pressure, reduces afterload)
- Emergent surgical repair/replacement is often required
Chronic Symptomatic MR (inoperable patients):
- ACE inhibitors/ARBs (e.g., lisinopril 20 mg/day): reduce LV volume, improve symptoms
- Beta-blockers: observational benefit
- Diuretics: relieve congestion
- Rate/rhythm control if AF present; anticoagulation mandatory with AF
Chronic Asymptomatic MR:
- Vasodilators have not been shown to benefit patients with normal afterload
- Treat hypertension aggressively per guidelines
Secondary MR: Prioritize treatment of the underlying HF — GDMT (ACEI/ARB, beta-blocker, MRA, SGLT2i as appropriate)
Surgical Therapy
Indications (ACC/AHA 2020 Guidelines):
- Symptomatic severe primary MR with any LVEF
- Asymptomatic severe primary MR with:
- LVEF ≤60%, or
- LV end-systolic diameter (LVESD) ≥40 mm
- New-onset AF attributable to MR
- Pulmonary hypertension (PASP >50 mmHg at rest)
Mitral Valve Repair vs. Replacement:
- Repair is strongly preferred when technically feasible:
- Preserves LV geometry and function
- Lower operative mortality (~1%), better long-term survival
- No need for long-term anticoagulation (unlike mechanical valve)
- Annuloplasty ring ± leaflet/chordal procedures
- Replacement (MVR) indicated when leaflets are fused, calcified, have shortened/fused chordae, or when durable repair is not achievable
Transcatheter Edge-to-Edge Repair (TEER) — MitraClip
TEER replicates the Alfieri stitch — joining the A2 and P2 scallops to create a double-orifice valve, reducing regurgitant orifice without annular intervention.
Indications:
- Primary MR: prohibitive surgical risk (FDA approved)
- Secondary MR: symptomatic severe MR despite GDMT in HFrEF patients (LVEF 20–50%, LVESD <70 mm, PASP <70 mmHg) — shown to reduce HF hospitalizations and all-cause mortality vs. medical therapy alone (COAPT trial)
Outcomes:
- Reduction of MR to grade <2 in 84–93% of patients
- 30-day mortality: ~5–6% in high-risk elderly cohorts
- Significant improvement in NYHA class and QoL at 12 months
Complications
| Complication | Mechanism |
|---|---|
| Atrial fibrillation | LA dilation → substrate for re-entry |
| Pulmonary hypertension | Chronic elevation in LA/pulmonary venous pressure |
| Right heart failure | Secondary to pulmonary HTN |
| Infective endocarditis | Turbulent flow damages endothelium |
| LV systolic dysfunction | Chronic volume overload → decompensation |
| Sudden cardiac death | Rare; higher risk with MVP + thickened leaflets |
Prognosis
- Severe asymptomatic MR with preserved LV function: ~10% per year risk of developing symptoms, LV dysfunction, or AF
- Surgery before LVEF declines below 60% or LVESD exceeds 40 mm is associated with better postoperative outcomes and potentially reversible LV dysfunction
- Once LV contractile dysfunction develops, it may not be fully reversible even after surgery
Sources: Goldman-Cecil Medicine, p. 691–697 | Braunwald's Heart Disease | Harrison's Principles of Internal Medicine, 22e | Textbook of Clinical Echocardiography
This is a shared conversation. Sign in to Orris to start your own chat.