rheumatoid arthritis detailed
Reading File
Finding Sources
Searching PubMed
"rheumatoid arthritis"[MeSH Terms] AND treatment
Searching Images
rheumatoid arthritis joint pathology hands X-ray
Finding Sources
Finding Sources
Searching PubMed
"rheumatoid arthritis"[MeSH Terms] AND "biologic agents"[MeSH Terms]
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have comprehensive, authoritative content from multiple textbooks plus high-quality images. Let me compile the complete detailed answer.
Rheumatoid Arthritis — Detailed Overview
1. Definition
Rheumatoid arthritis (RA) is a chronic systemic inflammatory disease of unknown etiology that primarily targets synovial tissues. It produces a nonsuppurative proliferative and inflammatory synovitis that, if untreated, progresses to destruction of articular cartilage and, in some cases, joint fusion (ankylosis). Systemic inflammation underlies a variety of coexistent comorbidities including cardiovascular disease, osteoporosis, cognitive dysfunction, psychiatric disease, and cancer.
— Goldman-Cecil Medicine; Robbins & Kumar Basic Pathology
2. Epidemiology
| Feature | Detail |
|---|---|
| Global prevalence | 0.5–1% of adults |
| Sex ratio | ~3× more common in women (before menopause) |
| Age | Can occur at any age; peak incidence in the 3rd–5th decades |
| Incidence (women) | ~40 per 100,000/year |
| Incidence (men) | ~20 per 100,000/year |
| Genetic extremes | Near 0% in rural Nigerian cohorts; ~5% in Chippewa/Yakima/Inuit Native Americans |
Prevalence increases with each decade of life because RA is a lifelong disease. The incidence may be decreasing, perhaps reflecting reductions in smoking. Geographic and phenotypic variation likely reflect differences in genetics, the gastrointestinal or respiratory microbiome, and other environmental factors.
— Goldman-Cecil Medicine
3. Pathogenesis
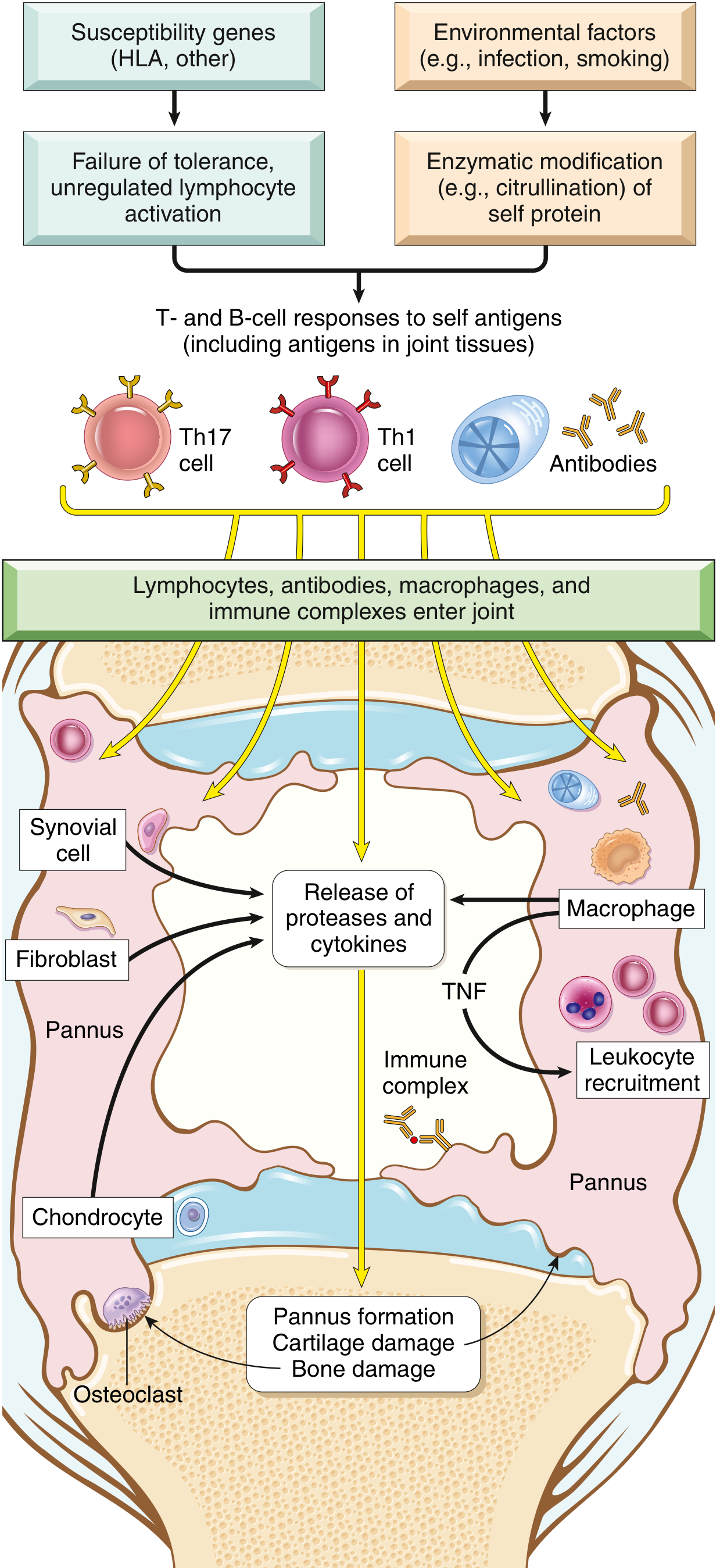
Fig. 19.34 — Major processes in the pathogenesis of rheumatoid arthritis (Robbins & Kumar Basic Pathology)
3a. Genetics
- Heritability is ~60%; concordance is 12–15% in monozygotic twins and 2–5% in dizygotic twins
- RA is a polygenic disorder; over 100 informative SNPs have been identified, most implicating immune genes
- ~40% of genetic risk accrues in the HLA region; key alleles include DRB*0401, DRB*0404, DRB*0101, DRB*1402
- HLA-DR4 is specifically associated with ACPA-positive RA
3b. Environmental Triggers
- Smoking is the best-established modifiable risk factor — promotes citrullination of self proteins
- Periodontitis (Porphyromonas gingivalis) and other infections can also drive citrullination
- Citrullinated epitopes (arginine → citrulline, posttranslational modification) are presented by HLA-DR4, creating neoantigens recognized by autoreactive T cells (molecular mimicry)
3c. Cellular & Molecular Mechanisms
T cells: CD4+ helper T cells (Th1 and Th17) react against joint antigens. Th17 cells produce IL-17, which recruits neutrophils and monocytes.
Macrophages: Release TNF, IL-1, and IL-6, the key cytokines that:
- Recruit and activate leukocytes
- Stimulate secretion of proteases (MMPs) that destroy hyaline cartilage
- TNF is considered the master mediator — the basis for the efficacy of TNF inhibitors
B cells & autoantibodies:
- Rheumatoid factor (RF): IgM or IgA autoantibodies against the Fc portion of IgG (~80% of patients); promote complement activation and macrophage activation via FcR binding
- Anti-citrullinated peptide antibodies (ACPA/anti-CCP): Detected in up to 70% of RA patients; targets include citrullinated fibrinogen, type II collagen, α-enolase, and vinculin; correlate with more severe erosive disease and extra-articular features
Fibroblast-like synoviocytes (FLS): Partially transformed cells with anchorage independence, loss of contact inhibition, and TLR expression — the main drivers of cartilage destruction at the cartilage-pannus junction, releasing MMPs (MMP1, MMP3, MMP13) in excess of their tissue inhibitors
Osteoclasts: Activated by RANKL (expressed on T cells), IL-1, TNF, and IL-17 → localize in periarticular bone → cause the characteristic marginal bone erosions visible on X-ray
Systemic effects: Circulating cytokines and immune complexes activate endothelium, accelerate atherosclerosis, drive systemic osteoporosis, and contribute to fatigue, cognitive impairment, and depression.
4. Morphology / Pathology
- Early: Synovium becomes edematous and hyperplastic (villous projections); infiltration by CD4+ T cells, plasma cells, macrophages, and mast cells; formation of lymphoid follicles with germinal centers
- Pannus: A destructive layer of fibrovascular granulation tissue that erodes cartilage and bone at joint margins
- Late: Fibrous or bony ankylosis (joint fusion)
- Articular cartilage: Progressive loss — joint space narrowing on X-ray
- Bone: Marginal ("punched-out") erosions, periarticular osteopenia
5. Clinical Features
Articular Manifestations
- Symmetric polyarthritis of small joints (PIPs, MCPs, wrists, MTPs) — hallmark presentation
- Morning stiffness lasting >1 hour (reflects inflammatory activity)
- Joints are warm, swollen, tender; range of motion restricted
- C1–C2 cervical involvement is common; subluxation (usually minor, occasionally causing cord compression and death)
- Temporomandibular, cricoarytenoid (hoarseness, dysphagia, rarely acute respiratory distress), and sternoclavicular joints may also be involved
Late deformities:
- Ulnar deviation of MCP joints
- Swan-neck deformity (PIP hyperextension, DIP flexion)
- Boutonnière deformity (PIP flexion, DIP hyperextension)
- Z-thumb deformity
- Baker (popliteal) cyst — synovial fluid dissects into the calf
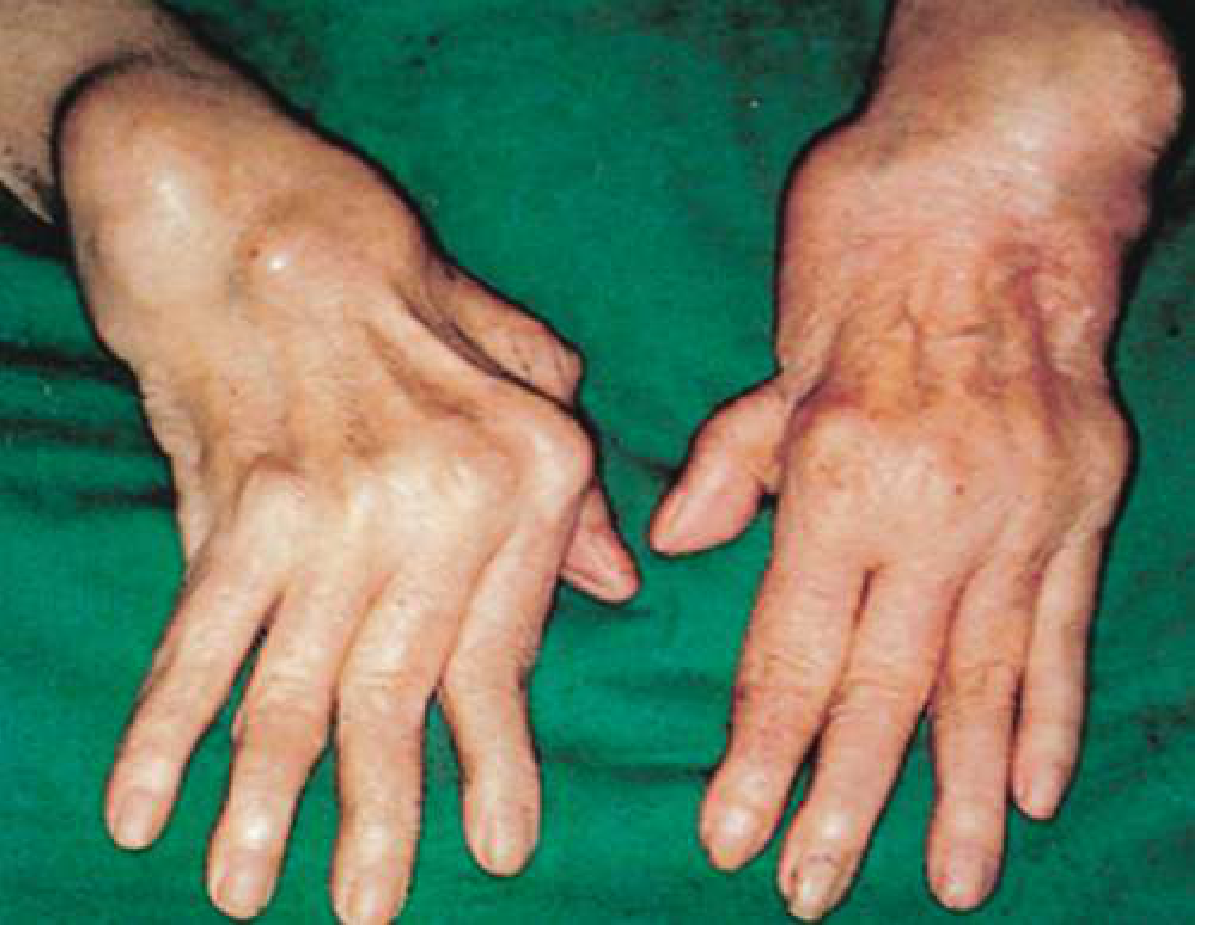
Severe advanced RA: massive tendon swelling over dorsal wrists, muscle wasting, ulnar deviation of MCPs, and swan-neck deformity (Goldman-Cecil Medicine, Fig. 243-3)
Extra-articular Manifestations
All are more common in RF/ACPA-positive patients:
| Organ/System | Manifestation |
|---|---|
| Skin | Subcutaneous nodules (~20%; extensor surfaces, joints, pressure points); palmar erythema; vasculitis |
| Eyes | Keratoconjunctivitis sicca (secondary Sjögren's); episcleritis; scleritis; scleromalacia perforans |
| Lungs | Interstitial lung disease (ILD); pleuritis; pleural effusion; rheumatoid nodules; Caplan syndrome (coal workers) |
| Heart | Accelerated atherosclerosis; pericarditis; pericardial effusion; valvular disease; conduction abnormalities; myocarditis |
| Blood | Anemia of chronic disease (proportional to disease activity); thrombocytosis; Felty syndrome (RA + splenomegaly + neutropenia) |
| Neurologic | Peripheral neuropathy; mononeuritis multiplex; cervical myelopathy (C1-C2 subluxation); carpal tunnel syndrome |
| Systemic | Fatigue, weight loss, low-grade fever; secondary amyloidosis (long-term) |
6. Laboratory & Investigations
| Test | Findings |
|---|---|
| Rheumatoid Factor (RF) | Positive in ~80%; also elevated in SLE, Sjögren's, infections, healthy elderly — not specific |
| Anti-CCP (ACPA) | Positive in ~70%; highly specific (~95%) for RA; may precede symptoms by years |
| ESR / CRP | Elevated, parallel disease activity |
| CBC | Normocytic normochromic anemia of chronic disease; thrombocytosis; neutropenia in Felty's |
| Synovial fluid | Inflammatory (WBC 5,000–50,000/mm³, predominantly neutrophils); poor mucin clot; low glucose |
| X-ray (hands/feet) | Periarticular osteopenia → joint space narrowing → marginal erosions → deformity/ankylosis |
| MRI | Most sensitive for early synovitis, bone edema (subclinical erosions), and tendon pathology |
| Ultrasound | Detects subclinical synovitis and Power Doppler signal |
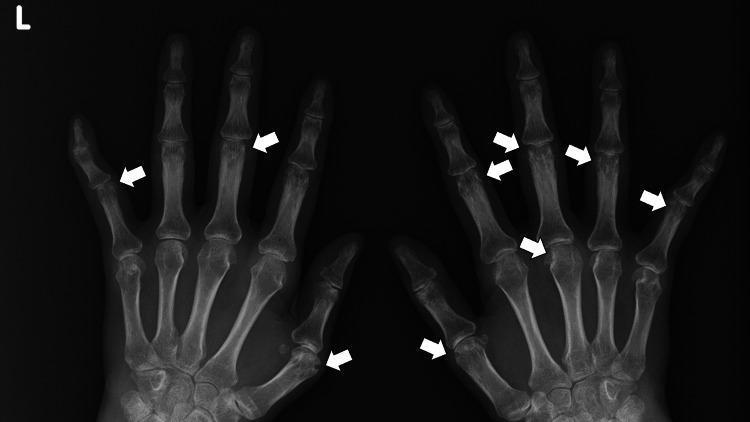
PA X-ray of both hands: symmetric marginal erosions at MCPs and PIPs, periarticular osteopenia, and joint space narrowing — characteristic of RA
7. Diagnosis — 2010 ACR/EULAR Classification Criteria
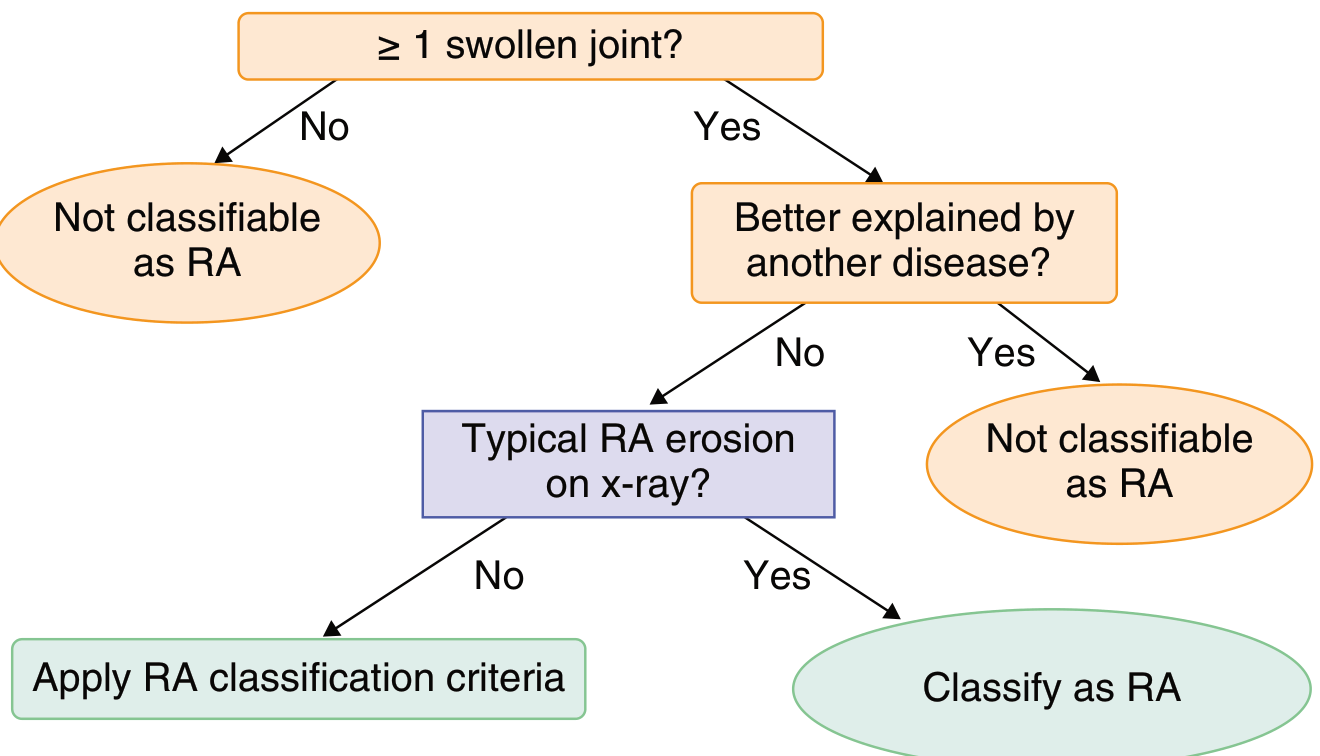
Score ≥ 6/10 = Definite RA
| Domain | Points |
|---|---|
| Joint involvement | 0–5 |
| 1 medium-large joint | 0 |
| 2–10 medium-large joints | 1 |
| 1–3 small joints | 2 |
| 4–10 small joints | 3 |
| >10 joints (≥1 small) | 5 |
| Serology | 0–3 |
| Neither RF nor ACPA positive | 0 |
| Low positive (>ULN, ≤3× ULN) | 2 |
| High positive (>3× ULN) | 3 |
| Acute phase reactants | 0–1 |
| Duration of synovitis | 0–1 (≥6 weeks = 1) |
(Data from Aletaha et al., Arthritis Rheum 2010;62:2569)
Differential Diagnosis
Conditions to exclude include: SLE, psoriatic arthritis, reactive arthritis, gout/pseudogout, OA, viral arthritis (parvovirus, hepatitis B/C), polymyalgia rheumatica, adult-onset Still's disease, and chronic tophaceous gout (especially in seronegative nodular disease).
8. Disease Activity Assessment
Several validated indices are used:
- DAS28 (Disease Activity Score — 28 joints) — most widely used in clinical practice
- SDAI (Simplified Disease Activity Index)
- CDAI (Clinical Disease Activity Index)
- ACR 20/50/70 — dichotomous improvement criteria used in clinical trials (20/50/70% improvement in joint counts, physician/patient global assessments, CRP/ESR, functional questionnaire)
- RAPID3 — patient-reported outcome measure
Treatment targets: Remission or low disease activity (LDA), guided by the "treat-to-target" strategy.
9. Treatment
9a. NSAIDs
Once the cornerstone of therapy, now considered adjunctive agents only. Inhibit COX-1 and COX-2. Minimize chronic use due to GI (gastritis, peptic ulcer), renal, and cardiovascular risks. Some patients respond preferentially to a particular NSAID.
9b. Glucocorticoids
- Bridge therapy: Low–moderate doses for rapid control while DMARDs take effect (weeks to months)
- Flare management: 1–2 week bursts, guided by severity
- Chronic low-dose: 5–10 mg/day prednisone for inadequate DMARD response — minimize to reduce osteoporosis, infection risk, metabolic effects
9c. Conventional (Synthetic) DMARDs (csDMARDs)
| Drug | Dose | Key Toxicities | Monitoring |
|---|---|---|---|
| Methotrexate (first-line) | 10–25 mg/week PO or SQ + folic acid 1 mg/day | Hepatotoxicity, myelosuppression, pneumonitis, teratogenicity | CBC, LFTs, creatinine every 2–3 months |
| Hydroxychloroquine | 200–400 mg/day (≤5 mg/kg) | Irreversible retinal toxicity, cardiotoxicity | OCT + visual fields annually |
| Sulfasalazine | 500 mg BID → 1000–1500 mg/day | Granulocytopenia, hemolytic anemia (G6PD) | CBC every 2–4 weeks × 3 months, then every 3 months |
| Leflunomide | 10–20 mg/day | Hepatotoxicity, myelosuppression, teratogenicity | CBC, LFTs every 2–3 months |
Triple therapy (methotrexate + hydroxychloroquine + sulfasalazine) has demonstrated efficacy comparable to some biologic combinations in early RA.
9d. Biologic DMARDs (bDMARDs)
TNF-α inhibitors (most-used class):
- Infliximab (3 mg/kg IV weeks 0, 2, 6, then q8 weeks)
- Etanercept (50 mg SQ weekly)
- Adalimumab, certolizumab pegol, golimumab
- Key risks: Serious infections (bacterial, fungal), reactivation of latent TB (screen before starting), lymphoma risk (controversial), drug-induced lupus, demyelination
Other biologics:
| Drug | Target | Notes |
|---|---|---|
| Abatacept | CD80/86 co-stimulation (T cell) | First-line alternative to TNF inhibitors |
| Rituximab | CD20 (B cells) | Preferred in lymphoma patients; interstitial lung disease |
| Tocilizumab / Sarilumab | IL-6 receptor | Effective monotherapy; can mask fever of infection |
| Anakinra | IL-1 receptor | Less commonly used due to daily injection and modest effect |
| Secukinumab/Ixekizumab | IL-17 | Less evidence in RA vs. PsA |
9e. Targeted Synthetic DMARDs (tsDMARDs) — JAK Inhibitors
- Tofacitinib (JAK1/3), Baricitinib (JAK1/2), Upadacitinib (JAK1), Filgotinib (JAK1)
- Oral; efficacy comparable to biologics
- Risks: infections, herpes zoster reactivation, venous thromboembolism, cardiovascular events, malignancy (post-marketing data; use with caution in patients >65, smokers, and those at high CV/thromboembolic risk)
9f. Treatment Algorithm ("Treat-to-Target")
Step 1: csDMARD (methotrexate ± HCQ ± SSZ) ± short-term glucocorticoids
↓ (if inadequate response in 3–6 months)
Step 2: Add bDMARD (TNF inhibitor first-line, or abatacept, tocilizumab)
OR tsDMARD (JAK inhibitor)
↓ (if inadequate response or toxicity)
Step 3: Switch within or between biologic classes; consider combination therapy
Goal: Remission or LDA. Reassess every 1–3 months until target reached, then every 3–6 months.
10. Monitoring & Complications
- Cardiovascular disease is the leading cause of premature death in RA — screen and aggressively manage CV risk factors
- Osteoporosis — DEXA scan, calcium/vitamin D supplementation, consider bisphosphonates in patients on chronic glucocorticoids
- Infections — immunosuppression increases risk of bacterial, fungal, and opportunistic infections; screen for TB/hepatitis B/C before biologics
- Malignancy — slightly elevated lymphoma risk (particularly in highly active disease); JAK inhibitors carry additional warnings
- Secondary amyloidosis (AA) — rare, complicates long-standing undertreated disease
11. Prognosis
Poor prognostic factors:
- High RF/ACPA titres
- Elevated CRP/ESR at baseline
- Early erosions on imaging
- Functional disability at presentation
- Extra-articular manifestations
- Smoking
With modern treat-to-target strategies and the biologic/JAK inhibitor era, remission is achievable in 20–40% of patients. Early aggressive treatment significantly reduces joint destruction and improves long-term functional outcomes.
Sources:
- Goldman-Cecil Medicine, International Edition — Ch. 243 (Rheumatoid Arthritis)
- Harrison's Principles of Internal Medicine, 22E (2025) — Ch. 370 (Table 370-2, DMARD monitoring)
- Robbins & Kumar Basic Pathology — Ch. 19 (RA pathogenesis and morphology)
- Robbins, Cotran & Kumar Pathologic Basis of Disease — Ch. 12
This is a shared conversation. Sign in to Orris to start your own chat.