Laurens classification of stomach cancer
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
Lauren classification gastric cancer intestinal diffuse histology
Searching PubMed
Lauren classification gastric cancer
I now have everything needed for a comprehensive answer. Let me compile it.
Laurén Classification of Gastric Cancer
Proposed by Pekka Laurén in 1965, this is the most widely used clinicopathological classification of gastric adenocarcinoma. It divides tumours into two primary types based on histological appearance, with a third mixed category added later.
1. Intestinal Type
| Feature | Detail |
|---|---|
| Architecture | Well-differentiated; forms recognizable gland-like tubular or papillary structures (resembles carcinoma elsewhere in the GI tract) |
| Gross appearance | Polypoid or ulcerating mass (Borrmann types I–III) |
| Precursor lesion | Arises on a background of chronic atrophic gastritis → intestinal metaplasia (Correa cascade) |
| H. pylori | Strongly linked — H. pylori drives the metaplasia–dysplasia sequence |
| Epidemiology | More common in males; incidence increases with age; predominant type in high-incidence regions (Japan, Korea, Eastern Europe) — suggesting environmental/dietary causation |
| Metastasis pattern | Tends to metastasize hematogenously to the liver |
| Prognosis | More favourable than diffuse type |
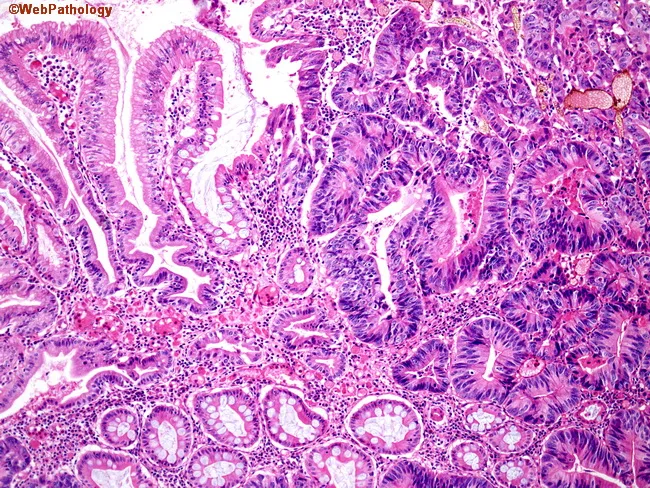
2. Diffuse Type
| Feature | Detail |
|---|---|
| Architecture | Poorly differentiated; lacks glands; composed of poorly cohesive single cells or tiny clusters infiltrating the gastric wall |
| Hallmark cell | Signet ring cells — intracellular mucin displaces the nucleus peripherally |
| Gross appearance | Diffuse infiltration without obvious mass; can cause linitis plastica (rigid, leather-bottle stomach — entire wall thickened and stiff) |
| Precursor | Not clearly associated with intestinal metaplasia or chronic gastritis |
| Molecular link | Associated with CDH1 mutations (loss of E-cadherin) — same pathway as lobular breast cancer; also associated with blood group A |
| Epidemiology | Equal sex distribution; affects a younger age group; found at similar frequency worldwide (not geographically clustered) |
| Metastasis pattern | Spreads transmurally and via lymphatics; frequently metastasizes to the peritoneal cavity (carcinomatosis) |
| Prognosis | Significantly worse than intestinal type |
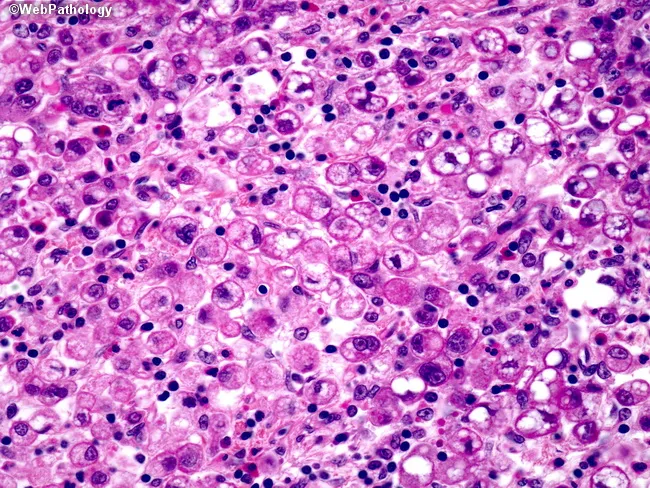
3. Mixed Type
- Contains both intestinal and diffuse components within the same tumour.
- Intermediate to poor prognosis.
- Constitutes a small but significant proportion of cases.
Comparison at a Glance
| Feature | Intestinal | Diffuse |
|---|---|---|
| Differentiation | Well → moderate | Poor |
| Gland formation | Yes | No |
| Signet ring cells | No | Yes (characteristic) |
| Gross pattern | Polypoid / ulcerating | Infiltrating / linitis plastica |
| Precursor | Intestinal metaplasia | None clearly defined |
| H. pylori link | Strong | Weak |
| Sex | Male > Female | Equal |
| Age at onset | Older | Younger |
| Blood group A | No | Yes |
| Metastasis | Liver (haematogenous) | Peritoneum (lymphatic/transmural) |
| Geographic distribution | Clustered (endemic regions) | Worldwide, uniform |
| Prognosis | Better | Worse |
Composite Histology & Gross Appearance
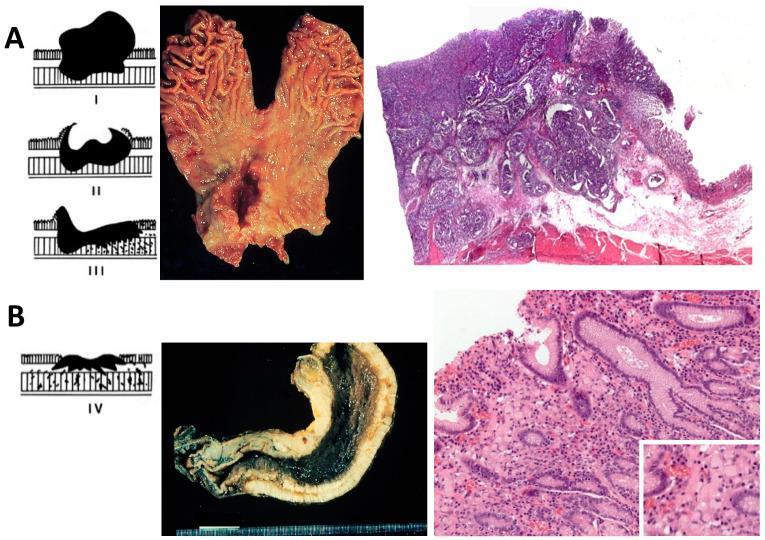
Relationship to Other Classifications
- WHO classification: Tubular and papillary carcinomas correspond to Laurén intestinal type; poorly cohesive carcinomas (including signet ring cell) correspond to diffuse type.
- Borrmann classification: Classifies advanced gastric cancer by gross morphology (types I–IV); broadly aligns but is separate from Laurén.
- Modified Laurén classification: Combines histologic type with tumour location (proximal vs. distal) → classifies as diffuse, proximal nondiffuse, or distal nondiffuse. Distal nondiffuse has the best prognosis; diffuse has the worst. This is a better predictor of survival than histology alone.
- TCGA molecular classification (2014): Defined 4 molecular subtypes — EBV+, microsatellite instable (MSI), genomically stable (GS), and chromosomal instability (CIN). GS subtype corresponds most closely to Laurén diffuse/signet ring; CIN to intestinal type.
Sources: Bailey & Love's Short Practice of Surgery 28e, p. 1192; Sabiston Textbook of Surgery; Sleisenger & Fordtran's Gastrointestinal and Liver Disease
T staging of gastric cancer
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
Searching Images
gastric cancer T staging depth of invasion wall layers diagram
Excellent — I have all the content and images needed. Here is the comprehensive answer:
T Staging of Gastric Cancer
Based on AJCC/UICC TNM Classification, 8th Edition (2017)
The T (Tumour) category describes the depth of wall invasion — the single most important determinant of resectability and prognosis. The gastric wall has five layers (mucosa → muscularis mucosae → submucosa → muscularis propria → serosa), and the T stage tracks how deeply the tumour penetrates these.
T Classification — Full Criteria
| Stage | Layer Invaded | Detail |
|---|---|---|
| Tis | Intraepithelial | Carcinoma in situ; tumour within epithelium without invasion of lamina propria; includes high-grade dysplasia |
| T1 | Mucosa or submucosa | Does not penetrate muscularis propria |
| T1a | Lamina propria or muscularis mucosae | Confined within mucosa |
| T1b | Submucosa | Invades through muscularis mucosae into submucosa |
| T2 | Muscularis propria | Invades through submucosa into muscular wall, but not beyond |
| T3 | Subserosa (subserosal connective tissue) | Penetrates through muscularis propria into subserosa without perforating the visceral peritoneum |
| T4 | Serosa or adjacent organs | Perforates outermost layers |
| T4a | Serosa (visceral peritoneum) | Tumour perforates/invades the peritoneal surface |
| T4b | Adjacent structures/organs | Invades liver, pancreas, spleen, colon, diaphragm, kidney, adrenal, small intestine, aorta, abdominal wall, etc. |
Note on T0 and TX: T0 = no evidence of primary tumour; TX = primary tumour cannot be assessed.
Diagram — Depth of Invasion with EUS Correlation
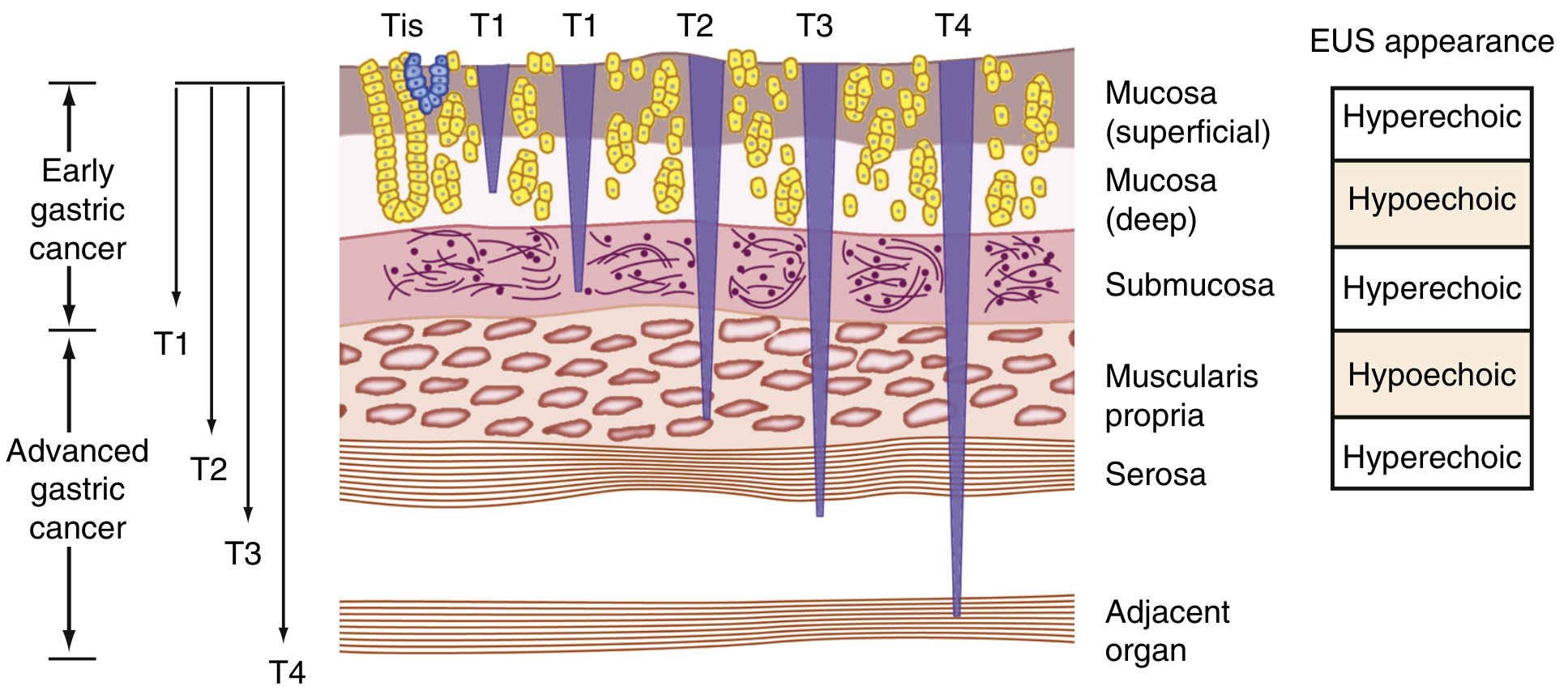
The five gastric wall layers alternate hyperechoic/hypoechoic on EUS. T1 = mucosa/submucosa; T2 = muscularis propria; T3 = subserosa; T4 = serosa or beyond.
Key Clinical Correlates
Early vs. Advanced Gastric Cancer
- Early gastric cancer (EGC): Tumour confined to mucosa or submucosa (T1), regardless of nodal status → 5-year survival >90% in Japan, >80% in the West
- Advanced gastric cancer (AGC): Tumour invades muscularis propria or deeper (T2–T4)
T3 vs. T4a — a Critical Surgical Distinction
- T3 = subserosa intact (serosa not breached) → potentially curative R0 resection more achievable
- T4a = serosa perforated → peritoneal seeding risk increases dramatically
- T4b = adjacent organ involved → may require extended (multivisceral) resection or may be unresectable
OGJ Tumours (8th Edition Change)
Tumours involving the oesophagogastric junction (EGJ) with epicentre ≤2 cm into the proximal stomach are staged as oesophageal cancer; EGJ tumours with epicentre >2 cm into the stomach are staged as gastric cancer.
Staging Modalities for T Assessment
| Modality | Role |
|---|---|
| EUS (Endoscopic Ultrasound) | Gold standard for T staging — visualizes all 5 wall layers; best for T1/T2 differentiation |
| MDCT (Multi-detector CT) | Used for T3/T4 and to assess adjacent organ involvement; limited for T1 vs T2 |
| MRI | Emerging alternative; intermediate signal in tumour on T1WI; limited routine use |
| Staging laparoscopy | Detects peritoneal disease not visible on CT, especially important in T3–T4 |
CT limitations: massive submucosal infiltration makes T1/T2 distinction difficult; subserosa and serosa are not separately visible, hindering T2 vs. T3 distinction. Cachexia and fat plane effacement further impede T4 assessment.
Overall Staging (AJCC 8th ed. — T component)
| Clinical Stage | T | N | M |
|---|---|---|---|
| IA | T1 | N0 | M0 |
| IB | T1 N1 or T2 N0 | M0 | |
| IIA | T1 N2, T2 N1, or T3 N0 | M0 | |
| IIB | T1 N3, T2 N2, T3 N1, or T4a N0 | M0 | |
| IIIA | T2 N3, T3 N2, or T4a N1 | M0 | |
| IIIB | T3 N3, T4a N2, or T4b N0–N1 | M0 | |
| IIIC | T4a N3 or T4b N2–N3 | M0 | |
| IV | Any T | Any N | M1 |
Sources: Bailey & Love's Short Practice of Surgery 28e (UICC 8th ed. table); Sleisenger & Fordtran's GI & Liver Disease, Fig. 54.8; Grainger & Allison's Diagnostic Radiology, Table 20.3 (AJCC 8th ed.)
This is a shared conversation. Sign in to Orris to start your own chat.