Penis enlargement
penile enlargement surgery augmentation
penis enlargement surgical procedures medical review 2024 2025
penile enlargement surgery techniques outcomes complications
Penile Enlargement: Surgical & Procedural Overview
Patient Selection & Psychological Considerations
Procedures for Penile Length Enhancement
1. Suspensory Ligament Release (Ligamentolysis)
- Gain: 1–3 cm in flaccid length; minimal erect length gain
- Key limitation: Does not create new erectile tissue — it advances existing tissue
- Complications: Loss of penile support angle during erection, hematoma, scarring, reduced ability to achieve upward erection
- A pubic fat pad reduction (lipoplasty) is often performed simultaneously to maximize apparent length
2. V-Y and Z-Plasty Skin Advancement
3. Ventral Phalloplasty / Scrotoplasty
4. Sliding Elongation of the Corpora Cavernosa
- Greater length gains but significantly higher complication rates including erectile dysfunction
5. Penile Disassembly / Total Phalloplasty
Procedures for Penile Girth Enhancement
1. Hyaluronic Acid (HA) Filler Injection
- Gain: 1–2 cm increase in circumference
- Duration: 12–18 months; requires re-treatment
- Complication rate: ~4.3% — subcutaneous nodules, bruising, infection; rare allergic reactions
- Dissoluble with hyaluronidase if complications occur — a key safety advantage
2. Autologous Fat Grafting
- Gain: ~3–4 cm circumference increase when used as surgical grafts
- Limitation: Resorption rates are unpredictable (months to years); complications include irregular fat nodules, skin deformity, scarring, and "scrotalization" (penile skin migrating scrotal appearance)
- Injected fat has largely fallen out of favor; dermal fat grafts have fewer complications
3. Scrotal Dartos Flap (Scrotal Flap Method)
- Gain: ~3.59 ± 0.80 cm girth increase — among the largest reported for any technique
- No serious adverse events (fibrosis, flap infection) reported at 6-month follow-up in published series
4. Penuma® Silicone Implant
- Available in three sizes (14, 16, 18 cm)
- Implanted via subpubic or penoscrotal incision, placed superficial to Buck's fascia
- Provides permanent girth enhancement
- Complication profile includes implant removal rates, infection, and fibrosis
5. Acellular Dermal Matrix (ADM) / Allograft Grafting
- ADM combined with prostaglandin E showed mean girth gain of 1.1 cm but high complication rates in one study (47/78 patients had erectile discomfort), suggesting it is not an ideal standalone approach
6. Biodegradable Scaffolds / Tissue Engineering
Phalloplasty (Reconstructive / Gender-Affirming)
- Transmen (female-to-male gender-affirming surgery)
- Penile agenesis, traumatic loss, or severe micropenis
- Minor complication rate: 17–35%
- Major complication rate (urethral fistula, stenosis, flap necrosis): 7–15%
Summary of Evidence & Cautions
| Technique | Gain | Evidence Quality | Key Risk |
|---|---|---|---|
| Suspensory ligament release | 1–3 cm length | Moderate | Erection angle loss |
| HA filler injection | 1–2 cm girth | Moderate | Resorption, nodules (reversible) |
| Autologous fat grafting | 3–4 cm girth | Moderate | Unpredictable resorption, deformity |
| Scrotal dartos flap | ~3.6 cm girth | Limited | Relatively new technique |
| Penuma implant | Permanent girth | Limited | Infection, explantation |
| Penile disassembly / sliding elongation | >3 cm length | Low | Erectile dysfunction, high complications |
| Phalloplasty | Full reconstruction | Established (reconstructive) | High complexity, multiple stages |
Topical steroids?
topical corticosteroids clinical use
topical steroids potency classification indications side effects 2024
Topical Corticosteroids
Mechanism of Action
- Anti-inflammatory — suppress prostaglandins, leukotrienes, and cytokines
- Antiproliferative — reduce keratinocyte turnover
- Immunosuppressive — inhibit T-cell mediated reactions
- Vasoconstrictive — reduce erythema and oedema
Potency Classification
US System (7 Classes)
| Class | Potency | Key Examples |
|---|---|---|
| I (Super-high) | Strongest | Clobetasol propionate 0.05%, Halobetasol propionate 0.05%, Betamethasone dipropionate augmented 0.05% (ointment) |
| II (High) | — | Fluocinonide 0.05%, Halcinonide 0.1%, Desoximetasone 0.25% |
| III (Upper-mid) | — | Triamcinolone acetonide 0.5%, Amcinonide 0.1% |
| IV (Mid) | — | Mometasone furoate 0.1%, Triamcinolone acetonide 0.1%, Fluocinolone acetonide 0.025% |
| V (Lower-mid) | — | Fluticasone propionate 0.05% cream, Betamethasone valerate 0.1% cream, Hydrocortisone butyrate 0.1% cream |
| VI (Low) | — | Desonide 0.05%, Alclometasone dipropionate 0.05%, Fluocinolone acetonide 0.01% |
| VII (Least) | Weakest | Hydrocortisone 1% (OTC), Hydrocortisone 2.5% (Rx) |
UK/European System (4 Classes)
| Class | Potency | Example |
|---|---|---|
| Very potent | Up to 600× hydrocortisone | Clobetasol propionate 0.05% |
| Potent | 100–150× hydrocortisone | Betamethasone valerate 0.1% |
| Moderate | 2–25× hydrocortisone | Clobetasone butyrate 0.05% |
| Mild | — | Hydrocortisone 0.5–2.5% |
Vehicle matters: The same molecule can span two potency classes depending on formulation. Ointments > creams > lotions > gels for penetration and potency.
Common Clinical Indications
| Condition | Recommended Potency |
|---|---|
| Atopic dermatitis (body/limbs, maintenance) | Mild–Moderate (e.g. triamcinolone 0.1%) |
| Atopic dermatitis (face, flexures) | Mild only |
| Psoriasis (plaques) | Moderate–Very high |
| Lichen planus (cutaneous) | High; occlusion may be needed for hypertrophic lesions |
| Contact dermatitis (acute) | Moderate–High, short course |
| Seborrhoeic dermatitis (face/scalp) | Mild–Moderate |
| Pruritus scroti / anogenital pruritus | Low–Mild (caution re: "addicted scrotum syndrome") |
| Oral lichen planus | High-potency formulation (gels/pastes) |
Formulation Guide
- Ointment: Most potent, best for dry/lichenified skin; not suitable for wet/infected or hairy areas
- Cream: Versatile, suitable for moist and flexural areas
- Lotion/solution: Best for scalp and hairy areas
- Gel: Scalp; note propylene glycol base can irritate and dry skin
- Foam: Scalp psoriasis; cosmetically acceptable
- Shampoo: Scalp conditions with wash-off use
Site-Based Prescribing Rules
| Body Area | Guidance |
|---|---|
| Face, eyelids | Mild only; high risk of atrophy, rosacea, perioral dermatitis, glaucoma |
| Flexures (axillae, groin, submammary) | Mild only; increased absorption and occlusion |
| Palms/soles | Can tolerate potent–very potent (thick skin) |
| Trunk/extremities | Moderate–high for chronic inflammatory conditions |
| Scalp | Potent generally well-tolerated |
| Children | Use lowest effective potency; risk of systemic absorption proportionally higher |
Duration Guidelines
| Potency | Maximum Continuous Use |
|---|---|
| Very high (Class I) | 1–2 weeks only; never on face or flexures |
| High (Class II–III) | Up to 3 weeks for acute flares |
| Moderate (Class IV–V) | Longer-term acceptable for trunk/extremities; maintenance therapy validated (e.g. fluticasone twice weekly) |
| Mild (Class VI–VII) | Generally safe for longer periods; preferred for face and sensitive areas |
Adverse Effects
Local (with prolonged or inappropriate use)
- Skin atrophy — thinning, fragility, telangiectasia
- Striae — irreversible, especially in flexures
- Acneiform/rosacea-like eruption — especially facial use
- Perioral dermatitis
- Hypopigmentation
- Hypertrichosis
- Infection potentiation/masking — tinea incognita, bacterial, herpetic, molluscum; granuloma gluteale infantum (diaper area in infants)
- Tachyphylaxis — loss of efficacy with continuous use; address by rotating or taking breaks
Systemic (rare; high-potency, large surface area, prolonged use)
- HPA axis suppression — especially Class I agents; can cause adrenal insufficiency
- Cushing syndrome — reported with >50 g/week of clobetasol propionate or >500 g/week hydrocortisone
- Growth retardation — children receiving large quantities of potent agents
Topical Steroid Withdrawal (TSW)
Allergic Contact Dermatitis
Pregnancy & Special Populations
- Mild–moderate TCS in pregnancy: No significant risk to mother or fetus based on available evidence
- High-potency TCS in pregnancy: Small but real association with low birth weight — avoid over large areas or prolonged courses
- Paediatrics: Risk of systemic absorption is proportionally greater (higher body surface-to-weight ratio); prefer mild potency; avoid occlusion
Practical Prescribing Tips
- Fingertip unit (FTU): One FTU (≈0.5 g) covers an area equal to two adult palms
- Steroid ladder: Start with an appropriate potency for the site and severity — don't automatically start mild everywhere
- Step down: Once controlled, reduce to lowest effective potency and consider intermittent ("weekend") dosing for maintenance
- Emollients: Apply TCS to the lesion; emollient on uninvolved skin. Allow a few minutes between the two
- Combination products (TCS + antifungal): Useful for conditions where secondary candidal/dermatophyte infection is likely (e.g. flexural eczema)
Topical testosterone not steroids
topical testosterone gel transdermal testosterone therapy
topical testosterone gel formulations indications pharmacology 2024
Topical Testosterone
Why Topical? — Pharmacokinetic Rationale
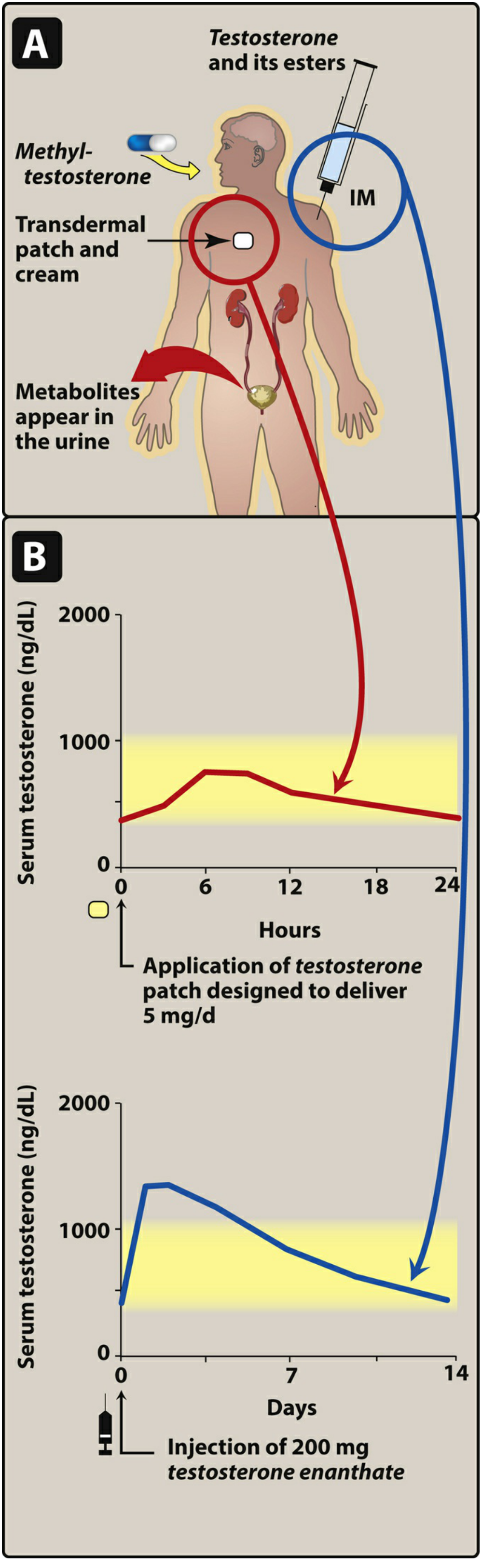
Available Topical Formulations
Gels
| Brand | Concentration | Starting Dose | Application Site |
|---|---|---|---|
| AndroGel 1% | 1% gel | 50 mg (5 g) once daily AM | Shoulders, upper arms, abdomen |
| AndroGel 1.62% | 1.62% gel | 40.5 mg (2 pump actuations) once daily AM | Shoulders, upper arms only |
| Testim | 1% gel | 50 mg (5 g tube) once daily AM | Shoulders, upper arms |
| Vogelxo | 1% gel | 50 mg (5 g) once daily AM | Shoulders, upper arms |
- Bioavailability: Only ~10% of applied dose is absorbed over 24 hours
- Dose range: 25–100 mg/day depending on serum levels
- Titrate based on pre-dose morning serum testosterone at ~14 and 28 days after starting
2% Solution
| Brand | Dose | Site |
|---|---|---|
| Axiron | 30 mg per axilla once daily (metered applicator) | Axilla |
- Axilla has high skin permeability and significant 5α-reductase activity
- Concealable application site
Transdermal Patches
| Brand | Dose | Notes |
|---|---|---|
| Androderm | 2.5 mg or 5 mg/day | Apply to arm, back, or buttock; avoid sun-exposed areas |
| Testoderm (scrotal, historical) | 4–6 mg/day | Required scrotal shaving; high DHT levels from scrotal 5α-reductase; largely discontinued |
- Patches are associated with skin irritation and contact dermatitis in a proportion of users; local cortisone cream can alleviate irritation
- Rotate sites to reduce chronic skin reactions
Nasal Gel
| Brand | Dose | Frequency |
|---|---|---|
| Natesto | 11 mg (1 actuation per nostril) | 2–3× daily (total 22–33 mg/day) |
- Advantages: Rapid onset, short half-life, no transfer risk to partners/children
- Disadvantages: Requires dosing up to 3×/day; nasal irritation possible
Indications
- Primary hypogonadism (congenital or acquired — e.g. Klinefelter syndrome, orchiectomy, cryptorchidism)
- Hypogonadotropic hypogonadism (congenital or acquired — e.g. pituitary/hypothalamic disease)
- Gender-affirming hormone therapy in transgender men — starting dose typically 50 mg/day gel, titrated
- Limited evidence for lichen sclerosus — topical testosterone cream may benefit loss of sexual sensitivity (Berek & Novak's Gynecology)
- Cirrhosis-related gonadal dysfunction — topical testosterone may improve muscle strength and survival in selected patients (Sleisenger & Fordtran's GI and Liver Disease)
Contraindications
- Known or suspected prostate cancer or breast cancer in men
- Pregnancy (absolute contraindication; androgens are teratogenic)
- Women (not indicated; risk of virilisation)
- Severe hepatic impairment (Child-Pugh C)
Monitoring
| Parameter | Timing |
|---|---|
| Serum testosterone (pre-dose AM) | 14 days, 28 days after starting/dose change, then periodically |
| Haematocrit/haemoglobin | Baseline, then periodically (polycythaemia risk) |
| PSA + digital rectal exam | Baseline and periodically in men >40 |
| Lipid profile | Periodically |
| Bone density | If long-standing hypogonadism |
Adverse Effects
Local
- Skin irritation, erythema, pruritus at application site
- Contact dermatitis (patches > gels)
- Acne at application site
Systemic
| Effect | Notes |
|---|---|
| Polycythaemia | Dose-dependent; monitor haematocrit; dose reduction or phlebotomy if Hct >54% |
| Suppression of spermatogenesis | TRT reduces FSH/LH → azoospermia; men desiring fertility should be counselled; enclomiphene citrate preserves sperm counts as an alternative |
| Gynaecomastia | From aromatisation to estradiol |
| Acne / oily skin | From DHT |
| Sleep apnoea — worsening | Known association |
| VTE / cardiovascular events | Black box warning: possible increased risk of MI, stroke, DVT, PE — especially in men with pre-existing CVD |
| Worsening BPH | Monitor lower urinary tract symptoms |
| Potential prostate cancer promotion | Contraindicated if suspected; PSA monitoring required |
⚠️ Secondary (Transfer) Exposure — Black Box Warning
- Wash hands immediately after application
- Cover the site with clothing once dry
- Wash the application site with soap and water before anticipated skin-to-skin contact
Testosterone Therapy and Fertility
- Clomiphene citrate (SERMs to stimulate endogenous production)
- hCG ± FSH injections (for hypogonadotropic hypogonadism)
- Rather than exogenous testosterone
For penis enlargement
topical testosterone penile growth micropenis
topical testosterone penis enlargement evidence clinical effectiveness
Topical Testosterone for Penis Enlargement
The Short Answer
Where It Does Work: Micropenis (Hypogonadal States)
Mechanism
Evidence
- Mean +60% increase in penile length
- Mean +52.9% increase in penile girth
"Studies have shown that topical application of 5% testosterone cream causes increased penile growth, but its effect is due to absorption of the hormone, which systemically stimulates genital growth."
Clinical Protocol (Micropenis)
- Goal: Provide sufficient testosterone to stimulate penile growth without premature epiphyseal closure
- Regimen: Testosterone enanthate 25 mg IM every 3 weeks × 4 doses is standard; transdermal testosterone has also been used
- Monitoring: Stretched penile length (pubis to glans) before and after each course; bone age X-ray to monitor epiphyseal status
- Timing: Therapy should begin by age 1 year; repeat courses may be needed as child grows
- In boys with micropenis treated with testosterone at birth and puberty, final stretched penile length averaged 10.3 cm (within normal range in all 8 cases reported) — Campbell-Walsh-Wein Urology
In Normal Adult Men
- Penile growth requires open androgen-sensitive growth pathways — these are active in fetal development, infancy, and puberty, but are largely closed in adults
- Penile length in adults has only a weak correlation with circulating testosterone levels (r=0.22 in one study) — and this reflects developmental exposure, not current levels
- The Translational Andrology and Urology study explicitly states: "A weak association between adult testosterone levels and penile length does not mean that men with higher testosterone levels have larger penises and by no means suggests that exogenous testosterone or testosterone replacement therapy will increase penile length"
- The Mayo Clinic notes that testosterone-containing "pills and lotions" marketed for enlargement have not been proved to work and may be harmful
- The AUA and Urology Care Foundation do not endorse topical testosterone for penile enlargement in normal adult men
Summary
| Population | Does Topical Testosterone Work? | Mechanism |
|---|---|---|
| Children/adolescents with hypogonadal micropenis | Yes — established use | Corrects androgen deficiency → restores developmental penile growth |
| Adults with hypogonadism | Limited data; may support normal penile function but not enlargement beyond baseline | Replaces deficient testosterone; growth windows are closed |
| Eugonadal adult men | No clinical evidence | Growth pathways closed; no androgen-receptor response to drive tissue expansion |
How can I enlarge my penis without surgery
nonsurgical penile enlargement traction extender hyaluronic acid
non-surgical penis enlargement evidence traction device filler 2024 2025 clinical review
penile traction device extender elongation clinical trial
hyaluronic acid penile girth injection outcomes
nonsurgical penile enlargement traction device vacuum pump filler evidence outcomes
Non-Surgical Penile Enlargement: What the Evidence Shows
1. Penile Traction Devices (Extenders)
- In one trial of 23 patients using a penile stretching device, mean flaccid length increased from 8.8 cm to 10.5 cm at 3 months. No significant girth improvement.
- A second study of 54 patients using traction 6 hours/day for 4 months showed significant increases in flaccid, stretched, and erect length at 6-month follow-up.
- The BJU International systematic review (2021) classified traction therapies as "mostly efficacious and safe"
- No adverse effects reported in published trials when devices were used as instructed
2. Hyaluronic Acid (HA) Filler Injection
- Girth increase: Consistently ~1–2 cm circumference gain
- A 2023 meta-analysis (Kusumaputra et al., PMID 37663705) of 283 patients found HA significantly superior to polylactic acid for penile girth enhancement (p=0.01) and sexual satisfaction at 12 weeks (p=0.0004)
- Complication rate ~4%: subcutaneous nodules, bruising, minor infection — all manageable
- Key safety advantage: Reversible — complications can be dissolved with hyaluronidase
- Duration: 12–18 months; repeat treatment required
3. Polylactic Acid (PLA) Filler Injection
- Less effective than HA for girth increase in head-to-head comparisons
- Longer-lasting but not reversible — disadvantage compared to HA if complications occur
- Similar complication rates to HA
4. Vacuum Erection Devices (VED / Pumps)
- Increases penile size temporarily only — effect reverses within ~30 minutes
- No permanent enlargement in normal men
- Best evidence for use post-surgery (e.g. post-penile prosthesis, post-prostatectomy) to maintain penile length and prevent fibrosis
- Used excessively or incorrectly, VEDs can cause bruising, petechiae, and damage to elastic tissue → less firm erections over time
5. Jelqing (Manual Stretching/Milking Exercises)
6. Pills, Supplements, and Herbal Products
7. Pubic Fat Pad Reduction (Lifestyle / Body Composition)
Summary Table
| Method | Effectiveness | Evidence Quality | Permanent? | Key Risk |
|---|---|---|---|---|
| Penile traction device | Modest length gain (~1–2 cm) | Moderate | Yes (sustained use) | Compliance burden |
| HA filler injection | Reliable girth gain (~1–2 cm) | Moderate | No (12–18 months) | Nodules; needs qualified provider |
| PLA filler injection | Girth gain; less than HA | Low–Moderate | Longer-lasting but not permanent | Not reversible |
| Vacuum pump | Temporary only | Low (for enlargement) | No | Tissue damage with overuse |
| Jelqing | Unproven | None | Unknown | Fibrosis, ED |
| Pills/supplements | None | None | No | Drug interactions, toxicity |
| Weight loss / fat pad reduction | Improves appearance | Practical | Yes | None |
Bottom Line
- Penile traction devices — for modest length gains with sustained daily use
- HA filler injections by a qualified clinician — for temporary girth enhancement