Aminoglycosides
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive content from multiple authoritative textbooks. Let me compile the full answer.
Aminoglycosides
Chemistry & Members
Aminoglycosides consist of amino sugars linked via glycosidic bonds to an aminocyclitol ring. Members are derived from two genera:
- Streptomyces: streptomycin, neomycin, kanamycin, tobramycin
- Micromonospora: gentamicin, sisomicin
- Semisynthetic: amikacin (from kanamycin), netilmicin (from sisomicin), plazomicin
The most clinically used are gentamicin, tobramycin, and amikacin.
Mechanism of Action
Aminoglycosides are concentration-dependent bactericidal agents. They cross the bacterial outer membrane, cell wall, and cytoplasmic membrane via an oxygen-dependent, energy-dependent transport process to reach the cytoplasm where they:
- Bind irreversibly to the 30S ribosomal subunit
- Cause misreading of mRNA → production of aberrant proteins
- Cause premature dissociation of the ribosome from mRNA → interruption of protein synthesis
The three key effects on the ribosome — block of the initiation complex, generation of miscoded peptide chains, and block of translocation — are illustrated here:
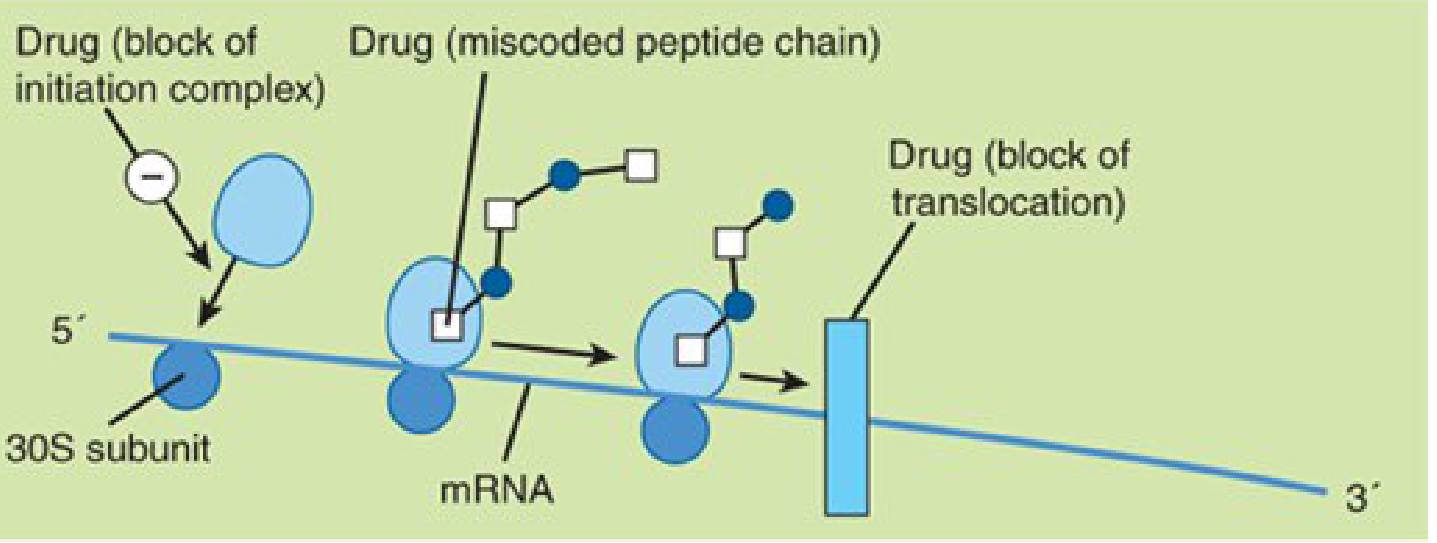
Because entry requires aerobic respiration, anaerobes are intrinsically resistant (no functional oxidative transport). For the same reason, organisms in an abscess cavity are not effectively killed.
Spectrum of Activity
| Organism Type | Activity |
|---|---|
| Aerobic gram-negative rods (Enterobacteriaceae, Pseudomonas, Acinetobacter) | Excellent |
| Some gram-positive organisms | Yes (with combination) |
| Mycobacterium tuberculosis | Streptomycin, amikacin |
| Anaerobes | None (intrinsic resistance) |
| Streptococci / Enterococci (alone) | Poor — fail to penetrate cell wall |
Synergy with cell-wall–active agents: When given alongside a β-lactam or vancomycin, the disrupted cell wall allows aminoglycosides to penetrate streptococci and enterococci, achieving bactericidal synergy. This is the basis for treating enterococcal endocarditis (ampicillin + gentamicin, or the preferred current regimen of ampicillin + ceftriaxone).
Tobramycin has the most potent activity against P. aeruginosa. Amikacin has the broadest activity and is often reserved for gentamicin/tobramycin-resistant gram-negatives. — Goldman-Cecil Medicine
Pharmacodynamics
- Concentration-dependent killing: Higher peak concentrations → faster, greater bacterial kill
- Postantibiotic effect (PAE): Bactericidal activity persists for several hours beyond measurable drug levels
- PD index: AUC/MIC ratio (or peak:MIC ratio) is the target linked to bacterial killing
- These properties justify once-daily dosing (extended-interval dosing)
Pharmacokinetics
| Parameter | Details |
|---|---|
| Absorption | Negligible orally (highly polar cations); IV infusion over 30–60 min or IM |
| Distribution | Largely excluded from CNS, eye, and intracellular compartments; high concentrations in renal cortex |
| CSF penetration | ~20% of plasma even with inflammation; requires intrathecal injection for meningitis |
| Half-life | 2–3 hours (normal renal function); 24–48 hours in renal failure |
| Elimination | Renally excreted (GFR-dependent); dose reduction required for creatinine clearance < 60 mL/min |
| Dialysis | Only 40–60% removed by hemodialysis; less by peritoneal dialysis |
— Katzung's Basic and Clinical Pharmacology, 16th Ed.
Dosing Strategies
Once-Daily (Extended-Interval) Dosing
- Preferred in most clinical settings (normalrenal function, CrCl > 60 mL/min)
- Gentamicin/tobramycin: 5–7 mg/kg/day; amikacin/plazomicin: 15 mg/kg/day
- Target: serum concentration < 1 mcg/mL at 18–24 hours post-dose (Hartford nomogram)
- Toxicity risk is equal or lower compared to conventional dosing due to reduced time above toxic trough threshold
- Exceptions: Not well-defined for endocarditis (enterococcal/staphylococcal on prosthetic valve), pregnancy, neonates, obesity
Conventional (Multiple Daily) Dosing
- Gentamicin/tobramycin: peak 8–10 mcg/mL, trough < 2 mcg/mL (optimal < 1 mcg/mL)
- Peak drawn 30–60 min post-dose; trough drawn just before next dose
- Required when CrCl < 60 mL/min
Adverse Effects
1. Nephrotoxicity
- Most common serious adverse effect
- Accumulation in the proximal tubular cells of the renal cortex
- Manifests as non-oliguric renal failure, rising creatinine
- Risk factors: prolonged therapy (>5 days), high doses, elderly, pre-existing renal insufficiency, concurrent nephrotoxins (NSAIDs, contrast, amphotericin B, vancomycin)
- Trough > 2 mcg/mL predicts toxicity
- Generally reversible with discontinuation
2. Ototoxicity
- Cochleotoxicity (hearing loss) and/or vestibulotoxicity (vertigo, ataxia)
- Streptomycin preferentially causes vestibular damage
- Other aminoglycosides preferentially cause cochlear damage
- Often irreversible due to destruction of hair cells in the organ of Corti
- Risk factors: same as nephrotoxicity + loop diuretics (furosemide)
- AUC/MIC graphs below show the overlap between therapeutic effect and nephrotoxicity probability:
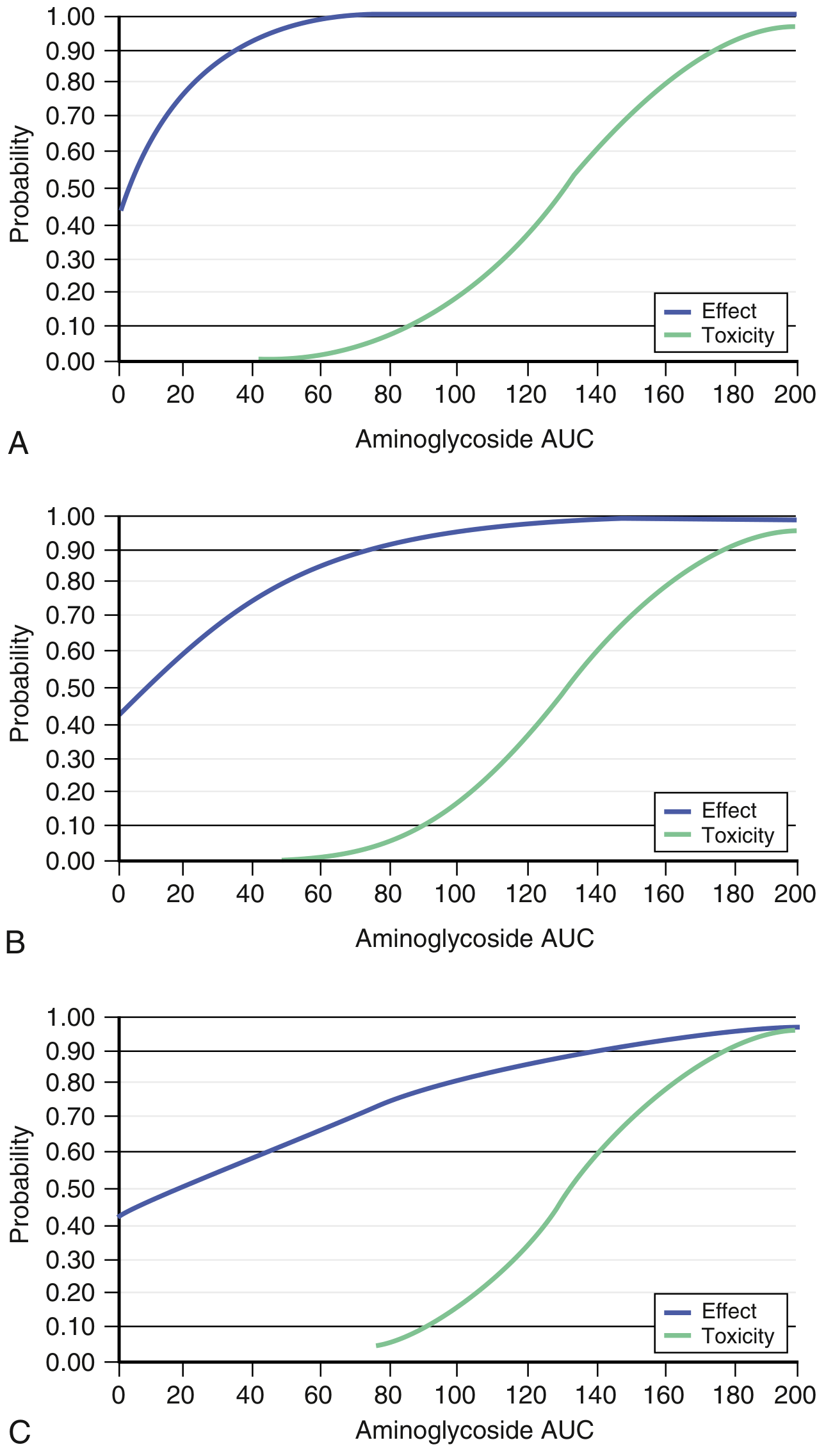
3. Neuromuscular Blockade
- Rare; occurs at very high doses
- Curare-like effect → respiratory paralysis
- Reversed by calcium gluconate (promptly) or neostigmine
- Caution in patients with myasthenia gravis or receiving neuromuscular blockers
Resistance Mechanisms
Four mechanisms, in order of clinical importance:
-
Enzymatic modification (most common):
- Phosphotransferases (APHs), adenylyltransferases (ANTs), acetyltransferases (AACs) modify amino/hydroxyl groups → drug no longer binds ribosome
- Encoded on plasmids or transposons
- Amikacin is less susceptible to these enzymes due to structural modifications; plazomicin is the most resistant to modifying enzymes
-
Decreased uptake / permeability defect: Mutation or deletion of outer membrane porins; seen with Pseudomonas; produces low-level cross-resistance to all aminoglycosides
-
Ribosomal binding site mutation: Requires mutation of multiple rRNA gene copies — uncommon; limits streptomycin (single binding site)
-
Active efflux: Occurs only in gram-negative bacteria; rarely clinically significant
— Medical Microbiology 9e; Katzung's Basic and Clinical Pharmacology, 16th Ed.
Clinical Uses
| Indication | Aminoglycoside Used |
|---|---|
| Serious gram-negative infections (empiric/sepsis) | Gentamicin or tobramycin + β-lactam |
| Pseudomonas aeruginosa infections | Tobramycin preferred |
| Drug-resistant gram-negatives (gentamicin/tobramycin-R) | Amikacin |
| Enterococcal/streptococcal endocarditis (synergy) | Gentamicin (+ ampicillin or penicillin) |
| Tuberculosis (second-line) | Streptomycin or amikacin |
| Tularemia, plague, brucellosis | Streptomycin (+ tetracycline) |
| Complicated UTI (CRKP) | Plazomicin |
| Mycobacterial infections | Amikacin (± streptomycin) |
| Cystic fibrosis (Pseudomonas) | Tobramycin (inhaled) |
Key "Must-Know" Points
- Bactericidal, concentration-dependent killers with postantibiotic effect → support once-daily dosing
- No oral bioavailability — parenteral only (or topical/inhaled for local infections)
- Inactive in anaerobic conditions and against obligate anaerobes
- Synergistic with β-lactams against gram-positives (enterococci, streptococci)
- Monitoring: renal function + drug levels (especially with conventional dosing or prolonged therapy)
- Major toxicities: nephrotoxicity (reversible) and ototoxicity (often irreversible)
- Plazomicin: newest agent, retains activity against many aminoglycoside-modifying enzyme–expressing organisms
Sources: Medical Microbiology 9e (Murray); Katzung's Basic and Clinical Pharmacology 16e; Goldman-Cecil Medicine
Source from KD TRIPATHI BOOK
Finding Sources
Finding Sources
KD Tripathi's Essentials of Medical Pharmacology is not available in the textbook library. The library's pharmacology collection includes only:
- Goodman & Gilman's The Pharmacological Basis of Therapeutics
- Katzung's Basic and Clinical Pharmacology, 16th Edition
- Lippincott Illustrated Reviews Pharmacology
KD Tripathi is a widely used South Asian pharmacology textbook but it is not part of this library's licensed collection.
What I can do instead:
- Source the aminoglycosides topic from Goodman & Gilman or Lippincott — both are highly detailed and cover the same content as Tripathi (mechanism, spectrum, PK, toxicity, clinical uses)
- Lippincott in particular uses a presentation style similar to Tripathi — concise, exam-oriented, with clear tables
Would you like me to pull the aminoglycoside content from Goodman & Gilman, Lippincott, or both?
Lippincott
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the Lippincott content. Here is the complete presentation:
Aminoglycosides
Lippincott Illustrated Reviews: Pharmacology
Members: Amikacin, Gentamicin, Neomycin, Plazomicin, Streptomycin, Tobramycin
A. Mechanism of Action
Aminoglycosides diffuse through porin channels in the outer membrane of susceptible organisms. An oxygen-dependent transport system then carries the drug across the cytoplasmic membrane. Inside the cell, they bind the 30S ribosomal subunit, where they:
- Interfere with assembly of the functional ribosomal apparatus
- Cause the 30S subunit to misread the genetic code → aberrant, nonfunctional proteins
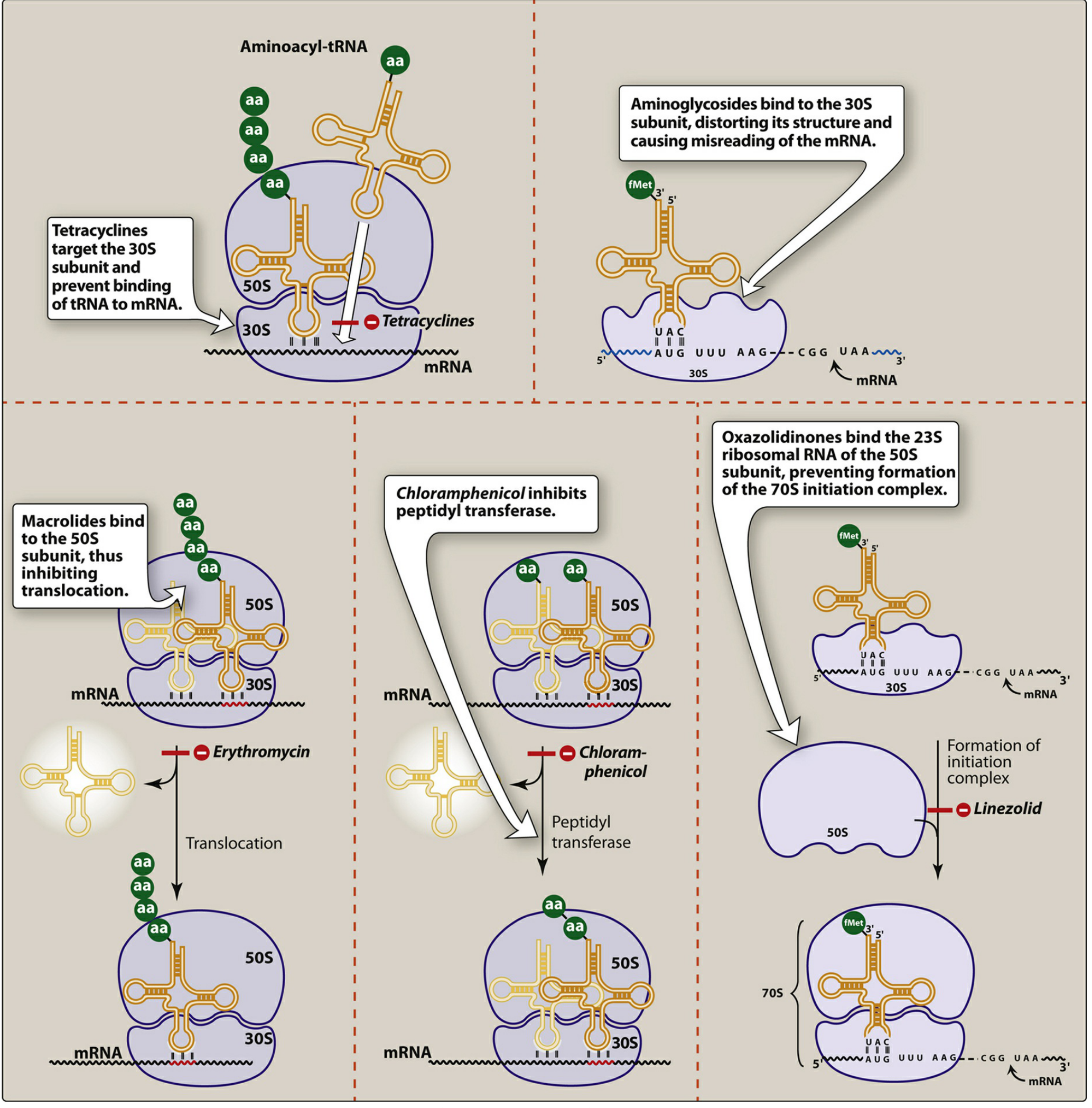
Pharmacodynamic properties:
- Concentration-dependent bactericidal activity — efficacy depends on peak (C
max) / MIC ratio - Target C
max: 8–10 × MIC - Exhibit a postantibiotic effect (PAE) — continued bacterial suppression even after drug falls below MIC; the larger the dose, the longer the PAE
- These two properties are the rationale for high-dose extended-interval (once-daily) dosing, which also reduces nephrotoxicity and increases convenience
B. Antibacterial Spectrum
Aminoglycosides are effective against aerobic gram-negative bacilli, including multidrug-resistant organisms:
| Organism | Drug of Choice |
|---|---|
| Pseudomonas aeruginosa | Tobramycin (alone for UTI; + anti-pseudomonal β-lactam for pneumonia) |
| Klebsiella pneumoniae, Enterobacter spp. | Amikacin or gentamicin |
| Acinetobacter baumannii | Gentamicin/amikacin |
| Francisella tularensis (tularemia) | Gentamicin |
| Brucella spp. | Gentamicin + doxycycline |
| Yersinia pestis (plague) | Streptomycin |
| Enterococcus faecalis / faecium endocarditis | Ampicillin + gentamicin (synergy) |
| Streptococcus agalactiae | Ampicillin + gentamicin (synergy) |
Synergy: Aminoglycosides are combined with β-lactams for serious gram-positive infections (particularly enterococcal endocarditis). The β-lactam disrupts the cell wall, allowing aminoglycoside entry → bactericidal synergy.
Anaerobes, Streptococcus, Enterococcus are intrinsically resistant when aminoglycosides are used alone (no oxygen-dependent transport possible).
C. Resistance
Three mechanisms:
- Efflux pumps — active export of drug out of the cell
- Decreased uptake — altered outer membrane permeability
- Enzymatic modification and inactivation (most common clinically) — plasmid-encoded enzymes (phosphotransferases, acetyltransferases, adenylyltransferases) inactivate the drug
Each modifying enzyme has its own aminoglycoside specificity, so cross-resistance cannot be assumed. Amikacin and plazomicin are less vulnerable to these enzymes than other members of the class.
D. Pharmacokinetics
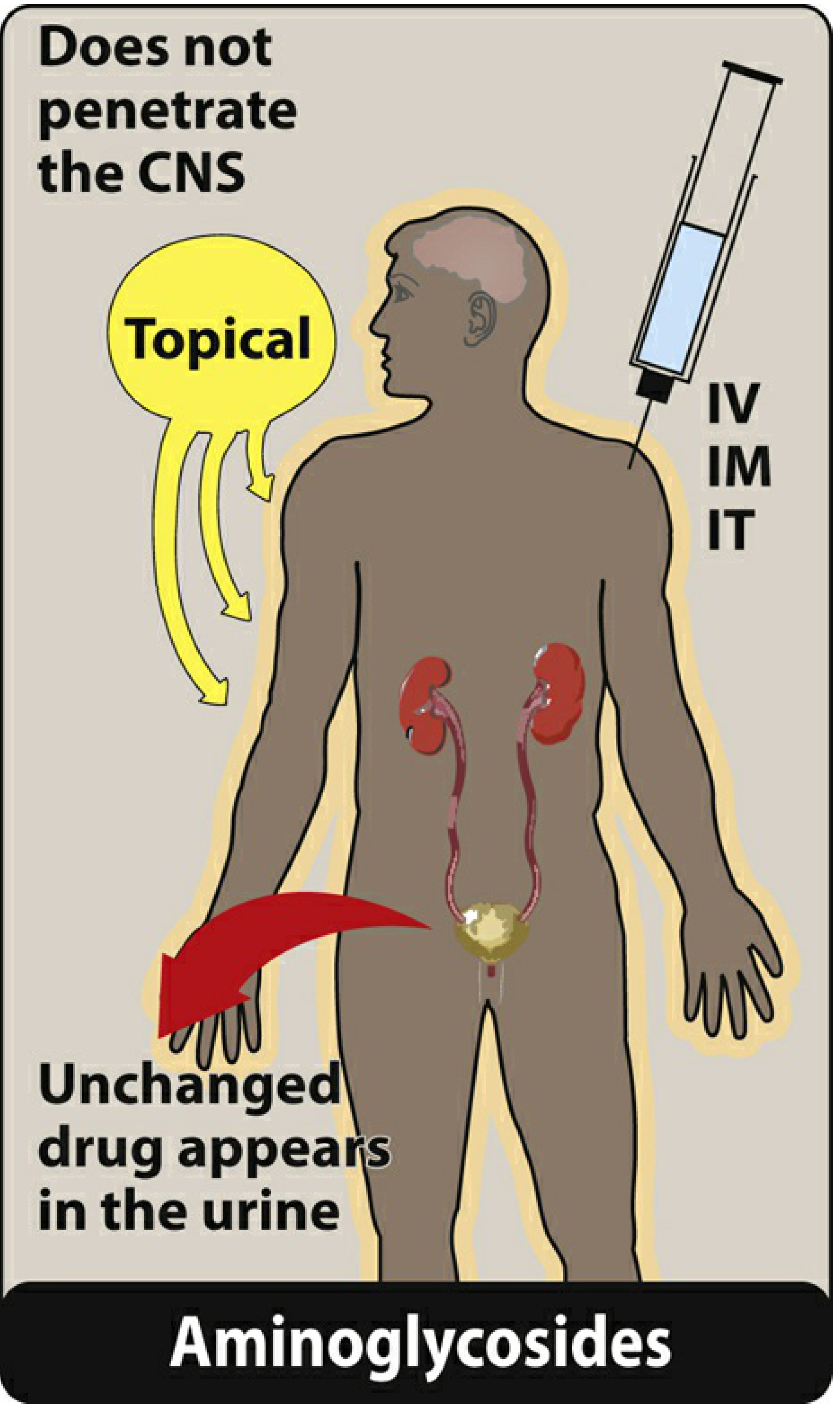
1. Absorption
- Highly polar, polycationic structure → not absorbed orally
- Must be given parenterally (IV, IM) to achieve adequate serum concentrations
- Neomycin exception: Never given parenterally (severe nephrotoxicity); administered topically (skin infections) or orally (GI decontamination before colorectal surgery)
- Intrathecal (IT) route used for CNS infections
2. Distribution
- Hydrophilic → poor tissue penetration; subtherapeutic concentrations in most body fluids
- Does not penetrate the CNS — concentrations inadequate even with inflamed meninges → intrathecal or intraventricular routes required for CNS infections
- Cross the placental barrier → accumulate in fetal plasma and amniotic fluid (teratogenic risk)
3. Elimination
-
90% excreted unchanged in the urine (renal filtration)
- Accumulates in renal dysfunction → dose adjustment mandatory
- Neomycin (oral): excreted unchanged in feces
E. Adverse Effects
Therapeutic drug monitoring of gentamicin, tobramycin, amikacin, and plazomicin plasma concentrations is essential. The elderly are especially vulnerable.
1. Ototoxicity
- Vestibular (vertigo, loss of balance) and auditory (hearing loss/deafness) damage
- Directly related to high peak plasma concentrations and duration of treatment
- Drug accumulates in endolymph and perilymph of the inner ear
- Deafness may be irreversible and can affect the developing fetus
- Streptomycin preferentially causes vestibular toxicity (vertigo)
- Potentiated by concurrent cisplatin or loop diuretics (furosemide)
2. Nephrotoxicity
- Retention by proximal tubular cells disrupts calcium-mediated transport processes
- Ranges from mild, reversible renal impairment to severe acute tubular necrosis (potentially irreversible)
- Risk increases with prolonged therapy, high doses, elderly patients, pre-existing renal disease
3. Neuromuscular Blockade
- Associated with rapid concentration increase (high doses given over a short period) or concurrent use of neuromuscular blocking agents
- Patients with myasthenia gravis are particularly at risk
- Reversed promptly by calcium gluconate
4. Allergic Reactions
- Contact dermatitis is a common reaction to topically applied neomycin
Summary Table
| Property | Detail |
|---|---|
| Class | Aminocyclitol + amino sugars |
| Action | Binds 30S subunit → mRNA misreading; bactericidal |
| PD type | Concentration-dependent; exhibits PAE |
| PD target | C |
| Route | IV, IM (parenteral only); topical (neomycin) |
| Distribution | Poor CNS penetration; crosses placenta |
| Elimination | >90% unchanged in urine; dose-adjust for renal failure |
| Spectrum | Aerobic gram-negatives; synergy with β-lactams for gram-positives |
| Key toxicities | Ototoxicity (irreversible), nephrotoxicity, neuromuscular blockade |
| Resistance | Enzymatic modification (main), efflux, decreased uptake |
| Amikacin/Plazomicin | Less susceptible to resistance enzymes |
— Lippincott Illustrated Reviews: Pharmacology, Chapter 30 (Protein Synthesis Inhibitors)
This is a shared conversation. Sign in to Orris to start your own chat.