Endometriosis
Reading File
Asking for Preferences
Finding Sources
Searching PubMed
endometriosis
Searching Images
endometriosis pathology laparoscopy
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Loading Image
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content from multiple authoritative sources. Here is the full overview:
Endometriosis
Definition
Endometriosis is the presence of endometrial glands and stroma outside the uterine cavity. It affects approximately 10% of reproductive-age women, 15–20% of infertile women, and more than 30% of women with chronic pelvic pain (CPP). Confirmation requires direct operative visualization, so true prevalence is unknown. — Berek & Novak's Gynecology; Robbins & Kumar Basic Pathology
Classification (Types)
Three recognized subtypes:
| Type | Description |
|---|---|
| Superficial peritoneal endometriosis | Implants on peritoneal surfaces; early lesions appear red/flame-like or colorless vesicles |
| Ovarian endometriosis (endometrioma) | Cystic ovarian lesions filled with old blood ("chocolate cysts"), 3–5 cm |
| Deep infiltrating endometriosis (DIE) | Invades >5 mm below peritoneum; involves uterosacral ligaments, rectovaginal septum, bowel, bladder |
Risk of malignant transformation is mainly confined to deep infiltrating endometriosis. — Robbins & Kumar
The ASRM staging system (I–IV: minimal, mild, moderate, severe) is based on laparoscopic findings — extent of implants, adhesions, and adnexal involvement.
Pathogenesis
The exact mechanism remains elusive. Four leading theories:
-
Regurgitation (Sampson's) theory — Retrograde menstrual flow carries viable endometrial cells through the fallopian tubes, which implant on peritoneal surfaces. This is the most widely accepted mechanism.
-
Benign metastasis theory — Endometrial tissue spreads via blood vessels and lymphatics to distant sites (lung, bone, brain).
-
Coelomic metaplasia theory — Peritoneal mesothelium (sharing embryonic origin with the müllerian ducts) undergoes metaplastic change into endometrial-type tissue.
-
Extrauterine stem/progenitor cell theory — Bone marrow-derived stem cells differentiate into ectopic endometrial tissue.
Molecular mechanisms sustaining implants
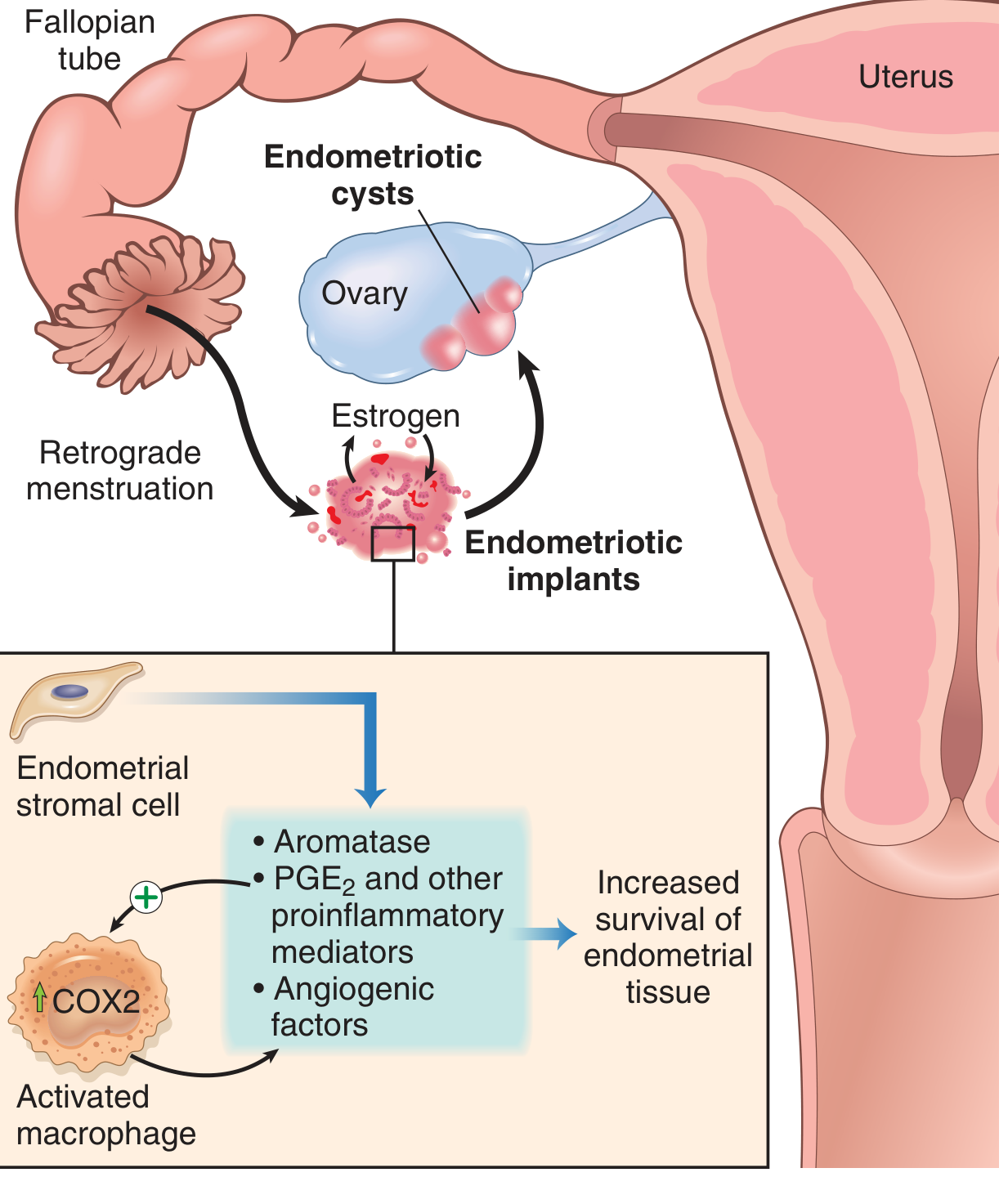
Once implanted, lesions are not merely misplaced uterine tissue — they have distinct biology:
- Aromatase overexpression in stromal cells → elevated local estrogen production (self-sustaining estrogen loop)
- ↑ PGE₂, VEGF, and matrix metalloproteinases (MMPs) → proinflammatory and proangiogenic environment
- Activated macrophages recruited to implants upregulate COX-2 → more PGE₂
- Progesterone resistance — ectopic implants fail to undergo normal decidualization and regression
Sites of Involvement
- Most common (pelvic): Ovaries, uterosacral ligaments, posterior cul-de-sac (pouch of Douglas), broad ligament, fallopian tubes, pelvic peritoneum
- GI tract: Sigmoid, rectum, small bowel (12–37% of cases); causes fibrosis and wall contraction, intact mucosa — rare rectal bleeding
- Urinary tract: Bladder, ureters
- Thoracic: Diaphragm, pleura (catamenial pneumothorax)
- Distant: Umbilicus, laparotomy scars, rarely bone, lung, brain
Gross & Histologic Morphology
Lesions grossly appear as red-brown nodules (early = red/vesicular; late = "powder-burn" dark fibrotic lesions) on serosal surfaces. Chronic bleeding causes fibrous adhesions that can obliterate the cul-de-sac, fix the uterus in retroversion, and distort adnexal anatomy.
Chocolate cysts (endometriomas): Large ovarian cysts filled with dark brown inspissated blood from repeated hemorrhage.
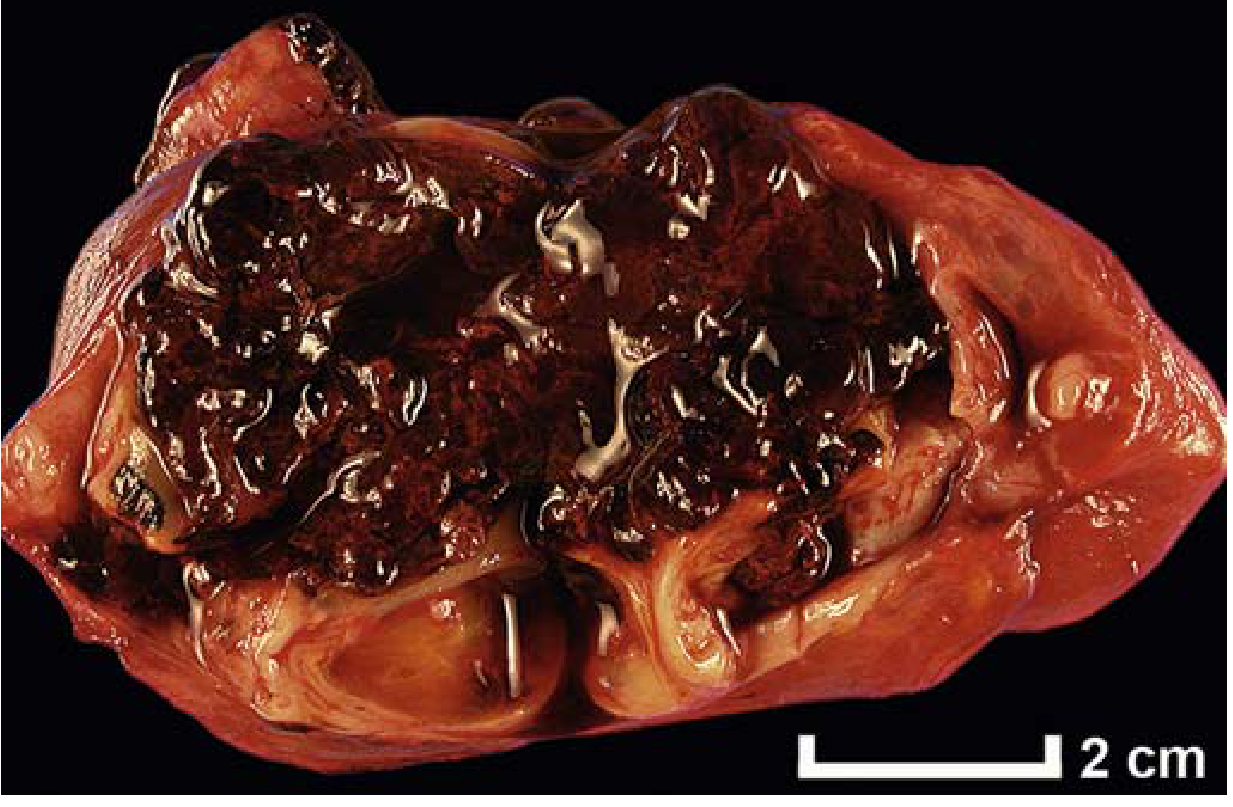
Histologic diagnosis requires both endometrial glands AND stroma at ectopic sites.
Clinical Features
Symptoms
- Dysmenorrhea — severe, often begins 1–2 weeks before menses (hallmark)
- Deep dyspareunia — due to involvement of uterosacral ligaments and rectovaginal septum
- Chronic pelvic pain — can be constant or cyclic
- Infertility — presenting complaint in 30–40% of patients; mechanisms include tubal distortion, impaired oocyte maturation, peritoneal inflammation
- Dyschezia (painful defecation), bloating, urinary urgency/frequency — especially with bowel/bladder involvement
- Catamenial symptoms — cyclic chest pain, hemoptysis, or pneumothorax (thoracic endometriosis)
- Abnormal uterine bleeding despite ovulatory cycles
Signs (Examination)
- Uterosacral nodularity on rectovaginal exam
- Fixed retroverted uterus or laterally deviated cervix (adhesions)
- Adnexal mass (endometrioma)
- Focal tenderness in uterosacral/broad ligament regions
- Cyclically tender abdominal wall mass (scar endometriosis)
The severity of visible disease often does NOT correlate with symptom severity — extensive disease can be asymptomatic, and minimal disease can cause debilitating pain.
Diagnosis
| Modality | Details |
|---|---|
| Laparoscopy (gold standard) | Direct visualization ± histologic biopsy; definitive diagnosis |
| Transvaginal ultrasound (TVUS) | First-line imaging — detects endometriomas (homogeneous "ground glass" echogenicity); poor sensitivity for peritoneal implants |
| MRI pelvis | Best for mapping DIE — low-signal fibrotic plaques, rectovaginal nodule, obliteration of pouch of Douglas; guides surgical planning |
| CA-125 | May be elevated but low sensitivity and specificity; not used alone for diagnosis |
| Endometrial biopsy | Detection of endometrial nerve fibers — promising but unproven |
| Clinical diagnosis | Accurate in ~50% of cases only |
Homogeneous hemorrhagic ovarian cysts that fail to resolve after 1–2 menstrual cycles are suspicious for endometriomas and warrant further workup. — Berek & Novak's
Management
Treatment is guided by the patient's symptoms, desire for fertility, and extent of disease.
Pharmacologic (Medical)
| Drug Class | Mechanism | Examples |
|---|---|---|
| Combined oral contraceptives (COCs) | Suppress gonadotropins → ↓ estrogen biosynthesis; first-line | Ethinyl estradiol/progestin |
| Progestins | Decidualize and atrophy ectopic tissue; block proliferative effect of estrogen | Medroxyprogesterone, dienogest, norethindrone; levonorgestrel IUS (off-label) |
| GnRH agonists | Medical castration — suppress pituitary gonadotropins → profound hypoestrogenism | Leuprolide, goserelin, nafarelin |
| GnRH antagonists | Immediate competitive receptor blockade without initial flare | Elagolix (FDA-approved for endometriosis) |
| Aromatase inhibitors | Block local estrogen production in implants; used for refractory DIE | Letrozole, anastrozole (with add-back) |
| NSAIDs/COX-2 inhibitors | Symptomatic pain relief; reduce PGE₂ | |
| Danazol | Synthetic androgen; inhibits pituitary-gonadal axis; rarely used now (hirsutism, hepatotoxicity) |
Add-back therapy: When GnRH agonists are used >6 months, low-dose estrogen (CEE 0.625 mg) or high-dose progestin (norethindrone 5 mg) is added to prevent bone loss and menopausal symptoms. — Goodman & Gilman's
Surgical
| Approach | Indication |
|---|---|
| Laparoscopic excision (cystectomy) | Preferred for endometriomas — lower recurrence, better spontaneous pregnancy rates than ablation (Cochrane evidence) |
| Ablation/coagulation | Superficial peritoneal implants |
| Radical excision (DIE) | Rectovaginal, bowel, or ureteral involvement — multidisciplinary approach with colorectal/urologic surgery |
| Hysterectomy ± bilateral oophorectomy | Definitive treatment in women who have completed childbearing with severe refractory disease |
Excision is superior to ablation for pain recurrence and spontaneous pregnancy rates. Endometriomas should be approached excisionally where possible. Cases with extensive disease should be performed by surgeons trained in retroperitoneal dissection. — Berek & Novak's
Fertility & Complications
- Infertility affects up to 40% — mechanisms include tubal occlusion by adhesions, impaired folliculogenesis, peritoneal inflammatory milieu inhibiting fertilization
- Endometrioma — repeated hemorrhage destroys ovarian cortex → reduced ovarian reserve (↓ AMH)
- Malignant transformation — rare; mainly from DIE and ovarian endometriomas → endometrioid or clear cell ovarian carcinoma
- Adhesions — post-inflammatory and post-surgical; risk of bowel obstruction, fistula formation
- Ureteral involvement — silent hydronephrosis/renal damage possible
Recent Evidence (PubMed, 2024–2026)
- A 2024 systematic review (PMID 39067455) in Human Reproduction Update highlights the central role of fibrosis in endometriosis — fibrotic remodeling drives pain and organ dysfunction and is increasingly recognized as a therapeutic target.
- Evidence on anti-fibrotic approaches and novel agents (antifibrotics, anti-VEGF, immunomodulators) is an emerging area of research.
- Nutritional interventions (omega-3, antioxidants) show potential but require further RCT evidence (PMID 38047410).
Sources: Robbins & Kumar Basic Pathology, p. 689 | Berek & Novak's Gynecology | Goodman & Gilman's Pharmacological Basis of Therapeutics | Harrison's Principles of Internal Medicine 22E | Grainger & Allison's Diagnostic Radiology
This is a shared conversation. Sign in to Orris to start your own chat.