properties of cardiac muscle
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I have everything needed for a comprehensive, well-illustrated answer. Here it is:
Properties of Cardiac Muscle
1. Syncytial (Functional Syncytium)
Cardiac muscle fibers are made up of individual cells connected in series and in parallel. At each intercalated disc, adjacent cell membranes fuse to form gap junctions — permeable communicating channels that allow rapid diffusion of ions. This allows action potentials to travel from one cardiac cell to the next with negligible resistance, making the heart muscle behave as a syncytium even though individual cells are anatomically separate.
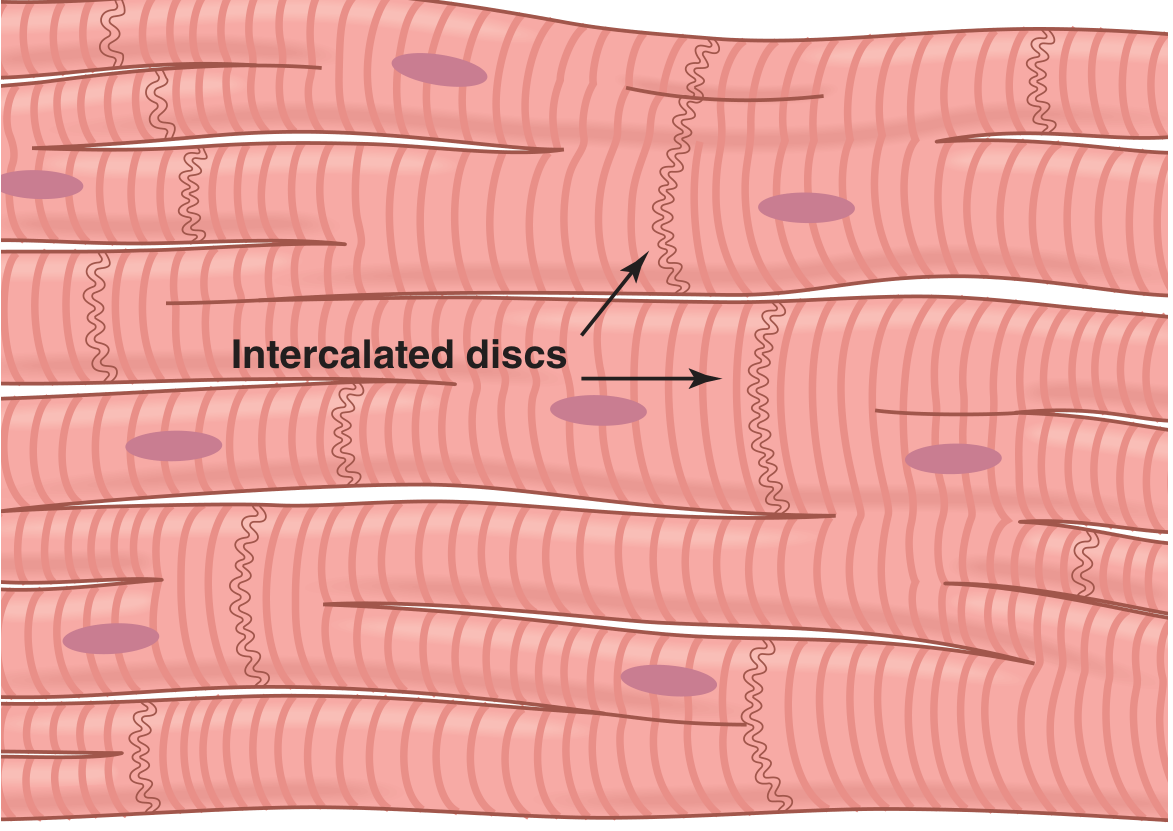
The heart is divided into two functional syncytia: the atrial syncytium (walls of both atria) and the ventricular syncytium (walls of both ventricles), separated by fibrous AV rings. Conduction between them occurs only via the AV bundle, allowing the atria to contract slightly before the ventricles.
2. Automaticity (Rhythmicity)
Cardiac muscle — particularly SA node pacemaker cells — can spontaneously generate action potentials without any external neural input. This property is called automaticity or autorhythmicity. After each impulse, the membrane potential in pacemaker cells gradually depolarizes (the prepotential or pacemaker potential) driven by:
- Declining I
K(potassium current falls after repolarization) - Activation of I
h(funny current, a mixed Na⁺/K⁺ current through HCN channels activated by hyperpolarization) - T-type Ca²⁺ channel current completing the prepotential
- L-type Ca²⁺ channel current producing the actual upstroke
The hierarchy of automaticity: SA node > AV node > His-Purkinje > ventricular muscle.
3. Action Potential with Plateau
The ventricular action potential averages ~105 mV amplitude (from −85 mV resting to ~+20 mV peak), with a characteristic plateau phase lasting ~0.2–0.3 seconds — making contraction ~15× longer than skeletal muscle.
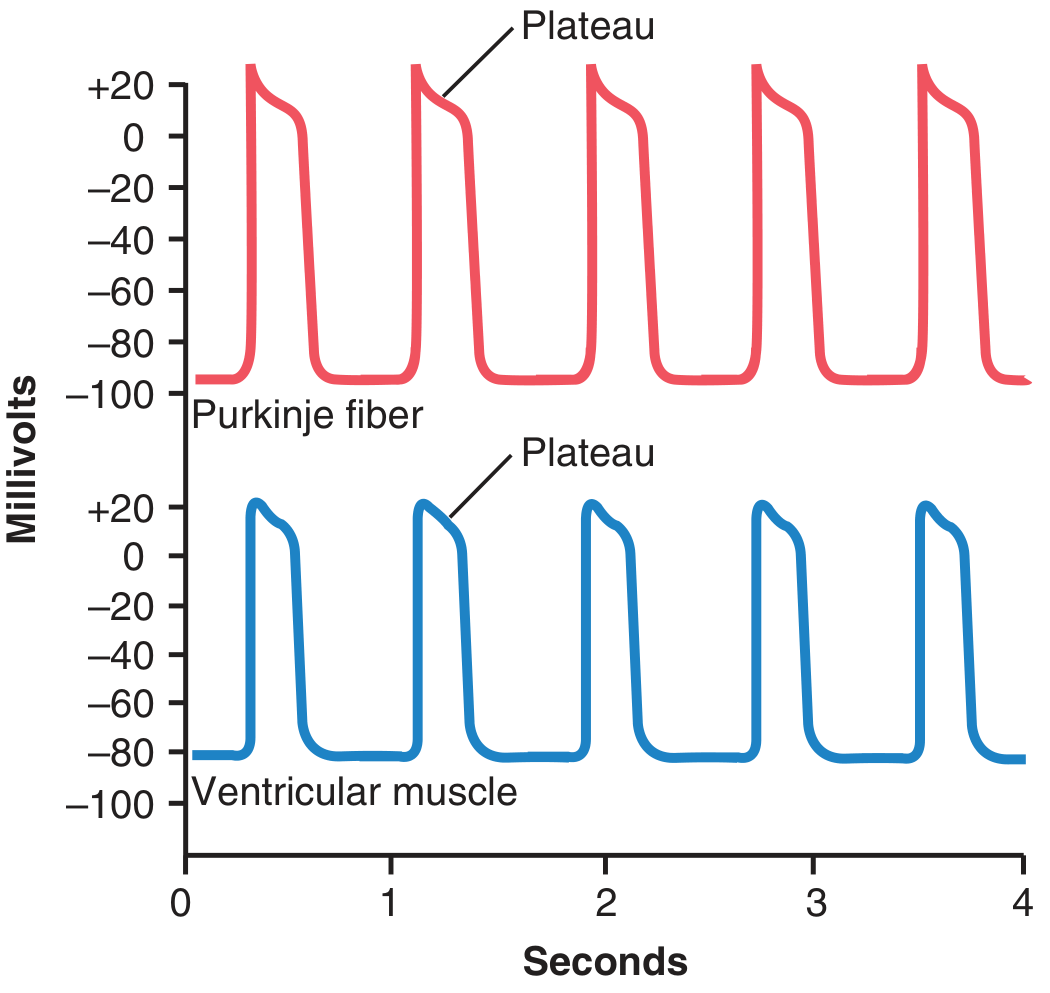
Phases:
| Phase | Event | Ion responsible |
|---|---|---|
| 0 — Depolarization | Fast voltage-gated Na⁺ channels open | Na⁺ influx (I |
| 1 — Initial repolarization | Fast Na⁺ channels close; transient K⁺ efflux | K⁺ efflux |
| 2 — Plateau | Slow L-type Ca²⁺ channels open; K⁺ channels close | Ca²⁺ influx, ↓ K⁺ efflux |
| 3 — Rapid repolarization | Ca²⁺ channels close; slow K⁺ channels open | K⁺ efflux |
| 4 — Resting potential | ~−80 to −90 mV in working myocardium | Stable |
Two key reasons the plateau exists in cardiac but not skeletal muscle:
- Cardiac membranes have slow voltage-gated Ca²⁺ channels (L-type) that remain open for 0.2–0.3 s.
- Cardiac membrane K⁺ permeability decreases ~5-fold immediately after onset of the action potential (partly due to Ca²⁺ influx), preventing early repolarization.
4. Excitation-Contraction Coupling (Ca²⁺-Dependent)
Cardiac E-C coupling uses calcium-induced calcium release (CICR):
- Action potential spreads along the sarcolemma and into T-tubules (5× wider than in skeletal muscle, with mucopolysaccharide-bound Ca²⁺).
- L-type Ca²⁺ channels (dihydropyridine receptors, DHPR) in the T-tubule membrane open → Ca²⁺ influx from extracellular fluid.
- This triggers ryanodine receptor (RyR2) channels in the sarcoplasmic reticulum to release a larger Ca²⁺ store into the sarcoplasm.
- Cytosolic Ca²⁺ binds troponin C → cross-bridge formation → contraction.
Unlike skeletal muscle, cardiac muscle depends on extracellular Ca²⁺ for full contraction because the SR is less well developed. Without Ca²⁺ influx via T-tubules, contraction would be markedly reduced.
5. Refractory Period (Long Absolute Refractory Period)
Cardiac muscle remains in its absolute refractory period from phase 0 through ~mid-phase 3 (until membrane potential reaches approximately −50 mV during repolarization). The relative refractory period lasts until phase 4.
- Ventricular ARP: 0.25–0.30 s (nearly as long as the contraction itself)
- Atrial ARP: ~0.15 s
- Relative refractory period: additional ~0.05 s
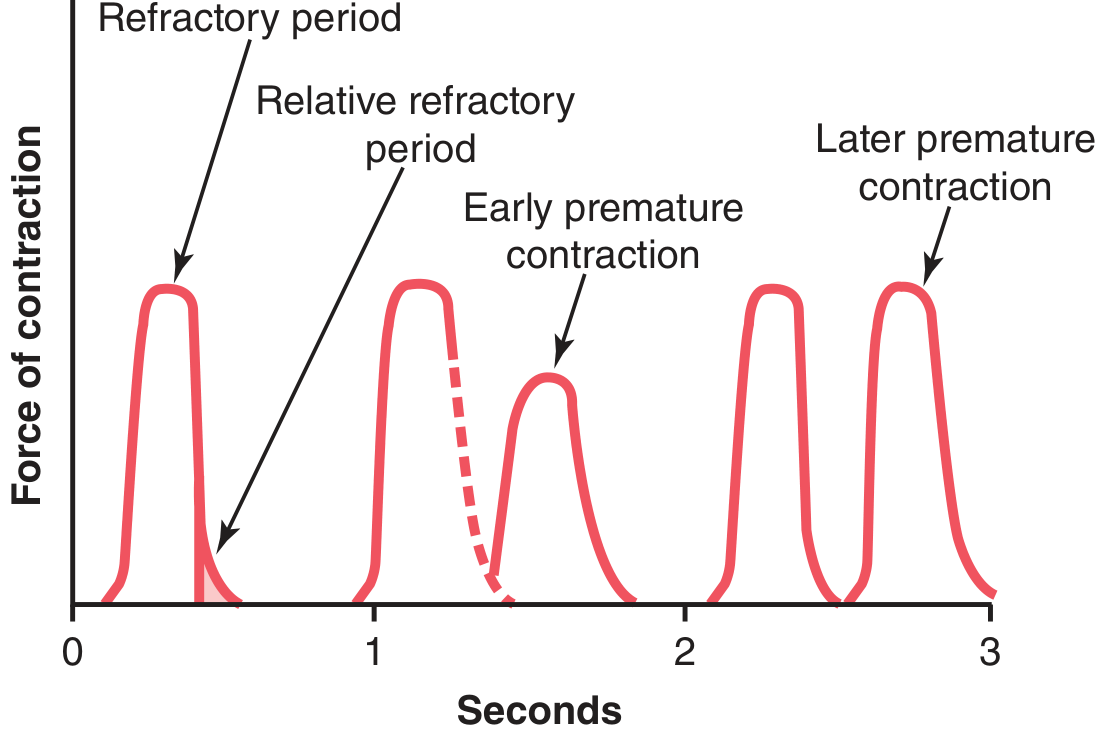
Clinical significance: Because the ARP lasts almost as long as the contraction, tetanic summation is impossible in cardiac muscle — this is an essential safety feature, as sustained tetanus would be fatal.
6. All-or-None Contraction
When an action potential is initiated, the entire myocardium contracts maximally given the current physiological conditions. There is no graded twitch response as in skeletal motor units. The strength of contraction can be modulated (e.g., by preload, afterload, catecholamines, Ca²⁺ concentration), but each individual fiber either contracts fully or not at all.
7. Frank-Starling Mechanism (Length-Tension Relationship)
Within physiological limits, the force of cardiac contraction increases with increasing initial fiber length (preload). Increased venous return → greater end-diastolic volume → greater stretch of sarcomeres → increased overlap of actin-myosin filaments at optimal length → more forceful contraction. This allows the heart to automatically match output to venous return.
8. Conduction Velocity
| Structure | Conduction velocity |
|---|---|
| Atrial/ventricular muscle | 0.3–0.5 m/s |
| AV node | 0.02–0.05 m/s (slow, causes the AV delay) |
| Purkinje fibers | Up to 4 m/s (rapid spread to ventricles) |
Summary
| Property | Key feature |
|---|---|
| Syncytium | Gap junctions at intercalated discs allow electrical spread |
| Automaticity | SA > AV > His-Purkinje hierarchy |
| Action potential | Plateau due to L-type Ca²⁺ channels + ↓ K⁺ permeability |
| E-C coupling | CICR (Ca²⁺ influx triggers SR Ca²⁺ release) |
| Refractory period | Long ARP prevents tetanus |
| All-or-none | Each cell contracts maximally or not at all |
| Frank-Starling | Length-force relationship; output matches venous return |
Sources: Guyton and Hall Textbook of Medical Physiology, pp. 122–126; Ganong's Review of Medical Physiology, 26th ed., p. 522
This is a shared conversation. Sign in to Orris to start your own chat.