Type if nystagmus, mechanisms, physiology of nystagmus, how it’s generate , different type, peripheral vs central , Patho physiology in disease, with example using standard article and book
nystagmus types mechanisms classification
nystagmus peripheral central differentiation vestibular
nystagmus pathophysiology vestibular ocular motor
What Was Covered
Physiology & Generation
Types
Peripheral vs. Central
Disease Pathophysiology
Vestibolo ocular response and its physiology and Patho physiology and disease with examination. Use standard article and textbook
vestibulo-ocular reflex physiology pathophysiology
video head impulse test vHIT vestibular function
HINTS exam head impulse nystagmus skew stroke
HINTS bedside examination acute vestibular syndrome
What Was Covered
Physiology
Types of VOR
Cerebellar Adaptation
Velocity Storage
Clinical Examination (10 tests detailed)
Pathophysiology in 10 diseases
Different between giddiness, dizziness and vertigo , peripheral vs central vertigo , Patho physiology, test and treatment, explain all this in detail using standard article and textbook
Different between giddiness, dizziness and vertigo , peripheral vs central vertigo , Patho physiology, test and treatment, explain all this in detail using standard article and textbook
dizziness vertigo diagnosis classification management
BPPV Epley maneuver treatment vertigo
vestibular neuritis treatment corticosteroids vertigo
Meniere disease diagnosis treatment guidelines
vestibular rehabilitation exercise vertigo balance
persistent postural perceptual dizziness PPPD treatment
Summary of What Was Covered
1. Definitions Distinguished
2. Peripheral vs Central (Full Comparison Table)
3. Pathophysiology
4. Examination (10 Tests)
5. Treatment
Please provide answer
- A follow-up on the previous topics (nystagmus, VOR, vertigo)?
- A new medical topic?
- A specific disease, mechanism, or clinical scenario?
Different between giddiness, dizziness and vertigo , peripheral vs central vertigo , Patho physiology, test and treatment, explain all this in detail using standard article and textbook
- Definitions — Giddiness vs Dizziness vs Vertigo (with the 4-subtype classification from Adams & Victor)
- Peripheral vs Central Vertigo — full 12-feature comparison table from Harrison's, Rosen's, and Neuroanatomy through Clinical Cases
- Pathophysiology — BPPV (canalithiasis), Vestibular Neuritis (HSV-1 deafferentation), Ménière's (endolymphatic hydrops + K⁺ toxicity), Posterior Circulation Stroke (PICA/Wallenberg, AICA), Vestibular Migraine, PPPD, and more
- Examination — 10 clinical tests with full technique and interpretation: Dix-Hallpike, Head Impulse Test, HINTS exam (with 2026 meta-analysis evidence), Romberg, Fukuda, caloric testing, audiometry, and neuroimaging
- Treatment — Epley maneuver (with diagram), vestibular neuritis management (3 systematic reviews on steroids + VRT), Ménière's stepwise treatment, PPPD (Cochrane-level evidence for SSRIs + CBT + VRT), posterior fossa stroke emergency management, and a complete drug summary table
Can please provide it again
Giddiness, Dizziness, and Vertigo: A Complete Clinical Review
PART 1 — DEFINITIONS AND DISTINCTIONS
"The term dizziness is applied by the patient to a number of different sensory and psychic experiences — a feeling of rotation or whirling as well as non-rotatory swaying, weakness, faintness, light-headedness, or unsteadiness… These experiences fall into four categories: (1) vertigo; (2) near syncope; (3) disequilibrium; and (4) ill-defined light-headedness, or 'giddiness.'" — Adams and Victor's Principles of Neurology, 12th Ed.
1.1 Vertigo
- Implies an acute asymmetry of vestibular inputs from the two labyrinths or their central pathways
- Most specific form of dizziness for vestibular disease
- Accompanied by nausea, vomiting, tendency to fall
- Worsened by head movement and (in peripheral causes) by eye opening
- Can be peripheral (labyrinth / vestibular nerve) or central (brainstem / cerebellum)
"Vertigo — an illusion of self or environmental motion — implies an acute asymmetry of vestibular inputs from the two labyrinths or in their central pathways." — Harrison's Principles of Internal Medicine, 22nd Ed. (2025)
1.2 Dizziness (Non-Vertiginous Subtypes)
| Subtype | Core Sensation | Primary Mechanism |
|---|---|---|
| Presyncope / near-syncope | Faintness, blackout feeling, about to faint | Brain hypoperfusion — cardiac arrhythmia, orthostatic hypotension, vasovagal, aortic stenosis |
| Disequilibrium | Off-balance, unsteadiness when walking (no spinning), better when sitting | Balance system failure — multisensory deficits, cerebellar ataxia, proprioceptive loss, parkinsonism |
| Non-specific light-headedness | Floating, swimming, fuzzy, unreal | Anxiety, hyperventilation, hypoglycaemia, anaemia, medications |
1.3 Giddiness
- Not a rotational vertigo
- Not presyncope
- Fuzzy, floating, unreal sensation
- Mechanism: hyperventilation → ↓PaCO₂ → cerebral vasoconstriction → light-headedness + perioral/digital paraesthesiae
- Associated: anxiety disorders, panic attacks, somatoform disorders
- Diagnosis of exclusion after organic causes are ruled out
"Ill-defined light-headedness, or 'giddiness,' a symptom that often accompanies anxiety." — Adams and Victor's Principles of Neurology, 12th Ed.
1.4 Bedside Differentiation: Key History Questions
| Question | Vertigo | Presyncope | Disequilibrium | Giddiness |
|---|---|---|---|---|
| "Is the room spinning?" | YES | No | No | No |
| "Feel like you'll faint/black out?" | No | YES | No | Sometimes |
| "Only when walking, better sitting?" | No | Sometimes | YES | No |
| Positional head-change trigger | YES (BPPV) | No | No | No |
| Duration | Seconds (BPPV) / Hours (Ménière) / Days (neuritis) | Seconds | Chronic | Variable |
| Nausea | Prominent | Mild | Absent | Absent |
| Orthostatic BP drop | No | Yes | No | No |
PART 2 — PHYSIOLOGY OF BALANCE
- Vestibular system — semicircular canals (angular acceleration), otoliths/maculae (linear acceleration + gravity). Dominant for rapid head signals.
- Visual system — retinal slip, optic flow, horizon reference
- Somatosensory/Proprioceptive system — joint position sensors, muscle spindles, plantar mechanoreceptors, cervical spine afferents
"Any disease that disrupts these neural mechanisms may give rise to vertigo and disequilibrium. The interdependence of the two schemata (self and environment) is ascribed to the fact that the various sense organs are usually activated simultaneously by any body movement." — Adams and Victor's Principles of Neurology, 12th Ed.
PART 3 — PERIPHERAL vs. CENTRAL VERTIGO
3.1 Full Comparison Table
| Feature | Peripheral | Central |
|---|---|---|
| Site of lesion | Labyrinth, vestibular nerve (CN VIII) | Brainstem, cerebellum, vestibular cortex |
| Onset | Sudden | Gradual or sudden |
| Severity of vertigo | Often severe initially | Usually mild–moderate (but can be severe in stroke/haemorrhage) |
| Duration | Seconds (BPPV); hours (Ménière); days (neuritis) | Weeks–months continuous; seconds–minutes (TIA) |
| Nystagmus direction | Unidirectional, horizontal ± torsional | Direction-changing on lateral gaze; purely vertical; purely torsional |
| Fixation effect on nystagmus | Suppressed by visual fixation | Not suppressed (may worsen) — use Frenzel lenses |
| Head Impulse Test (HIT) | Positive (catch-up saccade toward lesion) | Negative (normal VOR — alarming central sign) |
| Skew deviation | Absent | May be present — vertical ocular misalignment on cover test |
| Hearing loss / tinnitus | Common (labyrinthitis, Ménière's) | Rare (unless AICA territory stroke) |
| Associated neurological signs | Absent | Diplopia, dysarthria, dysphagia, facial numbness, Horner syndrome, hemiplegia, limb ataxia |
| Postural instability | Can stand (uncomfortable) | May be unable to stand at all (cerebellar lesion) |
| Alexander's law | Follows it | Often does not follow |
| Examples | BPPV, vestibular neuritis, Ménière's, labyrinthitis | Posterior fossa stroke/haemorrhage, MS (INO), cerebellar degeneration, Wallenberg syndrome, tumour |
3.2 Critical Pearls for Peripheral vs Central
- Pure vertical nystagmus → always central
- Direction-changing nystagmus with change of gaze → always central
- Normal head impulse test in acute constant vertigo → red flag for posterior fossa stroke
- Inability to stand unaided → suggests central cause
- Neurological deficits + vertigo → treat as posterior fossa emergency
- "Acute dizziness with normal head impulse testing may indicate cerebellar ischemia rather than vestibular neuritis." — Cummings Otolaryngology HNS
PART 4 — PATHOPHYSIOLOGY IN MAJOR CONDITIONS
4.1 Benign Paroxysmal Positional Vertigo (BPPV)
- Otoconia (CaCO₃ crystals) detach from utricular macula (due to head trauma, aging, osteoporosis, viral infection, or idiopathic)
- Migrate into a semicircular canal — posterior canal ~90%, horizontal canal ~8%, anterior canal rare
- Head repositioning → gravity moves the free-floating canalith plug → hydrodynamic pressure on cupula → anomalous vestibular signal → VOR mismatch → brief intense vertigo + nystagmus
- Cupulolithiasis variant: debris adheres to cupula → making it gravity-sensitive; no latency, longer duration, less fatigable
- Posterior canal → upbeat + torsional (upper poles beating toward the down-ear)
- Horizontal canal → horizontal geotropic (canalithiasis) or apogeotropic (cupulolithiasis)
4.2 Vestibular Neuritis (Acute Unilateral Vestibulopathy)
- Presumed HSV-1 reactivation in vestibular ganglia → inflammation/demyelination of the superior division of the vestibular nerve
- Sudden complete/partial unilateral vestibular deafferentation
- Intact side fires at ~90 spikes/sec; affected side fires less → brain interprets this as continuous head rotation away from the lesion
- Spontaneous horizontal-torsional nystagmus (fast phase toward intact side), severe vertigo, nausea, oscillopsia
- No cochlear involvement = no hearing loss (key differentiator from labyrinthitis)
- Recovery: central vestibular compensation over days–weeks via brainstem/cerebellar plasticity; nystagmus resolves even without peripheral recovery
4.3 Labyrinthitis
4.4 Ménière's Disease (Endolymphatic Hydrops)
- Excess endolymph → distension of the membranous labyrinth (endolymphatic hydrops)
- Periodic ruptures of Reissner's membrane → K⁺-rich endolymph enters perilymph space → K⁺ intoxication of hair cells and CN VIII afferents
- Irritative phase: burst of excitation → nystagmus toward affected ear
- Paretic phase: inhibitory block → nystagmus away from affected ear
- Attack self-terminates as K⁺ is reabsorbed and Reissner's membrane re-heals
- ≥2 spontaneous vertigo episodes, each lasting 20 min–12 hours
- Audiometrically documented low-to-mid frequency SNHL in affected ear (at least once)
- Fluctuating aural symptoms (hearing loss, tinnitus, or fullness) in affected ear
- Not better explained by another vestibular diagnosis
4.5 Vestibular Migraine
4.6 Posterior Circulation Stroke / TIA
- Vestibular nuclei + inferior cerebellar peduncle involved
- Features: severe vertigo, vomiting, dysphagia, hoarseness (CN IX, X), ipsilateral facial pain/temperature loss (CN V), contralateral body pain/temperature loss, ipsilateral Horner syndrome, ipsilateral limb ataxia
- Nystagmus: horizontal-torsional + skew deviation + ocular tilt reaction
- HIT: often negative (central pattern — no catch-up saccade)
- Labyrinthine artery arises from AICA → sudden unilateral end-organ failure + ipsilateral hearing loss
- "Central mimic" of vestibular neuritis — acute hearing loss + positive HIT (peripheral pattern but central cause)
- HINTS+ (adding hearing loss) specifically improves AICA stroke detection
- Sudden severe vertigo + severe headache + inability to stand
- Ipsilateral CN VI palsy (brainstem compression by expanding hematoma)
- Neurosurgical emergency — can cause tonsillar herniation
- Episodic vertigo lasting minutes + other posterior fossa symptoms (diplopia, dysarthria, dysphagia)
- Risk factors: age >60, hypertension, atherosclerosis, diabetes
4.7 Persistent Postural-Perceptual Dizziness (PPPD)
4.8 Perilymph Fistula
PART 5 — CLINICAL EXAMINATION
5.1 Approach (Three Core Questions)
- Is it dangerous? → arrhythmia? stroke? cerebellar haemorrhage?
- Is it vestibular? → spinning (vertigo) vs fainting vs imbalance
- If vestibular — peripheral or central?
5.2 Orthostatic Blood Pressure
- Measure supine → standing at 1 and 3 minutes
- Normal: systolic drops ≤10 mmHg, pulse rises ≤10 bpm
- Orthostatic hypotension: systolic drop >20 mmHg or diastolic >10 mmHg → presyncope mechanism confirmed
5.3 Otoscopy
- Perforated/scarred TM → perilymph fistula
- Cholesteatoma, fluid → labyrinthitis source
- Vesicles in EAC (herpes zoster) → Ramsay Hunt syndrome (VZV)
5.4 Dix-Hallpike Test (Gold Standard for BPPV)
- Patient sits upright; head turned 45° toward the ear being tested
- Rapidly lower patient to supine with head extended 20–30° below horizontal (over table edge)
- Patient keeps eyes open; examiner observes nystagmus and asks about vertigo
- Hold 30–60 seconds; return to seated; repeat other side
| Feature | Peripheral (BPPV) | Central |
|---|---|---|
| Latency to nystagmus | 2–5 seconds | Immediate or none |
| Nystagmus direction | Upbeat + torsional (toward down-ear) | Any; pure vertical is always central |
| Duration | <30 seconds | Prolonged / persistent |
| Fatigability with repetition | Yes (habituates) | No |
| Vertigo accompanying nystagmus | Always present | Nystagmus can occur without vertigo |
5.5 Head Impulse Test (Halmagyi-Curthoys)
- Positive (catch-up saccade after impulse): VOR gain reduced → peripheral vestibular lesion on that side → reassuring in acute vertigo
- Negative (eyes remain locked on target): VOR intact → in patient with acute constant vertigo → central cause must be excluded
5.6 HINTS Exam
| Component | Peripheral (Safe) | Central (Dangerous — stroke) |
|---|---|---|
| H — Head Impulse Test | Positive (catch-up saccade) | Negative (normal VOR) |
| N — Nystagmus type | Unidirectional horizontal ± torsional | Direction-changing with gaze |
| T — Test of Skew (cover-uncover) | No skew deviation | Vertical skew (hypertropia) present |
- Xu et al., Am J Emerg Med 2026 (PMID 41045791) — HINTS family meta-analysis: high sensitivity/specificity for stroke in AVS
- Anburajan et al., J Neurol 2026 (PMID 41665728) — HINTS+ systematic review: acute hearing loss component critical for AICA infarct detection
5.7 Romberg Test
- Patient stands feet together, arms at sides
- Test 1: eyes open; Test 2: eyes closed
- Positive Romberg: can stand eyes open but sways/falls eyes closed → vestibular or proprioceptive dysfunction (visual compensation present when eyes open)
- Cannot stand even with eyes open → cerebellar lesion or severe bilateral vestibular failure
- Falls toward the side of the lesion in peripheral vestibular disease
5.8 Fukuda (Unterberger) Stepping Test
- Patient marches in place 60 steps with eyes closed, arms extended
- Normal: moves <0.5 m, rotates <30°
- Unilateral vestibular hypofunction: rotates/deviates toward side of lesion
- Low sensitivity but useful as adjunct
5.9 Head-Shaking Nystagmus Test
- Frenzel lenses applied; examiner shakes head horizontally at 2 Hz for 20 cycles; observe on stopping
- Normal: no post-shaking nystagmus
- Unilateral vestibular hypofunction: post-shaking nystagmus beating toward intact side (velocity storage asymmetry)
- Tests VOR at 1–2 Hz (complementary to HIT and caloric testing)
5.10 Caloric Testing (Bithermal)
- Head tilted back 30° (supine) to align horizontal canal vertically
- Warm (44°C) → convection mimics ampullopetal flow → nystagmus same side (WARM = SAME)
- Cold (30°C) → convection mimics ampullofugal flow → nystagmus opposite side (COLD = OPPOSITE)
- COWS mnemonic: Cold-Opposite, Warm-Same
- Canal paresis (CP): [(RC+RW) − (LC+LW)] / (RC+RW+LC+LW) × 100%; CP >25% = unilateral hypofunction
- Tests each horizontal SCC independently at very low frequency (~0.003 Hz)
5.11 Audiometry (PTA + Tympanometry)
- Fluctuating low-to-mid frequency SNHL (250–1000 Hz) → Ménière's disease
- Sudden unilateral SNHL → labyrinthitis or AICA stroke
- Conductive hearing loss → otitis media, cholesteatoma
- Type B flat tympanogram → effusion
- MRI IAM with gadolinium → if vestibular schwannoma suspected (progressive unilateral SNHL without vertigo)
5.12 Neuroimaging
- MRI brain with DWI/FLAIR (posterior fossa sequences): mandatory if central cause suspected
- Critical caveat: MRI DWI sensitivity for posterior fossa infarct is only ~50% in the first 24–48 hours — HINTS by trained examiner outperforms early MRI DWI in this window
- Urgent CT head: if cerebellar haemorrhage suspected (sudden severe headache + vertigo + unable to stand)
PART 6 — TREATMENT
6.1 BPPV — Repositioning Maneuvers
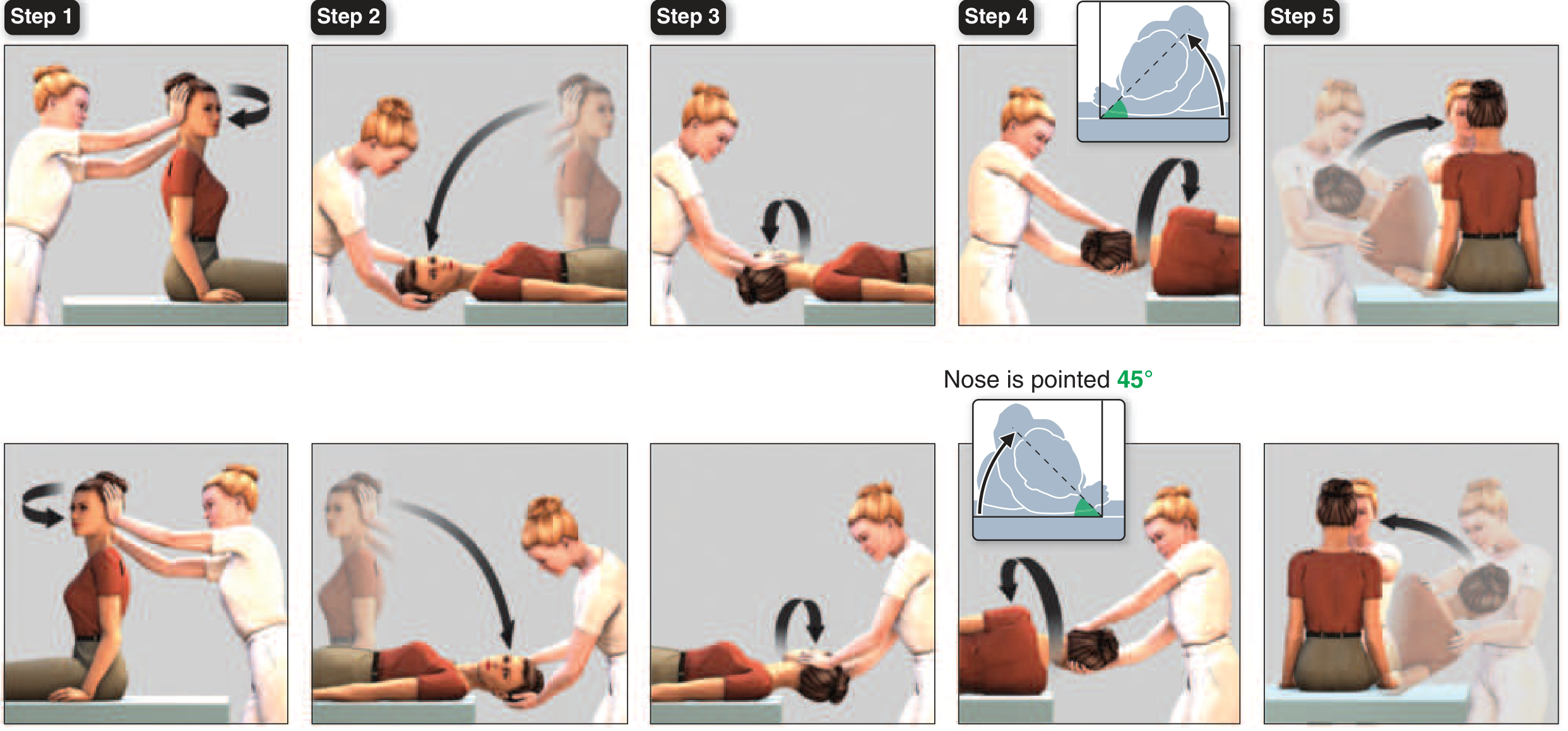
- Seated, head turned 45° right → rapidly recline to head-hanging 30° below horizontal; hold 30 sec
- Turn head 90° to left (now 45° left); hold 30 sec
- Roll body onto left side, turn head additional 90° (nose pointing down 45°); hold 30 sec
- Sit up on left side; repeat to confirm resolution
- Semont liberatory maneuver: rapid 270° lateral body rotation; comparable efficacy to Epley
- BBQ Roll (Lempert/360° maneuver): for horizontal canal BPPV
- Gufoni maneuver: for apogeotropic horizontal canal BPPV
- Brandt-Daroff exercises: self-administered habituation; useful for mild/recurrent BPPV
- Valsted et al., Am J Audiol 2024 (PMID 38900988) — systematic review of 4 repositioning maneuvers: Epley maneuver most effective; all 4 superior to sham
- Si et al., BMC Neurol 2025 (PMID 40098079) — network meta-analysis: confirms Epley as first choice
6.2 Vestibular Neuritis
- Vestibular suppressants (limit to first 3–5 days only):
- Prochlorperazine 5–10 mg IM/IV/oral (dopamine antagonist + antiemetic)
- Meclizine 25–50 mg TDS (H₁ antihistamine)
- Dimenhydrinate (Dramamine)
- Ondansetron for refractory vomiting
- Prolonged use impairs central compensation — must stop after Day 3–5
- Prednisolone 60–100 mg/day for 3 days, tapered over 3 weeks
- Three key systematic reviews:
- Leong et al., Otolaryngol HNS 2021 (PMID 33525978): improves caloric testing recovery but clinical benefit mixed
- Oliveira et al., Acad Emerg Med 2023 (PMID 35975654) — GRACE review: no definitive evidence for improved patient-reported outcomes; decision should be individualised
- Hidayati et al., Medicina 2022 (PMID 36143898): VRT + steroids together superior to either alone
- Gaze stabilisation exercises (head movements while fixating)
- Balance retraining
- Habituation exercises
- Encourage early mobilisation — immobility delays central compensation
6.3 Ménière's Disease
- Low-sodium diet (<1500 mg/day) — reduces endolymph volume fluctuations
- Diuretics: hydrochlorothiazide 25 mg + triamterene 37.5 mg (or acetazolamide)
- Avoid caffeine, alcohol, tobacco, stress
- Betahistine 16–24 mg TDS (H₁ agonist/H₃ antagonist) — increases cochlear blood flow; widely used in Europe/Asia; evidence mixed (BEMED trial showed no superiority over placebo for attack frequency)
- For refractory cases; preserves hearing; can be repeated
- Preferred in patients wishing to preserve hearing
- Selectively destroys vestibular hair cells while relatively sparing cochlear function
- Very effective for vertigo control (>90%) but carries risk of hearing loss
- Used when hearing is already severely compromised
- Endolymphatic sac decompression/shunting: non-ablative; modest evidence
- Vestibular nerve section: ablative, preserves hearing; highly effective
- Labyrinthectomy: removes labyrinth entirely; only if hearing already lost; definitive
6.4 Posterior Circulation Stroke — Emergency
- Immediate CT brain: exclude haemorrhage
- If ischaemic and eligible: IV alteplase (tPA) within 4.5 hours
- Endovascular thrombectomy for large vessel occlusion within 24 hours
- Admit to stroke unit; aspirin 300 mg loading immediately
- Dual antiplatelet (aspirin + clopidogrel) for 21 days after non-cardioembolic minor stroke/TIA
- Statin therapy; aggressive vascular risk factor control
- Cerebellar haemorrhage >3 cm or with brainstem compression: emergency suboccipital craniectomy
6.5 Vestibular Migraine
- Triptans (sumatriptan 50–100 mg oral), antiemetics (prochlorperazine), NSAIDs
- Benzodiazepines for severe brief attacks
- Amitriptyline 10–50 mg, topiramate, propranolol, valproate, venlafaxine
- Lifestyle: trigger avoidance (sleep hygiene, dietary triggers, stress management)
6.6 PPPD
- Sertraline, escitalopram, venlafaxine
- Cochrane systematic review (Webster et al., PMID 36906836): confirms SSRIs/SNRIs benefit
- Response takes 4–8 weeks
- Cochrane systematic review (Webster et al., PMID 36912784): VRT significantly reduces dizziness handicap
- Habituation, gaze stabilisation, balance retraining, gradual exposure to provocative environments
- Zang et al. meta-analysis (PMID 38350404): CBT + VRT superior to VRT alone
- Addresses fear-avoidance, hypervigilance to balance signals, catastrophising
6.7 Orthostatic Hypotension / Presyncope
- Increase fluid and salt intake; compression stockings
- Counter-manoeuvres (leg crossing, squatting, tensing leg muscles before standing)
- Slow position changes; raise head of bed
- Review medications (antihypertensives, diuretics)
- Pharmacological: fludrocortisone 0.1–0.3 mg/day (mineralocorticoid); midodrine 2.5–10 mg TDS (α₁ agonist); pyridostigmine; droxidopa (neurogenic OH)
PART 7 — DRUG SUMMARY TABLE
| Drug Class | Example | Mechanism | Indication |
|---|---|---|---|
| Antihistamines | Meclizine, cinnarizine, dimenhydrinate | H₁ block → reduces vestibular input to brainstem | Short-term acute vertigo, motion sickness |
| Anticholinergics | Scopolamine patch | Muscarinic block → reduces vestibular input | Motion sickness prevention |
| Phenothiazines | Prochlorperazine | D₂ block + antiemetic | Acute vomiting with vertigo |
| Benzodiazepines | Diazepam, lorazepam | GABA-A potentiation → vestibular sedation | Severe acute vertigo (short-term only) |
| Betahistine | Betahistine 16–24 mg TDS | H₁ agonist / H₃ antagonist → increases cochlear blood flow | Ménière's prophylaxis |
| Corticosteroids | Prednisolone 60 mg tapered | Anti-inflammatory | Vestibular neuritis (controversial) |
| Diuretics | HCTZ + triamterene | Reduce endolymph volume | Ménière's management |
| SSRIs/SNRIs | Sertraline, venlafaxine | ↓ cortical vestibular hypervigilance | PPPD first-line |
| Triptans | Sumatriptan | 5-HT₁B/₁D agonist → craniovascular vasoconstriction | Vestibular migraine acute |
| 4-Aminopyridine | Dalfampridine | K⁺ channel blocker → improves cerebellar Purkinje cell firing | Downbeat nystagmus, episodic ataxia type 2 |
PART 8 — RED FLAGS ("DANGEROUS DIZZINESS")
| Red Flag | Likely Cause |
|---|---|
| Sudden severe headache ("worst of life") | Cerebellar haemorrhage, subarachnoid haemorrhage |
| Cannot stand unaided | Cerebellar infarct or haemorrhage |
| Diplopia, dysarthria, dysphagia, facial numbness | Posterior fossa stroke (Wallenberg) |
| Ipsilateral facial palsy + vesicles in EAC | Ramsay Hunt syndrome (VZV) |
| Acute unilateral hearing loss + vertigo | AICA infarct (NOT just vestibular neuritis) |
| Normal head impulse test in acute constant vertigo | Central cause until proven otherwise |
| First episode in elderly + multiple vascular risk factors | Vertebrobasilar TIA |
| Progressive unilateral SNHL + no vertigo | Vestibular schwannoma |
KEY TEXTBOOK SOURCES
| Textbook | Sections Used |
|---|---|
| Harrison's Principles of Internal Medicine, 22nd Ed. (2025) | Chapter 24: Dizziness and Vertigo |
| Adams and Victor's Principles of Neurology, 12th Ed. | Dizziness and Vertigo chapter |
| Rosen's Emergency Medicine | Chapter 15, Tables 15.2 & 15.3 |
| Neuroanatomy through Clinical Cases, 3rd Ed. | Chapter 12.6 |
| Localization in Clinical Neurology, 8th Ed. | Systemic causes of vertigo |
| Cummings Otolaryngology HNS | Vestibular physiology and management |
KEY ARTICLES
| PMID | Authors | Journal/Year | Key Finding |
|---|---|---|---|
| 41045791 | Xu et al. | Am J Emerg Med 2026 | HINTS family meta-analysis — high sensitivity for stroke in AVS |
| 41665728 | Anburajan et al. | J Neurol 2026 | HINTS+ systematic review — hearing loss improves AICA detection |
| 38900988 | Valsted et al. | Am J Audiol 2024 | Epley most effective among 4 BPPV maneuvers (systematic review) |
| 40098079 | Si et al. | BMC Neurol 2025 | Network meta-analysis confirms Epley as first-choice for posterior canal BPPV |
| 33525978 | Leong et al. | Otolaryngol HNS 2021 | Steroids improve VOR recovery in vestibular neuritis (meta-analysis) |
| 35975654 | Oliveira et al. | Acad Emerg Med 2023 | GRACE review: no definitive evidence for steroids improving patient outcomes |
| 36143898 | Hidayati et al. | Medicina 2022 | VRT + steroids superior to either alone in vestibular neuritis (meta-analysis) |
| 36912784 | Webster et al. | Cochrane 2023 | Non-pharmacological (VRT) significantly reduces PPPD dizziness handicap |
| 36906836 | Webster et al. | Cochrane 2023 | SSRIs/SNRIs effective pharmacological treatment for PPPD |
| 38350404 | Zang et al. | BJORL 2024 | CBT + VRT superior to VRT alone in PPPD (meta-analysis) |
| 40210232 | Hudson et al. | Otol Neurotol 2025 | 15–40% bilateral progression in Ménière's over 10–20 years |
| 41147266 | Moreno-Ajona | Headache 2026 | PPPD vs vestibular migraine differentiation — narrative review |