Approach to a child with fever and rash in 10marks format for 3rd yr dermatology post graduate theory examination
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching Images
fever rash child maculopapular exanthem differential diagnosis
I now have sufficient material to write a comprehensive, exam-standard 10-mark answer. Here is the full structured response:
Approach to a Child with Fever and Rash
Introduction (0.5 marks)
Fever with rash in a child is a common yet diagnostically challenging presentation. It spans a wide spectrum from benign self-limiting viral exanthems to life-threatening emergencies (meningococcaemia, toxic shock syndrome). A systematic approach — combining a detailed history, morphological characterisation of the rash, and targeted investigations — is essential for accurate diagnosis and timely management.
I. History (1.5 marks)
A. Fever Details
- Onset, duration, pattern (continuous, remittent, intermittent)
- Height of fever: very high (>40°C) suggests bacterial sepsis or Kawasaki disease
- Relation to rash: Did the rash appear with fever or after defervescence?
- Rash appears after fever subsides → Roseola infantum (HHV-6)
- Rash appears with fever → measles, rubella, scarlet fever, dengue
B. Rash Details
- Onset, site of onset, direction of spread (cephalocaudal, centrifugal)
- Evolution (fixed vs. migratory), pruritus, desquamation
C. Associated Symptoms
| System | Clues |
|---|---|
| URTI (3 Cs) | Cough, coryza, conjunctivitis → Measles |
| Sore throat, strawberry tongue | Scarlet fever |
| Lymphadenopathy (postauricular/occipital) | Rubella |
| Arthralgia, retro-orbital pain | Dengue |
| Red eyes, cracked lips, swollen hands | Kawasaki disease |
| Headache, neck stiffness, photophobia | Meningococcal meningitis |
| Slapped-cheek appearance | Erythema infectiosum (Fifth disease) |
D. Epidemiological History
- Vaccination status (MMR, varicella)
- Contact with ill individuals, outbreaks
- Travel history (dengue-endemic areas)
- Drug history (drug hypersensitivity rash mimics viral exanthems)
- Insect/tick bites (Rocky Mountain spotted fever, rickettsial diseases)
II. Examination (2 marks)
A. General Assessment
- Toxic vs. non-toxic appearance: toxic child = bacterial sepsis; well-appearing = viral exanthem
- Vital signs: BP (hypotension → septic shock), pulse, temperature
- Assess for signs of systemic compromise: altered sensorium, poor perfusion, petechiae with fever = emergency
B. Morphological Classification of Rash
| Morphology | Key Diagnoses |
|---|---|
| Maculopapular (blanching) | Measles, rubella, roseola, drug rash, dengue |
| Vesicular/bullous | Varicella, herpes zoster, HSV, hand-foot-mouth (HFMD) |
| Petechial/Purpuric (non-blanching) | Meningococcaemia, ITP, HSP (IgAV), rickettsial |
| Urticarial | Drug hypersensitivity, viral, serum sickness |
| Desquamating/Scarlatiniform | Scarlet fever, Kawasaki, SSSS, TSS |
| Target lesions | Erythema multiforme, Stevens-Johnson syndrome |
The glass tumbler test (blanching): A non-blanching petechial/purpuric rash + fever = meningococcal disease until proved otherwise — treat empirically.
C. Distribution of Rash
| Pattern | Diagnosis |
|---|---|
| Starts at hairline → spreads cephalocaudal | Measles (rubeola) |
| Face first → trunk → limbs; fades in 3 days | Rubella |
| Trunk → spreads peripherally after defervescence | Roseola |
| "Slapped cheeks" + lacy reticular rash on limbs | Erythema infectiosum (B19) |
| Palms, soles, oral mucosa | HFMD, Rocky Mountain spotted fever, secondary syphilis |
| Acral (hands, feet, periungual) | Kawasaki disease |
D. Enanthem (Oral Mucosa)
- Koplik's spots (white spots on buccal mucosa opposite lower molars) → Pathognomonic of measles
- Strawberry tongue → Scarlet fever, Kawasaki disease
- Oral vesicles/ulcers → HFMD (Coxsackievirus A16), herpangina
E. Other Examination Findings
- Lymphadenopathy: cervical (measles, KD, mono), postauricular/occipital (rubella), generalised (EBV, HIV)
- Eye changes: conjunctival injection (KD, measles); uveitis (JIA)
- Hepatosplenomegaly: EBV, dengue, typhoid
- Meningism: Neisseria meningitidis, viral meningitis
- Joint swelling: HSP, reactive arthritis, rheumatic fever
- Desquamation of fingertips (1–2 weeks later): Kawasaki, SSSS, scarlet fever
III. Classification: Fever-Rash Timing
| Pattern | Disease |
|---|---|
| Fever → Rash (simultaneous) | Measles, rubella, scarlet fever, dengue, rickettsia |
| Fever → Defervescence → Rash | Roseola (HHV-6) |
| Rash before fever | Drug eruption |
| Fever + non-blanching rash = EMERGENCY | Meningococcaemia, HSP with sepsis |
IV. Differential Diagnosis by Aetiology (1 mark)
Viral
| Disease | Pathogen | Rash Type |
|---|---|---|
| Measles | Paramyxovirus | Maculopapular, cephalocaudal |
| Rubella | Togavirus | Maculopapular, fades in 3 days |
| Roseola (6th disease) | HHV-6/7 | Macular, post-defervescence |
| Erythema infectiosum (5th disease) | Parvovirus B19 | Slapped-cheek, reticular lacy |
| Varicella | VZV | Pleomorphic vesicular (dew drops on rose petal) |
| HFMD | Coxsackievirus A | Vesicles on palms, soles, oropharynx |
| Dengue | Flavivirus | Islands of white in sea of red |
Bacterial
| Disease | Organism | Rash Type |
|---|---|---|
| Scarlet fever | Group A Streptococcus | Sandpaper, pastia lines, perioral pallor |
| Meningococcaemia | Neisseria meningitidis | Petechial → purpuric → purpura fulminans |
| SSSS | S. aureus (exfoliative toxin) | Superficial bullae, Nikolsky sign + |
| Rickettsial | Rickettsia spp. | Petechial, centripetal (palms/soles) |
Other
- Kawasaki disease: Fever >5 days + ≥4 of 5 criteria (conjunctival injection, oral changes, rash, lymphadenopathy, extremity changes)
- HSP (IgA vasculitis): Palpable purpura on buttocks/extensor limbs + arthritis + abdominal pain + nephritis
- Drug hypersensitivity: Maculopapular, 7–10 days after first dose; pruritic; drug history essential
- SJS/TEN: Target lesions, mucosal involvement, epidermal detachment
V. Investigations (1.5 marks)
| Investigation | Purpose |
|---|---|
| CBC with differential | Leucocytosis (bacterial), leucopenia (viral/dengue), thrombocytopenia (dengue, ITP) |
| Blood culture | Bacterial sepsis |
| CRP, ESR, Procalcitonin | Sepsis biomarkers |
| NS1 antigen, dengue IgM/IgG | Dengue |
| Serology (IgM/IgG) | Measles, rubella, EBV, CMV, parvovirus B19 |
| Throat swab + RADT | Group A Strep (scarlet fever) |
| Paul-Bunnell / Monospot | Infectious mononucleosis |
| Tzanck smear, Viral swab PCR | Varicella, HSV |
| Urine analysis | HSP nephritis, KD |
| ECHO, ECG | Kawasaki disease (coronary artery aneurysm screening) |
| CSF analysis | Meningococcal/viral meningitis |
| Widal, Blood culture | Typhoid (rose spots) |
| Skin biopsy | If diagnosis uncertain (vasculitis, SJS) |
VI. Management Principles (1.5 marks)
A. Emergency Red Flags → Immediate Action
- Non-blanching petechiae/purpura + fever: IV Ceftriaxone 100 mg/kg stat → admit ICU
- Toxic-appearing child: secure IV access, blood cultures, empirical antibiotics
- Suspected SJS/TEN: stop all drugs, admit, ophthalmology, dermatology, ICU
B. Disease-Specific Treatment
| Condition | Treatment |
|---|---|
| Measles | Supportive; Vitamin A (WHO protocol) |
| Varicella | Oral acyclovir (immunocompromised: IV); avoid salicylates (Reye's) |
| Scarlet fever | Penicillin V/amoxicillin ×10 days |
| Meningococcaemia | IV Ceftriaxone; prophylaxis contacts (rifampicin) |
| Kawasaki disease | IVIG 2 g/kg single dose + Aspirin; follow-up ECHO |
| Dengue | Supportive; monitor platelets, haematocrit |
| SSSS | IV anti-staphylococcal antibiotics; wound care |
| Drug eruption | Withdraw offending drug; antihistamines; steroids if severe |
| HSP (IgA vasculitis) | NSAIDs; steroids for severe abdominal/renal involvement |
C. General Supportive Care
- Antipyretics (paracetamol/ibuprofen)
- Adequate oral/IV hydration
- Isolation precautions as appropriate (airborne: measles/varicella; droplet: meningococcal)
- Notification to public health authorities for notifiable diseases (measles, meningococcal)
VII. A Practical Algorithm (0.5 marks)
FEVER + RASH in a Child
│
├── Non-blanching (petechial/purpuric)?
│ └── YES → EMERGENCY: rule out meningococcaemia
│
├── Blanching maculopapular?
│ ├── Koplik's spots → Measles
│ ├── Post-defervescence → Roseola
│ ├── Postauricular LN + faint rash → Rubella
│ ├── Slapped cheeks → Erythema infectiosum (B19)
│ ├── Sandpaper + strawberry tongue → Scarlet fever
│ └── Island of white + thrombocytopenia → Dengue
│
├── Vesicular?
│ ├── Dew drops on rose petal → Varicella
│ └── Palms + soles + mouth → HFMD
│
└── Fever >5 days + ≥4 criteria → Kawasaki disease
Conclusion (0.5 marks)
A meticulous history and rash morphology are the cornerstones of diagnosis. The single most critical discriminator is blanching vs. non-blanching: non-blanching purpura with fever mandates emergency treatment for meningococcaemia. Vaccination status, enanthem, timing of rash relative to fever, and systemic features help narrow the differential. Investigations should be targeted and treatment must be disease-specific with appropriate isolation, contact tracing, and public health notification.
References:
- Tintinalli's Emergency Medicine, Chapters 12 & 142 (Viral Exanthems, Kawasaki Disease)
- Red Book 2021: Report of the Committee on Infectious Diseases (AAP) — Kawasaki Disease, Meningococcal Infections
- Harrison's Principles of Internal Medicine, 22nd Ed. — Meningococcal Disease
- Fitzpatrick's Dermatology, 9th Ed. — Petechial/Purpuric Rashes
- Sherris & Ryan's Medical Microbiology, 8th Ed. — Roseola, Exanthems
Illustrative images for reference:
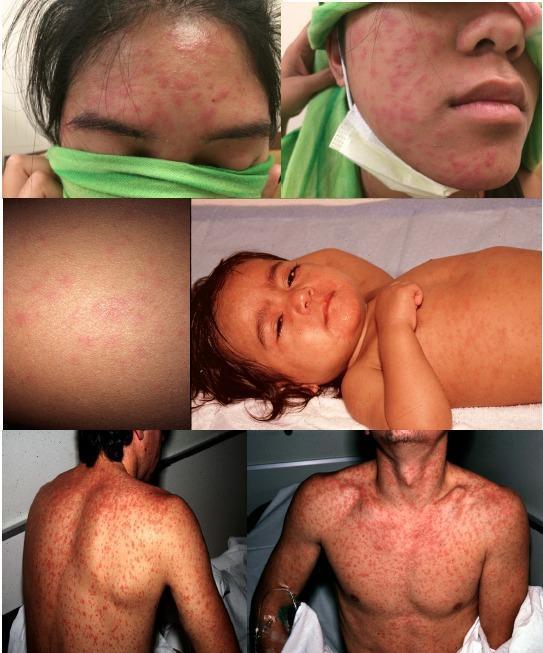
Measles: erythematous maculopapular rash beginning at hairline, spreading cephalocaudally, with coalescence
Scarlet fever: diffuse sandpaper-like erythematous blanching rash; note uniform distribution
Rubella: discrete pink maculopapular lesions; rapid spread; fades by day 3; postauricular lymphadenopathy is characteristic
Dengue: confluent erythema with characteristic "islands of white in a sea of red"
Enumerate Neutrophillic dermatoses .Discuss pyoderma gangrenosum in 10marks format for 3rd yr dermatology post graduate theory examination
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Searching Images
pyoderma gangrenosum ulcer violaceous undermined border
I now have comprehensive textbook data. Let me compile the full 10-mark answer.
Neutrophilic Dermatoses — Enumeration & Pyoderma Gangrenosum
PART A: Enumeration of Neutrophilic Dermatoses (2 marks)
Neutrophilic dermatoses are a group of inflammatory skin conditions characterised histologically by a sterile, non-infectious neutrophilic infiltrate in the skin (epidermis, dermis, or subcutaneous tissue), without primary vasculitis or infection.
Classification
I. Classic Neutrophilic Dermatoses
- Sweet Syndrome (Acute febrile neutrophilic dermatosis) — Dermal neutrophilic infiltrate
- Pyoderma Gangrenosum (PG) — Neutrophilic ulcerating dermatosis
- Behçet Disease — Neutrophilic vasculitis + systemic involvement
- Bowel-Associated Dermatosis-Arthritis Syndrome (BADAS)
- Rheumatoid Neutrophilic Dermatitis
- Subcorneal Pustular Dermatosis (Sneddon-Wilkinson disease)
- IgA Pemphigus (Intraepidermal neutrophilic type)
II. Neutrophilic Dermatoses Associated with Hematologic Malignancies
8. Bullous PG / Bullous Sweet Syndrome
9. Neutrophilic eccrine hidradenitis
10. Histiocytoid Sweet syndrome
III. Neutrophilic Dermatoses Associated with Bowel Disease
11. Pustular psoriasis
12. Pyostomatitis vegetans
IV. Autoinflammatory Syndromes with Neutrophilic Skin Involvement
13. PAPA syndrome (Pyogenic Arthritis, PG, Acne)
14. PASH syndrome (PG, Acne, Suppurative Hidradenitis)
15. PAPASH syndrome
16. SAPHO syndrome
V. Others
17. Erythema elevatum diutinum
18. Acute generalized exanthematous pustulosis (AGEP)
19. Blastomycosis-like pyoderma
20. Reactive arthritis–associated neutrophilic dermatosis
PART B: Pyoderma Gangrenosum (PG) in Detail (8 marks)
1. Definition & Historical Note
Pyoderma gangrenosum is a rare, painful, recurrent, ulcerating neutrophilic dermatosis associated with systemic disease in ~50% of patients. The name is a misnomer — it is neither pyogenic in aetiology nor truly gangrenous.
- First described by Brocq (1908) as "geometric phagedism"
- Term coined by Brunsting, Goeckerman & O'Leary (1930)
2. Epidemiology
- Incidence: ~3–10 cases/million/year
- Affects all ages; peak incidence in the 4th–6th decade
- Female predominance in classic form
- ~4% of cases occur in children (>40% have underlying IBD; 18% have leukemia)
- ~25–50% of cases are idiopathic
3. Pathogenesis
PG is believed to result from dysregulation of innate immunity rather than primary infection. Key mechanisms include:
| Pathway | Role |
|---|---|
| IL-1β overexpression | Central driver; canakinumab (anti-IL-1β) effective clinically |
| IL-8, IL-17, TNF overexpression | Found in lesional skin biopsies |
| Th1/Th17 pathway | IL-23 and JAK/STAT pathway implicated |
| Pathergy | IL-8 and IL-36 overexpression trigger lesions at trauma sites |
| PTPN6 splice variants | Murine model data; links to Sweet syndrome spectrum |
| Autoinflammatory genes | PAPA, PASH syndromes — aberrant inflammasome activation |
Pathergy (development of lesions at sites of minor trauma) occurs in 20–30% of patients — surgeons must be warned; avoid unnecessary debridement.
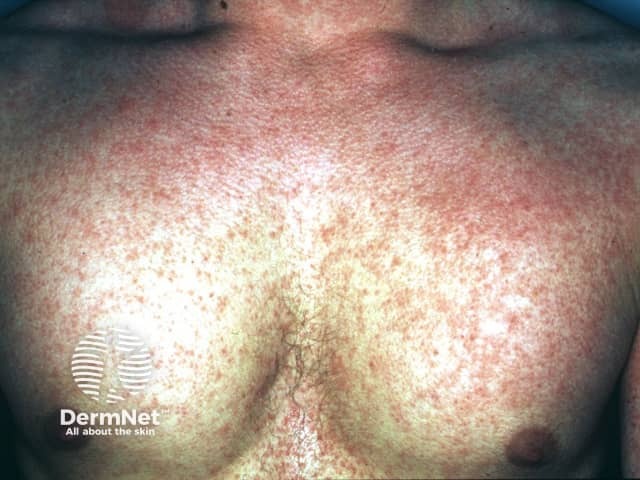
4. Clinical Features
A. Primary Lesion
- Begins as a painful inflammatory pustule on erythematous/violaceous base
- Rapidly ulcerates and expands
B. Classic/Fully Developed Lesion (Ulcerative PG)
- Deeply painful ulcer with:
- Undermined, overhanging, violaceous/gray-purple borders ← pathognomonic sign
- Surrounding erythematous halo
- Purulent or vegetative base
- Most common on lower extremities (pretibial area) and trunk
- Heals with characteristic cribriform (sieve-like) atrophic scars
- Satellite violaceous papules may appear peripherally and fuse ("cheesecloth" pattern early on)
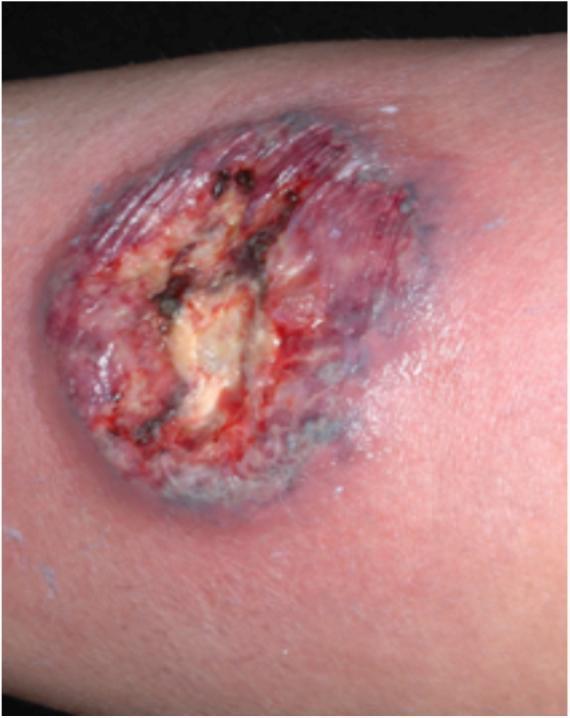
Classic PG: deep ulcer with undermined violaceous border, purulent base, and surrounding erythema
5. Clinical Variants
| Variant | Features | Associated Condition |
|---|---|---|
| Ulcerative (Classic) | Painful, deep, undermined violet-bordered ulcer; lower limbs | IBD, RA |
| Bullous (Atypical) | Superficial blue-gray bullae; dorsal hands and face; overlaps with bullous Sweet syndrome | AML, MDS, IgA gammopathy |
| Pustular | Multiple sterile pustules; rarely ulcerate; resolve without scar | IBD, Behçet |
| Vegetative (Superficial granulomatous) | Superficial, slowly enlarging, cribriform ulceration; verrucous border; trunk; least aggressive | Often no systemic disease |
| Peristomal PG | Around surgical stomas; erosions and ulcers | Post-colectomy, IBD |
| Postsurgical PG | At surgical wounds; misdiagnosed as necrotising fasciitis | Post-breast surgery, etc. |
| Mucosal (Pyostomatitis vegetans) | Labial/buccal mucosa; vegetative plaques studded with pustules | IBD |
6. Associated Systemic Conditions (50% of cases)
| Category | Specific Diseases |
|---|---|
| Inflammatory bowel disease (most common) | Ulcerative colitis > Crohn's disease (1.5–5% of IBD patients) |
| Arthritis | Seronegative/seropositive RA, axial arthropathy |
| Hematologic disorders | AML, CML, CLL, MDS, IgA monoclonal gammopathy, myeloma, polycythemia vera |
| Autoinflammatory syndromes | PAPA, PASH, PAPASH, SAPHO |
| Other | Sarcoidosis, SLE, thyroid disease, HIV |
IBD is more common in patients <65 years; malignancy more common >65 years — Dermatology 2-Volume Set 5e
7. Histopathology
PG has no pathognomonic histology — biopsy is primarily used to exclude mimics.
| Stage | Histological Findings |
|---|---|
| Early lesion | Suppurative folliculitis; follicular rupture |
| Active expanding lesion | Massive dermal oedema; neutrophilic abscess; epidermal neutrophilic abscesses at violaceous border |
| Mature lesion | Fibrosing inflammation at ulcer edge |
| All stages | Sterile infiltrate (no organisms on culture/special stains); no primary vasculitis |
Biopsy should be taken from the active, undermined edge and must include adequate depth (down to panniculus). Tissue must also be sent for bacterial, mycobacterial, fungal, and viral cultures.
8. Diagnosis
PG is a diagnosis of exclusion — no single specific serological or histological test exists.
Three Validated Diagnostic Criteria Systems:
| Criteria | Requirements |
|---|---|
| Su Criteria | Both major criteria + ≥2 minor criteria |
| PARACELSUS Score | Score >10 points (progressive course + violaceous border + other features) |
| Delphi Consensus Criteria | Major criterion (biopsy with neutrophilic infiltrate) + ≥4/8 minor criteria |
Su Criteria:
- Major: (1) Rapid progression of painful necrolytic ulcer with irregular violaceous undermined border; (2) Other causes excluded
- Minor: (1) Pathergy; (2) History of IBD/arthritis; (3) History of pustule/papule ulcerating within 4 weeks; (4) Cribriform scarring; (5) Response to systemic steroids or CSA; (6) Peripheral neutrophilia on biopsy
Investigations
| Investigation | Purpose |
|---|---|
| Skin biopsy (edge + depth) + cultures | Exclude infection, tissue diagnosis |
| CBC, peripheral smear, bone marrow | Exclude haematological malignancy |
| Colonoscopy + stool occult blood | Evaluate for IBD |
| SPEP + immunofixation electrophoresis | IgA/IgG gammopathy |
| ANA, ANCA, antiphospholipid Ab, VDRL | Exclude vasculitis, autoimmune, syphilis |
| CXR, urinalysis | Screen for systemic disease |
9. Differential Diagnosis
Early (non-ulcerative) stage:
- Sweet syndrome, folliculitis, insect bite, panniculitis, Behçet disease, cellulitis, halogenoderma
Ulcerative stage (most important):
| Category | Mimics |
|---|---|
| Vascular | Venous stasis ulcer, arterial occlusive disease, antiphospholipid syndrome |
| Vasculitis | Cutaneous PAN, ANCA vasculitis, SLE |
| Infection | Ecthyma gangrenosum, atypical mycobacteria, deep fungi (blastomycosis, sporotrichosis), leishmaniasis, amebiasis |
| Malignancy | SCC, cutaneous lymphoma, leukemia cutis |
| Other | Calciphylaxis, factitial disease, brown recluse spider bite, hydroxyurea ulcer |
Critical warning: PG misdiagnosed as necrotising fasciitis → aggressive surgical debridement → pathergy → expansion of ulcer. Always consider PG before surgery on chronic ulcers.
10. Treatment
Key principle: Halt inflammation + promote wound healing. Treat underlying disease.
A. Local/Topical (mild/localised disease)
| Treatment | Notes |
|---|---|
| Potent topical corticosteroids | First-line for solitary or slowly progressive lesions |
| Intralesional triamcinolone | Pathergy risk at injection sites |
| Topical tacrolimus 0.1% | Effective; monitor levels if large surface area |
| Topical timolol | Emerging evidence |
| Wound care | Occlusive dressings, whirlpool baths; avoid debridement |
B. Systemic (moderate–severe disease)
| Drug | Dose | Mechanism |
|---|---|---|
| Prednisolone (first-line) | 1 mg/kg/day PO | Blocks IL-1α/β, impairs neutrophil migration |
| Cyclosporine (first-line) | 3–5 mg/kg/day PO | Calcineurin inhibitor; reduces IL-2, TNF-α, IFN-γ |
| IV Methylprednisolone (very severe) | 1 g/day × 3–5 days | Rapid induction |
| Dapsone | 50–200 mg/day | Inhibits neutrophil myeloperoxidase; suppresses IL-8, TNF-α |
| Minocycline | 100 mg BD | Suppresses neutrophil chemotaxis, MMP, cytokines |
| Mycophenolate mofetil | 500 mg–1.5 g BD | Steroid-sparing; inhibits T/B cell proliferation |
| Azathioprine | 2–2.5 mg/kg/day | Purine analogue; steroid-sparing |
| Methotrexate | Up to 30 mg/week | Folate antimetabolite; anti-neutrophil chemotaxis |
C. Biologics (refractory disease)
| Drug | Target | Notes |
|---|---|---|
| Infliximab (best evidence) | TNF-α | 5 mg/kg IV at 0, 2, 6 weeks then 6–8 weekly; RCT evidence (MAPP trial) |
| Adalimumab | TNF-α | 40 mg SC every 1–2 weeks |
| Canakinumab | IL-1β | Especially in autoinflammatory PG (PAPA, PASH) |
| Secukinumab/Ixekizumab | IL-17 | Emerging data |
| Ustekinumab | IL-12/23 | IBD-associated PG |
| Tofacitinib | JAK1/3 | JAK-STAT pathway; refractory cases |
D. Wound Care Principles
- Avoid aggressive debridement — triggers pathergy
- Moist wound environment; non-adherent dressings
- Skin grafting only under adequate systemic immunosuppression
11. Prognosis
- Variable — disease may remit, recur, or pursue a chronic course
- Cribriform scarring is characteristic of healed lesions
- Activity of PG and IBD may run independent courses — colectomy does not reliably cure PG
- Reappearance of PG lesions in malignancy-associated cases may signal cancer relapse
- Mortality directly from PG is rare; morbidity from associated systemic diseases is significant
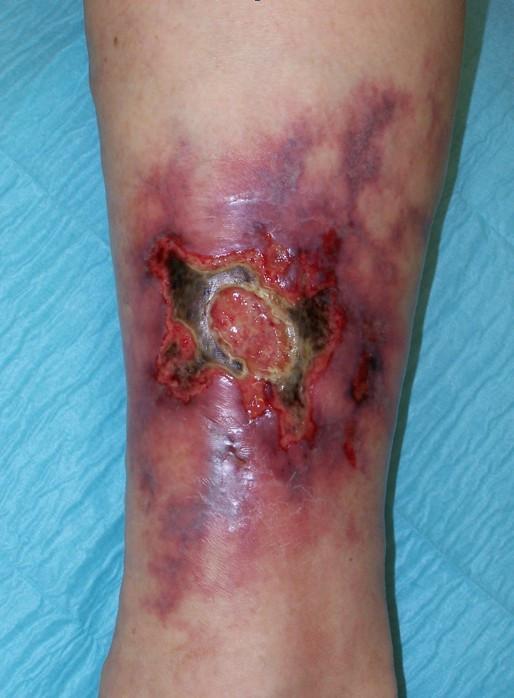
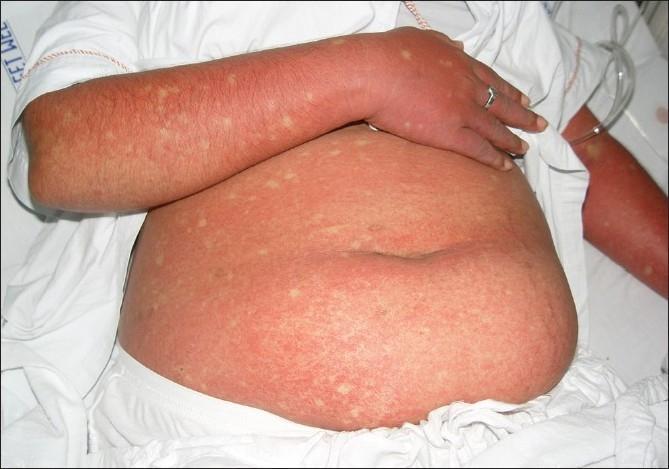
PG: Large irregular ulcer with ragged violaceous undermined borders, erythematous halo, and granulating base — lower extremity
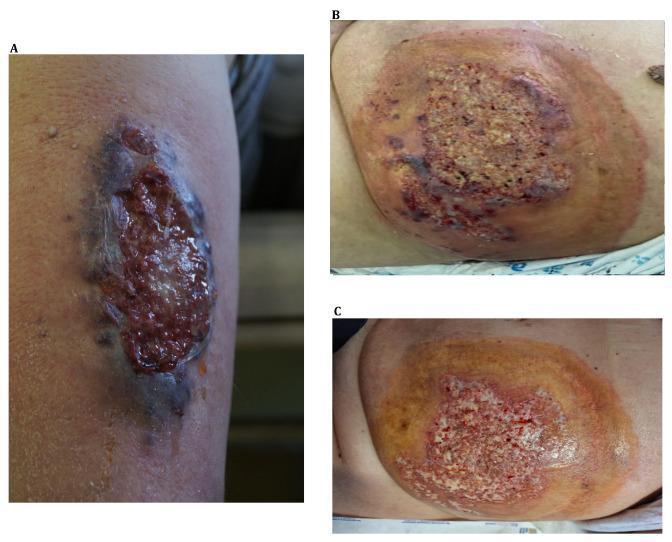
A: Localised PG on leg; B: Extensive abdominal PG in AML patient; C: Same lesion after 2 weeks of cyclosporine + prednisolone — healthy granulation tissue emerging
Summary Table — PG at a Glance
| Feature | Details |
|---|---|
| Aetiology | Neutrophilic dysregulation; idiopathic in 25–50% |
| Primary lesion | Pustule → necrotic ulcer |
| Pathognomonic sign | Undermined violaceous border |
| Most common site | Lower limbs, pretibial |
| Most common association | Inflammatory bowel disease |
| Pathergy | 20–30% |
| Biopsy | Sterile neutrophilic infiltrate; no vasculitis; exclude mimics |
| First-line systemic Rx | Prednisolone, cyclosporine |
| Best biologic evidence | Infliximab (MAPP trial) |
| Healing scar | Cribriform (sieve-like) atrophic scar |
References:
- Dermatology 2-Volume Set, 5th Ed. (Bolognia et al.) — Chapter 25: Pyoderma Gangrenosum, pp. 547–555
- Andrews' Diseases of the Skin, 13th Ed. — Chapter 7: Reactive Neutrophilic Dermatoses, pp. 171–174
- Fitzpatrick's Dermatology, 9th Ed. — Chapter 37: PG Differential Diagnosis & Treatment Tables
- Dermatology 2-Volume Set, 5th Ed. — Table 25.8: PG Diagnostic Criteria (Su, PARACELSUS, Delphi Consensus)
Approach to a patient coming with pruritus in 10marks format for 3rd yr dermatology post graduate theory examination
Finding Sources
Finding Sources
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.