How can we detect?Hiv what’s the proper.test for hiv
Reading File
Finding Sources
Searching PubMed
HIV diagnosis laboratory testing
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have everything needed for a complete, authoritative answer. Here it is:
HIV Detection: The Proper Tests
Overview
HIV diagnosis relies on demonstrating antibodies to HIV and/or directly detecting the virus or its components. Antibodies generally appear in circulation 3–12 weeks after infection. The CDC recommends HIV screening as a routine part of health care for all individuals aged 13–64 at least once, and annually (or every 3–6 months) for high-risk groups.
The Three Categories of HIV Tests
1. 4th-Generation Antigen/Antibody Combination Test ✅ (Recommended First-Line)
This is the current standard recommended by the CDC for initial HIV screening.
- Detects both HIV-1 p24 antigen (a core viral protein) AND HIV-1/HIV-2 antibodies (IgM and IgG) simultaneously
- Can detect infection as early as 2–6 weeks after exposure (window period ~16–18 days for lab-based; ~19–20 days for rapid version)
- Uses ELISA-based technology
- Sensitivity: >99.5%
- Available in lab-based and rapid point-of-care formats (results in ≤30 minutes)
The Western blot, which was previously used for confirmation, is no longer recommended for this purpose.
2. Nucleic Acid Test (NAT) — HIV RNA PCR
Used when:
- The 4th-generation screen is positive but the differentiation immunoassay is negative/indeterminate (to detect acute infection)
- Screening blood products for transfusion
- Diagnosing HIV in infants born to HIV-positive mothers (since maternal IgG crosses the placenta, making antibody tests unreliable in newborns)
| Method | Sensitivity | Window Period |
|---|---|---|
| Quantitative HIV-1 RNA (viral load) | Detects ≥20–40 copies/mL | 6–10 days |
| Qualitative HIV-1 RNA | Detects ≥100 copies/mL | 10–12 days |
| HIV RNA by RT-PCR | Reliable to 20 copies/mL | 7–28 days |
| HIV RNA by bDNA | Reliable to 50 copies/mL | — |
NAT is also critical for monitoring response to antiretroviral therapy (ART) — a rising or non-suppressed viral load signals treatment failure or resistance.
3. Antibody-Only Tests (3rd-Generation)
- Detect IgM and IgG antibodies only (no antigen detection)
- Window period: 22–24 days (lab); 26–32 days (rapid)
- Less preferred than 4th-generation due to longer window period
- Includes rapid tests using oral fluid (saliva) or blood — results in ~20–30 minutes
- FDA-approved home test: OraQuick® In-Home HIV Test (oral fluid, results in ~20 minutes)
The CDC Recommended Testing Algorithm
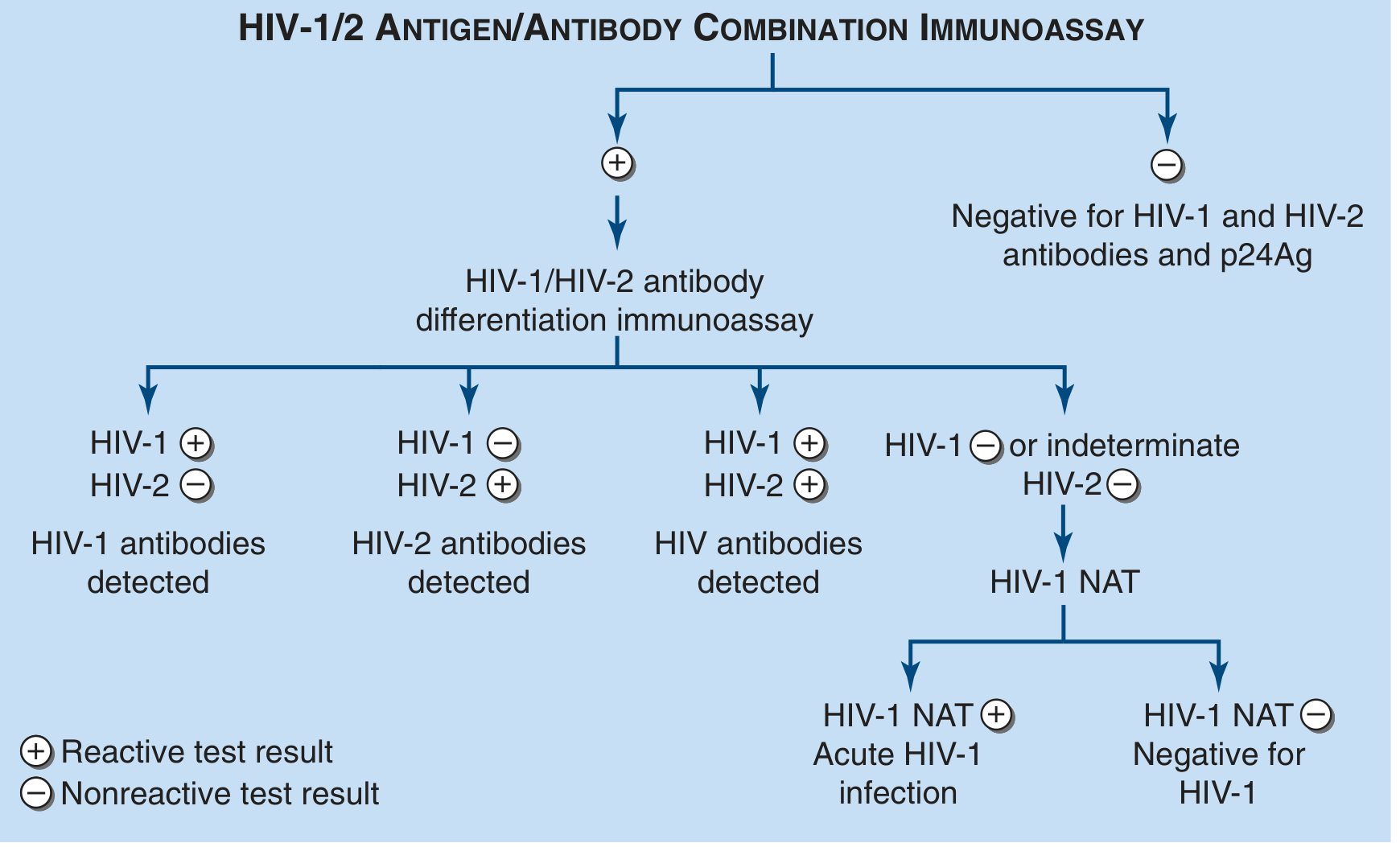
Step 1: Perform a 4th-generation HIV-1/2 antigen/antibody immunoassay
- Negative → HIV ruled out (retest if recent exposure within 3 months)
- Positive/indeterminate → Proceed to Step 2
Step 2: HIV-1/HIV-2 antibody differentiation immunoassay (e.g., Bio-Rad Geenius)
- HIV-1 positive → Confirms HIV-1 infection
- HIV-2 positive → Confirms HIV-2 infection
- Both positive → Confirms HIV infection
- Negative/indeterminate → Proceed to Step 3
Step 3: HIV-1 NAT (nucleic acid test)
- NAT positive → Acute HIV-1 infection confirmed
- NAT negative → HIV-1 negative (Step 1 was a false positive)
Special Situation: Infants Born to HIV-Positive Mothers
Maternal IgG crosses the placenta, so antibody tests cannot be used to diagnose HIV in infants. Instead, use:
- HIV-1 DNA/RNA PCR (or HIV-1 culture)
- Confirm with repeat testing
- HIV DNA PCR positivity rates: 38% at 48 hours of life → 93% at 14 days → 98% at 4 weeks
Summary: Window Periods at a Glance
| Test | What It Detects | Window Period |
|---|---|---|
| Qualitative NAT (RNA PCR) | HIV RNA | ~10–12 days |
| 4th-gen Ag/Ab combo (lab) | p24 antigen + IgM/IgG | ~16–18 days |
| 4th-gen Ag/Ab combo (rapid) | p24 antigen + IgM/IgG | ~19–20 days |
| 3rd-gen antibody EIA (lab) | IgM + IgG | ~22–24 days |
| 3rd-gen antibody EIA (rapid) | IgM + IgG | ~26–32 days |
| Western blot (no longer recommended) | IgG | ~35–40 days |
Key Points
- A negative test during the window period does not rule out infection — repeat testing 3 months after exposure is advised
- During the window period, an infected person can still transmit the virus via sexual contact or blood donation
- All positive rapid/home tests must be confirmed by a healthcare professional with laboratory testing
- HIV RNA quantification (viral load) is essential for monitoring ART efficacy — goal is suppression to <50 copies/mL
— Harrison's Principles of Internal Medicine 22E, pp. 1632–1633; Sherris & Ryan's Medical Microbiology, 8th Ed., pp. 733–735; Dermatology 2-Volume Set, 5th Ed., p. 4100
This is a shared conversation. Sign in to Orris to start your own chat.