Write a detailed note for college exam on fetal circulation and its physiology.
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content from four major textbooks and two excellent diagrams. Let me compile the detailed exam note.
Fetal Circulation and Its Physiology
Sources: The Developing Human (Moore & Persaud), Langman's Medical Embryology, Ganong's Review of Medical Physiology, Medical Physiology (Boron & Boulpaep)
1. Introduction
The fetal cardiovascular system is designed exclusively to serve prenatal needs. Because the lungs do not perform gas exchange in utero, the fetal circulation is fundamentally different from the adult circulation in three key ways:
- The placenta serves as the combined lung, gut, and kidney of the fetus.
- The heart pumps in parallel (not in series as in adults) — thanks to two right-to-left shunts.
- Three specialized shunts divert blood away from the non-functional lungs and reduce hepatic first-pass.
The fetal heart begins beating and circulating blood by the 4th week of gestation.
2. The Placenta as the Gas Exchange Organ
The placenta is the "fetal lung." Its maternal portion is a large blood sinus into which the fetal villi project, containing small branches of the fetal umbilical arteries and vein. O₂ is taken up by fetal blood and CO₂ is discharged across the walls of the villi. However, this exchange is less efficient than in the lungs because the cellular layers covering the villi are thicker and less permeable than alveolar membranes.
- Umbilical vein carries oxygenated blood from placenta to fetus (O₂ saturation ~80%)
- Umbilical arteries (×2) carry deoxygenated blood from fetus to placenta (O₂ saturation ~58–60%)
Memory tip: "Umbilical Vein carries O₂-rich blood" — the Vein is the exception to the rule that veins carry deoxygenated blood.
3. The Three Fetal Shunts
These are the most important structural features of fetal circulation:
| Shunt | Location | Direction | Bypasses |
|---|---|---|---|
| Ductus venosus | Umbilical vein → IVC | Right-to-right | Liver (hepatic sinusoids) |
| Foramen ovale | Right atrium → Left atrium | Right-to-left | Pulmonary circulation |
| Ductus arteriosus | Pulmonary trunk → Descending aorta | Right-to-left | Lungs |
4. Step-by-Step Blood Flow in the Fetus
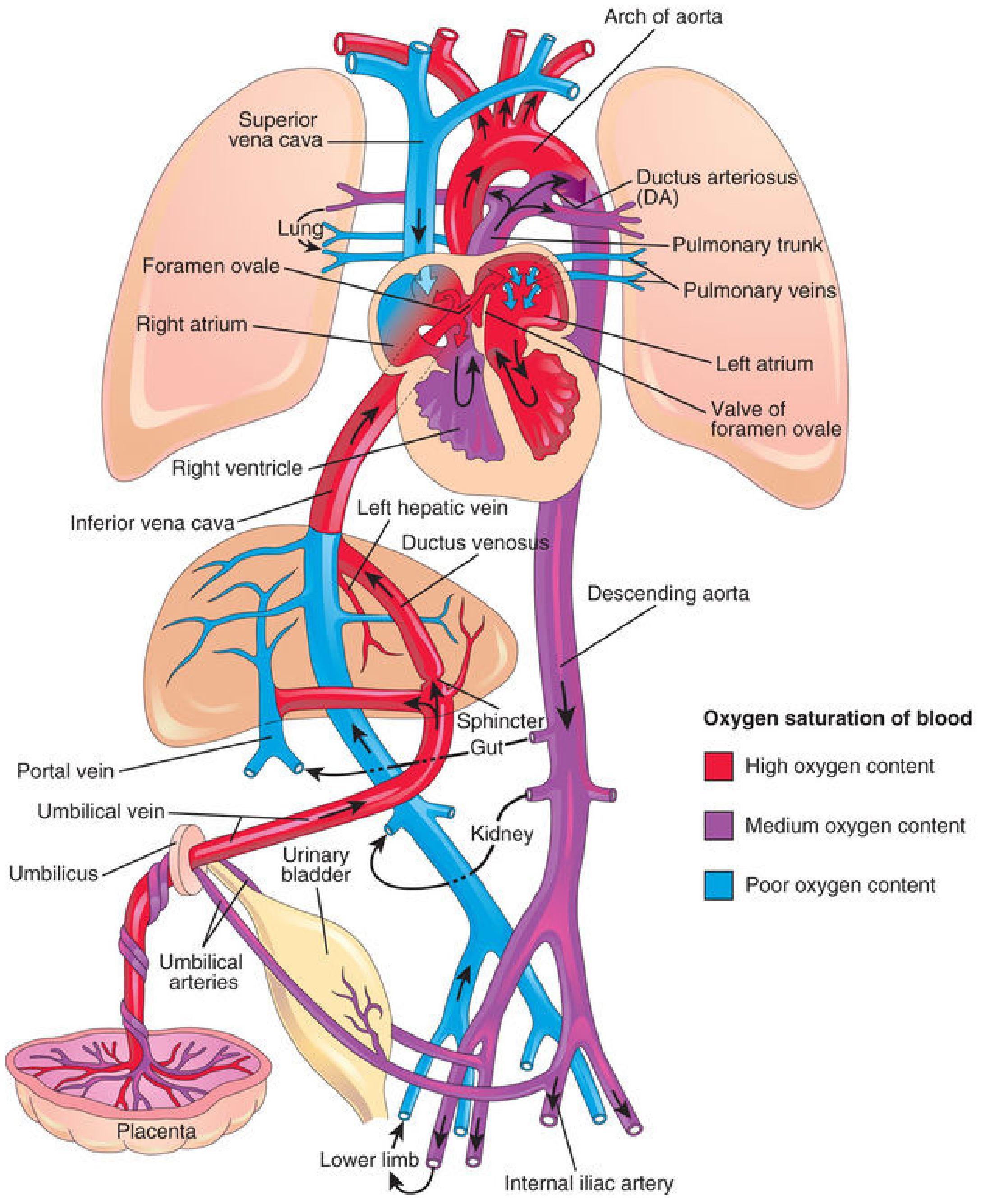
Fig. 1 – Fetal circulation. Red = high O₂, purple = medium O₂, blue = low O₂. (The Developing Human)
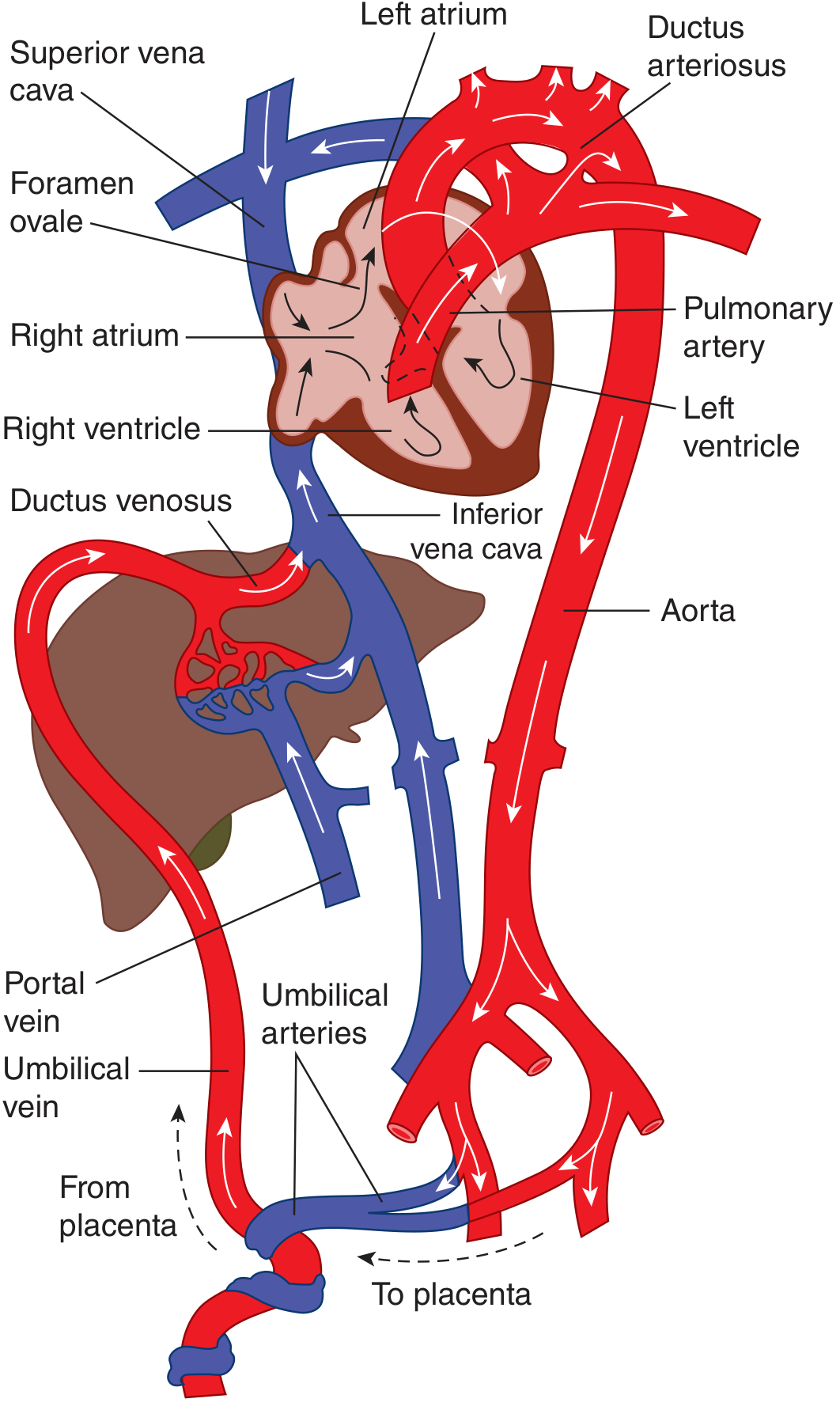
Fig. 2 – Circulation in the fetus. Most oxygenated blood from IVC is diverted via foramen ovale to left atrium → aorta → head. Deoxygenated blood from SVC goes via pulmonary artery → ductus arteriosus → descending aorta → umbilical arteries. (Ganong's)
Step-by-step route:
① Placenta → Umbilical Vein
- Highly oxygenated blood (~80% saturation) leaves the placenta via the single umbilical vein under high pressure.
② Umbilical Vein → Liver / Ductus Venosus
- On approaching the liver, the blood splits:
- ~50% passes through the ductus venosus directly into the IVC (bypasses the liver)
- ~50% enters the hepatic sinusoids via portal circulation and reaches the IVC through hepatic veins
- Both streams merge in the IVC, mixing with deoxygenated blood from the lower limbs → IVC saturation ~67%
③ IVC → Right Atrium
- The blood enters the right atrium and is guided toward the foramen ovale by the valve of the inferior vena cava (Eustachian valve).
- The crista dividens (lower edge of septum secundum) diverts most IVC blood through the foramen ovale into the left atrium.
- A small portion remains in the right atrium and mixes with poorly oxygenated blood from the SVC and coronary sinus.
④ Foramen Ovale → Left Atrium → Left Ventricle → Ascending Aorta
- Blood that crosses the foramen ovale mixes with a small amount of deoxygenated pulmonary venous return in the left atrium.
- From the left ventricle, it enters the ascending aorta.
- The first branches of the ascending aorta are the coronary and carotid arteries → therefore the heart and brain receive the best-oxygenated blood in the fetus.
⑤ Right Atrium → Right Ventricle → Pulmonary Trunk
- Mixed (medium-saturation) blood from the right atrium (SVC + small IVC component) passes to the right ventricle and is ejected into the pulmonary trunk.
- Pulmonary vascular resistance is high in fetal life (vasoconstriction of unexpanded lung vessels) → only ~10% enters the lungs.
⑥ Pulmonary Trunk → Ductus Arteriosus → Descending Aorta
- ~90% of blood in the pulmonary trunk passes through the ductus arteriosus into the descending aorta, mixing with blood from the proximal aorta.
- From the descending aorta:
- ~65% → umbilical arteries → back to the placenta for reoxygenation
- ~35% → viscera and inferior body (kidneys, lower limbs, gut)
Sites of Blood Mixing (Langman's Classification)
| Site | What mixes |
|---|---|
| I. Liver | Umbilical vein + small portal blood |
| II. IVC | Oxygenated blood + deoxygenated blood from lower limbs/pelvis |
| III. Right atrium | IVC blood + SVC blood (head and arms) |
| IV. Left atrium | Foramen ovale blood + pulmonary venous return |
| V. Descending aorta | Proximal aortic blood + ductus arteriosus flow |
5. Why the Heart Pumps in Parallel
Due to the patent foramen ovale and ductus arteriosus, the left and right ventricles pump in parallel in the fetus. The right ventricular output is slightly greater than the left (possible because of the parallel system). After birth, the serial system requires both ventricular outputs to be equal before shunts can close.
6. Fetal Hemoglobin (Hb F) — Physiological Basis
The fetal tissues would suffer hypoxic damage given the low placental O₂ saturation were it not for fetal hemoglobin (Hb F):
- Hb F has a higher O₂ affinity than adult Hb A → its O₂–dissociation curve is shifted left
- Mechanism: Hb F binds 2,3-DPG less effectively than Hb A → less right-shifting of the curve → greater O₂ uptake at any given PO₂
- This allows fetal red cells to load O₂ from maternal blood in the placenta even at relatively low PO₂ values
7. Fetal Pulmonary Circulation
- Pulmonary vasculature is vasoconstricted in utero due to low PO₂ and high CO₂
- Pulmonary vascular resistance is much higher than systemic resistance
- Pulmonary blood flow is minimal (~10% of right ventricular output)
- The walls of pulmonary arteries are thick; thinning begins at birth with lung expansion
8. Circulatory Changes at Birth (Transitional Neonatal Circulation)
At birth, the parallel fetal circuit must switch to a serial adult circuit — largely within minutes.
Trigger: Lung Expansion
With the first breath:
- Lungs expand → pulmonary vascular resistance falls to <20% of the in-utero value
- PO₂ rises → oxygen acts as a vasodilator via nitric oxide (NO) production
- Pulmonary blood flow increases dramatically
Sequential Events at Birth
| Event | Mechanism | Effect |
|---|---|---|
| Umbilical artery constriction | Cold, handling, high O₂ in cord blood; completely constricted within 3–5 min | Prevents blood loss from neonate |
| Ductus venosus constriction | Sphincter contraction | All portal blood now passes through hepatic sinusoids |
| Foramen ovale functional closure | ↑ Left atrial pressure > Right atrial pressure (more pulmonary venous return) presses septum primum against septum secundum | Eliminates right-to-left atrial shunt |
| Ductus arteriosus constriction | ↑ O₂ tension; ↓ prostaglandin E₂ (PGE₂) and prostacyclin (PGI₂); bradykinin from initial lung inflation | Blood flow reverses briefly (left-to-right) then ceases |
Timeline of Ductus Arteriosus Closure:
- Functional closure: within hours of birth
- 24 hours: 20% ducts functionally closed
- 48 hours: ~80% closed
- 96 hours: 100% closed in term neonates
- Anatomic closure (ligamentum arteriosum): by 12th postnatal week
Role of Prostaglandins
- In utero: PGE₂ and PGI₂ keep the ductus arteriosus open (vasodilation of ductal smooth muscle)
- At birth: cyclooxygenase activity falls, PG synthesis drops → ductus constricts
- In prematurity: ductus may stay patent → treated with indomethacin (COX inhibitor)
- TGF-β is involved in the subsequent anatomic closure
Foramen Ovale Permanent Closure:
- Anatomic closure by the 3rd postnatal month — septum primum proliferates and adheres to left margin of septum secundum
- Leaves the oval fossa (fossa ovalis) as a permanent anatomical remnant
9. Adult Derivatives of Fetal Structures
| Fetal Structure | Adult Derivative |
|---|---|
| Umbilical vein (intra-abdominal) | Ligamentum teres hepatis (round ligament of liver) |
| Ductus venosus | Ligamentum venosum (in fissure of liver) |
| Foramen ovale | Fossa ovalis (floor formed by septum primum; border by septum secundum) |
| Ductus arteriosus | Ligamentum arteriosum (left pulmonary artery to arch of aorta) |
| Umbilical arteries (distal) | Medial umbilical ligaments |
| Umbilical arteries (proximal) | Superior vesical arteries (supply urinary bladder) |
10. Clinical Correlates
Patent Ductus Arteriosus (PDA)
- Failure of ductus arteriosus to close after birth
- 2–3× more common in females
- Creates a left-to-right shunt (aorta → pulmonary artery) → pulmonary vascular overload
- Associated with maternal rubella infection and prematurity
- Treatment: Indomethacin (COX inhibitor → ↓ PGE₂ → ductal constriction); surgical ligation if unresponsive
- Continuous "machinery" murmur
Patent Foramen Ovale (PFO)
- ~25–30% of adults have a probe-patent foramen ovale
- Usually asymptomatic but can cause paradoxical embolism (venous thrombus → systemic circulation)
Fetal Asphyxia
- Any disturbance interfering with placental O₂/CO₂ exchange (maternal hypotension, abruption, cord prolapse, severe anemia)
- Causes: low PO₂, high PCO₂, acidosis → depressed myocardial function and reduced cardiac output
- Brain forced to shift to anaerobic metabolism → ATP depletion, endothelial damage, hemorrhage
11. Summary Table: Key Oxygen Saturations in Fetal Circulation
| Location | O₂ Saturation |
|---|---|
| Umbilical vein (from placenta) | ~80% |
| IVC (mixed blood) | ~67% |
| Portal/systemic venous blood | ~26% |
| Ascending aorta / cerebral vessels | Well-oxygenated (~65%) |
| Descending aorta / umbilical arteries | ~58–60% |
12. Quick Review — Key Exam Points
- Umbilical vein carries oxygenated blood (one vein, two arteries in cord)
- Ductus venosus bypasses the liver; ~50% of umbilical venous blood uses it
- Crista dividens directs IVC blood through the foramen ovale
- Heart and brain get the best-oxygenated blood (via ascending aorta)
- High pulmonary vascular resistance is the reason the ductus arteriosus is necessary
- Hb F has higher O₂ affinity than Hb A because it binds 2,3-DPG less
- At birth: lungs expand → ↑ PO₂ → ↑ pulmonary flow → ↑ LA pressure → foramen ovale closes
- O₂ constricts the ductus arteriosus; prostaglandins (PGE₂, PGI₂) dilate it
- Parallel → Serial pump transition is completed within minutes–hours of birth
- Right ventricular wall is thicker than left in fetus/neonate; reverses by end of first month
References: Moore KL et al., The Developing Human (10e), p. 869–880 | Sadler TW, Langman's Medical Embryology (14e), p. 236–238 | Ganong WF, Review of Medical Physiology (26e), p. 614–616 | Boron WF & Boulpaep EL, Medical Physiology (3e), p. 1689
Fetal physiology?
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
I now have comprehensive material from multiple authoritative sources. Let me compile a thorough, exam-ready note on fetal physiology.
Fetal Physiology — Detailed Exam Notes
Sources: Morgan & Mikhail's Clinical Anesthesiology (7e), Mulholland & Greenfield's Surgery (7e), Ganong's Review of Medical Physiology (26e), Medical Physiology — Boron & Boulpaep (3e), Creasy & Resnik's Maternal-Fetal Medicine
1. Overview
Fetal physiology differs fundamentally from adult physiology because:
- The fetus lives in an oxygen-poor, fluid-filled intrauterine environment
- All gas exchange, nutrition, and waste removal occur via the placenta — not the lungs, gut, or kidneys independently
- Multiple organ systems are structurally present but functionally immature, with maturation occurring in a carefully programmed sequence
- The fetus must undergo a profound physiological transition at birth to survive extrauterine life
2. Fetal Cardiovascular Physiology
(Covered in detail in the companion Fetal Circulation note — key points summarized here)
Unique Features
- Heart pumps in parallel (not series) due to two intracardiac/extracardiac shunts
- Three obligatory shunts: ductus venosus, foramen ovale, ductus arteriosus
- Right ventricle output > left ventricle output in fetal life
- Best-oxygenated blood preferentially directed to brain and myocardium via ascending aorta
Cardiac Output
- Neonatal cardiac output is heart-rate dependent (stroke volume is fixed due to non-compliant myocardium)
- Normal fetal heart rate: 110–160 bpm
- Bradycardia = myocardial ischemia, impending arrest
Persistent Fetal Circulation (PFC) / Persistent Pulmonary Hypertension of the Newborn (PPHN)
- Hypoxia or acidosis in the first days of life can prevent or reverse the normal transition → right-to-left shunting resumes
- Creates a vicious cycle: ↑ pulmonary vascular resistance → right-to-left shunt → hypoxemia → acidosis → more ↑ PVR
- Treatment: mechanical ventilation, supplemental O₂, inhaled nitric oxide (pulmonary vasodilator), milrinone, ECMO in refractory cases
3. Fetal Respiratory Physiology
Fetal Breathing Movements (FBM)
- Begin in the late first trimester (~10–12 weeks), confirmed by Doppler
- Occupy <50% of any 24-hour period; become more regular near term
- Purpose: development and conditioning of respiratory muscles and lung growth
- Hypoxia and tactile stimulation promote FBM
- FBM decrease just before labor — clinically important (absent FBM may precede labor)
Fetal Lung Development (5 Stages)
| Stage | Gestational Age | Key Events |
|---|---|---|
| Embryonic | 3–7 weeks | Lung bud from foregut endoderm |
| Pseudoglandular | 7–16 weeks | Airway branching up to terminal bronchioles; no gas exchange possible |
| Canalicular | 16–26 weeks | Vascularization begins; alveolar-capillary approximation begins |
| Saccular | 26–36 weeks | Saccules expand; type II pneumocytes begin surfactant production (~30 weeks) |
| Alveolar | 36 weeks → 2 years postnatal | True alveoli form; surfactant production matures |
Surfactant
- Produced by type II alveolar pneumocytes (alveolar epithelial cells)
- Composed of dipalmitoylphosphatidylcholine (DPPC, lecithin), phosphatidylglycerol, apoproteins
- Function: reduces alveolar surface tension → prevents alveolar collapse at end-expiration
- Production begins ~30 weeks; sufficient for extrauterine life by 34–36 weeks
- L:S ratio (lecithin:sphingomyelin) ≥ 2:1 → lung maturity (clinical test on amniotic fluid)
- Glucocorticoids (e.g., betamethasone given to mother) accelerate surfactant production → given between 24–34 weeks when preterm delivery is anticipated
Lung Fluid
- In utero, alveoli and airways are filled with ~90 mL of plasma ultrafiltrate (lung fluid)
- Volume approximates the functional residual capacity (FRC) of the neonatal lung
- Essential for lung growth — restricting it impairs lung expansion
- At onset of labor: catecholamines and arginine vasopressin (AVP) ↓ fluid production and activate epithelial Na⁺ channels (ENaCs) for active fluid reabsorption
- During vaginal delivery: "vaginal squeeze" expels remaining fluid
- Residual fluid reabsorbed by pulmonary capillaries and lymphatics
Respiratory Distress Syndrome (RDS) / Hyaline Membrane Disease
- Result of surfactant deficiency in premature neonates (<34 weeks)
- Alveolar collapse, progressive hypoxemia
- Treatment: exogenous surfactant therapy, CPAP/mechanical ventilation
Transient Tachypnea of the Newborn (TTN)
- Delayed reabsorption of fetal lung fluid
- Occurs in C-section babies (no vaginal squeeze) and small preterm neonates
- Improves within 2–48 hours; diagnosis of exclusion; supportive treatment
4. Fetal Hematology
Fetal Hemoglobin (Hb F)
- Composition: α₂γ₂ (two alpha + two gamma chains) — vs. adult Hb A (α₂β₂)
- Higher O₂ affinity than Hb A → O₂ dissociation curve shifted LEFT
- Mechanism: Hb F binds 2,3-DPG less effectively → less right-shifting effect → greater O₂ loading at lower PO₂
- Allows fetal RBCs to extract O₂ from maternal blood even at low PO₂ values in the placenta
O₂-Affinity Transition at Birth
- After birth, O₂ demands increase but Hb F impairs O₂ unloading to tissues
- During 1–2 month transition to Hb A: rising 2,3-DPG levels and mild acidosis reduce O₂ affinity → better O₂ delivery to tissues
Hematopoiesis — Sites (Developmental Sequence)
| Period | Primary Site | Secondary Sites |
|---|---|---|
| 3–8 weeks (embryonic) | Yolk sac (extraembryonic mesoderm) | — |
| 6 weeks – 7 months | Liver (main site in mid-gestation) | Spleen (5–8 months) |
| From ~5 months onwards | Bone marrow (takes over gradually) | — |
| Term and postnatal | Bone marrow exclusively | — |
Mnemonic: "Young Liver Synthesizes Blood" — Yolk sac → Liver → Spleen → Bone marrow
Fetal Blood Counts
- Fetal Hb (Hb F) constitutes ~80% of total Hb at birth, falling to <5% by 6 months
- Nucleated RBCs present in fetal circulation (erythroblasts) — marker of fetal stress if seen postnatally in excess
- Fetal polycythemia: Hct ~50–65% at term (compensates for low O₂ saturation)
5. Fetal Renal Physiology
Development and Function
- Definitive kidney (metanephros) begins forming at ~5 weeks; nephrons form between 8–36 weeks
- Fetal kidneys begin producing urine by ~10 weeks
- By 20 weeks, fetal urine is the main source of amniotic fluid
Amniotic Fluid
- Produced by: fetal urine (mainly), fetal lung fluid, umbilical cord, fetal skin (early)
- Swallowed by the fetus at ~500 mL/day (recycled)
- Normal volume at term: 800–1000 mL
- Oligohydramnios (↓ amniotic fluid): renal agenesis, obstructive uropathy → "Potter sequence" (pulmonary hypoplasia, limb deformities, facial features)
- Polyhydramnios (↑ amniotic fluid): impaired fetal swallowing (esophageal atresia, anencephaly), excess production
Renal Immaturity
- Fetal kidneys are physiologically immature — low GFR, poor concentrating ability
- Primary waste excretion occurs via placenta (not fetal kidneys)
- Kidneys gain functional significance only after birth
6. Fetal Gastrointestinal Physiology
Development Timeline
| Week | Event |
|---|---|
| 10 weeks | Intestinal villi with columnar epithelium; first digestive enzymes appear |
| 14 weeks | Peyer patches in ileum (lymphoid tissue) |
| 18 weeks | T and B lymphocytes appear in Peyer patches |
| 20 weeks | Gastric motility detectable |
| 34 weeks | Full intestinal motility; coordinated suck and swallow |
| Term | Mucin production reaches postnatal levels |
Meconium
- Composed of intestinal secretions, mucus, bile, swallowed amniotic fluid, desquamated cells
- Present from ~16 weeks onwards
- Normally expelled within 24–48 hours after birth
- Meconium-stained amniotic fluid indicates fetal distress → risk of meconium aspiration syndrome
GI Maturation Implications
- Many digestive enzymes not at normal levels until 34 weeks
- Premature feeding must be cautious; delayed feeding causes villous atrophy
- Incomplete gut barrier function + lack of IgA → risk of necrotizing enterocolitis (NEC) in premature neonates
7. Fetal Neurological Physiology
CNS Development
- Neural tube closes by week 4; failure causes neural tube defects (anencephaly, spina bifida)
- Neuronal proliferation: 10–18 weeks
- Neuronal migration: 12–24 weeks
- Synaptogenesis and myelination: begins ~24 weeks, continues until years after birth
Germinal Matrix
- High concentration of thin-walled blood vessels present between 24–34 weeks
- Poorly tolerant to blood pressure changes → prone to rupture → intraventricular hemorrhage (IVH) in premature neonates
- Premature infants cannot autoregulate cerebral blood flow → any hemodynamic instability raises IVH risk
Fetal Hypoxia and the Brain
- Fetal tissues have remarkable resistance to hypoxia (poorly understood)
- Chronic O₂ deficiency → forced shift to anaerobic metabolism → ATP depletion, ADP/inorganic phosphate accumulation → neuronal/glial injury → endothelial damage → cerebral/intraventricular hemorrhage
Retina Development
- Retinal angiogenesis: 16 weeks → complete by 40 weeks
- Dependent on hypoxic environment stimulating VEGF
- Premature birth + O₂ exposure inhibits VEGF → arrested retinal vasculogenesis → Retinopathy of Prematurity (ROP)
- Risk highest at <28 weeks; incidence ~84% in this group
8. Fetal Endocrine Physiology
Thyroid
- Fetal thyroid begins concentrating iodine by 10–12 weeks; TSH secretion by 10 weeks
- Fetal T₄ levels rise progressively from mid-gestation
- Maternal T₄ crosses the placenta to provide some hormonal support in early pregnancy
- Neonatal thyroid function is tested at birth (neonatal screening for congenital hypothyroidism)
Adrenal Cortex (Fetal Zone)
- The fetal adrenal gland has a unique "fetal zone" (large inner zone) absent in adults
- Produces DHEA-S → converted by placenta to estrogens (especially estriol)
- Estriol levels are used as a marker of fetoplacental unit function
- At birth: fetal zone involutes → replaced by adult cortex
Insulin and Glucose
- Fetal pancreatic β-cells functional from ~10–12 weeks
- Glucose crosses the placenta freely (facilitated diffusion) → fetal glucose = ~70–80% of maternal
- Fetal glucose stores (glycogen) at birth can last only ~24 hours
- Glucose turnover in neonates: 4–9 mg/kg/min (vs. 2–3 in adults)
- 90% of glucose used by the brain → hypoglycemia (glucose <45 mg/dL) causes neurological injury
Fetal Adrenal Medulla (Stress Response)
- Fetal adrenal medulla produces epinephrine and norepinephrine
- Catecholamine surge at birth: initiates lung fluid clearance, heart rate increase, and mobilization of glycogen
9. Fetal Immune System
- Passive immunity: IgG crosses the placenta from ~22 weeks onwards (active transport via FcRn receptors); maximal transfer in third trimester
- At birth, neonatal IgG levels ≈ maternal levels
- IgM does NOT cross placenta (large molecular weight) → presence of IgM in neonatal blood indicates intrauterine infection (e.g., TORCH)
- IgA absent in gut lumen until well after birth → provided by breast milk (secretory IgA)
- Cellular immunity: T lymphocytes identifiable in Peyer patches by week 18; functional but immature
10. Fetal Energy and Metabolic Physiology
| Parameter | Fetal/Neonatal Value |
|---|---|
| Total energy requirement | 80–120 kcal/kg/day |
| Energy for growth (of total) | Up to 1/3 |
| Glucose turnover | 4–9 mg/kg/min |
| Glycogen stores duration (at birth) | ~24 hours |
| Basal metabolic rate | Higher than adults |
| Primary energy substrate | Fats (energy storage) |
- Carbohydrates predominate in perinatal period; fats take over as primary fuel after early neonatal phase
- Infants unable to feed must receive dextrose-containing IV fluids immediately
11. Physiological Transition at Birth
Respiratory Initiation
- Initiated within 30 seconds of birth, sustained within 90 seconds
- Triggers: mild hypoxia/acidosis, cord clamping, cold, pain, touch, noise, light
- Lung expansion → ↑ alveolar and arterial O₂ tension → potent stimulus for pulmonary arterial vasodilation
- Pulmonary vascular resistance falls to <20% of fetal values with first few breaths
Cardiovascular Transition (Parallel → Serial)
| Event | Trigger | Timing |
|---|---|---|
| Umbilical artery constriction | Cold, handling, ↑ O₂ | 3–5 min |
| Ductus venosus closure | Umbilical cord clamping → ↓ mesenteric return | Minutes |
| Foramen ovale functional closure | ↑ Left atrial pressure > right atrial pressure | Minutes |
| Ductus arteriosus functional closure | ↑ O₂ tension, ↓ PGE₂/PGI₂, bradykinin | Hours (80% by 48h) |
| Ductus arteriosus anatomic closure | TGF-β, intimal proliferation | 2–12 weeks |
Role of Chemical Mediators at Birth
| Mediator | Effect on Ductus Arteriosus | Source |
|---|---|---|
| ↑ O₂ | Constriction | Lungs |
| Bradykinin | Constriction | Released from lung at first inflation |
| Acetylcholine | Constriction | Autonomic |
| PGE₂ / PGI₂ | Dilation (keeps patent in utero) | Locally produced |
| Indomethacin (COX inhibitor) | Constriction | Drug — used in PDA |
| Prostaglandins (exogenous) | Dilation | Drug — used to keep PDA open in duct-dependent CHD |
12. Fetal Asphyxia — Physiological Consequences
Definition: combined hypoxia (low PO₂) + hypercapnia (high PCO₂) + acidosis
Causes:
- Maternal hypotension
- Abruptio placentae
- Cord prolapse (compression in birth canal)
- Severe maternal anemia
- Twin-to-twin transfusion syndrome
Consequences:
- Depressed myocardial function → ↓ cardiac output → worse O₂ delivery
- Brain shifts to anaerobic metabolism: ATP ↓, ADP and phosphate ↑
- Neuronal/glial integrity disrupted
- Endothelial damage → cerebral hemorrhage, IVH
- Chronic deficiency → increased risk of metabolic derangements, cerebral palsy
13. Summary: Key Numbers to Remember
| Fact | Value |
|---|---|
| Umbilical vein O₂ saturation | ~80% |
| Umbilical artery O₂ saturation | ~58–60% |
| IVC O₂ saturation (mixed) | ~67% |
| Surfactant production begins | ~30 weeks |
| Surfactant sufficient for survival | ~34–36 weeks |
| Fetal urine production (main amniotic fluid source) | from ~20 weeks |
| Neonatal glucose turnover | 4–9 mg/kg/min |
| Glycogen stores last | ~24 hours |
| Hb F → Hb A transition | 1–2 months |
| IgG placental transfer starts | ~22 weeks |
| Ductus arteriosus 100% functionally closed | by 96 hours (term) |
| Ductus arteriosus anatomic closure | 2–12 weeks |
| Foramen ovale anatomic closure | ~3 months |
14. Quick Clinical Associations
| Condition | Underlying Physiological Defect |
|---|---|
| RDS (Hyaline Membrane Disease) | Surfactant deficiency (<34 wk) |
| Transient Tachypnea of Newborn | Failure to clear fetal lung fluid |
| Patent Ductus Arteriosus | Failure of ductal constriction (especially in prematurity, ↓ O₂) |
| Persistent Pulmonary Hypertension | Failed drop in PVR; reversion to fetal circulation |
| Retinopathy of Prematurity | O₂ exposure inhibits VEGF → abnormal retinal angiogenesis |
| Necrotizing Enterocolitis | Immature gut barrier + delayed colonization |
| Intraventricular Hemorrhage | Fragile germinal matrix vessels + loss of cerebral autoregulation |
| Potter Sequence | Renal agenesis → oligohydramnios → pulmonary hypoplasia + limb/facial deformities |
References: Morgan & Mikhail's Clinical Anesthesiology (7e), p. 1592–1596 | Mulholland & Greenfield's Surgery (7e), p. 5277–5297 | Ganong's Review of Medical Physiology (26e), p. 614–616 | Boron & Boulpaep Medical Physiology (3e), p. 1689
This is a shared conversation. Sign in to Orris to start your own chat.