Pathophysiology of ocd to explain in conference
Reading File
Finding Sources
Searching PubMed
"obsessive-compulsive disorder"[MeSH Terms] AND pathophysiology
Searching the Web
OCD pathophysiology cortico-striato-thalamo-cortical circuit neurobiology 2024 2025
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Searching Images
OCD cortico-striato-thalamo-cortical circuit pathophysiology diagram
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Reading File
Excellent — I now have rich, high-quality data from multiple authoritative sources. Let me compile the comprehensive conference presentation on OCD pathophysiology.
Pathophysiology of Obsessive-Compulsive Disorder (OCD)
A comprehensive framework for conference presentation
1. Overview & Epidemiology
OCD has a lifetime prevalence of 2–3% worldwide and ranks among the most disabling psychiatric conditions. Onset is typically in adolescence or early adulthood; the two sexes are equally affected. The course is usually waxing and waning, but some patients show progressive psychosocial deterioration. The disorder is strongly comorbid with depression, other anxiety disorders, eating disorders, and tic disorders.
2. Definition of Core Symptoms
| Feature | Description |
|---|---|
| Obsessions | Recurrent, intrusive, ego-dystonic thoughts, images, or impulses (contamination, harm, symmetry, blasphemy) recognized as irrational |
| Compulsions | Repetitive behaviors (washing, checking, ordering, counting) performed to neutralize obsessional anxiety |
| Threshold | Rituals occupy >1 hour/day and cause clinically significant distress or impairment |
| Insight | Patients recognize irrationality — this distinguishes obsessions from delusions |
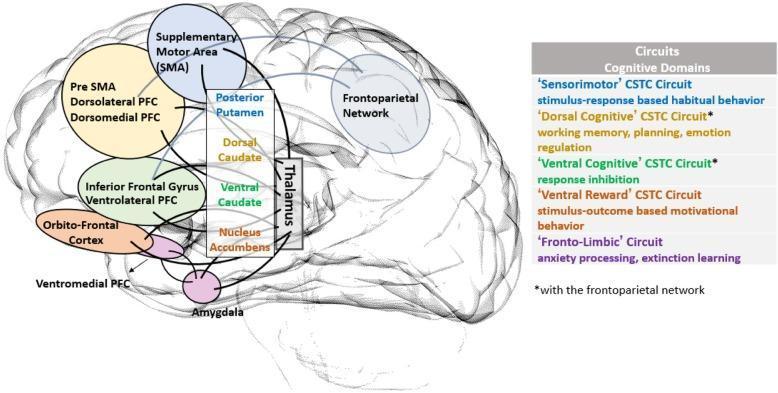
3. The Core Neuroanatomical Circuit: CSTC Loop
The Cortico-Striato-Thalamo-Cortical (CSTC) loop is the central circuit in OCD pathophysiology.
How the normal CSTC loop works:
- Cortex sends information → Striatum (caudate/putamen)
- Striatum modulates output via Globus Pallidus (internal and external) → Subthalamic nucleus → Thalamus
- Thalamus gates information back to Cortex
- The system filters out irrelevant thoughts and actions
The Five CSTC Sub-circuits Implicated in OCD
| Circuit | Cortical Node | Striatal Node | Function Disrupted in OCD |
|---|---|---|---|
| Sensorimotor | SMA, Pre-SMA | Posterior Putamen | Habitual, repetitive behaviors |
| Dorsal Cognitive | DLPFC, Dorsomedial PFC | Dorsal Caudate | Working memory, planning, emotion regulation |
| Ventral Cognitive | Inferior Frontal Gyrus, VLPFC | Ventral Caudate | Response inhibition |
| Ventral Reward | Orbitofrontal Cortex, VMPFC | Nucleus Accumbens | Motivational/goal-directed behavior |
| Fronto-Limbic | VMPFC | Amygdala | Anxiety processing, fear extinction |
4. Direct vs. Indirect Pathway Imbalance
The striatum feeds two competing pathways:
- Direct pathway (excitatory net effect on cortex): Striatum → GPi/SNr → Thalamus → Cortex (promotes action)
- Indirect pathway (inhibitory net effect): Striatum → GPe → STN → GPi/SNr → Thalamus (suppresses action)
In OCD: The direct pathway is hyperactive relative to the indirect pathway, leading to:
- Failure of thalamic gating (thalamus becomes overactive)
- Cortical hyperactivation, especially in the orbitofrontal cortex (OFC) and anterior cingulate cortex (ACC)
- Inability to suppress unwanted intrusive thoughts and repetitive behaviors
This is analogous to hyperkinetic movement disorders (e.g., Huntington's disease, Sydenham's chorea) — but instead of unwanted movements, patients experience unwanted thoughts and compulsions. Importantly, OCD co-occurs in ~50% of Tourette's syndrome patients and can arise in Huntington's disease and Sydenham's chorea, confirming the basal ganglia link. — Adams & Victor's Principles of Neurology, 12th Ed.
5. Key Brain Regions: Structural & Functional Findings
Orbitofrontal Cortex (OFC)
- Consistently shows increased metabolic activity (hyperactivation) in PET and fMRI studies during OCD symptoms
- The OFC encodes expected value and error signals — hyperactivity generates an erroneous "something is wrong" signal that persists despite reassurance
- Normalizes with successful pharmacotherapy or CBT
Caudate Nucleus (Head)
- Decreased volume bilaterally on MRI (subtle but reproducible)
- Shows increased metabolic activity during symptomatic states
- Plays a crucial role in habit and skill learning — dysfunction leads to rigid, habitual behavior replacing flexible, goal-directed responses
- Caudate glutamate (Glx) levels are elevated in OCD patients and decrease in proportion to symptom improvement with SSRIs — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
- Caudate activity normalizes after successful treatment (pharmacological or behavioral)
Thalamus
- Acts as the relay station — normally gated by basal ganglia output
- Treatment-naïve OCD patients have larger thalamic volumes than controls
- After SSRI therapy (paroxetine), thalamic volume decreases to normal levels; this reduction correlates with symptom improvement
- With CBT alone, thalamic volume does not change — suggesting SSRI-specific structural modulation — Kaplan & Sadock's Comprehensive Textbook
Anterior Cingulate Cortex (ACC)
- Hyperactive in OCD; involved in error detection and conflict monitoring
- Generates the "something is not right" feeling that drives compulsive checking
- Pediatric OCD patients show reversed neurodevelopmental volume trajectories vs. healthy controls — suggesting impaired neuronal pruning in early illness
6. Neurotransmitter Systems
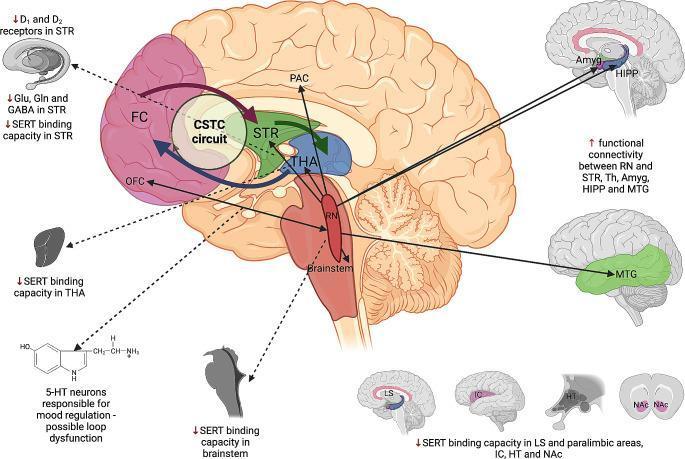
Serotonin (5-HT) — Primary System
- Decreased serotonin transporter (SERT) binding in brainstem, thalamus, striatum, limbic system, and nucleus accumbens
- Raphe nuclei (brainstem serotonin source) show increased functional connectivity to striatum, thalamus, amygdala, hippocampus, and middle temporal gyrus — likely a compensatory response
- The central clinical evidence: selective serotonin reuptake inhibitors (SSRIs) are the only class with robust efficacy in OCD, and efficacy does not correlate with antidepressant effect (clomipramine's anti-OCD activity is serotonin-specific, not noradrenergic) — Neuroanatomy Through Clinical Cases, 3rd Ed.
- Fluoxetine, fluvoxamine, paroxetine, sertraline all approved; ~50–60% of patients achieve adequate improvement — Harrison's 22nd Ed.
Glutamate — Emerging Key Role
- Elevated glutamate (Glx) in the caudate nucleus of OCD patients
- Glutamate reduction in caudate tracks clinical improvement with SSRIs
- The CSTC loop uses glutamate as its primary excitatory neurotransmitter — excess corticostriatal glutamatergic drive may sustain compulsive circuits
- Ketamine (NMDA antagonist) has shown promise in treatment-refractory OCD, supporting this mechanism
GABA — Inhibitory Deficit
- Reduced GABA levels in the striatum — loss of inhibitory control over the direct pathway
- Contributes to thalamic disinhibition and cortical hyperactivation
Dopamine
- D1 and D2 receptors altered in striatum
- Explains OCD overlap with Tourette's syndrome (dopamine-rich basal ganglia pathway)
- Dopamine antagonists (antipsychotics) are effective augmentation agents in OCD — particularly when tics coexist
7. Genetic & Immunological Factors
- Twin studies confirm a genetic contribution (heritability ~40–65%)
- No single OCD susceptibility gene identified — polygenic architecture
- Strong family aggregation with Tourette's disorder — shared genetic pathway involving basal ganglia circuits
- Insulin signaling genes recently implicated
- SAPAP3 gene variants associated with pathological grooming in humans
PANDAS (Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infections)
- Group A Streptococcus infection triggers autoantibodies that cross-react with basal ganglia (caudate/putamen), similar to Sydenham's chorea
- Results in acute-onset or dramatically worsening OCD/tics
- Functional imaging shows increased caudate and OFC activity
- Supports the causal role of striatal pathology in OCD — Adams & Victor's Neurology
8. Goal-Directed vs. Habit Learning: The Two-Stage Model
A key modern framework integrating circuit neuroscience:
Stage 1 (Early OCD):
- Behavior begins as goal-directed (OFC/caudate-mediated) — patient performs rituals to reduce anxiety
- Ventral striatum (goal-directed system) is initially dominant
Stage 2 (Established OCD):
- Chronic repetition transfers control to habit circuits (posterior putamen, sensorimotor CSTC)
- Behavior becomes automatic, less responsive to outcome — explaining why compulsions feel impossible to resist
- This shift from goal-directed to habit-based processing is a key target for behavioral therapy (ERP — Exposure and Response Prevention), which aims to re-engage goal-directed systems
9. Lesion Evidence & Neurosurgical Implications
| Lesion Location | Effect |
|---|---|
| OFC, cingulate, frontal/temporal cortex | Can precipitate OCD symptoms |
| Basal ganglia | OCD-like compulsive behavior |
| Cingulotomy (cingulate lesion) | Historical surgical treatment — effective but now largely replaced |
| Deep Brain Stimulation (DBS) of the anterior limb of internal capsule/nucleus accumbens | FDA-approved for refractory OCD; normalizes CSTC hyperactivity |
| rTMS over supplementary motor area or DLPFC | Effective in treatment-resistant cases |
10. Neural Circuitry Summary Schematic
Cortex (OFC, ACC, DLPFC)
↓ Glutamate ↑↑ (hyperactive)
Striatum (Caudate)
/Direct pathway↑ / Indirect pathway↓
↓
Globus Pallidus / SNr (insufficient inhibition)
↓
Thalamus ↑↑ (enlarged, overactive)
↓ (gates back, unfiltered)
Cortex → OFC error signal → "something is wrong" → anxiety
↓
Compulsion → temporary relief → reinforces the loop
11. Treatment Rationale Directly From Pathophysiology
| Pathophysiological Target | Treatment |
|---|---|
| ↓ SERT binding / serotonergic deficiency | SSRIs (fluoxetine, sertraline, fluvoxamine, paroxetine); clomipramine |
| CSTC hyperactivity (OFC, caudate, thalamus) | CBT / ERP normalizes OFC and caudate activity |
| Habit circuit dominance | ERP re-engages goal-directed circuits |
| Glutamate excess | Augmentation with riluzole, N-acetylcysteine (investigational) |
| Dopamine dysregulation / tic comorbidity | Antipsychotic augmentation (aripiprazole, risperidone) |
| Refractory CSTC loop | DBS of anterior internal capsule; rTMS over SMA |
Key Take-Home Messages for Conference
- OCD is a circuit disorder — not a character flaw or purely psychological condition. The CSTC loop is structurally and functionally abnormal.
- Three primary nodes: Orbitofrontal cortex (error signal), Caudate nucleus (habit gating), Thalamus (relay — enlarged, ungated).
- Serotonin is the primary neurotransmitter target, but glutamate and dopamine play important roles.
- The transition from goal-directed to habitual behavior explains symptom chronicity and informs treatment.
- OCD, Tourette's, Sydenham's chorea, and PANDAS share basal ganglia pathology — a spectrum of corticostriatal dysfunction.
- Successful treatment — whether pharmacological or behavioral — normalizes brain activity in the same circuit nodes.
Sources:
- Harrison's Principles of Internal Medicine, 22nd Ed. (McGraw Hill, 2025)
- Adams & Victor's Principles of Neurology, 12th Ed.
- Kaplan & Sadock's Comprehensive Textbook of Psychiatry
- Kaplan & Sadock's Synopsis of Psychiatry
- Neuroanatomy Through Clinical Cases, 3rd Ed. (Blumenfeld)
- Neuroscience: Exploring the Brain, 5th Ed. (Bear, Connors, Paradiso)
- 2025 web evidence: CSTC circuits beyond classical loops (Rewiring the OCD Brain, 2025); striatal transcriptomics (Translational Psychiatry, 2025)
Pathophysiology of ocd in short and easy words
OCD Pathophysiology — Simple & Short
Think of it like a broken alarm system in the brain
The Normal Brain
Your brain has a circuit (like an electrical loop) that:
- Notices something potentially wrong
- Checks it
- Sends a "all clear" signal → you move on
The OCD Brain — What Goes Wrong
🔴 3 Brain Areas Malfunction Together:
Orbitofrontal Cortex (OFC)
"Something is WRONG!"
↓
Caudate Nucleus
(supposed to filter this)
↓ ← FILTER IS BROKEN
Thalamus
(sends alarm back to cortex)
↓
Cortex screams louder: "STILL WRONG!"
↓
Person does a compulsion
(washing, checking, counting)
↓
Temporary relief... then loop restarts
In Even Simpler Terms
| Brain Part | Normal Job | In OCD |
|---|---|---|
| OFC (front of brain) | Detects errors | Stuck in "error mode" — alarm keeps ringing |
| Caudate (deep brain) | Filters/shuts off the alarm | Filter is broken — alarm never silenced |
| Thalamus (relay center) | Passes messages | Overloaded — keeps amplifying the alarm |
The Chemistry Problem
| Chemical | What It Does | In OCD |
|---|---|---|
| Serotonin | Calms the alarm | Too little effect → alarm stays loud |
| Glutamate | Excites the circuit | Too much → circuit stays hyperactive |
| Dopamine | Drives habits | Imbalanced → behavior becomes automatic |
This is why SSRIs (serotonin boosters) work — they turn the volume down on the alarm.
Why Compulsions Feel Impossible to Stop
Early OCD → rituals are done to reduce anxiety (a choice)
Over time → the habit circuit takes over → rituals become automatic, like breathing — the person can't just "stop"
This is why Exposure & Response Prevention (ERP) therapy works — it trains the brain to tolerate the alarm without acting on it, gradually resetting the circuit.
One-Line Summary
OCD = a brain circuit stuck in "error mode" that won't reset, driven by low serotonin and excess glutamate, forcing the person into repetitive rituals just to feel temporary relief.
This is a shared conversation. Sign in to Orris to start your own chat.