Cushing disease
Reading File
Finding Sources
Searching PubMed
"Cushing disease"[MeSH Terms] AND diagnosis
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
Reading File
I have all the essential information needed. Let me now compose a comprehensive, well-structured answer.
Cushing Disease
Cushing disease is a specific form of Cushing syndrome caused by an ACTH-secreting pituitary adenoma (corticotroph adenoma). The term is reserved for this pituitary-driven cause only — other causes of hypercortisolism are called "Cushing syndrome."
Epidemiology
- Annual incidence: 1.2–2.4 per million population
- Accounts for 60–70% of all endogenous Cushing syndrome cases
- Women affected 8–10× more than men
- Most tumors are microadenomas (<10 mm) at diagnosis due to early symptom development; only 10–15% are macroadenomas
— Goldman-Cecil Medicine
Pathophysiology
An autonomously functioning corticotroph adenoma secretes ACTH constitutively, bypassing normal hypothalamic-pituitary feedback. This drives bilateral adrenal hyperplasia and sustained, non-pulsatile cortisol hypersecretion that lacks the normal diurnal variation. ACTH also co-secretes melanocyte-stimulating hormone (MSH), causing mild hyperpigmentation. The molecular pathology of most corticotroph adenomas remains incompletely understood, though USP8 mutations are the most commonly identified somatic mutation.
Clinical Features
The presentation reflects prolonged glucocorticoid and mineralocorticoid excess:
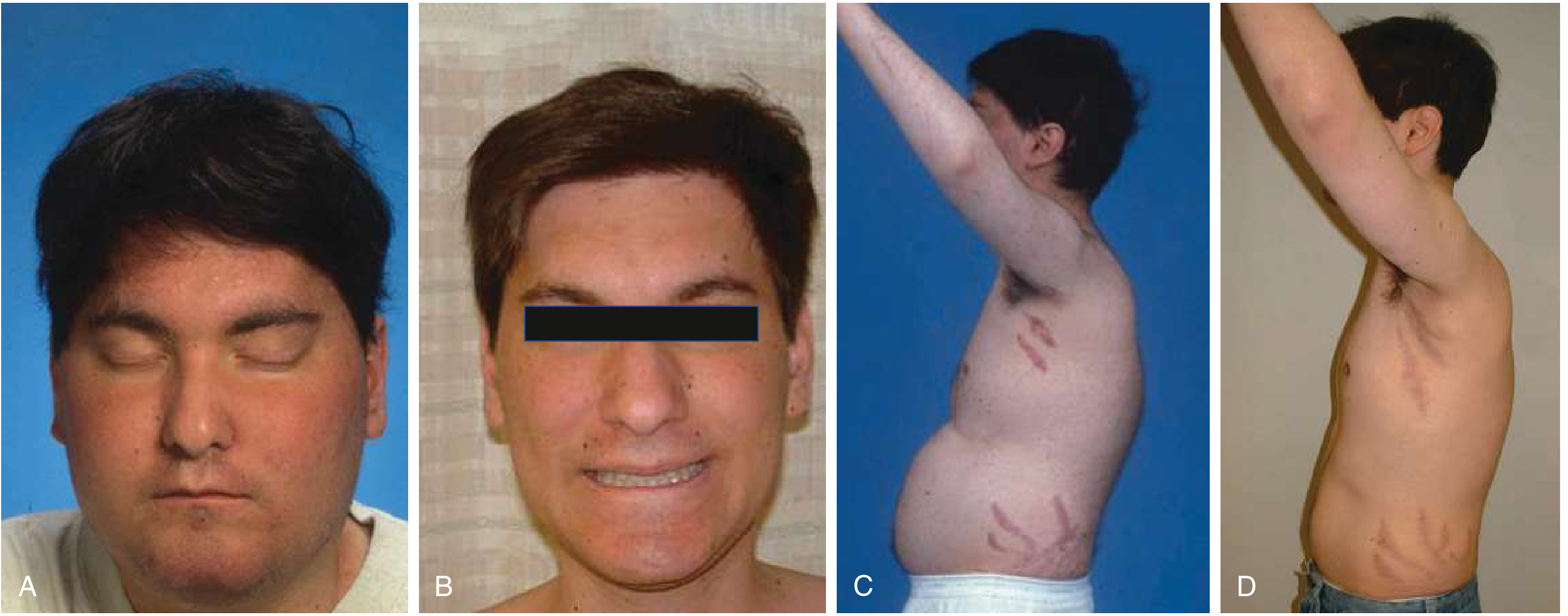
Clinical photographs (before and after transsphenoidal surgery in an 18-year-old male):
| System | Features |
|---|---|
| General | Central (centripetal) obesity, moon facies, buffalo hump (interscapular fat), supraclavicular fat pads |
| Skin | Wide violaceous striae (>1 fingerbreadth, purple), facial plethora, hirsutism, acne, thin skin, easy bruising/ecchymoses, hyperpigmentation |
| Musculoskeletal | Proximal muscle weakness, osteoporosis, vertebral fractures, back pain |
| Cardiovascular | Hypertension (ACTH stimulates mineralocorticoids), hypercoagulable state → DVT, PE |
| Metabolic | Glucose intolerance/diabetes, hypokalemia, hypercalciuria/renal stones, impaired wound healing, granulocytosis + lymphopenia |
| Reproductive | Oligomenorrhea/amenorrhea, decreased libido, female-pattern baldness |
| Neuropsychiatric | Depression (most patients), irritability, emotional lability, psychosis, disrupted sleep — suicidal ideation if untreated |
| Tumor mass effects | Headache, visual field defects, hypopituitarism (macroadenomas) |
The violaceous striae of Cushing disease are distinguishable from pregnancy striae by being wider than a fingerbreadth and purple, with subcutaneous bleeding contributing to pigmentation.
— Goldman-Cecil Medicine
Diagnosis
Diagnosis proceeds in three steps: (1) confirm hypercortisolism, (2) establish ACTH-dependence, (3) localize the source to the pituitary.
Step 1 — Confirm Hypercortisolism (screening tests, ≥2 required)
| Test | Threshold | Notes |
|---|---|---|
| Late-night salivary cortisol (23:00–24:00) | >0.09 µg/µL | Sensitivity 80–90%, reflects free cortisol; best screening test |
| 24-hour urine free cortisol (UFC) | >3× upper limit of normal | Reliable if properly collected and renal function normal |
| 1 mg overnight DST | Morning cortisol >1.8 µg/dL = failed suppression | Cortisol >10 µg/dL = high likelihood of hypercortisolism |
| Low-dose DST (2-day) | Same cut-off | Increases specificity |
Step 2 — Establish ACTH-Dependence
- Plasma ACTH >15 pg/mL → ACTH-dependent (pituitary or ectopic)
- Plasma ACTH undetectable → ACTH-independent (primary adrenal source)
- Very high ACTH (>100 pg/mL) → suggests ectopic ACTH or primary adrenal insufficiency
Step 3 — Localize to Pituitary vs. Ectopic
| Test | Finding favoring Cushing disease |
|---|---|
| Pituitary MRI (gadolinium) | Microadenoma visible in ~50% of cases (negative MRI does not exclude diagnosis) |
| High-dose DST (8 mg) | ≥50% suppression of UFC favors pituitary source |
| CRH stimulation test | ≥35% rise in ACTH at 15–30 min; sensitivity 93%, specificity 100% for pituitary vs. ectopic |
| Inferior petrosal sinus sampling (IPSS) | Central-to-peripheral ACTH ratio ≥2 basal, ≥3 post-CRH → confirms pituitary source; lateralization guides surgical approach |
IPSS is the gold standard when MRI is negative or equivocal, or when ectopic ACTH cannot be excluded.
— Goldman-Cecil Medicine; Harrison's Principles of Internal Medicine 22e
Management
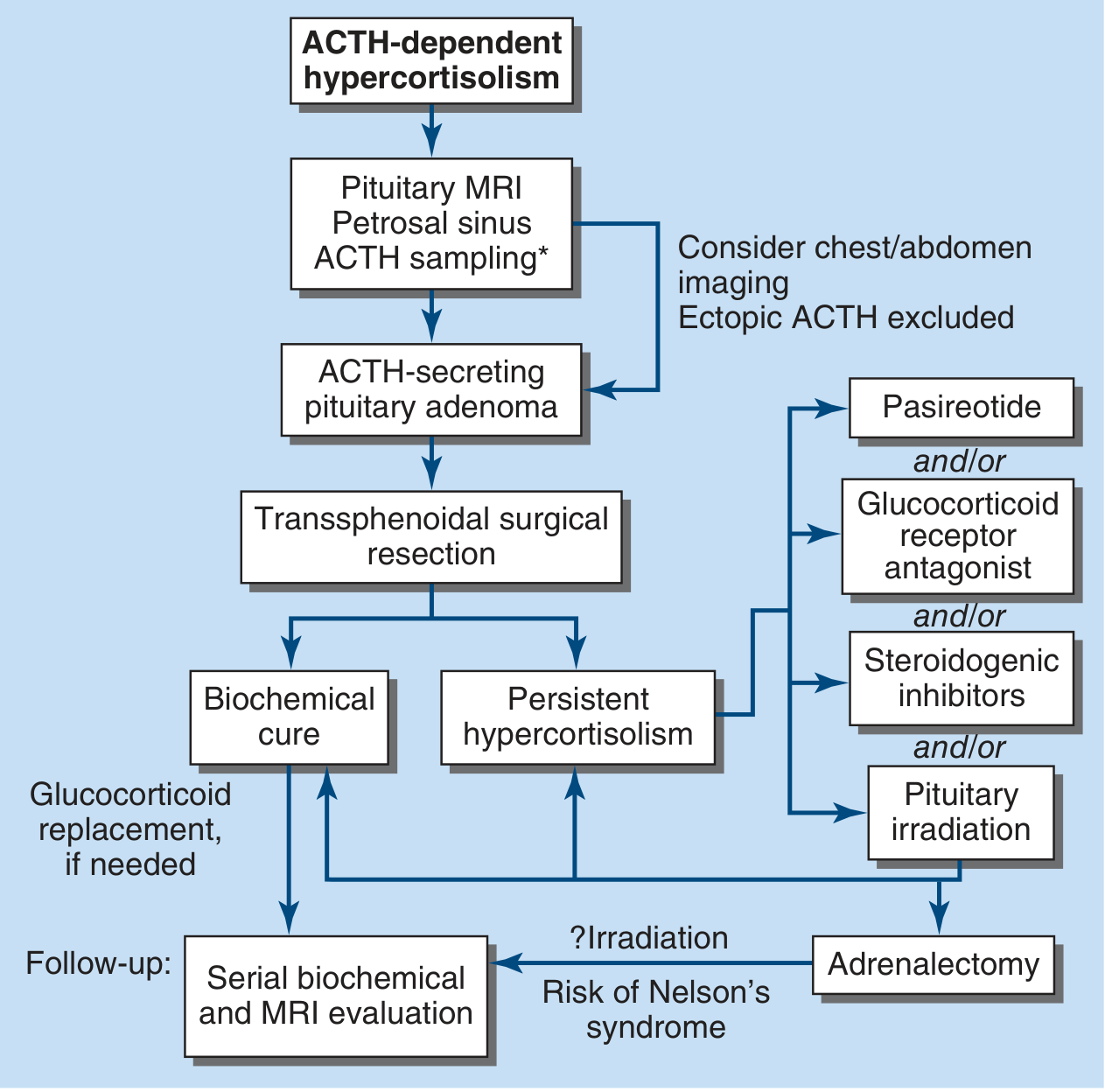
First-line: Transsphenoidal Surgery (TSS)
- Treatment of choice — selective adenomectomy via endoscopic or microscopic transsphenoidal approach
- Remission rates: ~80% for microadenomas, <50% for macroadenomas
- Surgery is rarely successful when no adenoma is visible on MRI
- Post-operative ACTH deficiency lasting up to 12 months is expected (HPA axis suppression) — requires low-dose cortisol replacement
- Biochemical recurrence: ~5% of initially cured patients
- Postoperative thromboprophylaxis is advocated due to hypercortisolism-related coagulopathy
— Harrison's Principles of Internal Medicine 22e
If Initial Surgery Fails or Recurs
Option 1 — Repeat surgery: Achieves remission in additional 50–70% when pituitary source is well-documented
Option 2 — Pituitary irradiation: Cures only ~15% of adults; slow onset; used in combination with medical therapy
Option 3 — Medical therapy (to bridge to surgery or as adjunct):
| Drug | Mechanism | Efficacy / Notes |
|---|---|---|
| Osilodrostat | 11β-hydroxylase inhibitor | Normalizes UFC in ~86%; monitor for hypocortisolism, hypokalemia, QTc prolongation |
| Ketoconazole | Multiple P450 inhibitors | 600–1200 mg/d; monitor LFTs; gynecomastia, impotence |
| Levoketoconazole | Same as ketoconazole (2S,4R enantiomer) | Similar efficacy and side-effect profile |
| Metyrapone | 11β-hydroxylase inhibitor | Normalizes cortisol in ~75%; nausea, acne/hirsutism exacerbation |
| Pasireotide LAR | Somatostatin receptor agonist (SST5>SST2) | Reduces ACTH/cortisol; tumor shrinkage in ~40%; hyperglycemia in up to 70% |
| Mifepristone | Glucocorticoid receptor antagonist | Blocks cortisol action peripherally; approved for hyperglycemia in Cushing's; ACTH and cortisol levels rise |
| Mitotane | Adrenocortical cell destruction + enzyme inhibition | Slow onset; significant side effects |
Option 4 — Bilateral adrenalectomy: Definitive cortisol reduction but requires lifelong steroid replacement. Major risk: Nelson's syndrome — rapid pituitary tumor enlargement with very high ACTH and severe hyperpigmentation, caused by loss of adrenal feedback. Prophylactic pituitary irradiation before adrenalectomy may reduce this risk.
— Harrison's Principles of Internal Medicine 22e; Goldman-Cecil Medicine
Complications to Monitor
- Cardiovascular: Hypertension, dyslipidemia, thromboembolism
- Skeletal: Osteoporosis → fragility fractures
- Metabolic: Diabetes mellitus, electrolyte abnormalities
- Infectious: Opportunistic infections (immunosuppressed state)
- Nelson's syndrome (post-adrenalectomy)
- Psychiatric: Depression, rarely psychosis — does not always resolve after biochemical cure
Key Distinctions
| Cushing Disease | Cushing Syndrome | |
|---|---|---|
| Cause | Pituitary corticotroph adenoma | Any cause of hypercortisolism |
| ACTH | Elevated (ACTH-dependent) | Elevated (ACTH-dep.) or suppressed (ACTH-indep.) |
| Dexamethasone | High-dose suppresses; low-dose does not | Depends on etiology |
| Treatment | Transsphenoidal surgery | Depends on cause |
Sources: Goldman-Cecil Medicine (International Ed.), Harrison's Principles of Internal Medicine 22e (2025), Mulholland & Greenfield's Surgery 7e
Cushing syndrome
Finding Sources
Searching PubMed
"Cushing syndrome"[MeSH Terms] AND diagnosis
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
I now have comprehensive material from multiple authoritative textbooks. Let me compose the full answer.
Cushing Syndrome
Cushing syndrome is the clinical state resulting from prolonged, inappropriate exposure to excess glucocorticoids, whether from an endogenous or exogenous source. It is distinguished from Cushing disease, which refers specifically to the pituitary ACTH-adenoma subtype.
Etiology & Classification
The three pathophysiologic mechanisms of hypercortisolism are illustrated below:
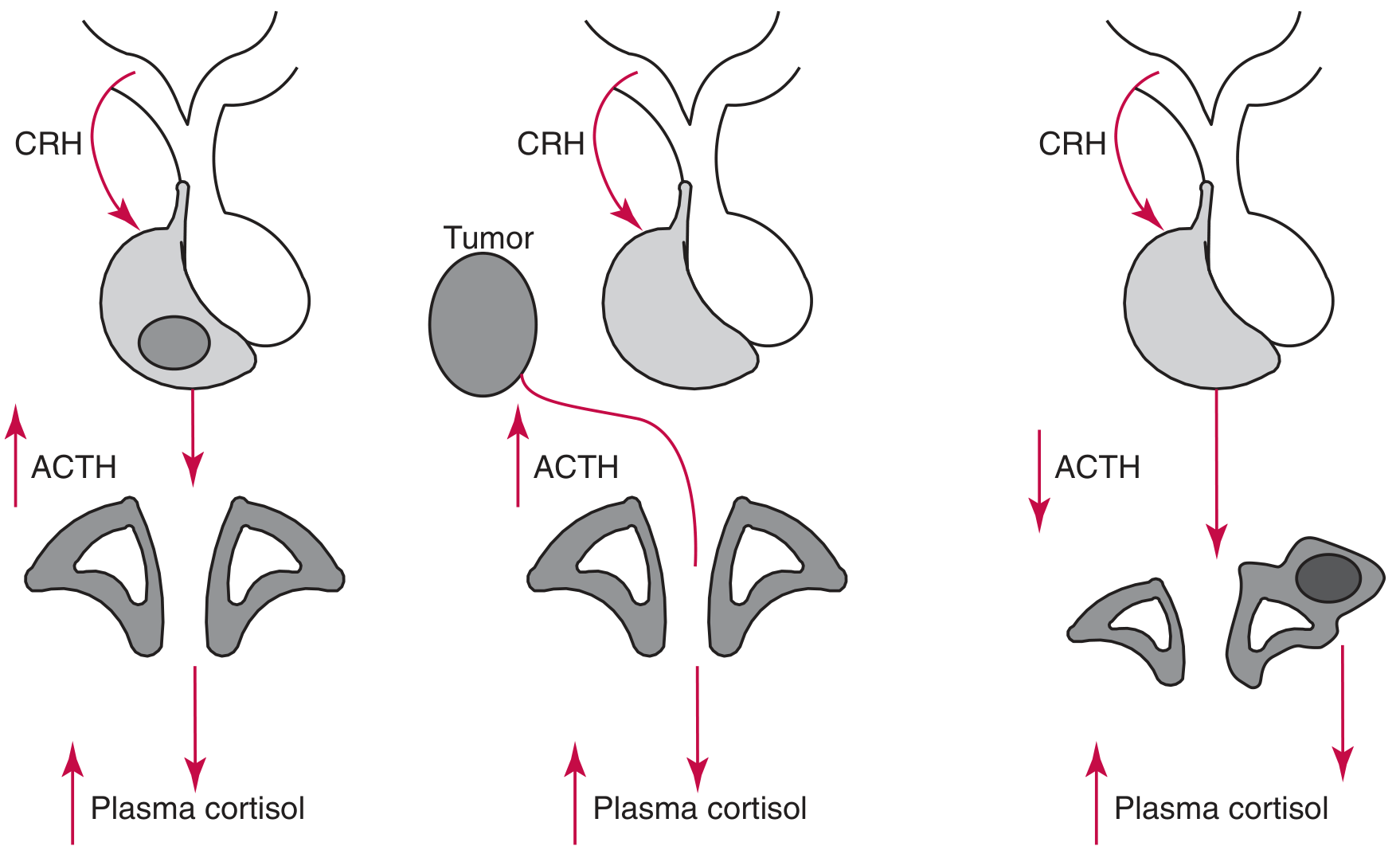
(Left: ACTH-dependent with bilateral adrenal hyperplasia; Center: Ectopic ACTH tumor; Right: ACTH-independent adrenal adenoma with contralateral atrophy)
1. Exogenous (Iatrogenic) — Most Common Overall
The most common cause of Cushing syndrome globally is therapeutic corticosteroid administration. This includes systemic, topical (skin, nasal, conjunctival, gingival — especially in children), and inhaled routes. Adrenal cortex is suppressed; endogenous cortisol is low.
2. ACTH-Dependent Endogenous (~80% of endogenous cases)
| Cause | Frequency | Details |
|---|---|---|
| Cushing disease (pituitary corticotroph adenoma) | ~80% of ACTH-dep. | Most common endogenous cause; predominantly women; microadenoma in ~50% |
| Ectopic ACTH syndrome (EAS) | ~20% of ACTH-dep. | ACTH from non-pituitary tumor → bilateral adrenal hyperplasia |
| Ectopic CRH secretion | Very rare | Lung, thymic, pancreatic, medullary thyroid tumors; most also co-secrete ACTH |
Tumors causing ectopic ACTH (in approximate frequency):
- Small cell lung carcinoma
- Pulmonary carcinoid
- Pancreatic and thymic neuroendocrine tumors
- Gastrinoma, medullary thyroid carcinoma, pheochromocytoma
3. ACTH-Independent Endogenous (~20% of endogenous cases)
| Cause | Notes |
|---|---|
| Adrenal adenoma | Most common; unilateral, <60 mm, <30 g; contralateral adrenal atrophies |
| Adrenocortical carcinoma (ACC) | Larger; ~50% non-functional; CRH/ACTH suppressed |
| PPNAD (Primary Pigmented Nodular Adrenocortical Disease) | Multiple pigmented micronodules (<10 mm); associated with Carney complex (PRKAR1A mutation); paradoxic cortisol rise on dexamethasone |
| PBMAH (Primary Bilateral Macronodular Adrenal Hyperplasia) | Nodules up to 4 cm; ARMC5 mutation; aberrant G-protein–coupled receptors sensitive to vasopressin, LH, serotonin, GIP |
— Tietz Textbook of Laboratory Medicine 7e; Campbell-Walsh-Wein Urology
Pseudo-Cushing Syndrome
Non-neoplastic conditions can mimic both the clinical and biochemical picture of Cushing syndrome:
With clinical features of Cushing: depression, alcohol dependence, glucocorticoid resistance, morbid obesity, poorly controlled diabetes, polycystic ovary syndrome
Unlikely to have clinical features (biochemical overlap only): physical stress, malnutrition/anorexia nervosa, intense chronic exercise, hypothalamic amenorrhea
The mechanism likely involves hypothalamic paraventricular nucleus activation → CRH hypersecretion.
— Tietz Textbook of Laboratory Medicine 7e
Clinical Features
Chronic glucocorticoid excess affects virtually every organ system:
| System | Features |
|---|---|
| General | Central (centripetal) obesity — face, neck, trunk, abdomen with limb sparing; moon facies; buffalo hump (interscapular fat); supraclavicular fat pads |
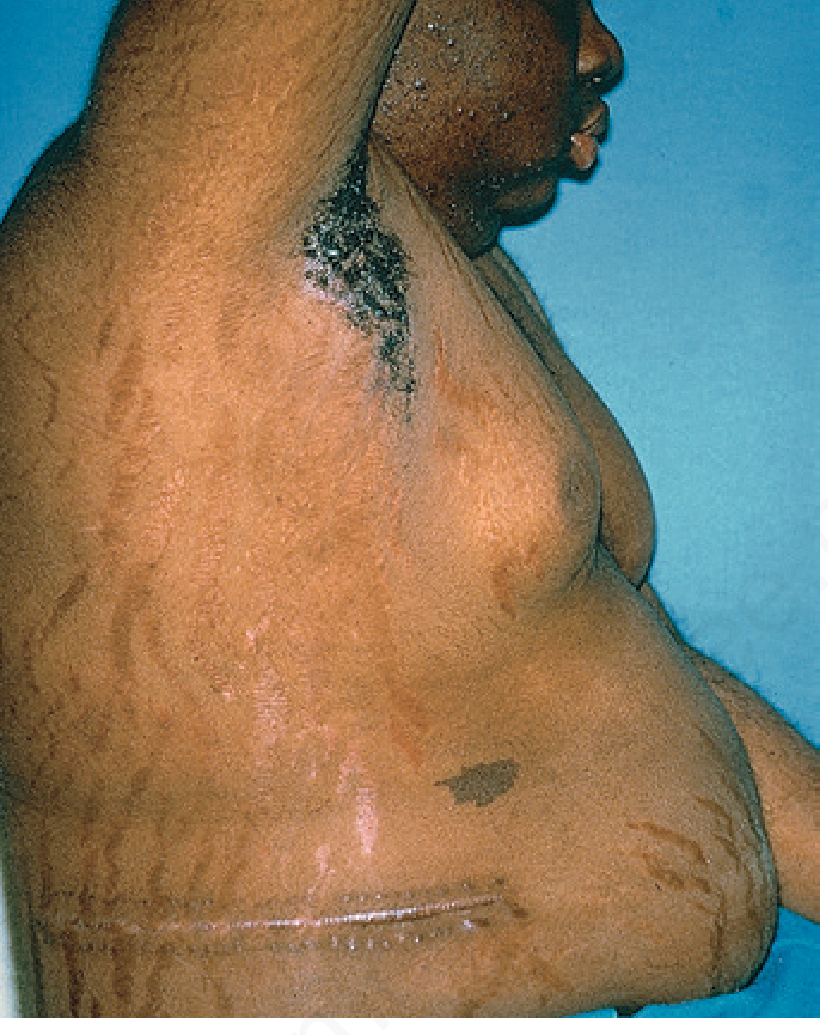
| Skin | Wide, purple/violaceous atrophic striae (abdomen, buttocks, breasts, upper arms, thighs); facial plethora; thin/fragile skin; easy bruising; Liddle sign (skin strips with tape removal); hypertrichosis; acne; female scalp thinning with lanugo; opportunistic fungal infections (Pityrosporum, dermatophytes) |
| Musculoskeletal | Proximal muscle weakness; osteoporosis; kyphosis; vertebral fractures; back pain |
| Cardiovascular | Hypertension; marked arteriosclerosis; hypercoagulability → DVT, PE |
| Metabolic | Glucose intolerance/DM (~20%); hypokalemia; hypercalciuria/renal stones; impaired wound healing |
| Reproductive | Decreased libido; oligomenorrhea/amenorrhea; female hirsutism and male-pattern baldness |
| Neuropsychiatric | Depression (most patients), irritability, emotional lability, psychosis, disrupted sleep |
| Immune | Lymphopenia, granulocytosis; susceptibility to infections, including unusual pathogens |
| Hyperpigmentation | Present in ACTH-dependent causes (co-secreted MSH); most pronounced in ectopic ACTH syndrome (very high ACTH) |
Peak age of onset: 20s–30s. Women affected ~4× more than men in non-iatrogenic cases.
— Andrews' Diseases of the Skin; Goldman-Cecil Medicine
Diagnosis
The diagnostic approach has three sequential phases:
Phase 1 — Confirm Hypercortisolism (Screening)
At least two positive tests are typically required:
| Test | Threshold | Notes |
|---|---|---|
| 1 mg overnight DST | Morning cortisol >1.8 µg/dL (failed suppression) | Best screening; cortisol <50 nmol/L essentially rules out (except iatrogenic) |
| Late-night salivary cortisol (23:00–24:00) | >0.09 µg/µL | Reflects free cortisol; sensitivity 80–90%; not affected by cortisol-binding globulin |
| 24-h urine free cortisol (UFC) | ≥3× upper normal limit = 95–100% sensitive and specific | Requires accurate collection; biologic variation is a limitation |
Phase 2 — Confirm and Classify (Low- then High-dose DST)
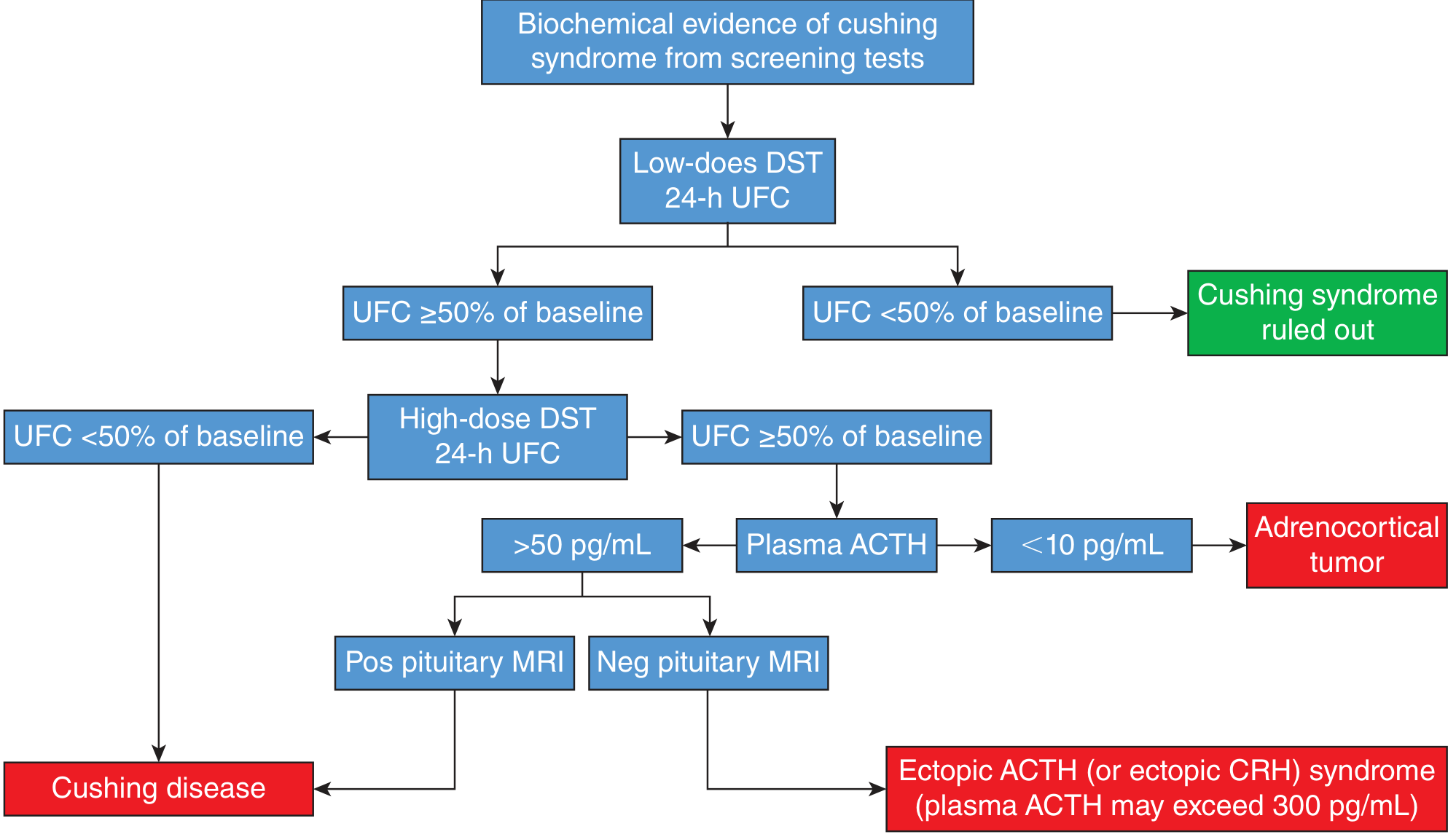
- Low-dose DST + 24-h UFC: If UFC suppresses to <50% of baseline → Cushing syndrome ruled out
- High-dose DST + 24-h UFC: If UFC suppresses → likely Cushing disease (pituitary source)
- If UFC does NOT suppress → measure plasma ACTH:
- ACTH <10 pg/mL → adrenocortical tumor (ACTH-independent)
- ACTH >50 pg/mL → ACTH-dependent (ectopic or pituitary)
- ACTH >300 pg/mL → strongly suggests ectopic ACTH
Phase 3 — Localize the Source
| Test | Purpose |
|---|---|
| Pituitary MRI (gadolinium) | Visible adenoma in ~50% of Cushing disease; incidentalomas occur in 10% of the general population — a positive MRI alone is insufficient |
| CRH stimulation | ≥35% ACTH rise → pituitary origin; no rise → ectopic |
| IPSS (inferior petrosal sinus sampling) | Gold standard for distinguishing Cushing disease from ectopic ACTH; central:peripheral ratio ≥2 basal or ≥3 post-CRH confirms pituitary; lateralization guides surgery |
| Abdominal CT/MRI | Adrenal mass in ACTH-independent disease; bilateral hyperplasia in ACTH-dependent |
| Chest/abdomen imaging | Identify occult ectopic ACTH-producing tumor (lung, pancreas, thymus) |
| Adrenal venous sampling (AVS) | Bilateral adrenal lesions — identifies the dominant secreting side |
Up to 50% of Cushing disease patients have no abnormality on pituitary MRI, making biochemical confirmation and IPSS essential.
— Campbell-Walsh-Wein Urology; Tietz Laboratory Medicine 7e
Treatment (by Cause)
Exogenous/Iatrogenic
- Gradual tapering and discontinuation of corticosteroids (risk of adrenal insufficiency)
Cushing Disease (Pituitary Adenoma)
- First-line: Transsphenoidal adenomectomy — ~80% remission for microadenomas
- Adjuncts/salvage: Repeat surgery, pituitary irradiation, steroidogenic inhibitors, pasireotide, mifepristone
- Last resort: Bilateral adrenalectomy (risk of Nelson syndrome — pituitary tumor enlargement + severe hyperpigmentation from uninhibited ACTH rise; occurs in 8–29%)
Ectopic ACTH Syndrome
- Resection of primary ACTH-producing tumor (possible in only ~10% of cases)
- For unresectable or unlocalized tumors: bilateral adrenalectomy with lifelong steroid replacement
ACTH-Independent (Adrenal)
- Adenoma: Ipsilateral (laparoscopic) adrenalectomy — curative; contralateral adrenal recovers over months
- ACC: Surgical resection ± mitotane, adjuvant therapy
- PPNAD/PBMAH: Bilateral adrenalectomy with lifelong replacement
Medical Therapy (Bridging or When Surgery Not Possible)
| Drug | Mechanism |
|---|---|
| Metyrapone | 11β-hydroxylase inhibitor |
| Ketoconazole / Levoketoconazole | Multiple CYP450 inhibitors |
| Osilodrostat | 11β-hydroxylase inhibitor |
| Mitotane | Adrenocortical cytotoxic + enzyme inhibitor |
| Aminoglutethimide | Cholesterol side-chain cleavage inhibitor |
| Etomidate (IV) | Rapid-acting for severe acute hypercortisolism |
| Pasireotide | SST5/SST2 agonist; pituitary-directed |
| Mifepristone | Glucocorticoid receptor antagonist |
— Campbell-Walsh-Wein Urology; Harrison's Principles of Internal Medicine 22e
Complications
- Cardiovascular: Hypertension, dyslipidemia, accelerated atherosclerosis, thromboembolism
- Skeletal: Osteoporosis with fragility fractures
- Infectious: Opportunistic and atypical infections
- Endocrine: Secondary diabetes mellitus, gonadal suppression
- Psychiatric: Depression, cognitive impairment — may persist after biochemical remission
- Nelson syndrome: Post-bilateral adrenalectomy → pituitary tumor expansion + marked hyperpigmentation
Summary: Cushing Syndrome vs. Cushing Disease
| Feature | Cushing Syndrome | Cushing Disease |
|---|---|---|
| Definition | Any cause of glucocorticoid excess | Pituitary corticotroph adenoma specifically |
| ACTH | Variable (elevated, normal, or suppressed) | Elevated |
| DST | Variable by cause | High-dose suppresses |
| Adrenal glands | Atrophy (exogenous/adrenal tumor), bilateral hyperplasia (ACTH-dep.) | Bilateral hyperplasia |
| Treatment | Depends on cause | Transsphenoidal surgery |
Sources: Campbell-Walsh-Wein Urology 3-Vol Set; Andrews' Diseases of the Skin; Tietz Textbook of Laboratory Medicine 7e; Goldman-Cecil Medicine; Harrison's Principles of Internal Medicine 22e
This is a shared conversation. Sign in to Orris to start your own chat.