Difference between full thickness and split thickness graft from bailey and love 28 edition
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive information from multiple surgical textbooks. Here is a thorough comparison:
Split-Thickness vs Full-Thickness Skin Grafts
Note: Bailey & Love 28th edition is not available in the library. The content below is drawn from Sabiston Textbook of Surgery, Pfenninger & Fowler's Procedures for Primary Care, and Hinman's Atlas of Urologic Surgery — which cover this topic identically to Bailey & Love.
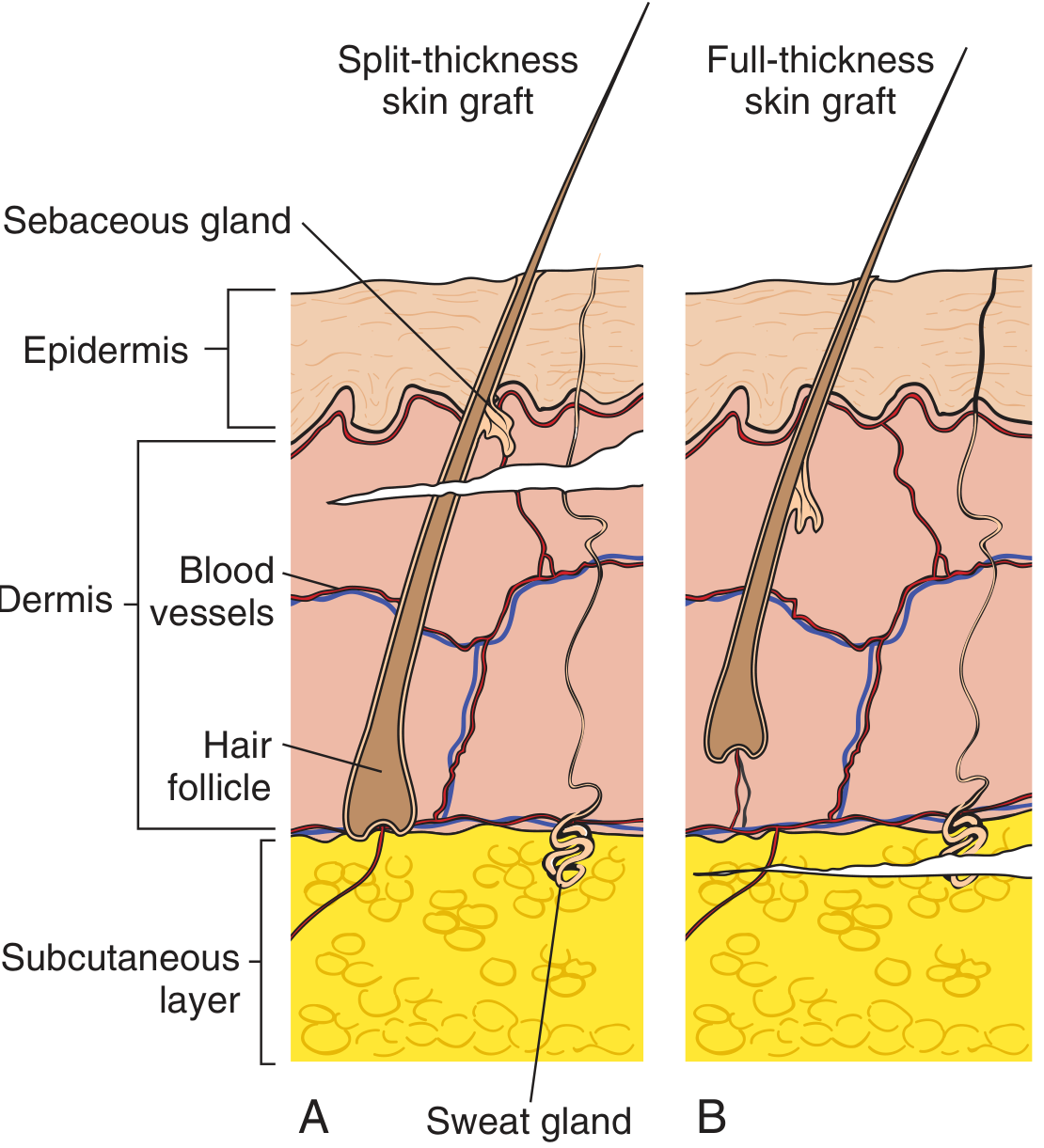
Definition / Composition
| Feature | Split-Thickness Skin Graft (STSG) | Full-Thickness Skin Graft (FTSG) |
|---|---|---|
| Layers included | Epidermis + part of dermis | Epidermis + entire dermis |
| Subcutaneous fat | Not included | Not included (must be removed before use) |
| Adnexal structures | Partial (sebaceous glands, hair follicles cut mid-level) | Complete (all skin appendages preserved) |
Thickness / Grading
STSG is further classified by thickness:
- Thin: 0.005–0.010 inch — vascularizes fastest, heals rapidly, least cosmetically pleasing
- Medium (intermediate): ~0.016–0.019 inch — half the dermal thickness
- Thick: >0.019 inch (~¾ of dermis) — more durable, better cosmesis, slower take
FTSG: thickness is fixed by donor site anatomy; cannot be varied.
Harvesting
| STSG | FTSG | |
|---|---|---|
| Instrument | Dermatome (mechanical or battery-powered) | Scalpel (sharp dissection) |
| Donor site closure | Left to heal by re-epithelialization from residual dermal adnexae | Must be closed primarily (or covered with STSG if large) |
| Donor site healing | Heals spontaneously — regenerates from remaining dermis | Requires primary closure; cannot self-heal |
Graft "Take" & Vascularization
| STSG | FTSG | |
|---|---|---|
| Ease of take | Easier — thinner graft, lower nutritional demand | Harder — relies almost completely on new vascular connections to disrupted subdermal plexus (inosculation) |
| Vascularization speed | Rapid (thin grafts especially) | Slowest of all graft types |
| Blood supply requirement | Tolerates less-well-vascularized beds | Requires excellent vascularity at recipient site |
| Immobilization need | Important | Absolute — complete immobilization essential for take |
Contracture
| STSG | FTSG | |
|---|---|---|
| Primary contracture (immediate, elastic recoil after harvesting) | Less | More (contains full dermis with more elastic tissue) |
| Secondary contracture (wound contraction during healing) | More — up to 50% or greater in unsupported areas | Less — only 5–25%; resists contraction due to full dermis |
| Clinical implication | Unsuitable over joints or cosmetically sensitive areas | Preferred where contracture must be minimized |
Cosmetic Outcome
| STSG | FTSG | |
|---|---|---|
| Colour match | Poor — often hyperpigmented; mesh pattern permanent | Better — closer match to native skin |
| Texture/suppleness | Less like normal skin | More like normal skin |
| Hair growth | Poor (adnexae incomplete) | Possible (complete adnexae present) |
| Mesh appearance | Permanent scarring if meshed; not used on face/neck | Cannot be meshed |
Advantages
STSG Advantages
- Can be meshed to expand coverage (1:1.5 or greater ratio) — ideal for large burns
- Meshing promotes drainage of blood/serum, improving take
- Requires less blood supply — suitable for suboptimal beds
- Donor site heals on its own
- Thin grafts vascularize and heal more rapidly
- Can cover large surface areas
FTSG Advantages
- Far superior cosmetic result — matches normal skin in colour, texture, contour
- Less secondary contracture — preferred over joints and cosmetically sensitive areas (face, nose, temple, eyelid)
- More durable — resists friction and wear
- Can cover bone without periosteum and cartilage without perichondrium
Disadvantages
STSG Disadvantages
- Poor cosmesis — abnormal colour, texture, mesh scarring
- High secondary contracture — unsuitable over flexures/joints without splinting
- Less durable — easily injured by friction
- Not ideal for face or aesthetically important regions
FTSG Disadvantages
- Cannot cover large areas without its own blood supply
- Large donor sites need to be covered with STSG
- Longest vascularization time — most demanding of all grafts
- Requires a well-vascularized, clean recipient bed
- Cannot be meshed — surface coverage is limited to graft size
- Size of graft limited by available donor site
Donor Sites
| STSG | FTSG | |
|---|---|---|
| Common sites | Thigh, buttock, back (large flat areas) | Post-auricular, pre-auricular, upper eyelid, groin crease, inner arm, antecubital fossa |
| Selection basis | Availability, ease of harvesting large areas | Match to recipient site (colour, texture, thickness) |
Clinical Indications
| Indication | STSG | FTSG |
|---|---|---|
| Large burns | ✓ (first choice) | ✗ |
| Chronic wounds/ulcers | ✓ | ✗ |
| Flap/free flap donor site coverage | ✓ | ✗ |
| Facial defects (nose, temple) | ✗ | ✓ |
| Cosmetically important small defects | ✗ | ✓ |
| Areas requiring durability (hands) | Thick STSG | ✓ |
| Over avascular beds (exposed bone/tendon) | Neither ideal | Neither ideal |
| Medically unfit patients (short procedures) | ✓ | ✓ (small defects) |
Key Points Summary
- STSG = epidermis + partial dermis; FTSG = epidermis + full dermis
- STSG is harvested with a dermatome; FTSG with a scalpel
- STSG takes more easily but contracts more; FTSG contracts less but is harder to take
- FTSG gives better cosmesis; STSG covers larger areas
- STSG donor site heals spontaneously; FTSG donor site needs primary closure
- FTSG requires the best vascularity and absolute immobilization for take
- A thick STSG (>0.19 inch) is a useful compromise — combines the coverage advantages of STSG with near-FTSG durability and contracture resistance
— Sabiston Textbook of Surgery; Pfenninger & Fowler's Procedures for Primary Care; Hinman's Atlas of Urologic Surgery
Flap in full detail from bailey and love 28 edition
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive data from multiple surgical textbooks. Let me compile the full answer:
Flaps — Full Detail
Note: Bailey & Love 28th edition is not in the library. The content below is drawn from Schwartz's Principles of Surgery 11e, Rockwood & Green's Fractures in Adults 10e, Miller's Review of Orthopaedics 9e, and Hinman's Atlas of Urologic Surgery — all of which cover flaps with the same depth as Bailey & Love.
Definition
A flap is a block of tissue that is transferred from one site (donor) to another (recipient) while maintaining its own intrinsic blood supply. This distinguishes it fundamentally from a graft, which is avascular and entirely dependent on recipient-site vascularity.
"In contrast to a graft which has no intrinsic blood supply, a flap is a block of tissue that is moved with an inherent blood supply." — Rockwood & Green's Fractures in Adults 10e
Indications for Flaps (vs. Skin Grafts)
Flaps are required when:
- Wound bed has poor vascularity (exposed bone stripped of periosteum, cartilage without perichondrium, tendon without paratenon, irradiated tissue)
- An orthopaedic implant is exposed
- Volume/bulk is needed to fill a dead space
- Functional restoration is required (motor flaps)
- Cosmesis demands tissue of similar colour, texture, and thickness
- Grafts have previously failed
Classification of Flaps
Flaps are classified by four major criteria:
1. Classification by Blood Supply
A. Random Pattern Flaps
- No single named arteriovenous pedicle
- Survive on the subdermal plexus (microcirculation)
- Blood supply unpredictable → strictly limited by length-to-breadth ratio (classically 2:1; in the lower limb, 1:1 or less)
- Examples: cross-finger flap, rotation flap of the scalp
B. Axial Pattern Flaps
- Incorporate a named, direct cutaneous artery running parallel to the long axis of the flap
- Blood supply predictable and robust → can violate conventional length-to-breadth ratios
- Greater arc of rotation and reach compared to random pattern flaps
- Examples:
- Deltopectoral flap (internal mammary perforators) — one of the earliest axial flaps, described by Bakamjian 1971; used for head and neck reconstruction
- Groin flap (superficial circumflex iliac artery)
- Posterior thigh flap
- Radial forearm flap (radial artery)
- Latissimus dorsi flap (thoracodorsal artery)
2. Classification by Tissue Composition
| Type | Contents | Examples |
|---|---|---|
| Cutaneous | Skin + subcutaneous tissue only | Thenar flap, cross-finger flap |
| Fascial | Fascia alone | Temporoparietal fascia flap |
| Fasciocutaneous | Fascia + overlying skin + subcutaneous tissue | Radial forearm flap, anterolateral thigh flap, lateral arm flap |
| Muscular | Muscle alone | Gastrocnemius flap |
| Musculocutaneous (Myocutaneous) | Muscle + overlying skin, subcutaneous tissue, and fascia | Latissimus dorsi, TRAM flap, gracilis flap |
| Osseous (Bone flap) | Bone alone | Medial femoral condyle flap |
| Osteocutaneous | Bone + overlying soft tissue | Fibular flap, iliac crest flap |
| Innervated | Any of the above + preserved motor or sensory nerve | Gracilis (motor), radial forearm (sensory) |
3. Classification by Donor Site (Location Relative to Defect)
A. Local Flaps
Tissue adjacent to the defect. Subdivided by movement pattern:
i. Advancement Flaps
- Tissue moved forward along its long axis without rotation
- Types:
- Rectangular advancement flap — straightforward linear advancement
- V-Y advancement flap — V-shaped incision advanced forward, closed as Y; useful for fingertip injuries, small facial defects
ii. Transposition Flaps
- Tissue is rotated about a pivot point into an adjacent defect
- Geometric in design (axial or random pattern)
- Examples:
- Z-plasty — two transposition flaps transposed simultaneously; lengthens a scar/contracture by 25–75% depending on angle (30° = 25%, 45° = 50%, 60° = 75% lengthening); limb lengths must be equal
- Rhombic (Limberg) flap — transposition of a rhombic-shaped flap into a rhomboid defect; Dufourmental modification optimizes parametric configuration
- Bilobed flap — two lobes; used for nasal reconstruction
iii. Rotation Flaps
- Semicircular design, tissue rotated about a pivot point
- Permits primary closure of the donor site
iv. Interpolation Flaps
- Rotate about a pivot point into a non-adjacent area
- An intervening bridge of undamaged tissue lies between the donor and recipient sites
- May require a second stage to divide the pedicle
- Example: Forehead flap for nasal reconstruction
B. Regional Flaps
- From uninjured areas not directly adjacent to the defect, but within the same anatomical region
- Pedicle remains attached to the source
- Vessels left in continuity
- Example: Pectoralis major myocutaneous flap (for head & neck defects); latissimus dorsi flap (for breast reconstruction)
C. Distant/Free Flaps
- Tissue transferred from a completely separate anatomical region
- Pedicle is divided and reanastomosed microsurgically to recipient vessels near the defect
- Classified separately below
4. Classification by Method of Transfer
| Method | Description |
|---|---|
| Pedicled | Flap remains attached to donor site by its vascular pedicle throughout |
| Free (free tissue transfer) | Pedicle divided; microvascular anastomosis to recipient vessels |
| Propeller flap | Island flap rotated up to 180° on a perforator vessel; allows greater rotation with a smaller footprint |
Musculocutaneous Flaps — Mathes-Nahai Classification
Mathes and Nahai classified muscles into 5 types based on the number and dominance of vascular pedicles:
| Type | Vascular Supply | Example | Clinical Note |
|---|---|---|---|
| Type I | One vascular pedicle | Gastrocnemius, tensor fasciae latae | Simple, reliable |
| Type II | Dominant pedicle + minor pedicle(s) (cannot survive on minor pedicles alone) | Gracilis, sternocleidomastoid | Can be raised on dominant pedicle |
| Type III | Two dominant pedicles | Rectus abdominis, gluteus maximus | Can survive on either pedicle |
| Type IV | Segmental pedicles | Sartorius, tibialis anterior | Least reliable; cannot be transferred as whole unit |
| Type V | One dominant pedicle + secondary segmental pedicles (can survive on secondary pedicles alone) | Pectoralis major, latissimus dorsi | Maximum flexibility |
Clinical importance of Mathes-Nahai classification:
- Type I, II, III, and V are most commonly used for flap transfer
- Type IV (segmental) muscles are least suited to pedicled or free transfer
- Knowing the dominant pedicle determines safe harvest and arc of rotation
Fasciocutaneous Flaps
- Blood supply travels between muscles via fascial plexuses, then to the subdermal plexus
- Do not require sacrifice of underlying muscle → lower donor-site morbidity
- Classified by Cormack and Lamberty (Types A–D) based on blood supply pattern
- Examples:
- Radial forearm flap (fasciocutaneous; radial artery pedicle)
- Anterolateral thigh (ALT) flap (descending branch of lateral femoral circumflex artery)
- Lateral arm flap (posterior branch of radial collateral artery)
- Medial fasciocutaneous flap of leg — proximally based; used for proximal/middle third tibial defects
- Lateral fasciocutaneous flap of leg — based on peroneal artery perforators; limited reach
- Reversed sural artery flap — distally based; uses neurocutaneous vessels along sural nerve; useful for ankle/heel; up to 21% partial necrosis rate
Perforator Flaps
- An evolution of fasciocutaneous and musculocutaneous concepts
- Blood supply from perforating vessels (musculocutaneous or septocutaneous) that pierce the fascia
- Only the skin paddle and the perforator vessel are harvested — underlying muscle is preserved intact
- Advantages: minimal donor-site morbidity, no muscle sacrifice, thinner/more pliable flaps
- Examples:
- DIEP flap (deep inferior epigastric artery perforator) — breast reconstruction without sacrificing rectus abdominis muscle
- ALT flap — most versatile workhorse free flap in current use
- Propeller flap — local perforator-based flap rotated up to 180°
Free Flaps (Free Tissue Transfer)
- A distant axial pattern flap raised on its named arteriovenous pedicle, then divided and microvascularly anastomosed to recipient vessels near the defect
- Considered the gold standard for complex reconstruction
- Requirements: patent recipient vessels, microsurgical expertise
Workhorse Free Flaps
| Flap | Pedicle | Tissue Type | Common Use |
|---|---|---|---|
| Radial forearm free flap | Radial artery | Fasciocutaneous | Oral/oropharyngeal reconstruction |
| Anterolateral thigh (ALT) | Descending branch of LFCA | Fasciocutaneous/musculocutaneous | Versatile — head & neck, limbs |
| Fibular free flap | Peroneal artery | Osteocutaneous | Mandibular, long bone reconstruction |
| Latissimus dorsi | Thoracodorsal artery | Musculocutaneous | Breast, chest wall, scalp |
| DIEP/TRAM | Deep inferior epigastric artery | Fasciocutaneous/musculocutaneous | Breast reconstruction |
| Gracilis | Medial circumflex femoral artery | Muscular | Facial reanimation, perineoplasty |
Postoperative Monitoring of Free Flaps
- Patient monitored in ICU / warm room (for vasodilation)
- Vasoconstrictors (nicotine, caffeine) restricted
- Maintain euvolaemia; avoid hypotension
- Clinical monitoring: colour, capillary refill, turgor, Doppler signal, temperature
Free Flap Complications
- Main cause of failure: inadequate arterial inflow
- Persistent vasospasm → thrombosis at anastomosis
- Seroma/haematoma → flap failure
- Venous congestion (presents early as purple/turgid flap)
- Partial or total flap loss
Tissue Expansion
A technique to increase available local flap tissue:
- Inflatable silicone elastomer expander placed beneath the planned flap
- Gradually inflated by serial saline injections over several weeks
- Histologic changes: thickened dermis, enhanced vasculature, reduced subcutaneous fat — actual new tissue generation occurs
- Expanded capsule is highly vascular → improves flap blood supply (acts as a "delay" phenomenon)
- Expander removed, expanded tissue transferred into defect
Advantages: tissue of identical colour, texture, thickness; minimal donor morbidity
Disadvantages/Complications: infection, haematoma, seroma, extrusion, implant failure, skin necrosis, pain, nerve injury, temporary cosmetic deformity
Applications: giant congenital naevi, burn scar reconstruction, scalp reconstruction, breast reconstruction
Reconstructive Ladder (Concept)
When planning wound coverage, surgeons follow the reconstructive ladder in ascending complexity:
- Primary closure
- Secondary intention
- Skin grafts (STSG/FTSG)
- Local flaps
- Regional pedicled flaps
- Free tissue transfer (free flaps)
The principle: use the simplest reliable option that achieves durable, functional, aesthetic reconstruction.
Sources: Schwartz's Principles of Surgery 11e; Rockwood & Green's Fractures in Adults 10e; Miller's Review of Orthopaedics 9e; Hinman's Atlas of Urologic Surgery
Decubitus ulcer from sabiston
Reading File
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.