Hepatocellular carcinoma
Reading File
Finding Sources
Searching PubMed
"hepatocellular carcinoma"[MeSH Terms] AND treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have rich, comprehensive content from multiple authoritative textbooks plus excellent imaging. I can compose a thorough, well-structured answer.
Hepatocellular Carcinoma (HCC)
Epidemiology
HCC is the most common primary liver tumor and the third leading cause of cancer death worldwide, accounting for approximately 900,000 new cases and 830,000 deaths annually. Over 80% of cases occur in developing countries, largely reflecting regional differences in risk factor prevalence. Incidence is rising in Western nations, driven by hepatitis C, non-alcoholic steatohepatitis (NASH), and alcohol-related cirrhosis. — Goldman-Cecil Medicine
Risk Factors
| Risk Factor | Key Points |
|---|---|
| Hepatitis B virus (HBV) | Most important worldwide; HCC can arise even without cirrhosis via direct viral DNA integration into host genome (particularly TERT promoter) |
| Hepatitis C virus (HCV) | Typically requires long-standing cirrhosis before HCC develops |
| Cirrhosis (any cause) | Alcohol, NASH, hemochromatosis, primary biliary cholangitis |
| Aflatoxin B1 | Environmental carcinogen (Aspergillus-contaminated food); acts synergistically with HBV |
| Non-alcoholic fatty liver disease (NAFLD/NASH) | Increasing contributor in Western populations |
| Fibrolamellar HCC | Rare subtype; young patients, no underlying liver disease |
Pathogenesis
HCC arises from malignant transformation of hepatocytes through a multi-step process:
- Chronic liver injury → inflammation, necrosis, regeneration
- Progressive accumulation of somatic mutations
- Key molecular events:
- TERT promoter mutations — ~45% of cases (telomerase activation)
- TP53 inactivation — ~30%
- β-catenin (CTNNB1) activation — ~25%
- Wnt, cell cycle, and chromatin-remodeling pathway alterations
In HBV-related HCC, direct viral DNA integration into the host genome provides an additional oncogenic mechanism. — Goldman-Cecil Medicine
Clinical Presentation
Presentation depends on tumor extent and degree of underlying liver dysfunction:
- Asymptomatic — detected by surveillance imaging or AFP elevation (most common in early disease)
- Decompensated liver disease — new ascites, encephalopathy, jaundice (tumor triggering hepatic decompensation)
- Right upper quadrant pain or a palpable mass
- Constitutional symptoms — weight loss, fever, night sweats (in advanced disease)
- Spontaneous tumor rupture — acute abdomen with hemoperitoneum (rare but life-threatening)
- Paraneoplastic syndromes — erythrocytosis (ectopic erythropoietin production), hypercalcemia
Diagnosis
Serum Biomarkers
- Alpha-fetoprotein (AFP): the key tumor marker; markedly elevated levels are diagnostic even without imaging confirmation, but HCC can occur with normal AFP. CA 19-9 and CEA are added if diagnosis is unclear.
Imaging
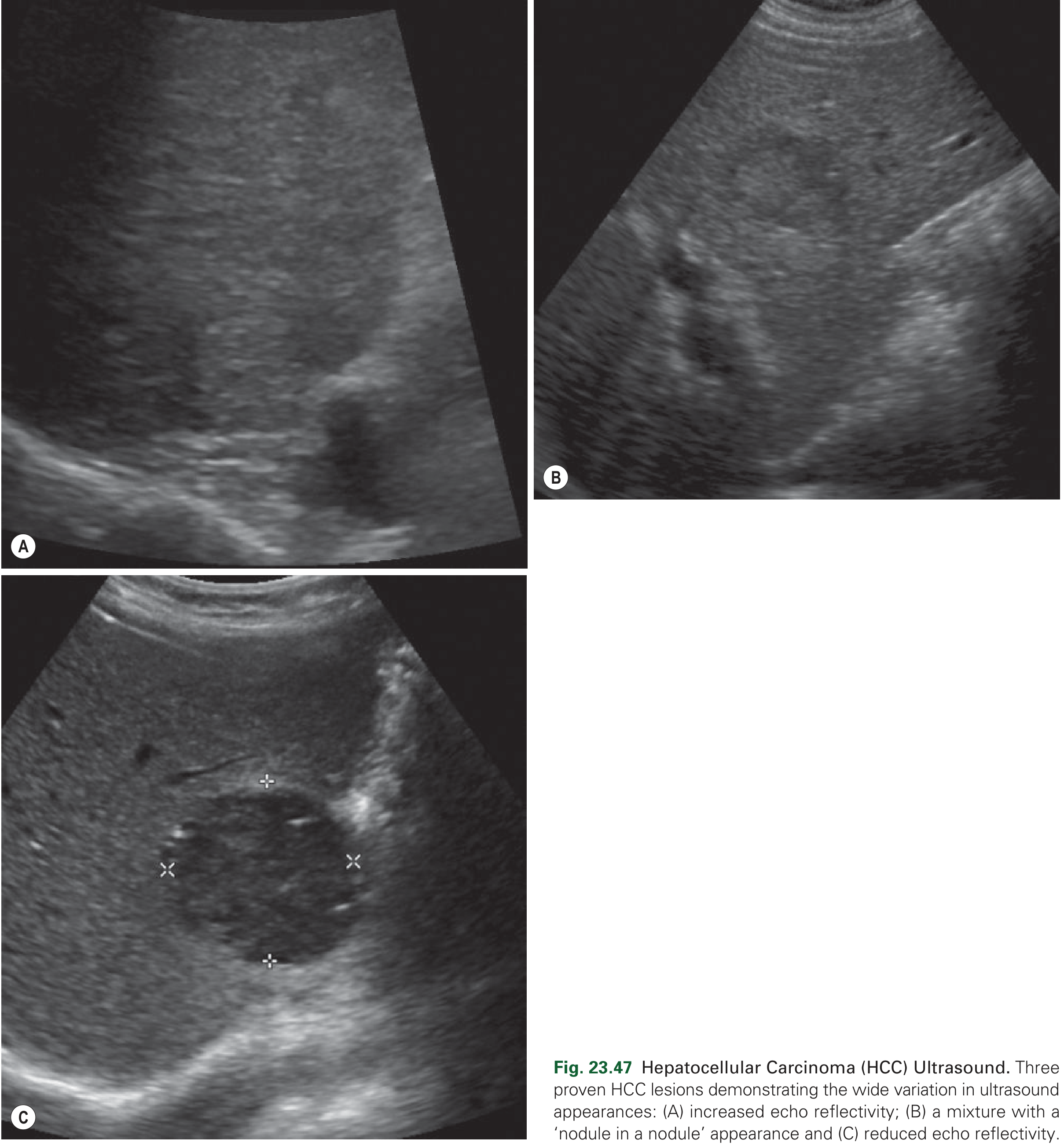
Ultrasound — first-line screening modality. HCC lesions show variable echogenicity (increased, decreased, or mixed "nodule in nodule" pattern). Portal vein thrombus and high-velocity Doppler signals from arterioportal shunting are important associated findings.
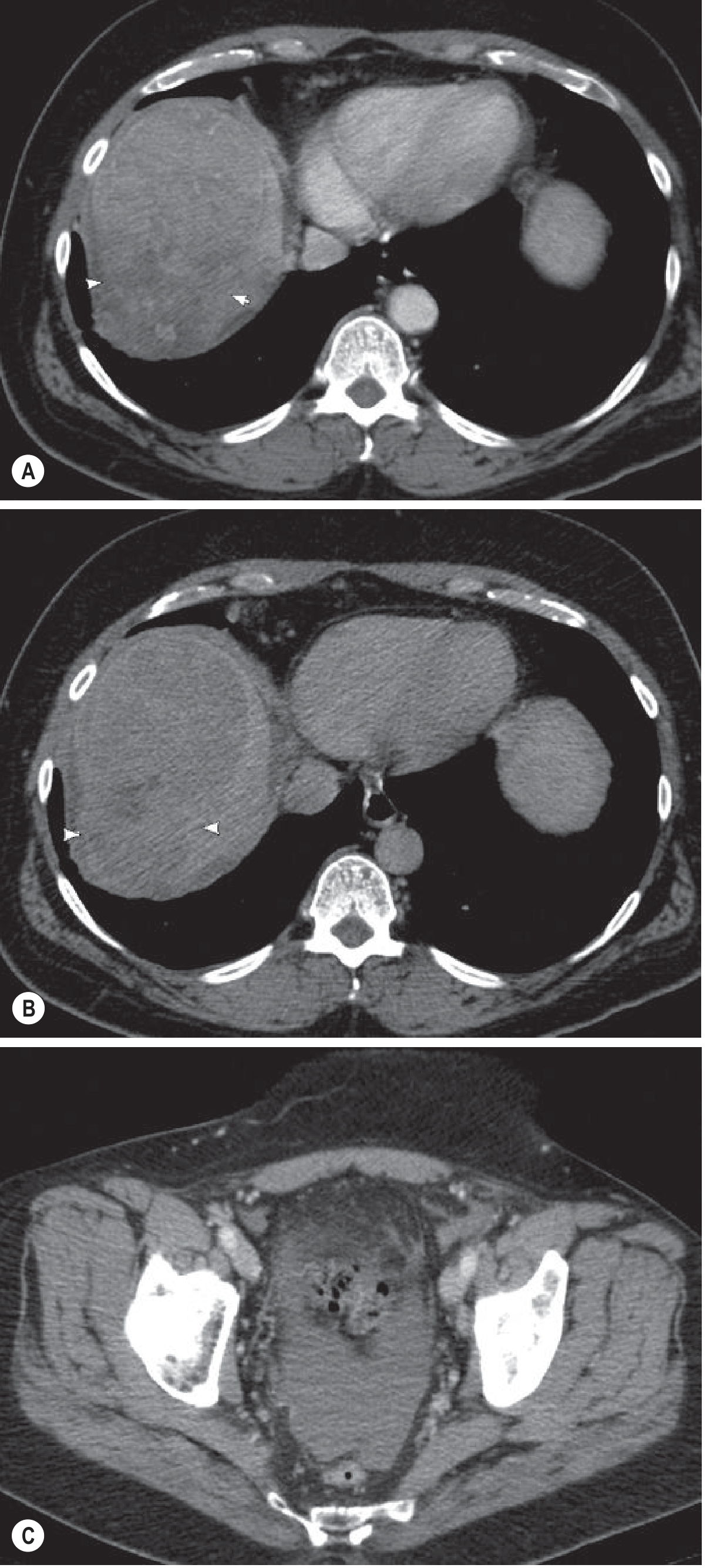
CT (multiphase) — Most HCCs are hypervascular, with characteristic arterial phase hyperenhancement followed by portal-venous phase "washout". Some show a "mosaic" enhancement pattern. Late arterial phase is essential as many lesions are only visible then. Calcification occurs in up to 7.5% of lesions; fat accumulation can also occur.
MRI — Most sensitive modality for HCC detection. Typical appearance: ↓T1, moderately ↑T2 signal, arterial phase hyperenhancement with portal/delayed phase washout. Portal venous invasion appears as vascular thrombus with portal vein dilatation. Hepatocyte-specific contrast agents (e.g., gadoxetate) enhance sensitivity further.
LI-RADS (Liver Imaging Reporting and Data System): standardizes lesion characterization in at-risk patients. LI-RADS 5 = diagnostic of HCC (no biopsy required for treatment planning in most cases). Biopsy is reserved for atypical cases or when molecular profiling for systemic therapy is needed.
Staging
Multiple staging systems are used; importantly, all must account for both the tumor burden and the degree of underlying liver disease:
| System | Basis | Use |
|---|---|---|
| BCLC (Barcelona Clinic Liver Cancer) | Tumor burden + liver function + performance status | Guides treatment allocation; most widely adopted |
| AJCC TNM | Anatomic tumor extent | Standard oncologic staging |
| Okuda | Tumor size + liver function parameters | Historical; less commonly used now |
| Child-Turcotte-Pugh (CTP) | Liver function (bilirubin, albumin, PT/INR, ascites, encephalopathy) | Critical for surgical risk stratification |
CTP Score Guide:
- Class A (5–6 pts): well-compensated cirrhosis — surgery feasible
- Class B (7–9 pts): significant functional compromise
- Class C (10–15 pts): decompensated — major surgery contraindicated
Staging workup: history & exam, AFP (+ CA 19-9/CEA if diagnosis unclear), chest CT, abdominal CT or MRI, selective bone scan. Common metastatic sites: lung, peritoneum, bone, adrenal gland, spleen, brain.
Treatment
1. Liver Transplantation
Treatment of choice for patients with advanced cirrhosis and limited HCC (meeting Milan criteria: single lesion ≤5 cm or ≤3 lesions none >3 cm, no vascular invasion or extrahepatic spread). Addresses both the malignancy and the underlying "field defect" of a cirrhotic liver. Limitations include organ availability and immunosuppression risks.
2. Surgical Resection
Best for patients with limited underlying liver disease (no cirrhosis, or CTP class A without portal hypertension). Perioperative mortality <5% in well-selected patients; rises significantly in CTP B/C or with portal hypertension. Anatomic (portal territory) resections preferred, as HCC spreads along portal venous tributaries. No proven adjuvant systemic therapy currently exists.
3. Thermal Ablation
For patients unfit for surgery, ablation (radiofrequency or microwave) of small HCC (<2–3 cm) achieves outcomes comparable to resection in randomized trials, with lower morbidity. Used as definitive therapy or bridge to transplantation.
4. Intra-arterial Therapies
For patients not candidates for curative options:
- TACE (transarterial chemoembolization) — most commonly used
- Bland hepatic artery embolization (HAE)
- Radioembolization (Y-90) — selective internal radiation therapy
- Often used as bridge to transplantation or before ablation of 3–5 cm tumors
5. Systemic Therapy
For extensive extrahepatic disease or when liver-directed therapy is insufficient:
| Line | Agents |
|---|---|
| First-line | Sorafenib, lenvatinib, atezolizumab + bevacizumab (anti-PD-L1 + anti-VEGF), nivolumab |
| Second-line | Regorafenib, cabozantinib, ramucirumab, nivolumab/pembrolizumab |
| Immunotherapy | Pembrolizumab or nivolumab (alone or nivolumab + ipilimumab) — can provide durable tumor responses |
Multikinase inhibitors (sorafenib, lenvatinib, regorafenib, cabozantinib) and checkpoint inhibitors have expanded survival options significantly. — Goldman-Cecil Medicine; Current Surgical Therapy 14e
Prognosis
- Overall 5-year survival remains <30% due to late presentation and underlying liver disease
- Early-stage disease treated by resection or transplantation can achieve 5-year survival >70%
- Surveillance programs (6-monthly liver ultrasound ± AFP in at-risk populations) are critical for detecting resectable disease
Surveillance Recommendations
High-risk groups warranting surveillance:
- All cirrhotics (regardless of etiology)
- HBV-infected patients (even without cirrhosis, particularly males >40 years, Africans >20 years, or those with family history of HCC)
Recommended: Liver ultrasound every 6 months, with or without AFP. — AASLD guidelines
Sources: Goldman-Cecil Medicine (International Edition); Current Surgical Therapy, 14th ed.; Grainger & Allison's Diagnostic Radiology.
This is a shared conversation. Sign in to Orris to start your own chat.