Define prematurity. Mention the clinical features of HMD (Hyaline membrane disease). Briefly outline the management of HMD.
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Loading Image
Loading Image
I now have all the material needed for a comprehensive answer. Here is the complete response:
Prematurity and Hyaline Membrane Disease (HMD)
1. Definition of Prematurity
Prematurity is defined as birth before 37 completed weeks of gestation, regardless of birth weight.
This is distinct from small for gestational age (SGA), which describes an infant (term or preterm) whose age-adjusted weight is below the 5th percentile — a distinction based on growth restriction rather than gestational age alone.
Sub-classifications by gestational age:
| Category | Gestational Age |
|---|---|
| Late preterm | 34–36⁺⁶ weeks |
| Moderately preterm | 32–33⁺⁶ weeks |
| Very preterm | 28–31⁺⁶ weeks |
| Extremely preterm | < 28 weeks |
Complications of prematurity (due to organ immaturity):
- Pulmonary: Hyaline membrane disease (RDS), apneic spells, bronchopulmonary dysplasia
- Cardiovascular: Patent ductus arteriosus → left-to-right shunt → pulmonary edema / congestive heart failure
- GI: Necrotizing enterocolitis (from hypoxia/ischemic gut)
- CNS: Intraventricular hemorrhage, periventricular leukomalacia
- Metabolic: Hypothermia, hypoglycemia, hypocalcemia, hyperbilirubinemia (immature liver)
- Ocular: Retinopathy of prematurity
- Immunologic: Increased susceptibility to infection, kernicterus
- Morgan & Mikhail's Clinical Anesthesiology, 7e, p. 1711; Medical Physiology, p. 463
2. Hyaline Membrane Disease (HMD) — Neonatal RDS
HMD, also called Neonatal Respiratory Distress Syndrome (RDS), is the most important and common cause of respiratory failure in preterm neonates. It is fundamentally a disease of surfactant deficiency resulting in widespread alveolar collapse.
Most cases occur in neonates born before 28 weeks gestational age; the risk decreases progressively as gestation advances toward 35 weeks, after which surfactant production by type II pneumocytes accelerates markedly.
Pathophysiology (Summary)
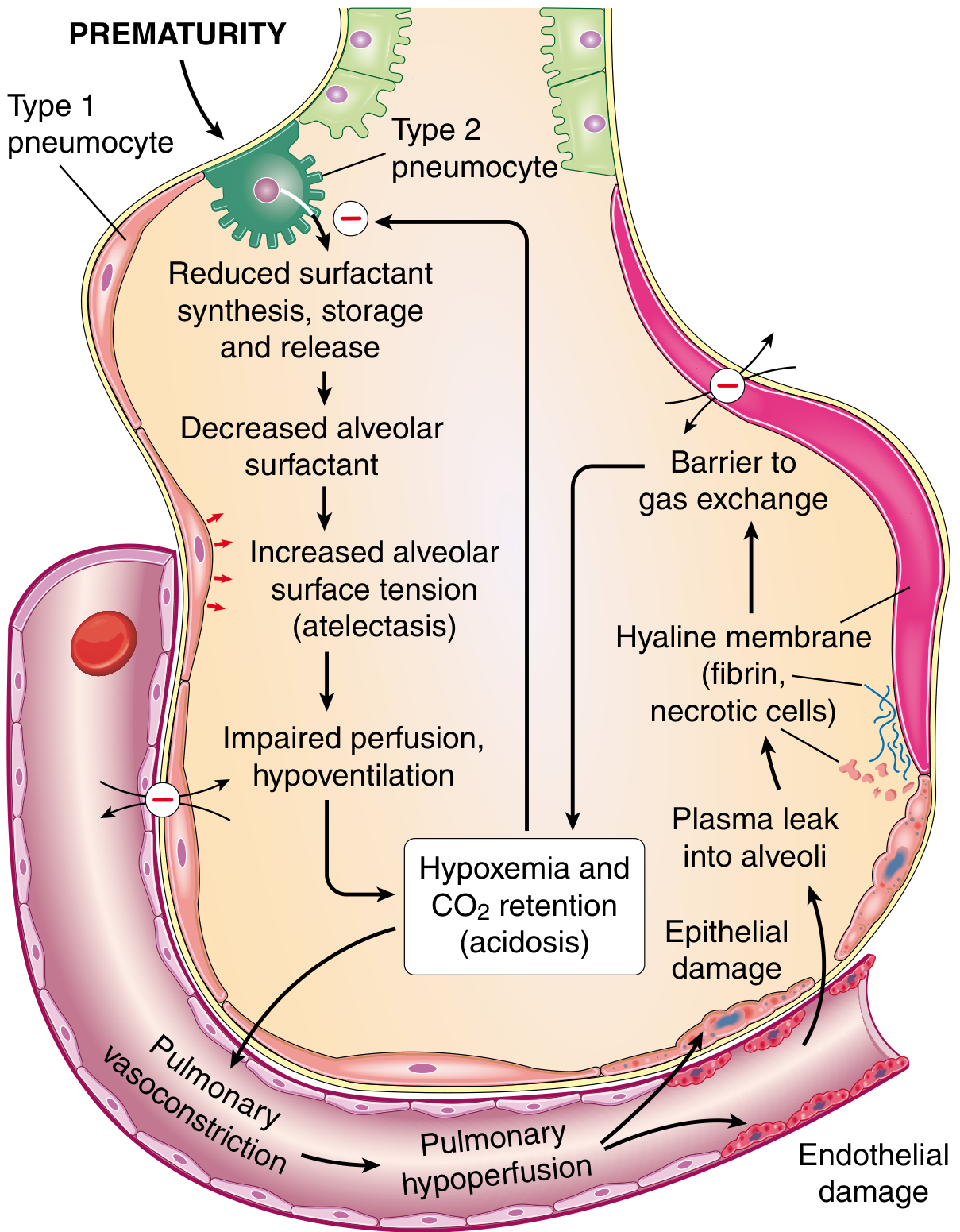
Fig. 10.6 — Pathophysiology of RDS (Robbins, Cotran & Kumar)
Prematurity → ↓ surfactant (dipalmitoyl phosphatidylcholine, SP-B, SP-C) → ↑ alveolar surface tension → atelectasis → impaired perfusion, hypoventilation → hypoxemia + CO₂ retention (acidosis) → pulmonary vasoconstriction + endothelial/epithelial damage → plasma leak into alveoli → hyaline membrane formation (fibrin + necrotic cells) → barrier to gas exchange → further ↓ surfactant synthesis — a vicious cycle.
3. Clinical Features of HMD
Risk Factors
- Preterm birth (strongest association; <28 weeks highest risk)
- Male sex
- Infant of a diabetic mother (insulin counteracts glucocorticoid-induced surfactant synthesis)
- Cesarean section (especially before onset of labour — labour itself stimulates surfactant synthesis)
Onset and Progression
- At birth: The infant may require resuscitation, but usually establishes rhythmic breathing within a few minutes and appears normal in color initially.
- Within 30 minutes of birth: Breathing becomes progressively more labored.
- Within a few hours: Frank cyanosis develops in the untreated infant.
Signs and Symptoms
| Feature | Detail |
|---|---|
| Tachypnea | Respiratory rate > 60/min |
| Grunting | Expiratory grunt (auto-PEEP to prevent alveolar collapse) |
| Subcostal / intercostal retractions | Due to high inspiratory effort against stiff, non-compliant lungs |
| Nasal flaring | Accessory muscle use |
| Cyanosis | Progressive; central cyanosis |
| Fine rales | Heard bilaterally on auscultation |
| Increasing oxygen requirement | Even 80% O₂ via ventilatory support may fail to improve oxygenation in severe cases |
Chest X-Ray (CXR) Findings
- Uniform minute reticulogranular (ground-glass) densities bilaterally
- Air bronchograms superimposed on the diffuse haziness
- In severe cases: complete "white-out" of lung fields
Gross Pathology
- Lungs are normal in size but solid, airless, and reddish-purple (liver-like in color)
- They sink in water (indicating absence of entrapped air)
Histopathology
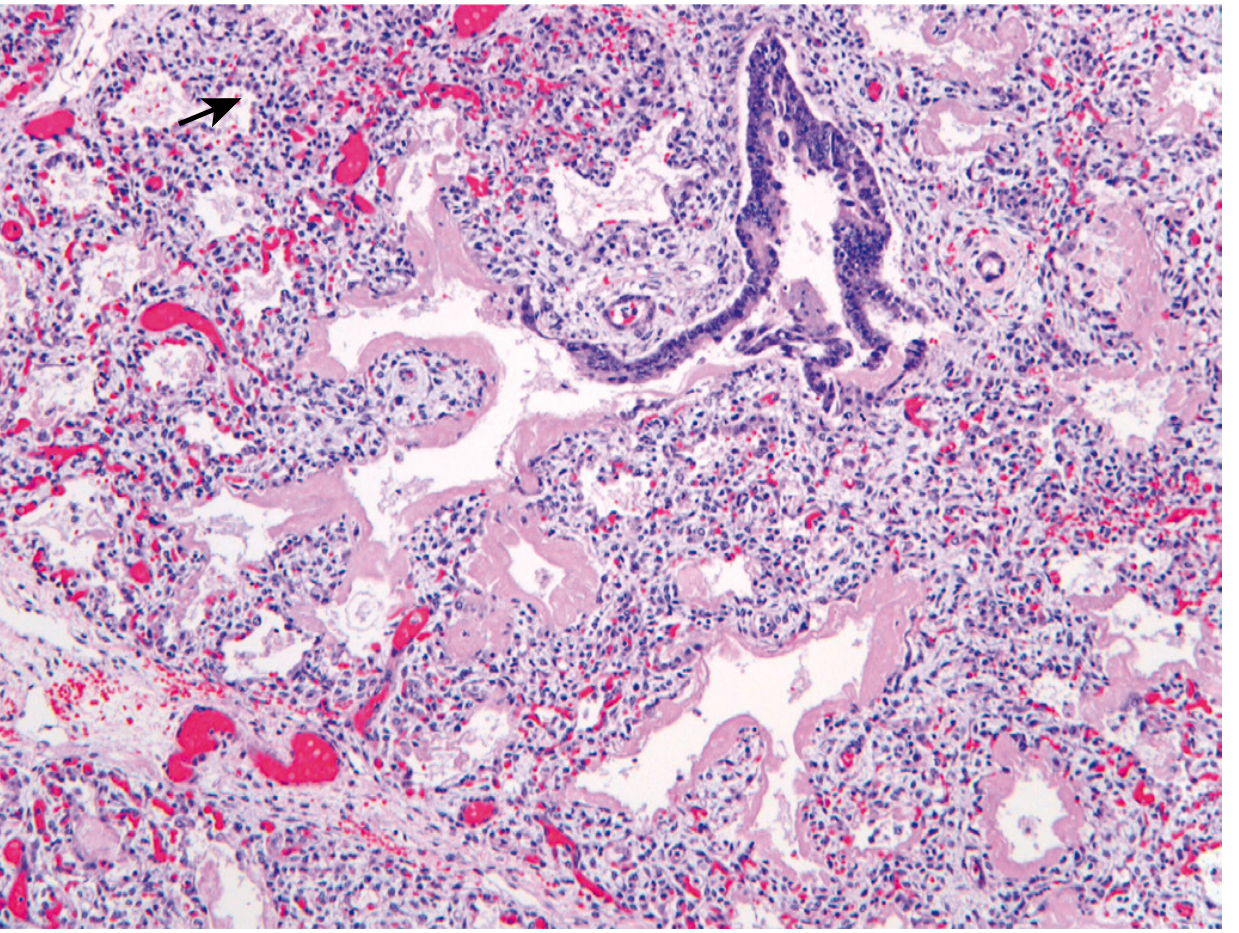
Fig. 10.7 — Hyaline membrane disease: eosinophilic hyaline membranes lining dilated alveoli; arrow = cuboidal epithelium indicating lung immaturity (Robbins, Cotran & Kumar)
- Alveoli are poorly developed and collapsed
- Eosinophilic hyaline membranes line the respiratory bronchioles, alveolar ducts, and alveoli
- Membranes composed of fibrin admixed with necrotic cellular debris
- Hyaline membranes are never seen in stillborn infants (they require a period of breathing)
Natural Course
- If untreated, death may occur within hours to days.
- If the infant survives beyond 3–4 days, recovery is likely.
- In survivors beyond 48 hours, alveolar epithelium proliferates beneath the membranes; macrophages phagocytose the debris.
- Robbins, Cotran & Kumar Pathologic Basis of Disease, pp. 432–434
4. Management of HMD — Outline
Management has three pillars: Prevention, Supportive care, and Specific (surfactant) therapy.
A. Prevention
| Intervention | Mechanism |
|---|---|
| Antenatal corticosteroids (betamethasone/dexamethasone IM, 24–34 weeks) | Accelerate fetal lung maturity; stimulate surfactant synthesis by type II pneumocytes |
| Delay premature labour | Prolongs gestation to allow further lung maturation |
| Fetal lung maturity assessment | Amniotic fluid phospholipid analysis (lecithin:sphingomyelin ratio ≥ 2:1 = maturity); phosphatidylglycerol presence |
B. Supportive Care
| Measure | Detail |
|---|---|
| Oxygen therapy | Supplemental O₂ to maintain SpO₂ 91–95%; avoid hyperoxia (risk of retinopathy of prematurity) |
| CPAP (Continuous Positive Airway Pressure) | First-line ventilatory support; keeps alveoli open, reduces work of breathing |
| Mechanical ventilation | For severe cases failing CPAP; use minimal pressures to avoid barotrauma |
| Thermoregulation | Incubator/radiant warmer; hypothermia worsens acidosis and ↓ surfactant synthesis |
| Fluid & electrolyte management | Careful IV fluids; avoid overhydration (worsens pulmonary edema) |
| Nutritional support | IV glucose initially; parenteral nutrition |
| Antibiotics | Cover for sepsis (clinical picture may overlap with neonatal pneumonia/GBS sepsis) |
| Treat PDA | Indomethacin or ibuprofen (COX inhibitors to close PDA); surgical ligation if needed |
C. Specific Therapy — Exogenous Surfactant
- Prophylactic surfactant at birth for extremely premature infants (<28 weeks gestational age) — highly beneficial.
- Rescue surfactant for infants who develop RDS — instilled directly into the trachea via endotracheal tube.
- Types: Natural/animal-derived (poractant alfa, beractant) — preferred over synthetic preparations.
- Results in dramatic improvement in pulmonary function, shortened clinical course, and markedly reduced mortality.
D. Complications to Monitor and Treat
| Complication | Management |
|---|---|
| Retinopathy of prematurity | Avoid oxygen fluctuations; ophthalmology screening; laser photocoagulation if needed |
| Bronchopulmonary dysplasia | Gentle ventilation strategies, diuretics, postnatal steroids (cautious) |
| Intraventricular hemorrhage | Antenatal steroids (also protective); avoid hemodynamic instability |
| Necrotizing enterocolitis | Early enteral feeding with breast milk; surgical if perforated |
- Robbins, Cotran & Kumar Pathologic Basis of Disease, pp. 432–434; Robbins & Kumar Basic Pathology, p. 152; Morgan & Mikhail's Clinical Anesthesiology, p. 1711; The Developing Human: Clinically Oriented Embryology, p. 954
thyroid function tests in early newborn periods
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Loading Image
I now have all the material needed for a comprehensive, well-structured answer.
Thyroid Function Tests in the Early Newborn Period
Background: Fetal Thyroid Development
Understanding neonatal thyroid function tests requires knowing the developmental timeline:
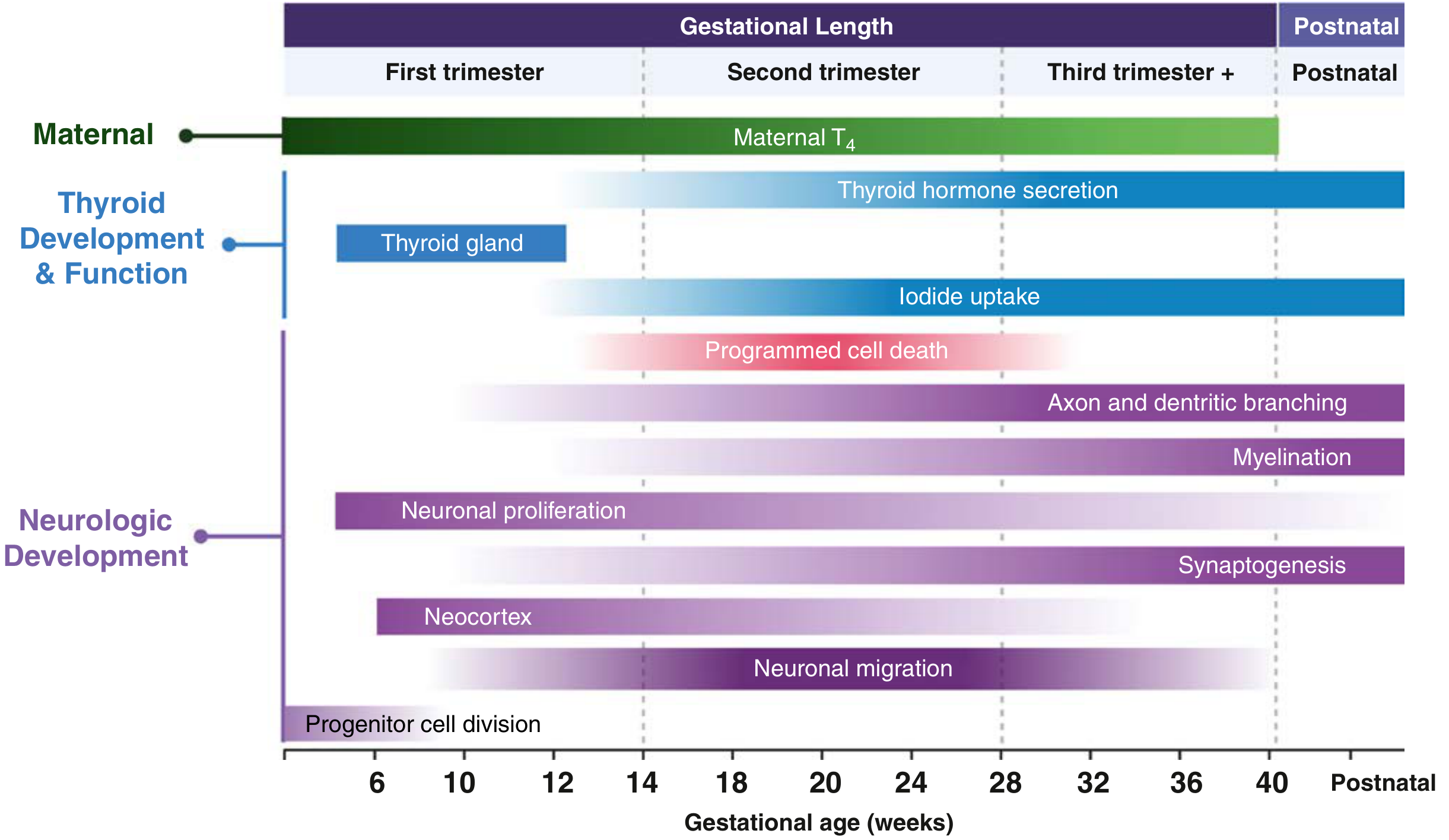
Fig. 61.5 — Thyroid development and neurological milestones (Creasy & Resnik)
| Gestational milestone | Thyroid event |
|---|---|
| 3rd week | Thyroid gland develops at base of tongue from endodermal cells |
| 5–7 weeks | Thyroid descends to pretracheal position; divides into lobes |
| 10–12 weeks | Hypothalamic–pituitary vascular maturation; TRH becomes detectable |
| 12–14 weeks | Iodide trapping, TH synthesis, and thyrocyte differentiation begin |
| <16 weeks | Fetus entirely dependent on maternal T₄ |
| 18–20 weeks | Iodine uptake, TSH, and T₄ concentrations start rising significantly |
| 30 weeks | T₃ begins rising (before this, high D3 activity converts T₄ → reverse T₃) |
| Term (~40 weeks) | T₄ reaches adult levels (~10 μg/dL); 30–50% of cord blood T₄ is still maternally derived |
The hypothalamic–pituitary–thyroid (HPT) axis does not fully mature until 1–2 months after birth.
The Neonatal TSH Surge — The Key Postnatal Event
At the moment of birth, extrauterine exposure to the cold environment triggers an immediate and dramatic physiological response in thyroid hormone axes:
Sequence of events immediately after birth:
- Cold exposure → stimulates hypothalamus → acute TRH surge
- TRH surge → anterior pituitary → massive TSH surge
- TSH peaks at 60–80 mIU/L (some sources: up to 70–100 mIU/L) within 30 minutes of birth
- High TSH → stimulates thyroid gland → T₄ rises moderately; increased peripheral conversion of T₄ → T₃ (3–4-fold rise in T₃)
- Over the next 3–5 days: Rising T₄ and T₃ exert negative feedback → TSH falls to normal adult levels
- By 4–6 weeks: T₄ and T₃ concentrations return to stable normal adult levels
This transient hyperthyroxinemia after birth represents a physiological thermogenic adaptation to extrauterine life.
Summary of TFT values at key time points:
| Hormone | Cord blood (at birth) | 30 min–24 h postnatal | Day 3–5 | 4–6 weeks |
|---|---|---|---|---|
| TSH | 7–10 mIU/L | Surge: 60–80 mIU/L | Falls rapidly | Adult range (~0.5–4.5 mIU/L) |
| Total T₄ | ~10 μg/dL | Moderate rise | Elevated | Normalizes |
| Free T₄ (FT₄) | Low-normal | Rises | Elevated vs adults | Normalizes |
| T₃ | Low (~50 ng/dL) | 3–4× surge | Elevated | Normalizes |
| Reverse T₃ (rT₃) | High (fetal pattern) | Rapidly falls | Low | Low |
Physiological Explanation for High rT₃ at Birth
In fetal life, a high ratio of deiodinase type 3 (D3) to type 1 (D1) preferentially converts T₄ to reverse T₃ (rT₃) — a biologically inactive metabolite. This prevents overexposure of the developing fetus to active T₃. After birth, D1 activity rises and rT₃ rapidly falls while T₃ rises.
TFT Interpretation Pitfalls in the Newborn Period
| Pitfall | Explanation |
|---|---|
| TSH appears elevated in first 24–48 h | Physiological surge — do NOT diagnose hypothyroidism on day 1 values |
| T₃ is low at birth / in preterm infants | Normal; fetal D3 keeps T₃ low until near term |
| rT₃ is elevated at birth | Normal fetal pattern; falls rapidly after birth |
| TBG (thyroxine-binding globulin) is higher in neonates | Causes total T₄ to be higher than in adults; FT₄ is more reliable |
| Premature infants have lower FT₄ and FT₃ with normal or low-normal TSH | Immature HPT axis — TSH does not mount an appropriate compensatory rise; can mimic central hypothyroidism |
Thyroid Function in Premature Neonates
Prematurity disrupts the normal postnatal TFT pattern:
- The physiological TSH surge is dramatically lower or absent in preterm infants
- FT₄ and FT₃ levels are low in the face of normal TSH — a pattern called transient hypothalamic hypothyroidism of prematurity
- It takes 3–8 weeks after birth for FT₄/FT₃ levels to reach term-equivalent values
- Seen in up to 50% of infants born < 28 weeks gestation
- Difficult to distinguish from true central hypothyroidism
- Associated with cognitive and neurological delays, though optimal T₄ target levels are not yet established and there is no universal treatment consensus
Newborn Screening for Congenital Hypothyroidism
Why it matters:
- Congenital hypothyroidism (CH) occurs in ~1:2,000–4,000 births
- The newborn looks clinically normal at birth (maternal T₄ partially protects)
- Untreated CH causes irreversible intellectual disability (cretinism)
- Early detection and treatment (within 2 weeks) leads to normal neurodevelopment
Screening strategy:
| Country approach | Method |
|---|---|
| Primary TSH (most common, e.g., UK, Australia) | Detects primary hypothyroidism; misses central hypothyroidism |
| Primary T₄ + backup TSH (Netherlands, parts of USA) | Detects both primary and central hypothyroidism |
| Combined TSH + FT₄ | Best — differentiates central vs primary CH, thyroid dysgenesis, dyshormonogenesis, thyroid hormone resistance, and delayed TSH elevation |
Timing:
- Term infants: ideally at 2–4 days of age (by day 5 of life at latest); avoids false-positive results from the physiological TSH surge
- Preterm infants: within 7 days of birth; may need repeat testing at 2–4 weeks due to the attenuated initial TSH response
- Specimen: dried blood spot on filter paper (Guthrie card), heel-prick capillary blood
Causes detected by screening:
| Cause | Mechanism |
|---|---|
| Thyroid dysgenesis (aplasia, hypoplasia, ectopia) | Most common; ~85% of CH cases |
| Dyshormonogenesis | TPO/TG/NIS/DUOX2 gene mutations |
| Central hypothyroidism | TSH deficiency (pituitary) or TRH deficiency (hypothalamic) — missed on TSH-only screening |
| Transient CH | Maternal TSH-receptor blocking antibodies, iodine excess/deficiency, antithyroid drug exposure |
Special Situations
Infants of Graves' disease mothers:
- Maternal TSH-receptor stimulating antibodies (TSI/TRAb) cross the placenta
- Can cause neonatal Graves' hyperthyroidism — transient but potentially dangerous
- Screen by measuring TRAb/TSI in at-risk mothers (history of Graves' disease) during pregnancy
Maternal hypothyroidism effect:
- Low maternal T₄ in first trimester (before fetal thyroid function begins) → impaired fetal neurodevelopment
- Associated with lower IQ, ADHD in offspring
- Maternal TSH screening is recommended before pregnancy or in the first trimester
Key Points Summary
- TSH surges to 60–80 mIU/L within 30 minutes of birth (cold stimulus) — a normal physiological event; do not interpret as pathological
- T₃ rises 3–4-fold in the first 24–48 hours; rT₃ falls postnatally
- T₄ and T₃ normalize by 4–6 weeks; the HPT axis matures by 1–2 months
- Prematurity blunts the TSH surge and causes transient low FT₄/FT₃ with normal TSH
- Newborn screening for congenital hypothyroidism uses dried blood spot TSH (± T₄) at 2–4 days in term infants
- TSH-only screening misses central hypothyroidism — combined TSH + FT₄ is more sensitive
- Neonatal TFT values must always be interpreted using age- and gestational-age-specific reference intervals
- Creasy & Resnik's Maternal-Fetal Medicine, pp. 1497–1499; Tietz Textbook of Laboratory Medicine, 7th ed., pp. 4767–4773
This is a shared conversation. Sign in to Orris to start your own chat.