Gestational trophoblastic neoplasia
Reading File
Finding Sources
Searching PubMed
"gestational trophoblastic neoplasia" AND treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
gestational trophoblastic neoplasia hydatidiform mole choriocarcinoma
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the full answer.
Gestational Trophoblastic Neoplasia (GTN)
Overview
Gestational trophoblastic disease (GTD) is a spectrum of interrelated tumors arising from abnormal proliferation of placental trophoblasts. The malignant subset — gestational trophoblastic neoplasia (GTN) — comprises invasive mole, choriocarcinoma, placental-site trophoblastic tumor (PSTT), and epithelioid trophoblastic tumor (ETT). GTN represents one of the most curable solid tumors in medicine, even in the presence of widespread metastasis. — Berek & Novak's Gynecology
Classification of GTD
| Category | Entities |
|---|---|
| Benign | Complete hydatidiform mole, Partial hydatidiform mole |
| Malignant (GTN) | Invasive mole, Choriocarcinoma, PSTT, ETT |
Hydatidiform Moles — The Precursor Lesions
Complete vs. Partial Mole
| Feature | Complete Mole | Partial Mole |
|---|---|---|
| Fetal/embryonic tissue | Absent | Present |
| Hydropic swelling of villi | Diffuse | Focal |
| Trophoblastic hyperplasia | Diffuse | Focal |
| Scalloping of villi | Absent | Present |
| Karyotype | 46,XX (90%); 46,XY | Triploid (69,XXY) or tetraploid |
| Risk of GTN | 15–20% (invasive mole); 2.5% (choriocarcinoma) | 1–4% |
Complete mole arises from fertilization of an empty (enucleate) egg by one sperm that duplicates its genetic material (androgenesis/monospermy, 90%) or by two sperm (dispermy, 10%). All nuclear DNA is paternally derived. No fetal development occurs.
Partial mole arises from fertilization of a normal egg by two sperm — karyotype is triploid (69,XXY). Fetal development can occur through early second trimester but the fetus is severely anomalous. — Robbins, Cotran & Kumar
Epidemiology & Risk Factors
- US/Europe: ~0.6–1.1 per 1,000 pregnancies; Japan: 2:1,000; Taiwan: 1:125
- Risk highest at extremes of reproductive age (< 16 or > 50 years); in women > 50, molar pregnancy may affect 1 in 3 conceptions
- Prior molar pregnancy, prior miscarriage, diets low in carotene and animal fat (geographic variation) — Harrison's Principles of Internal Medicine 22E
Clinical Presentation
- Vaginal bleeding (most common), amenorrhea, nausea
- Uterus large for dates, bilateral theca-lutein cysts (ovarian enlargement from hCG stimulation)
- Markedly elevated β-hCG
- Rare: hyperthyroidism (hCG cross-reacting with TSH receptor), hyperemesis, early-onset preeclampsia
- Ultrasound: "snowstorm" appearance (complete mole); focal cystic placental spaces (partial mole)
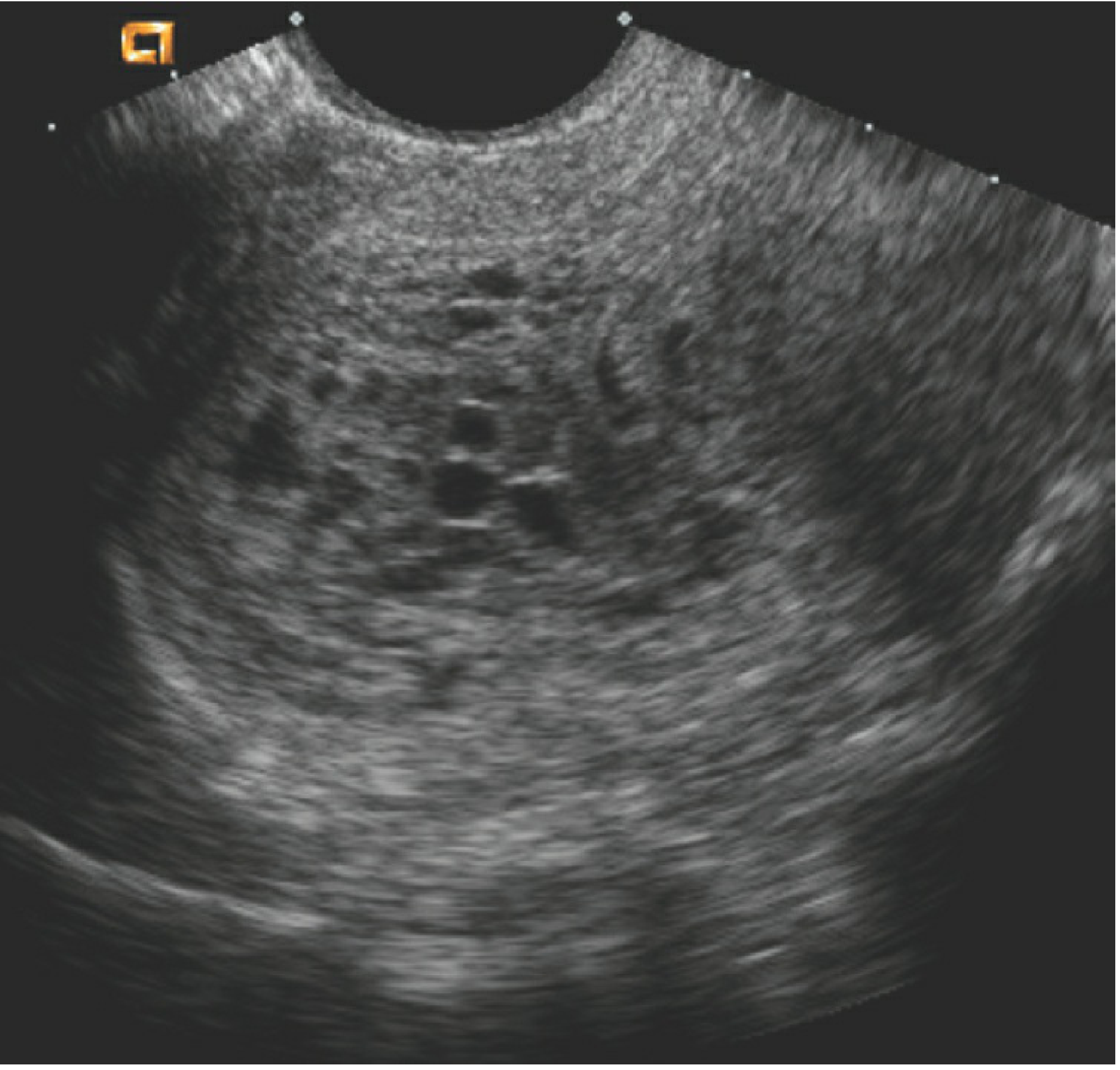
Ultrasonogram of a uterus showing the characteristic vesicular pattern of a complete hydatidiform mole. — Berek & Novak's Gynecology
Gestational Trophoblastic Neoplasia — Specific Entities
Invasive Mole
- Molar tissue that penetrates the myometrium; can perforate the uterus, causing intraperitoneal hemorrhage
- May erode uterine vessels → vaginal hemorrhage
- Arises in ~15% of complete moles; metastasizes in ~4% of post-molar cases
- Responds well to chemotherapy
Choriocarcinoma
- Highly malignant; sheets of anaplastic syncytiotrophoblast and cytotrophoblast without chorionic villi
- Can follow any pregnancy (molar, normal term, abortion, ectopic)
- Characteristically invades blood vessels → hematogenous spread; highly hemorrhagic metastases
- 2.5% risk after complete mole; post-nonmolar pregnancy GTN is always choriocarcinoma histologically
PSTT & ETT
- Composed predominantly of intermediate trophoblast; produce small amounts of hCG and human placental lactogen (hPL)
- Tend to remain confined to uterus and metastasize late
- Relatively insensitive to chemotherapy (unlike other GTN)
- Treatment: hysterectomy is primary modality
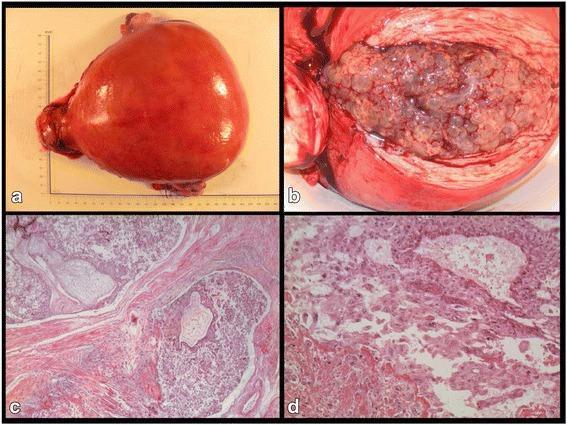
Composite: (a) gross specimen showing "bunch of grapes" appearance; (b) hydropic villi filling uterine cavity; (c) H&E showing trophoblastic invasion of myometrium; (d) high-power choriocarcinoma with atypical syncytio- and cytotrophoblasts.
Sites of Metastasis
| Site | Frequency |
|---|---|
| Lungs | 80% |
| Vagina | 30% |
| Pelvis | 20% |
| Liver | 10% |
| Brain | 10% |
Pulmonary patterns: alveolar "snowstorm," discrete rounded densities, pleural effusion, embolic pattern. Chest CT detects micrometastases in ~40% of presumed non-metastatic cases.
Vaginal metastases are highly vascular — do not biopsy (risk of severe hemorrhage).
CNS metastases: confirmed by brain CT/MRI; CSF:plasma hCG ratio < 1:60 suggests cerebral involvement.
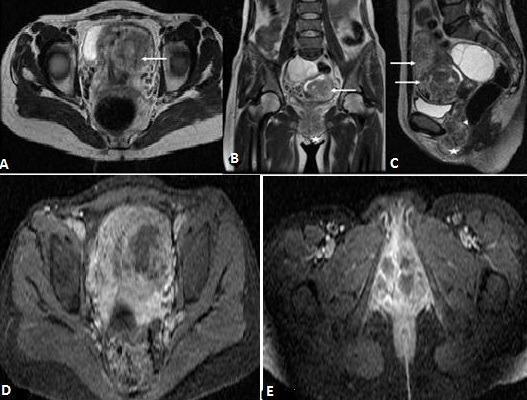
T2-weighted MRI showing enlarged uterus with bulky heterogeneous mass extensively invading the myometrium (arrows) with extension to vaginal wall (arrowhead).
Role of β-hCG
β-hCG is the cornerstone tumor marker:
- Diagnosis: elevated in all GTN (except PSTT/ETT where levels are relatively low)
- Monitor therapy response
- Detect relapse during surveillance
- Phantom/false-positive β-hCG: due to heterophilic antibodies. Excluded by: (1) simultaneous urine β-hCG (serum factors not excreted in urine), or (2) serial serum dilution (true hCG shows linear decrease)
Criteria for GTN (post-molar — WHO/FIGO): any of:
- Plateau (< 10% change) of β-hCG over 4 weekly measurements
- Rise ≥ 10% over 3 weekly measurements
- β-hCG still detectable at 6 months post-evacuation
- Histologic diagnosis of choriocarcinoma
FIGO Staging
| Stage | Description |
|---|---|
| I | Confined to uterus |
| II | Outside uterus but limited to genital structures |
| III | Pulmonary metastases (± genital involvement) |
| IV | Other distant metastases (brain, liver, kidney, GI) |
WHO Prognostic Scoring System
Scores are summed; total ≤ 6 = low risk; total ≥ 7 = high risk:
| Parameter | 0 | 1 | 2 | 4 |
|---|---|---|---|---|
| Age | < 40 | ≥ 40 | — | — |
| Antecedent pregnancy | Mole | Abortion | Term | — |
| Interval from index pregnancy | < 4 mo | 4–6 mo | 7–12 mo | > 12 mo |
| Pre-treatment hCG (IU/L) | < 1,000 | 1,000–10,000 | 10,000–100,000 | > 100,000 |
| Largest tumor size | < 3 cm | 3–4 cm | ≥ 5 cm | — |
| Site of metastases | Lung | Spleen/kidney | GI | Brain/liver |
| Number of metastases | 0 | 1–4 | 5–8 | > 8 |
| Prior failed chemotherapy | — | — | 1 drug | ≥ 2 drugs |
Management
Step 1 — Evacuate the mole
Suction curettage is preferred regardless of uterine size if fertility desired:
- Oxytocin infusion started before anesthesia
- Cervical dilation with progressive dilators
- Suction curettage (12-mm cannula preferred)
- Gentle sharp curettage to remove residual tissue
- Rh-negative patients receive Rh immune globulin
Hysterectomy if sterilization desired — ovaries can be conserved. Reduces risk of local invasion but does not prevent metastasis.
Prophylactic chemotherapy at evacuation: controversial; may be considered for high-risk patients (age > 40, uterus > gestational age, very high hCG) who cannot be followed closely.
Step 2 — Post-molar surveillance
- Serial weekly serum β-hCG until three consecutive normal values
- Then monthly for 6–12 months
- Contraception during surveillance period (OCPs safe and suppress LH which can cross-react with hCG assays)
Step 3 — Treatment of GTN
Pretreatment Workup
- Serum β-hCG, CBC, LFTs, TFTs, renal function
- Chest X-ray ± CT, pelvic US or CT, head CT/MRI (if metastatic)
- CSF hCG if brain imaging equivocal
Low-Risk GTN (FIGO Stage I–III, WHO score ≤ 6)
Single-agent chemotherapy → cure rate > 95%:
- Methotrexate (MTX): various regimens (MTX ± folinic acid rescue, 8-day MTX/FA, weekly IM MTX)
- Actinomycin-D (pulse or 5-day regimen): preferred if hepatic dysfunction or if MTX fails
High-Risk GTN (FIGO Stage II–IV, WHO score ≥ 7)
Combination chemotherapy as primary treatment:
- EMA-CO (etoposide, methotrexate, actinomycin-D, cyclophosphamide, vincristine): first-line regimen, complete response ~80%
- Multiagent salvage regimens (EMA-EP, BEP, paclitaxel-cisplatin/etoposide) for refractory disease
CNS metastases: whole-brain radiation + chemotherapy, or intrathecal MTX alongside systemic treatment; EMA-CO with high-dose MTX is used
Surgical adjuncts: hysterectomy for drug-resistant uterine disease, thoracotomy for isolated pulmonary resistant nodule
PSTT/ETT
Hysterectomy is primary treatment; EMA-EP if metastatic
Duration of Therapy
Chemotherapy continued for at least 3 cycles after the first normal β-hCG in low-risk disease; longer consolidation in high-risk.
Emerging Treatment: Immunotherapy
A 2025 systematic review and meta-analysis (PMID 40031424) evaluated pembrolizumab in refractory GTN, demonstrating meaningful response rates in chemotherapy-resistant cases, representing a potential new option for this rare scenario.
Fertility After GTN
- Normal subsequent pregnancies are expected after chemotherapy-induced remission
- No increased risk of congenital malformations or obstetric complications
- Patients advised to wait 12 months after achieving remission before attempting conception (to allow for reliable hCG surveillance)
Key Points Summary
| Feature | Detail |
|---|---|
| Curative even with metastases | Yes — one of the most curable solid tumors |
| Tumor marker | β-hCG (diagnostic + monitoring) |
| First-line Rx, low-risk | Single-agent MTX or Actinomycin-D |
| First-line Rx, high-risk | EMA-CO combination chemotherapy |
| PSTT/ETT treatment | Hysterectomy (chemo-resistant) |
| Fertility preserved | Yes, in most cases |
Sources: Berek & Novak's Gynecology; Robbins, Cotran & Kumar Pathologic Basis of Disease; Harrison's Principles of Internal Medicine 22E; Creasy & Resnik's Maternal-Fetal Medicine
This is a shared conversation. Sign in to Orris to start your own chat.