Spinal Anesthesia and epidural Anesthesia standard procedure- act as a professor of Anesthesia and teach me this topic
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
spinal anesthesia technique subarachnoid needle lumbar
Searching Images
epidural anesthesia loss of resistance technique Tuohy needle
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have everything needed for a comprehensive, professor-level lecture. Let me deliver it.
🏥 Neuraxial Anesthesia: Spinal and Epidural — A Professor's Lecture
Sources: Miller's Anesthesia 10e, Morgan & Mikhail's Clinical Anesthesiology 7e, Barash's Clinical Anesthesia 9e, Sabiston's Textbook of Surgery
PART I: FOUNDATIONAL ANATOMY — Know This Cold
Before touching a needle, you must own the anatomy. Every complication you will ever encounter traces back to imprecise knowledge of these layers.
Layers Traversed (Posterior → Anterior)
| Layer | Clinical Significance |
|---|---|
| Skin & subcutaneous fat | Skin wheal for local infiltration |
| Supraspinous ligament | Connects tips of spinous processes |
| Interspinous ligament | Between adjacent spinous processes; softer feel |
| Ligamentum flavum | Dense, rubbery — the key tactile landmark, thickest posteriorly (3–5 mm lumbar) |
| Epidural space | Contains fat, venous plexus (Batson's), lymphatics, nerve roots |
| Dura mater | Tough outer meningeal layer |
| Subdural space | Potential space — site of inadvertent subdural block |
| Arachnoid mater | True barrier to CSF |
| Subarachnoid space | Contains CSF — target for spinal anesthesia |
| Pia mater | Adheres to spinal cord |
Critical Landmarks
- Tuffier's line (intercristal/bicristal line): connects the two iliac crests → crosses at L4 spinous process or L4–L5 interspace in adults
- Conus medullaris terminates at L1 in adults (L3 in neonates) — always insert below L2 to avoid cord injury
- Dural sac terminates at S2 — caudal punctures above this risk dural puncture
The Epidural Space
The epidural space extends from the foramen magnum to the sacral hiatus. It is bounded:
- Posteriorly: Ligamentum flavum and laminae
- Anteriorly: Posterior longitudinal ligament + vertebral bodies
- Laterally: Pedicles and intervertebral foramina
📌 Professor's pearl: The epidural space is a potential space with sub-atmospheric (negative) pressure at lumbar levels — the basis of the "hanging drop" technique. It communicates freely with the paravertebral spaces through the intervertebral foramina, explaining why large epidural volumes can spread widely and why one-sided blocks occur when septa are present.
PART II: SPINAL ANESTHESIA (Subarachnoid Block)
Mechanism of Action
Local anesthetic injected into CSF acts directly on nerve roots and the spinal cord. Because drug is in CSF, onset is rapid (within 3–5 minutes), the block is dense and predictable, and only small drug volumes are needed.
Indications
- Lower abdominal surgery (hernia, appendectomy)
- Urological procedures (TURP, cystoscopy) — awake patient monitors for TUR syndrome
- Obstetric: Cesarean section, instrumental delivery
- Lower extremity orthopedic surgery (hip, knee arthroplasty)
- Perineal/anorectal/perianal surgery
- Gynecological procedures
Absolute Contraindications
- Patient refusal
- Local infection at needle site
- Severe hypovolemia/hemodynamic instability
- Raised intracranial pressure
- Coagulopathy (INR >1.4, platelets <80,000)
Relative Contraindications
- Fixed cardiac output states (severe AS, HOCM)
- Prior spine surgery
- Anatomical abnormalities (severe scoliosis, spina bifida)
- Demyelinating disease (controversial)
THE PROCEDURE — Step by Step
Step 1: Pre-procedure Checklist
- Informed consent, IV access established, monitoring applied (SpO₂, NIBP, ECG)
- Resuscitation equipment checked: airway cart, vasopressors (ephedrine 5–10 mg aliquots or phenylephrine 50–100 µg), atropine
- Preload with 500–1000 mL crystalloid (co-load is now preferred over pre-load)
Step 2: Positioning — Two Options
Option A: Sitting position (most common)
- Patient sits at edge of table, feet on a stool
- Arms folded over a pillow, chin tucked to chest
- Patient "arches back like an angry cat"
- Advantage: maximally opens interspaces, easier landmark palpation in obese patients, produces gravity-dependent pooling of hyperbaric solution (useful for saddle block)
Option B: Lateral decubitus
- Patient lies in fetal position, knees drawn to chest
- The side to be operated on placed dependently (for unilateral block with hyperbaric solution)
- Advantage: better tolerated in anxious or pre-sedated patients
Step 3: Sterile Preparation
- Full sterile gown and gloves, mask, hat — this is central neuraxial anesthesia; treat it as an operating procedure
- Cleanse skin with chlorhexidine 0.5–2% in alcohol (preferred over povidone-iodine; superior bacterial kill, but allow to dry completely — avoid intrathecal contamination)
- Drape the field
Step 4: Identify the Interspace
- Palpate Tuffier's line → L4–L5 interspace
- Preferred interspace in adults: L3–L4 or L4–L5 (below the conus)
- Identify midline using spinous process tips
Step 5: Local Infiltration
- Raise a skin wheal with 1–2 mL of 1–2% lidocaine using a 25G needle
- Infiltrate subcutaneous tissues and deeper interspinous ligament
Step 6: Needle Selection — Critical Choice
| Needle Type | Gauge | Tip Design | PDPH Risk |
|---|---|---|---|
| Quincke (cutting) | 22–25G | Beveled, cutting | Higher (dural fibres cut) |
| Whitacre (pencil-point) | 25–27G | Side-hole, pencil tip | Lowest |
| Sprotte | 24–27G | Side-hole, pencil tip | Lowest |
| Greene | 26–27G | Modified bevel | Low |
Rule: Always use a 25G or smaller pencil-point needle (Whitacre or Sprotte) in ambulatory patients and anyone at risk for PDPH (young women, obstetric patients).
For the standard adult single-shot spinal, a 25G Whitacre is the workhorse.
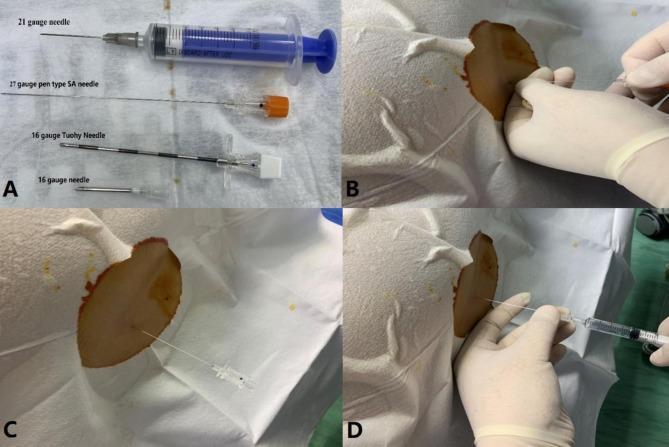
Standard spinal anesthesia equipment and technique — needle comparison, insertion, CSF confirmation, and injection (Miller's Anesthesia 10e)
Step 7: Needle Insertion — Midline Approach
- Insert the introducer (22G) first, anchored firmly in the interspinous ligament
- Insert the spinal needle through the introducer, bevel/opening oriented longitudinally (parallel to dural fibres) — this separates rather than cuts the longitudinal fibres, reducing PDPH
- Advance with slow, steady pressure — you will feel two distinct "pops":
- First pop: Through the ligamentum flavum
- Second pop: Through the dura-arachnoid (entry into subarachnoid space)
- Remove the stylet and wait for free flow of clear CSF — this confirms subarachnoid placement
📌 If no CSF appears: Rotate needle 90°; if still absent, suspect bevel against dura → slightly advance or withdraw. Never inject without confirming CSF.
Paramedian (Lateral) Approach
- Useful in elderly patients with calcified interspinous ligaments, kyphoscoliosis
- Insert 1 cm lateral and 1 cm caudal to spinous process tip, angle medially and cephalad
- Avoids supraspinous and interspinous ligaments
- Goes directly to ligamentum flavum and dura
Step 8: Drug Injection
- Attach pre-drawn syringe (or use the barbed/Luer-lock connection)
- Aspirate 0.2–0.5 mL CSF to confirm position ("barbotage" can help mixing but is not mandatory)
- Inject slowly at 0.2 mL/sec — too rapid → turbulent spread, unpredictable block height
- Re-aspirate at end to confirm continued intrathecal position
Step 9: Positioning After Injection
- Position the patient immediately for the desired block level (within 5–10 min before drug fixes to tissue)
- Hyperbaric solutions: dependent spread → patient positioning controls level
- Isobaric solutions: less gravity-dependent spread, more predictable height based on dose
Drugs and Doses for Spinal Anesthesia
| Drug | Concentration | Baricity | Typical Dose | Duration |
|---|---|---|---|---|
| Bupivacaine heavy (0.5%) | 0.5% in 8% dextrose | Hyperbaric | 10–15 mg (2–3 mL) | 2–4 hours |
| Bupivacaine plain | 0.5% | Isobaric | 10–15 mg | 2–4 hours |
| Lidocaine 5% heavy | 5% in 7.5% dextrose | Hyperbaric | 60–100 mg | 60–90 min (TNS risk!) |
| Ropivacaine | 0.5–1% | Slightly hypobaric | 15–25 mg | 2–4 hours |
| Tetracaine | 0.5% in 5% dextrose | Hyperbaric | 8–14 mg | 2–4 hours |
Intrathecal Adjuvants:
| Adjuvant | Dose | Effect |
|---|---|---|
| Fentanyl | 20–25 µg | Rapid onset, prolongs analgesia, ↓ LA dose |
| Sufentanil | 5–10 µg | More potent, faster onset |
| Morphine | 100–300 µg | Long duration (12–24 h), respiratory depression risk |
| Epinephrine | 100–200 µg | Prolongs duration especially of short-acting LAs |
| Clonidine | 15–45 µg | ↑ Duration, sedation, hypotension |
Factors Controlling Block Height (Miller's Table 41.3)
Most important (controllable):
- Baricity of solution — most reliable method of controlling spread
- Patient position during and after injection
- Dose — higher dose → more cephalad spread
- Site of injection — higher interspace → higher block
Less controllable:
- Age (elderly → smaller doses needed; reduced CSF volume)
- Height (minimal effect clinically)
- Pregnancy (raised intraabdominal pressure → reduced subarachnoid volume)
- Obesity → reduced subarachnoid space
- CSF volume varies: neonates 10 mL/kg → adults 1.5–2 mL/kg
Required Dermatomal Levels for Common Surgeries
| Surgery | Required Level |
|---|---|
| TURP, hip surgery | T10 |
| Lower extremity | L1–L2 |
| Cesarean section | T4 (nipple line — peritoneum is T4) |
| Upper abdominal | T4 |
| Perineal/anorectal | Saddle block (S3–S5) |
| Appendectomy | T6 |
PART III: EPIDURAL ANESTHESIA
How Epidural Differs from Spinal
| Feature | Spinal | Epidural |
|---|---|---|
| Target space | Subarachnoid (CSF) | Epidural space |
| Drug volume | Small (1.5–3 mL) | Large (10–20 mL) |
| Onset | Rapid (3–5 min) | Slow (10–20 min) |
| Block density | Dense, reliable | Variable, incomplete possible |
| Duration | Fixed (single shot) | Continuous via catheter |
| Dermatomal spread | Bulk distribution | Can achieve segmental block |
| Motor block | Usually dense | Titratable (dilute LA = analgesia without motor block) |
| Hypotension | Rapid, profound | Slower onset, more controllable |
| PDPH | Low (25G pencil-point) | Only if dura inadvertently punctured (wet tap ~1%) |
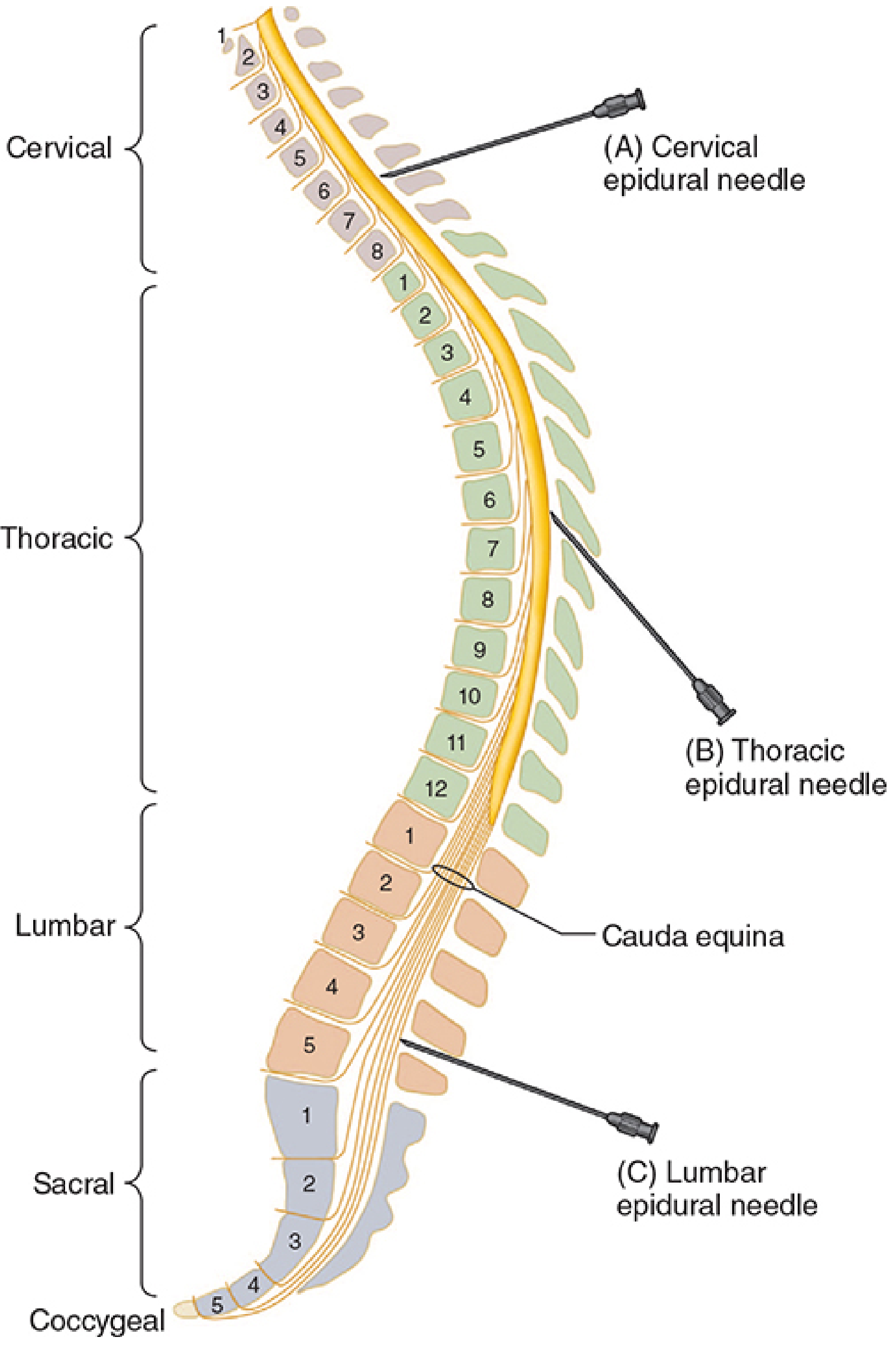
Angulation of the epidural needle at different spinal levels — Morgan & Mikhail's Clinical Anesthesiology 7e
Indications
- Labor analgesia (most common use worldwide)
- Major abdominal, thoracic, pelvic surgery
- Postoperative pain management (catheter technique)
- Chronic pain management
- Thoracic epidural for ICU patients (reduces post-thoracotomy pulmonary complications)
THE PROCEDURE — Step by Step
Step 1: Pre-procedure (Same as Spinal)
IV access, monitoring, resuscitation drugs ready. Informed consent.
Step 2: Positioning
- Lateral decubitus (preferred for catheter placement): Spine flexed maximally (chin to chest, knees to abdomen) → widens the interspinous spaces and pulls the spinal cord anteriorly, expanding the posterior epidural space
- Sitting position: Excellent for obese patients and thoracic epidurals
Step 3: Sterile Preparation
Identical to spinal — full asepsis. Chlorhexidine in alcohol; allow to completely dry. Never let chlorhexidine contact neural tissue.
Step 4: Needle — The Tuohy Needle
The Tuohy needle (16G, 17G, 18G for epidural catheter placement) is the standard:
- Curved Huber tip — deflects the catheter cephalad once the epidural space is entered
- Markings at 1-cm intervals — allow precise depth tracking
- Fitted with a Luer-lock cap for the LOR syringe
Other needles: Crawford (shorter bevel), Weiss (wings for one-handed technique)
Step 5: Identify the Epidural Space — Two Techniques
Both rely on the change in resistance as the needle traverses the ligamentum flavum into the low-pressure epidural space.
Technique A: Loss of Resistance (LOR) — Gold Standard
- Advance the Tuohy needle through skin and into the interspinous ligament (depth ~3–4 cm in average adult)
- Remove the stylet; attach the LOR syringe (glass syringe preferred — low friction, clear visualization) filled with:
- Air (2–3 mL): historically used; risk of pneumocephalus, venous air embolism, patchy block
- Saline (2–3 mL): now preferred — no compressibility artifacts, denser tactile feedback, no gas complications
- Apply constant gentle forward pressure on the plunger while simultaneously advancing the needle millimeter by millimeter
- As long as the needle tip is in the ligamentum flavum → high resistance → plunger springs back
- The moment the needle tip enters the epidural space → sudden loss of resistance → the syringe plunger advances freely with no springback
📌 Tactile teaching: Imagine pressing your thumb on a syringe filled with air — while in the ligament it feels like pressing against a hard rubber ball. When the epidural space is entered, that resistance vanishes completely in a split second. You will feel this the first time you do it correctly and never forget it.
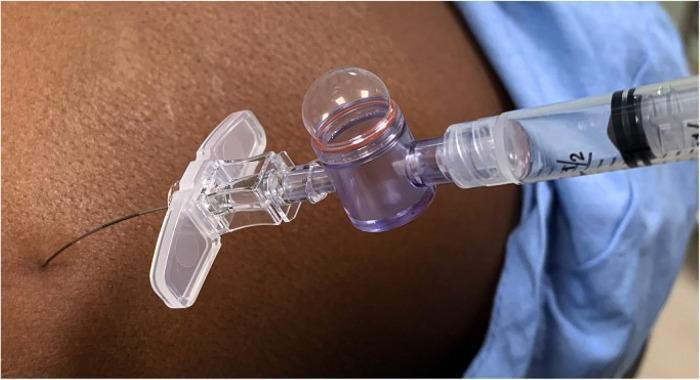
The Epidrum LOR device — inflated dome indicates resistance; collapse of dome signals epidural space entry
Technique B: Hanging Drop (Gutiérrez Sign)
- Fill the hub of the needle with a drop of saline
- As needle advances through ligamentum flavum, the drop remains stationary
- On entering the epidural space (sub-atmospheric pressure) → the drop is "sucked in" by negative pressure
- Less reliable, requires adequate negative pressure (works less well in thoracic region or obese patients)
- Historical use; LOR has largely replaced it
Step 6: Depth Assessment
- Average depth to epidural space in adults: 4–6 cm (midline lumbar)
- Rule of thumb: approximately equal to the distance from the skin to the ligamentum flavum
- Depth > 7–8 cm: high risk of false loss of resistance or intravascular placement
- Note the depth marking on the needle for subsequent catheter placement
Step 7: Catheter Insertion
- Through the Tuohy needle, thread a 20G epidural catheter
- Advance catheter 4–6 cm beyond the needle tip (so 2–4 cm remains in the epidural space)
- Hold the catheter firmly while removing the needle — catheter must not be pulled back through the needle (shearing risk)
- Fix catheter at skin — most institutions use transparent dressings with a strain-relief loop
Step 8: Test Dose — Non-Negotiable
Standard epidural test dose: 3 mL of lidocaine 2% with 1:200,000 epinephrine (15 µg)
The test dose serves to detect:
| What you're testing for | Positive result if... | Interpretation |
|---|---|---|
| Intravascular placement | Heart rate ↑ >20 bpm within 60–90 sec | Catheter is in a blood vessel |
| Intrathecal (dural) puncture | Dense motor block (leg weakness) within 3–5 min | Accidental spinal |
| Subdural placement | Patchy block, higher than expected level | Subdural catheter |
⚠️ Professor's warning: In parturients receiving oxytocin or during uterine contractions, tachycardia may be present at baseline, reducing the sensitivity of the epinephrine test dose. In beta-blocked patients, the HR criterion may be unreliable. Always combine with clinical assessment.
Step 9: Epidural Drug Administration
Fractionated dosing — always give the total dose in divided fractions (e.g., 5 mL aliquots every 5 minutes) while monitoring BP, HR, and sensory level. This allows you to:
- Detect intravascular/intrathecal catheter migration between doses
- Titrate to the desired level
- Avoid catastrophic total spinal or LAST
Standard epidural local anesthetics:
| Drug | Concentration | Onset | Duration | Use |
|---|---|---|---|---|
| Lidocaine 2% | 2% ± epi | Fast (5–10 min) | 60–90 min | Surgical, top-up |
| Bupivacaine 0.5% | 0.5% | Slow (15–20 min) | 2–4 h | Surgical |
| Bupivacaine 0.1–0.25% | Dilute | Slow | — | Labor analgesia (motor sparing) |
| Ropivacaine 0.2% | Dilute | Intermediate | — | Labor analgesia (less motor block than bupi) |
| Levobupivacaine | 0.5–0.75% | Slow | 2–4 h | Less cardiotoxic than racemic bupi |
| Chloroprocaine 3% | 3% | Fastest (3–5 min) | 30–45 min | Emergency CS, rapid top-up |
Epidural opioid adjuvants:
- Fentanyl 50–100 µg (bolus) or 2–4 µg/mL (infusion) — rapid analgesia, reduces LA requirements
- Morphine 2–5 mg — long duration (12–24 h postoperative analgesia), delayed respiratory depression
- Clonidine 75–150 µg — enhances analgesia, useful in opioid-resistant patients
Volume for epidural dosing: approximately 1.5–2 mL per dermatome to be blocked
PART IV: PHYSIOLOGICAL EFFECTS OF NEURAXIAL BLOCKADE
Understanding these is what separates a safe practitioner from a dangerous one.
Cardiovascular
- Sympathetic block → arterial vasodilation (↓ SVR) and venodilation (↓ preload)
- Block of T1–T4 (cardiac accelerator fibers) → bradycardia — can be profound and sudden
- Hypotension is the most common complication — treat with:
- Vasopressors: Phenylephrine (first-line in obstetrics — preserves uteroplacental flow) or ephedrine (mixed α/β — better for bradycardic patients)
- Volume: crystalloid co-load
- Positioning: left uterine displacement (obstetrics), Trendelenburg position
Respiratory
- Up to T4 level: minimal respiratory compromise — diaphragm (C3–C5) is unaffected
- Intercostal muscle paralysis begins at T1 — reduces forced expiration, cough effectiveness
- Total spinal anesthesia → diaphragmatic paralysis → respiratory arrest — manage with IPPV
- Patient subjective feeling of "can't breathe" is almost always from proprioceptive loss (can't feel chest moving), not true hypoventilation
Gastrointestinal
- Sympathetic block → parasympathetic dominance → increased peristalsis, contracted gut (good for surgical exposure)
- Nausea often from hypotension → treat hypotension first
Urinary
- S2–S4 block → detrusor paralysis → urinary retention — catheterize all surgical patients receiving neuraxial anesthesia; monitor ambulatory patients carefully
PART V: COMPLICATIONS — Recognition and Management
Hypotension
- Most common (20–33% of spinals for CS)
- Treat: phenylephrine infusion (100–150 µg/min titrated) now preferred over ephedrine in obstetrics; crystalloid co-load
Post-Dural Puncture Headache (PDPH)
- Occurs after dural puncture → CSF leaks → intracranial hypotension → traction on meningeal vessels and nerves
- Character: bilateral, frontal/occipital, postural (worse upright, better supine), may radiate to neck, tinnitus, diplopia (CN VI palsy)
- Incidence: 0.1–0.5% with 25–27G pencil-point spinal needles; up to 70% with 16G Tuohy (wet tap)
- Treatment:
- Conservative: bed rest, hydration, caffeine, analgesics
- Definitive: Epidural Blood Patch (EBP) — 15–20 mL autologous blood injected epidurally at the puncture level; 85–98% success rate
High/Total Spinal
- Excessive cephalad spread → hypotension, bradycardia, respiratory arrest, loss of consciousness
- Management: Secure airway immediately, CPR if needed, vasopressors, atropine
Local Anesthetic Systemic Toxicity (LAST)
- Most commonly from accidental intravascular injection
- Prodrome: circumoral numbness, metallic taste, tinnitus, agitation
- Severe: seizures, cardiovascular collapse (bupivacaine most cardiotoxic)
- Management: Airway/ABC, Intralipid 20% (1.5 mL/kg bolus), ACLS (avoid vasopressin, calcium channel blockers, lidocaine)
Epidural Hematoma
- Rare but catastrophic — most common with anticoagulated patients
- Presentation: back pain + progressive leg weakness/sensory loss
- Emergency: MRI, neurosurgical decompression within 6–8 hours to prevent permanent paraplegia
- Prevention: follow ASRA guidelines on anticoagulation timing
Transient Neurological Symptoms (TNS)
- Buttock/leg pain after spinal anesthesia (most common with lidocaine 5% hyperbaric)
- Self-limiting (24–72 hours), no permanent deficit
- Management: NSAIDs, reassurance
- Reason lidocaine is largely replaced by bupivacaine or chloroprocaine for ambulatory spinal
Cauda Equina Syndrome
- Permanent motor/sensory deficit of sacral nerve roots
- Associated with continuous spinal catheters and repeated dosing of 5% hyperbaric lidocaine
- Mechanism: pooling of high-concentration local anesthetic around sacral roots
Epidural Abscess
- Back pain + fever + neurological deficit days after epidural
- Diagnosis: MRI (most sensitive)
- Management: urgent surgical drainage ± antibiotics
PART VI: COMBINED SPINAL-EPIDURAL (CSE) TECHNIQUE
The CSE combines the rapid, dense onset of spinal anesthesia with the flexibility and prolonged duration of an epidural catheter.
Needle-Through-Needle Technique
- Place Tuohy needle in epidural space (LOR confirmed)
- Pass a long spinal needle (127 mm Whitacre) through the Tuohy needle until dura is punctured → CSF confirmed
- Inject spinal dose (reduced — e.g., 7.5–10 mg hyperbaric bupivacaine for CS instead of 12.5–15 mg)
- Remove spinal needle; thread epidural catheter
- If spinal block inadequate → extend via epidural catheter
Applications:
- Cesarean section (most common obstetric use — denser faster block than epidural alone)
- Long orthopedic cases (spinal provides initial block; epidural extends it)
- Labor analgesia (CSE provides fastest onset)
PART VII: COMPARISON TABLE — SPINAL vs. EPIDURAL
| Feature | Spinal | Epidural |
|---|---|---|
| Drug volume | 1.5–3 mL | 10–20 mL |
| Onset | 3–5 min | 10–20 min |
| Quality of block | Dense, complete | Variable (titratable) |
| Duration | Fixed (drug-dependent) | Unlimited (catheter) |
| Level of control | Limited (by baricity/dose) | Highly titratable |
| Motor block | Dense | Concentration-dependent |
| PDPH risk | 0.1–0.5% (pencil-point) | ~1% if wet tap |
| Cardiovascular changes | Sudden, significant | Slower, more controllable |
| Segmental block possible? | No | Yes |
| Standard needle | 25–27G Whitacre | 16–18G Tuohy |
| Catheter | No (single shot) | Yes |
| Typical use | CS, TURP, hip/knee, hernia | Labor, major abdominal/thoracic, post-op pain |
PART VIII: PROFESSOR'S KEY EXAMINATION POINTS
🧠 The examiner will ask these — know them precisely:
-
Why inject below L2? Spinal cord ends at L1 — below this is cauda equina, which is more resilient to needle injury than the cord itself
-
Why use pencil-point needles? They separate dural fibres longitudinally rather than cutting them → less CSF leak → less PDPH
-
What confirms subarachnoid placement? Free flow of clear CSF — never inject without this
-
What is the test dose and why? 3 mL lidocaine 2% + 15 µg epinephrine → detects intravascular (tachycardia) and intrathecal (motor block) placement
-
Hyperbaric bupivacaine mechanism? Denser than CSF → sinks dependently → gravity + patient position control spread
-
Why does epidural onset take 10–20 min? Drug must diffuse across dura, traverse epidural fat, and penetrate nerve roots — not in CSF, so no direct bathing of neural tissue
-
Ephedrine vs. phenylephrine for neuraxial hypotension? Phenylephrine = pure α-agonist = vasoconstriction without tachycardia, preserves uteroplacental flow → first-line in obstetrics. Ephedrine = mixed α+β = may cause fetal acidosis if used as primary agent in obstetrics, but preferred if bradycardia accompanies hypotension
-
ASRA anticoagulation guidelines: LMWH: wait 12 h (prophylactic) or 24 h (therapeutic) after last dose before neuraxial; catheter removal also requires these intervals
-
What is Batson's plexus? The valveless epidural venous plexus — explains why intravascular catheter placement is common (veins are thin-walled, multiple, engorged in pregnancy), and also why metastatic cancer can spread via this route
-
What is cauda equina syndrome? Permanent sacral nerve root damage from pooling of concentrated local anesthetic — avoid continuous spinal with lidocaine 5%
Textbook Sources:
- Miller's Anesthesia, 10e, Chapters 41 & 45
- Morgan & Mikhail's Clinical Anesthesiology, 7e, Chapter 45
- Barash's Clinical Anesthesia, 9e, Chapter 10
- Sabiston's Textbook of Surgery, Chapter 20
This covers the full scope of what you need to understand, perform safely, and discuss at the highest level in any exam or clinical setting. The anatomy is the foundation; the physiology explains your complications; the technique steps are your safety protocol; and the comparison between spinal and epidural guides your clinical decision-making for every patient.
Retry
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Searching Images
spinal anesthesia subarachnoid space anatomy layers needle technique
Reading File
Loading Image
Loading Image
Reading File
Searching Images
epidural space anatomy cross section ligamentum flavum dura mater layers
Searching Images
post dural puncture headache epidural blood patch complication spinal
Loading Image
Loading Image
Loading Image
I now have everything needed for a comprehensive, richly illustrated professor-level lecture. Let me deliver it now.
Neuraxial Anesthesia: Spinal & Epidural — A Complete Lecture
Your professor. Your textbooks: Miller's Anesthesia 10e · Morgan & Mikhail's Clinical Anesthesiology 7e · Barash's Clinical Anesthesia 9e · Sabiston's Textbook of Surgery
LECTURE 1: ANATOMY — The Foundation of Everything
You cannot perform neuraxial anesthesia safely without owning the anatomy. This is not optional background — it is your roadmap every single time you pick up a needle.
Surface Landmarks
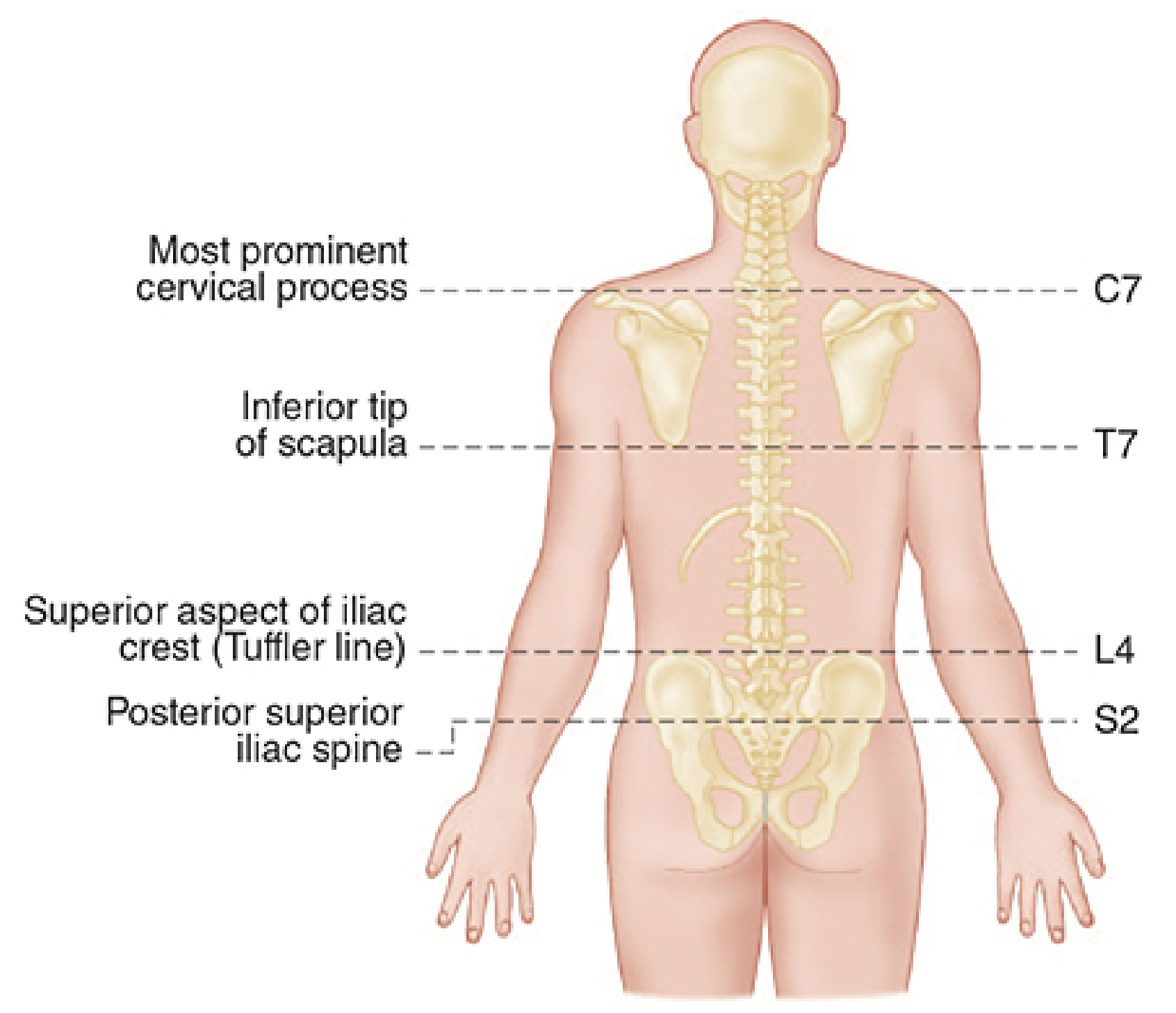
Fig. 1 — Surface landmarks for spinal level identification (Morgan & Mikhail, Fig. 45-13)
Memorise these permanently:
| Landmark | Spinal Level |
|---|---|
| Most prominent cervical process (vertebra prominens) | C7 |
| Inferior tip of scapula | T7 |
| Tuffier's line (superior aspect of both iliac crests) | L4 spinous process / L4–L5 interspace |
| Posterior superior iliac spines | S2 — marks the end of the dural sac |
| Nipple line | T4 — minimum sensory level for Cesarean section |
| Umbilicus | T10 — level for hip, TURP, obstetric procedures |
| Inguinal ligament | L1 |
📌 Professor's pearl: Tuffier's line crosses L4, not L5 as many students say. It is the spinous process of L4, so the interspace immediately below it is L4–L5, and the interspace above is L3–L4. Your safest working interspaces in adults are L3–L4 and L4–L5 — both are below the conus medullaris.
Layers Traversed — Posterior to Anterior
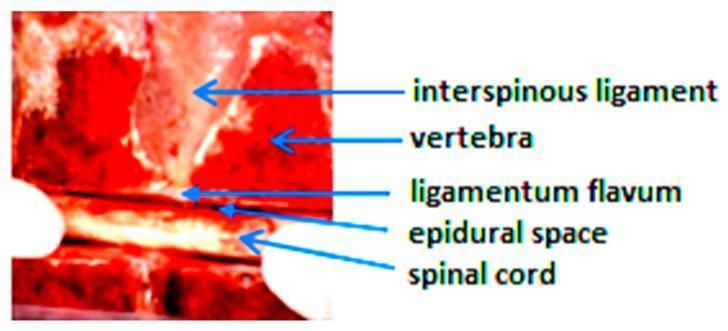
Fig. 2 — Sagittal cross-section specimen showing the sequential anatomical layers for neuraxial access
A needle placed at L3–L4 (midline approach) traverses in order:
- Skin → local anesthetic wheal here
- Subcutaneous fat → minimal resistance
- Supraspinous ligament → connects tips of spinous processes; firmer resistance begins
- Interspinous ligament → between adjacent spinous processes; softer, "spongy" feel
- Ligamentum flavum → the most important tactile landmark: dense, rubbery yellow elastic fibers; thickest posteriorly (3–5 mm at lumbar level); produces a distinct, gritty resistance you will feel as "pushing through cartilage"
- Epidural space → low/sub-atmospheric pressure; contains fat, Batson's venous plexus, lymphatics, nerve roots
- Dura mater → tough, fibrous outer meningeal layer
- (Subdural space — potential; clinically hazardous if a catheter enters here)
- Arachnoid mater → delicate, the true barrier to CSF
- Subarachnoid space → contains CSF — the target of spinal anesthesia
- Pia mater → adherent to the cord
The Effect of Patient Flexion — Why We Flex the Spine
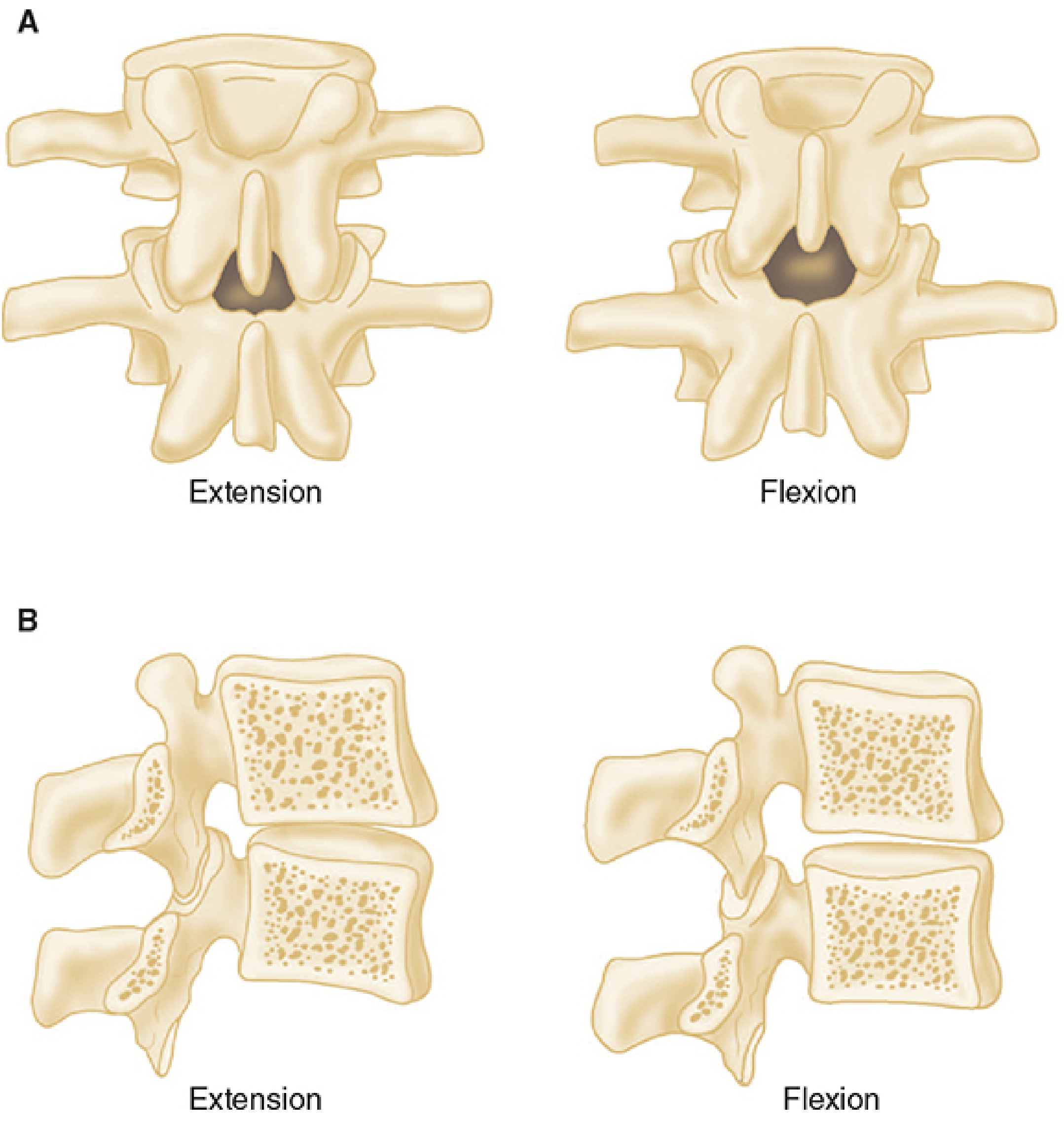
Fig. 3 — Flexion opens the interlaminar window, the target area for neuraxial blocks (Morgan & Mikhail, Fig. 45-15)
This is why you ask your patient to "arch your back like an angry cat" — it opens the interspinous gap from millimeters to centimeters.
LECTURE 2: SPINAL ANESTHESIA (Subarachnoid Block)
What It Is and How It Works
Local anesthetic injected directly into cerebrospinal fluid in the subarachnoid space bathes the nerve roots and spinal cord directly. Because the drug is already in the CNS environment:
- Onset is rapid — 3–5 minutes
- Block is dense — profound sensory and motor blockade
- Only tiny volumes are needed (1.5–4 mL)
- Duration is fixed — one shot, cannot extend it (unless catheter used)
Indications
- Urological: TURP, cystoscopy (awake patient monitors for TUR syndrome/hyponatremia)
- Lower abdominal: hernia repair, appendectomy
- Orthopedic: hip arthroplasty, knee arthroplasty, femur fixation
- Obstetric: Cesarean section (most common use worldwide), instrumental delivery
- Gynecological: hysterectomy, D&C
- Perineal/anorectal: hemorrhoidectomy, pilonidal sinus (saddle block)
- Vascular: lower limb bypass
Contraindications
Absolute:
- Patient refusal
- Infection at the injection site (local sepsis)
- Uncorrected hypovolemia / hemodynamic instability
- Raised intracranial pressure
- Coagulopathy: INR >1.4, platelets <80,000, therapeutic anticoagulation
Relative:
- Fixed cardiac output (severe aortic stenosis, HOCM) — sudden vasodilation may be lethal
- Prior back surgery at the same level
- Pre-existing neurological disease (controversial — document carefully)
- Severe scoliosis or spinal deformity
- Uncooperative patient
STANDARD PROCEDURE — Step by Step
✅ Step 1: Pre-Procedure Setup
Never skip this — it is your safety net:
- IV access secured (18G minimum — you may need to give vasopressors fast)
- Monitoring: SpO₂ continuous, NIBP q2 minutes, ECG, capnography if sedated
- Resuscitation drugs drawn up and labeled:
- Ephedrine 5–10 mg/mL aliquots
- Phenylephrine 50–100 µg/mL aliquots
- Atropine 0.6 mg
- Intralipid 20% available
- Airway equipment immediately accessible
- Fluid co-load: 500–1000 mL crystalloid (Hartmann's or normal saline) running as you start — co-load is now preferred over pre-load as it is more effective
- Discuss with patient: procedure, expected sensations (warmth, heaviness in legs), what to report
✅ Step 2: Patient Positioning
Two equally valid positions:
A. Sitting Position (most common for single-shot spinal)
Patient sits at the edge of the table, feet supported on a stool. Arms folded across a pillow on their lap, or resting on thighs. Head bent forward, chin to chest. An assistant (nurse or technician) stands in front to support the patient and encourage maintained flexion.
Advantages: Easiest to identify midline (even in obese patients), best interspinous opening, gravity keeps hyperbaric solutions in the sacral/lumbar region if you need a saddle block.
B. Lateral Decubitus ("Fetal Position")
Patient lies on their side, knees drawn up to chest, chin tucked to chest. The dependent side is the operative side if using hyperbaric solution (unilateral block).
Advantages: Better tolerated by anxious patients, useful when a pre-sedated or very weak patient cannot sit, preferred by some for catheter placement (epidural/CSE).
✅ Step 3: Sterile Preparation
This is a sterile procedure — treat it as an OR operation:
- Sterile gown, sterile gloves, mask, hat — all mandatory
- Skin preparation: chlorhexidine 0.5–2% in 70% isopropyl alcohol (preferred over povidone-iodine — superior bacterial kill, reduced PDPH risk with iodine contamination of CSF)
- CRITICAL: Allow the solution to completely air dry (30–60 seconds) before proceeding — wet chlorhexidine is neurotoxic
- Apply fenestrated sterile drape
✅ Step 4: Landmark Identification
- Palpate Tuffier's line → finger on L4 spinous process
- Count down to identify L4–L5 space or up to L3–L4 space
- Mark or indent the target interspace with a thumbnail
- Confirm midline by symmetrical palpation of both sides of the spinous processes
✅ Step 5: Local Anesthetic Skin Infiltration
- 25G needle, 1–2 mL of 1–2% lidocaine
- Raise a skin wheal at the marked entry site
- Infiltrate deeper into the interspinous ligament — this significantly reduces procedural pain and patient movement
✅ Step 6: Needle Selection

Fig. 4 — Spinal anesthesia equipment and technique steps: needle selection, insertion, CSF confirmation, injection
| Needle Type | Design | PDPH Risk | When to Use |
|---|---|---|---|
| Whitacre (pencil-point) | 25–27G, side-hole | Lowest | Standard for all patients |
| Sprotte (pencil-point) | 24–27G, large side-hole | Lowest | Standard alternative |
| Quincke (cutting) | 22–25G, beveled tip | Moderate–High | Avoid in young/obstetric patients |
Rule: Always use a 25G Whitacre or Sprotte — the pencil-point tip separates dural fibers longitudinally rather than cutting them, dramatically reducing the risk of post-dural puncture headache (PDPH). An introducer needle (22G, 2 cm) is used to guide the fine spinal needle through the skin and superficial tissues.
✅ Step 7: Needle Insertion — Midline Approach
Fig. 5 — Correct hand position for midline spinal needle insertion
- Insert the introducer (22G, 2 cm) firmly into the interspinous ligament, angled slightly cephalad (5–15°) to follow the downward angulation of the spinous processes
- Pass the spinal needle through the introducer
- Orient the bevel/opening parallel to the long axis of the spine (longitudinal) — this ensures dural fibers are spread, not cut, minimizing CSF leak
- Advance with steady, slow pressure:
- Supraspinous ligament → slight resistance
- Interspinous ligament → firmer; needle feels "anchored like an arrow in a target"
- Ligamentum flavum → distinct gritty, firm resistance
- Pop #1 (subtle): through ligamentum flavum
- Pop #2 (distinct): through dura-arachnoid — entry into subarachnoid space
- Remove the stylet and wait for free-flow of clear CSF — this is your non-negotiable confirmation of correct placement
- If no CSF: rotate needle 90° (bevel may face bone), aspirate gently, advance 1 mm
- Never inject without confirming CSF
✅ Paramedian Approach (When Midline Fails)
Used for elderly patients with calcified interspinous ligaments, kyphoscoliosis, prior lumbar surgery, or thoracic epidurals:
- Entry point: 1–2 cm lateral and 1 cm caudal to the inferior edge of the superior spinous process
- Needle directed medially and cephalad (10–25°) toward the midline
- Bypasses supraspinous and interspinous ligaments entirely — goes directly to ligamentum flavum
- If bone encountered shallowly: redirected upward (hitting lower lamina medially)
- If bone encountered deeply: redirected slightly more craniad toward midline (hitting lower lamina laterally)
✅ Step 8: Drug Injection
- Attach the pre-drawn syringe with a Luer-lock connection — prevents accidental dislodgment
- Aspirate 0.2–0.5 mL CSF to reconfirm placement
- Inject at 0.2 mL/second (slow, steady) — rapid injection creates turbulent flow and unpredictable spread
- At completion, re-aspirate a small volume of CSF to confirm continuous intrathecal position
- Remove the needle and the introducer as a unit
✅ Step 9: Positioning After Injection
- Position the patient immediately — the drug is mobile in CSF for 5–10 minutes before it fixes to neural tissue
- Hyperbaric solution + supine: drug gravitates to the natural lumbar lordosis → mid-lumbar level; tilting table Trendelenburg raises the level
- Hyperbaric + sitting for 3 min: drug pools in sacrum → saddle block
- Isobaric solution: less gravity-dependent, height determined primarily by dose
- Assess sensory level at 5 and 10 minutes using cold (ice or alcohol swab) or pinprick
Drug Doses for Spinal Anesthesia
| Drug | Formulation | Baricity | Dose | Duration |
|---|---|---|---|---|
| Hyperbaric bupivacaine 0.5% | 0.5% in 8% dextrose | Hyperbaric | 10–15 mg (2–3 mL) | 2–4 hours |
| Isobaric bupivacaine 0.5% | 0.5% plain | Isobaric | 10–15 mg | 2–4 hours |
| Hyperbaric lidocaine 5% | 5% in 7.5% dextrose | Hyperbaric | 60–100 mg | 60–90 min |
| Ropivacaine 0.5–1% | Plain | Slightly hypobaric | 15–25 mg | 2–4 hours |
| Tetracaine 0.5% | In 5% dextrose | Hyperbaric | 8–14 mg | 2–4 hours |
⚠️ Lidocaine and TNS: Lidocaine 5% hyperbaric carries a high risk of Transient Neurological Symptoms (burning buttock/leg pain for 24–72h). It has largely been replaced by bupivacaine or chloroprocaine for ambulatory procedures. Never use concentrated lidocaine for continuous spinal — this causes cauda equina syndrome.
Intrathecal Adjuvants
| Drug | Dose | Effect | Duration Added |
|---|---|---|---|
| Fentanyl | 20–25 µg | Rapid analgesia, ↓ LA dose required, ↓ TNS | +1–2 hours |
| Sufentanil | 5–10 µg | More potent than fentanyl | +1–2 hours |
| Morphine | 100–300 µg | Long analgesia (12–24h postop) | 12–24 hours |
| Epinephrine | 100–200 µg | Prolongs duration (especially short-acting LA) | +30–60 min |
| Clonidine | 15–45 µg | Enhanced analgesia, sedation | +1–2 hours |
📌 Pearl on morphine: Hydrophilic → stays in CSF longer → delayed respiratory depression up to 24 hours. Use only when close monitoring is available. Fentanyl is lipophilic → rapid uptake into neural tissue → limited spread, minimal delayed respiratory depression.
Factors Controlling Block Height
Controllable (use these clinically):
| Factor | How to Use |
|---|---|
| Baricity | Most reliable — hyperbaric sinks with gravity, hypobaric rises |
| Patient position | Tilt table or maintain sitting to direct spread |
| Dose/volume | Higher dose → more cephalad spread |
| Injection site | Higher interspace → higher block |
| Speed of injection | Rapid injection → more turbulent, higher spread |
Less controllable (anticipate in these patients):
- Pregnancy/obesity: Raised intraabdominal pressure compresses epidural veins → reduced CSF volume → higher than expected block → use reduced doses
- Elderly: Reduced CSF volume and spinal compliance → more sensitive → reduce dose 20–30%
- Height: Minimal clinical effect compared to baricity
Required Dermatomal Levels
| Procedure | Required Sensory Level |
|---|---|
| Cesarean section | T4 (peritoneum = T4, remember this!) |
| Intra-abdominal surgery | T4–T6 |
| TURP, hip surgery, lower abdominal | T10 |
| Lower extremity | L1–L2 |
| Perineal, anorectal, saddle block | S3–S5 |
LECTURE 3: EPIDURAL ANESTHESIA
How Epidural Differs Conceptually from Spinal
The drug is placed outside the dura, not in CSF. It must:
- Diffuse through the ligamentum flavum
- Traverse the epidural fat
- Penetrate the dura and nerve root sheaths
This explains why epidural anesthesia has:
- Slow onset (10–20 minutes vs 3–5 min for spinal)
- Variable block density (not bathing in CSF)
- Need for large volumes (10–20 mL vs 1.5–4 mL)
- Ability to do segmental blocks — drug confined to the injected level
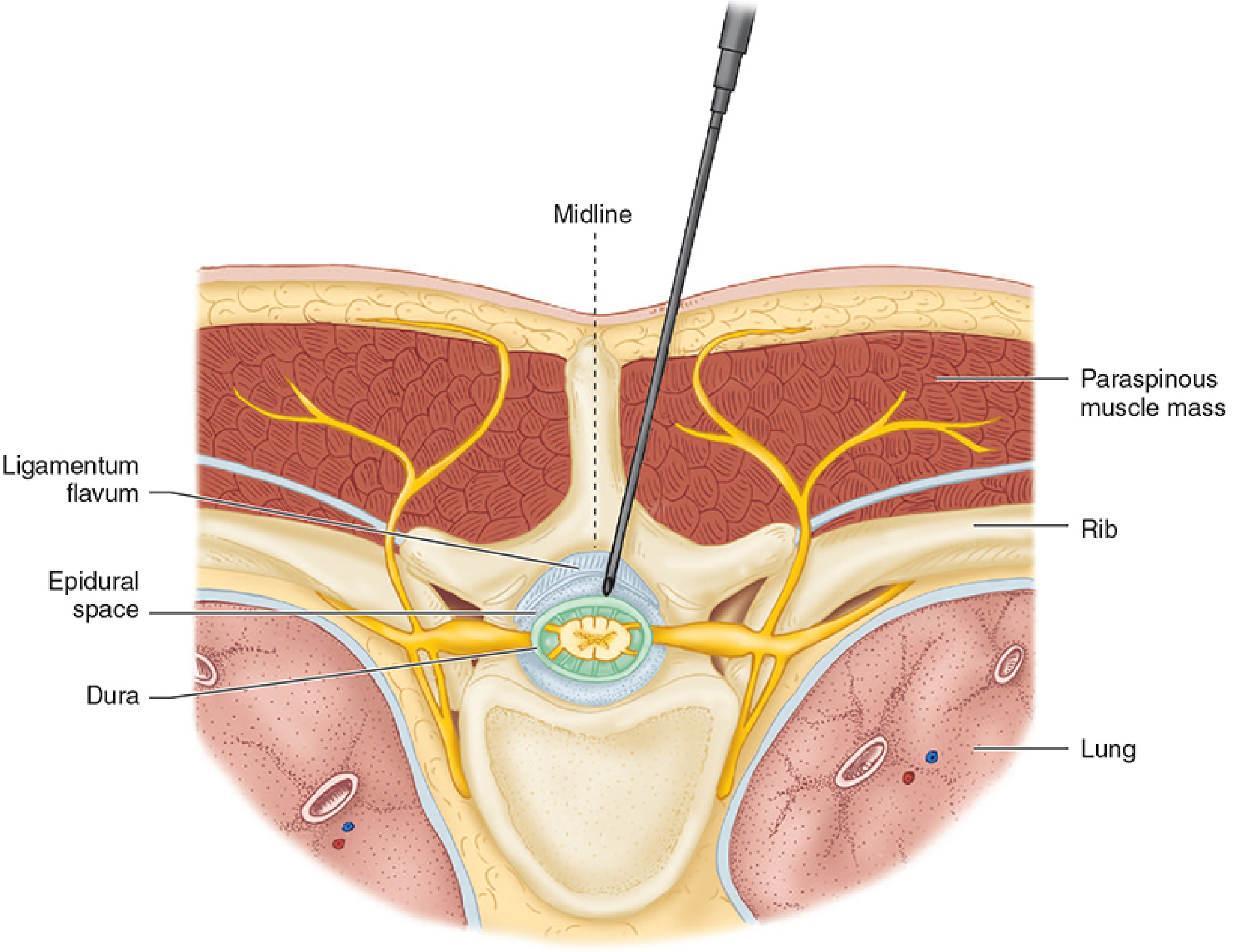
Fig. 6 — Cross-sectional anatomy of thoracic epidural needle placement: ligamentum flavum → epidural space → dura (Morgan & Mikhail, Fig. 45-17)
The Epidural Space — Anatomy Detail
The epidural space extends from the foramen magnum to the sacral hiatus. Contents:
- Batson's venous plexus — valveless, distensible veins (engorged in pregnancy) → most common site of inadvertent intravascular catheter placement
- Epidural fat (filled with fat in children → explains weight-based dosing)
- Lymphatics
- Nerve root sleeves as they exit the foramina
📌 Why do one-sided epidural blocks happen? Fluoroscopic and cadaveric studies have demonstrated fibrous septa and connective tissue bands within the epidural space that can restrict local anesthetic spread to one side. This explains patchy or unilateral blocks and is not a technique failure — it's anatomy.
Indications
- Labor analgesia — the most common use globally
- Major abdominal surgery (provides superior postoperative analgesia vs. parenteral opioids)
- Thoracic epidural for thoracotomy, esophagectomy, major lung resection — reduces postoperative ventilation time and pulmonary complications
- Major pelvic surgery
- Postoperative pain management (catheter-based, multi-day)
- Chronic pain management
STANDARD PROCEDURE — Step by Step
✅ Step 1: Pre-Procedure Setup
Identical to spinal. IV access, monitoring, vasopressors drawn up, Intralipid 20% available, airway equipment at hand.
✅ Step 2: Positioning
Lateral decubitus is preferred for catheter placement — in this position, spine flexion pulls the cord forward and expands the posterior epidural space, making catheter threading easier and reducing the chance of vascular puncture (the cord and its vessels move away from the needle).
Sitting is excellent for thoracic epidurals and obese patients.
✅ Step 3: Sterile Preparation
Full asepsis — chlorhexidine in alcohol, allow to dry completely. Gown, gloves, mask, hat.
✅ Step 4: The Tuohy Needle — Know Your Tool
The Tuohy needle (standard: 16G or 18G) is the epidural workhorse:
- Curved Huber tip — deflects the catheter cephalad once it exits the needle tip into the epidural space
- 1-cm graduation markings on the shaft — track exact depth
- Wings on some (Weiss needle) for one-handed technique
- Supplied with a stylet to prevent tissue coring during insertion
Other needles: Crawford (shorter bevel, more flexible catheter direction), used in some centers.
✅ Step 5: Needle Insertion to Ligamentum Flavum
- After skin wheal and deep infiltration with lidocaine, insert the Tuohy needle (with stylet in place) at the target interspace — L3–L4 or L4–L5 for lumbar; direct slightly cephalad
- Advance through supraspinous ligament, interspinous ligament, until increased resistance of the ligamentum flavum is distinctly felt
- Remove the stylet
- Attach the Loss of Resistance (LOR) syringe
✅ Step 6: Identifying the Epidural Space — Loss of Resistance
This is the heart of the epidural technique. Two media are used:
LOR with Saline (Preferred):
- Glass syringe (low friction, precise feedback) filled with 2–3 mL saline + small air bubble
- The air bubble must compress → tells you the fluid is not compressible when in a closed space
- Apply constant gentle forward pressure on the plunger with thumb while advancing the needle 1–2 mm at a time
- In the ligamentum flavum: high resistance — plunger springs back, the air bubble compresses
- The moment the tip enters the epidural space: sudden, complete loss of resistance — the saline flows freely, the air bubble no longer compresses
- Stop advancing immediately — you are in the epidural space
LOR with Air (Historical, less preferred):
- Same technique with 2–3 mL air
- Risk: pneumocephalus, venous air embolism, patchy block from air pockets
- LOR with saline has replaced this in most centers
📌 Teaching moment: LOR with saline can cause confusion if you cannot tell whether free-flowing fluid is saline from your syringe or CSF from an accidental dural puncture (wet tap). This is why some practitioners leave a small air bubble in the syringe — if you get wet tap, you will see CSF mixed with saline flowing freely and clear.
Hanging Drop Technique (Gutiérrez Sign — Alternative):
- Fill the Tuohy needle hub with a saline drop
- In the ligamentum flavum: drop remains stationary
- On entering epidural space (negative/sub-atmospheric pressure): drop is sucked inward
- Less reliable in obese patients (positive epidural pressure), and during general anesthesia, and at thoracic level
- Largely replaced by LOR
✅ Step 7: Depth of the Epidural Space
- Average depth skin-to-epidural space (lumbar, midline): 4–6 cm in adults
- Greater in obese patients (up to 8 cm)
- Note the depth marking on the needle
- A mnemonic: "4, 5, 6 cm deep in most adults" — if you are beyond 8 cm without LOR, stop and reassess
✅ Step 8: Catheter Insertion and Fixation
- Stabilise the Tuohy needle carefully — any movement at this point risks dural puncture or vascular entry
- Thread the 20G epidural catheter through the needle with a smooth, continuous motion
- Advance the catheter 4–6 cm beyond the needle tip (so 2–4 cm lies within the epidural space — just enough for secure placement without coiling into a vessel or nerve root)
- Hold the catheter firmly while withdrawing the needle — never pull the catheter back through the needle (shearing risk)
- If the patient reports sharp radiating pain or paresthesia down a leg during catheter threading → catheter is contacting a nerve root → withdraw 0.5–1 cm
- Secure catheter at skin with adhesive tape; draw the catheter up the back and over the shoulder; apply transparent dressing with strain-relief loop
✅ Step 9: The Test Dose — Never Skip This
This is your safety filter for two catastrophic misplacements:
Standard test dose: 3 mL of 1.5% lidocaine with 1:200,000 epinephrine (= 15 µg epinephrine)
| What it Tests | Positive Response | Action |
|---|---|---|
| Intravascular placement | Heart rate ↑ ≥20% within 60–90 seconds | Withdraw catheter; resite |
| Intrathecal (dural) puncture | Dense motor block (leg weakness, inability to move legs) within 3–5 min | Convert to spinal dose management; resite |
| Subdural catheter | Patchy, unusually extensive or delayed block | Resite |
⚠️ Limitations of the epinephrine test dose: In parturients, uterine contractions cause natural tachycardia — false positive rate is higher. In patients on beta-blockers, the heart rate criterion is abolished — false negatives occur. Rely also on clinical assessment and incremental dosing, not the test dose alone.
✅ Step 10: Incremental Drug Administration
Never give the full intended epidural dose as a single bolus. Always fractionate:
- Divide total dose into 5 mL aliquots
- Aspirate before each aliquot (even negative aspiration is not 100% reliable — catheters can be partly intravascular)
- Wait 3–5 minutes between aliquots, monitoring blood pressure, heart rate, sensory level
- If signs of intravascular injection at any point (tinnitus, metallic taste, numbness of tongue, agitation, HR rise): stop immediately
- If signs of intrathecal injection (rapidly ascending motor block): manage as high spinal
📌 Morgan & Mikhail's principle: "Each incremental dose is its own test dose." This incremental approach is the single most important safety practice in epidural anesthesia.
Epidural Drug Doses
Volumes and Levels:
- 1 to 2 mL per spinal segment to be blocked (e.g., L4–L5 to T4 = ~10–12 segments = 10–24 mL)
- Shorter patients: 1 mL/segment; taller patients: 2 mL/segment
- Elderly patients: dose requirements decrease with age (reduced epidural space compliance)
| Drug | Concentration | Use | Onset |
|---|---|---|---|
| Lidocaine | 2% ± epinephrine | Surgical, rapid top-up, emergency CS | 5–10 min |
| Bupivacaine | 0.5% | Surgical anesthesia | 15–20 min |
| Bupivacaine | 0.1–0.125% | Labor analgesia (motor sparing) | Slower |
| Ropivacaine | 0.2% | Labor analgesia (less motor block than bupivacaine) | Intermediate |
| Levobupivacaine | 0.5% | Equivalent to bupi, less cardiotoxic | 15–20 min |
| Chloroprocaine | 3% | Fastest onset (3–5 min) — emergency CS, rapid extension of labor epidural | Fastest |
Epidural Opioid Adjuvants:
| Drug | Dose (Bolus) | Infusion | Notes |
|---|---|---|---|
| Fentanyl | 50–100 µg | 2–4 µg/mL | Rapid analgesia, motor-sparing, reduces LA concentration needed |
| Sufentanil | 10–20 µg | 0.5–1 µg/mL | More potent, useful in labor |
| Morphine | 2–5 mg | — | 12–24h postoperative analgesia; monitor for delayed respiratory depression |
LECTURE 4: PHYSIOLOGICAL EFFECTS OF NEURAXIAL BLOCKADE
Every clinical sign you will manage post-block flows from these.
Cardiovascular — The Dominant Effect
Sympathetic nerve fibers (preganglionic: T1–L2) travel in the anterior white rami alongside the somatic nerve roots. Blocking them causes:
- Arteriolar vasodilation → ↓ SVR → ↓ blood pressure
- Venous vasodilation → ↓ venous return → ↓ preload → ↓ cardiac output
- Bradycardia when T1–T4 (cardiac accelerator fibers) are blocked — can be sudden and profound
📌 The Bezold-Jarisch reflex: In high spinal with severe preload reduction, decreased right heart filling activates ventricular mechanoreceptors → paradoxical bradycardia and vasodilation → can cause cardiac arrest even in healthy young patients. Treat aggressively with atropine, ephedrine, or epinephrine.
Management of neuraxial hypotension:
| Drug | Mechanism | When to Use |
|---|---|---|
| Phenylephrine | Pure α₁-agonist (vasoconstriction) | First-line in obstetrics — preserves uteroplacental blood flow, prevents fetal acidosis |
| Ephedrine | Mixed α+β (vasoconstriction + HR increase) | Preferred when bradycardia accompanies hypotension |
| Epinephrine | α+β₁+β₂ | Cardiac arrest, refractory hypotension |
| Crystalloid co-load | Volume expansion | Always run during and after block |
Respiratory
- Diaphragm (C3, C4, C5) is never blocked by lumbar neuraxial anesthesia
- Intercostal muscles (T1–T11) → paralyzed in very high blocks → reduces cough, forced expiration
- Patient subjectively feels they cannot breathe → this is almost always proprioceptive loss (cannot feel chest move), not true hypoxia → reassure and give oxygen
- True respiratory arrest occurs only with total spinal (cervical/brainstem level)
Gastrointestinal
- Sympathetic block (T5–L1) → parasympathetic dominance → increased peristalsis, small contracted bowel
- Excellent surgical conditions for abdominal surgery
- Nausea most commonly from hypotension — treat the hemodynamics first
Urinary
- S2–S4 block → detrusor muscle paralysis → urinary retention
- All patients receiving neuraxial anesthesia for surgical procedures should be catheterized
- For ambulatory patients: must void spontaneously before discharge
LECTURE 5: COMPLICATIONS — Recognition and Management
1. Hypotension (Most Common)
- Incidence: 15–33% for spinal anesthesia; 20–30% for epidural
- Prevention: crystalloid co-load + vasopressor infusion in high-risk patients (obstetric CS)
- Treatment: phenylephrine infusion, ephedrine, IV fluids, Trendelenburg
2. Post-Dural Puncture Headache (PDPH)
Pathophysiology: Dural puncture → CSF leaks through the hole → intracranial hypotension → traction on pain-sensitive intracranial structures (meningeal vessels, cranial nerves)
Clinical features:
- Bilateral, frontal or occipital, may extend to neck
- Postural — the hallmark: worse within 15 minutes of sitting/standing, relieved within 30 minutes of lying flat
- May include: tinnitus, diplopia (CN VI palsy from traction), photophobia, nausea
Incidence:
- 25–27G pencil-point (Whitacre/Sprotte): < 1%
- 25G Quincke cutting: ~2–5%
- 16G Tuohy wet tap: up to 70%
Treatment:
- Conservative: bed rest, adequate hydration, regular analgesics (paracetamol, NSAIDs, caffeine 300–500 mg oral or IV)
- Definitive: Epidural Blood Patch (EBP)
- 15–20 mL autologous blood drawn aseptically, injected epidurally at the puncture site or one level below
- Blood clots within the epidural space, sealing the dural tear
- Success rate: 85–98% after first patch
- Wait 24–48 hours before performing (earlier patches have lower success rates)
- Always obtain informed consent — risks include repeat dural puncture, backache, very rarely neural injury
3. High / Total Spinal
Cause: Excessive cephalad spread of local anesthetic (overdose, accidental intrathecal injection of epidural dose)
Progression: High sensory level → bilateral arm numbness → dyspnea (patient cannot feel breathing) → bradycardia → hypotension → loss of consciousness → apnea → cardiac arrest
Management:
- Call for help immediately
- Secure the airway — rapid sequence intubation
- CPR if cardiac arrest
- Vasopressors (epinephrine, phenylephrine)
- Atropine for bradycardia
- Maintain supine position (avoid Trendelenburg which worsens cephalad spread of remaining drug)
📌 Pearl: Total spinal is almost entirely preventable. Use correct dose, confirm intrathecal placement before injection, use incremental epidural dosing, always use the test dose.
4. Local Anesthetic Systemic Toxicity (LAST)
Cause: Accidental intravascular injection (most common with epidural)
Symptoms (in order of severity):
- CNS prodrome: circumoral numbness, metallic taste, tinnitus, lightheadedness, visual disturbance, agitation
- CNS toxicity: seizures, loss of consciousness
- Cardiovascular toxicity: arrhythmias (bupivacaine most dangerous — binds cardiac sodium channels with high affinity), VT, VF, cardiac arrest
Management:
- Stop injecting immediately
- Airway, 100% oxygen
- Seizures: benzodiazepines (midazolam 2–5 mg IV) or propofol (low dose)
- Intralipid 20% — the antidote:
- Bolus: 1.5 mL/kg over 1 minute
- Infusion: 0.25 mL/kg/min for at least 10 minutes
- "Lipid sink" theory — the lipid emulsion sequesters the hydrophobic local anesthetic
- ACLS for cardiac arrest — prolonged resuscitation may be needed (bupivacaine cardiotoxicity is reversible with lipid rescue)
- Avoid: vasopressin, calcium channel blockers, beta-blockers, high-dose propofol during LAST
5. Epidural Hematoma
Cause: Bleeding into epidural space, most commonly in anticoagulated patients
Risk factors: LMWH, heparin, warfarin, novel anticoagulants, thrombocytopenia, coagulopathy
ASRA Guidelines (4th Edition) — Key Intervals:
| Drug | Wait Before Needle | Wait After Removal of Catheter |
|---|---|---|
| LMWH (prophylactic) | 12 hours | 12 hours |
| LMWH (therapeutic) | 24 hours | 24 hours |
| Unfractionated heparin (SQ prophylactic) | 4–6 hours | 4–6 hours |
| Warfarin | INR ≤ 1.4 | INR ≤ 1.4 |
| Aspirin/NSAIDs | No mandatory wait | No mandatory wait |
Presentation: New-onset back pain + progressive lower extremity weakness/sensory loss + bladder/bowel dysfunction
Emergency: MRI immediately → neurosurgical decompression within 6–8 hours → delays beyond 8 hours have high rates of permanent paraplegia
6. Transient Neurological Symptoms (TNS)
- Burning/aching pain in buttocks, thighs, calves; begins 6–24 hours after spinal recovery
- Most associated with 5% hyperbaric lidocaine (and lithotomy position)
- No neurological deficit — purely sensory, self-limiting (24–72 hours)
- Treatment: NSAIDs, reassurance
- Prevention: avoid lidocaine 5% — use bupivacaine or chloroprocaine instead
7. Cauda Equina Syndrome
- Permanent motor and sensory deficit of sacral nerve roots
- Classic association: continuous spinal catheter + repeated doses of 5% hyperbaric lidocaine
- Drug pools around sacral roots (dependent, immobile in lateral/prone position) → direct neurotoxicity
- Prevention: never use concentrated lidocaine for continuous spinal; use bupivacaine
8. Epidural Abscess
- Back pain + fever + progressive neurological deficit, typically days to weeks after epidural
- Risk factors: prolonged catheter in situ (>96h), immune compromise, diabetes, bacteremia
- Organism: most commonly Staphylococcus aureus
- Diagnosis: MRI with gadolinium (most sensitive)
- Treatment: urgent neurosurgical drainage + IV antibiotics
LECTURE 6: COMBINED SPINAL-EPIDURAL (CSE) TECHNIQUE
The CSE gives you the best of both:
- Rapid, dense onset of spinal anesthesia
- Unlimited duration and flexibility of epidural
Needle-Through-Needle Technique (Standard)
- Identify epidural space with Tuohy needle via LOR — but do not inject anything yet
- Pass a long spinal needle (127 mm, 25–27G Whitacre) through the Tuohy needle until the dura is punctured — CSF flows
- Inject the intrathecal dose (typically reduced — e.g., 7.5–10 mg hyperbaric bupivacaine for CS rather than 12.5–15 mg for spinal alone)
- Remove the spinal needle, thread the epidural catheter
- If the spinal block is inadequate (insufficient height or premature regression): extend via epidural catheter
Applications
- Cesarean section — most common obstetric application
- Labor analgesia (CSE gives faster, more complete onset than epidural alone for the first 15 minutes)
- Long orthopedic procedures (total hip/knee in elderly) — spinal for initial block, epidural for extension and postoperative analgesia
LECTURE 7: MASTER COMPARISON TABLE
| Feature | Spinal | Epidural |
|---|---|---|
| Target space | Subarachnoid (CSF) | Epidural space |
| Drug volume | 1.5–4 mL | 10–20 mL |
| Onset | 3–5 min | 10–20 min |
| Block quality | Dense, reliable | Variable — titratable |
| Duration | Fixed (drug-dependent) | Unlimited with catheter |
| Segmental block | No | Yes |
| Motor block | Usually dense | Concentration-dependent |
| Cardiovascular effect | Rapid, significant | Slower, more controllable |
| PDPH risk | <1% (pencil-point) | Only if wet tap (~0.5–1%) |
| Standard needle | 25G Whitacre | 16–18G Tuohy |
| Catheter | No (usually) | Yes |
| Test dose | Not required (CSF confirms placement) | Mandatory |
| Typical use | CS, TURP, hip/knee arthroplasty | Labor, major abdominal/thoracic, postop pain |
LECTURE 8: EXAMINATION ESSENTIALS — 10 Questions Your Examiner Will Ask
🧠 Know these exactly. These are the questions that separate pass from distinction.
1. Why must you inject below L2 in adults?
The conus medullaris ends at L1. Below L1, the spinal canal contains only the cauda equina (free-floating nerve roots in CSF), which are more resilient to needle contact than the spinal cord itself. Injection at or above L2 risks direct cord trauma.
2. Why use pencil-point spinal needles?
Whitacre and Sprotte needles have a pencil tip with a side hole. They push dural fibers apart longitudinally rather than cutting them as a Quincke bevel does. This creates a smaller, self-sealing defect → dramatically reduced CSF leak → lower PDPH rate.
3. What confirms subarachnoid needle placement?
Free flow of clear CSF. Always confirm before injecting. Rotate the needle if no flow. Never inject without CSF confirmation.
4. What is the standard epidural test dose?
3 mL of 1.5% lidocaine with 1:200,000 epinephrine (15 µg epinephrine). Detects intravascular placement (HR ↑ ≥20%) and intrathecal placement (motor block within 3–5 min).
5. Why does epidural have slower onset than spinal?
Drug is not in CSF. It must diffuse across the dura, through epidural fat, and penetrate nerve root sheaths — multiple diffusion barriers versus direct bathing of nerve roots in CSF.
6. Phenylephrine vs. ephedrine for neuraxial hypotension in obstetrics?
Phenylephrine is first-line. It is a pure α-agonist → vasoconstriction without tachycardia → better uteroplacental perfusion, less fetal acidosis (better umbilical artery pH). Ephedrine (mixed α+β) causes more fetal acidosis as it crosses the placenta and stimulates fetal metabolism. Use ephedrine when bradycardia accompanies hypotension.
7. What is baricity and how do you use it clinically?
Baricity = density of LA solution ÷ density of CSF. Hyperbaric solutions (denser than CSF, e.g., bupi in 8% dextrose) sink dependently with gravity; hypobaric (less dense) rise to non-dependent areas; isobaric solutions spread relatively independent of gravity. You control block height by combining the correct baricity with patient positioning after injection.
8. What is an epidural blood patch and when is it indicated?
15–20 mL autologous blood injected epidurally at the site of dural puncture. Indicated for PDPH not responding to 24–48 hours of conservative treatment. Blood clots and seals the CSF leak. First-line definitive treatment with 85–98% success rate.
9. What are the ASRA interval rules for LMWH?
Prophylactic LMWH: wait 12 hours after last dose before needle/catheter placement and 12 hours after removal. Therapeutic LMWH: wait 24 hours before and 24 hours after. These rules apply to both insertion and removal of epidural catheters.
10. What is the Bezold-Jarisch reflex and why is it dangerous in spinal anesthesia?
Severe preload reduction (venous pooling from sympathectomy) → decreased right heart filling → activates ventricular mechanoreceptors → paradoxical vagal activation → bradycardia + vasodilation despite hypotension. Can cause sudden cardiac arrest in apparently healthy patients under spinal anesthesia. Treat aggressively: atropine 0.6 mg, ephedrine, epinephrine, and volume.
Sources: Morgan & Mikhail's Clinical Anesthesiology 7e (Ch. 45) · Miller's Anesthesia 10e (Ch. 41) · Barash's Clinical Anesthesia 9e · Sabiston's Textbook of Surgery · ASRA Guidelines 4th Edition
This is a shared conversation. Sign in to Orris to start your own chat.