How to differentiate between Post operative changes and Residual tumour in MRI brain ?Can we differentiate it by the MRI brain ?
Reading File
search_medical_text
MRI brain postoperative changes vs residual tumour differentiation
search_medical_text
advanced MRI techniques perfusion diffusion spectroscopy residual glioma post surgery
search_medical_image
postoperative MRI brain residual tumour enhancement versus surgical changes
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive breakdown of how to differentiate postoperative changes from residual tumour on MRI brain.
Differentiating Postoperative Changes vs. Residual Tumour on MRI Brain
Yes — MRI can differentiate the two, but it requires a multiparametric approach combining conventional sequences with advanced techniques. No single sequence is sufficient.
1. The Core Challenge
The critical window is the first 72 hours post-surgery. Beyond this, reactive/inflammatory enhancement appears at the resection margins and overlaps significantly with tumour enhancement — making differentiation increasingly difficult on conventional MRI alone.
Key Rule: The "72-hour window" — an early postoperative MRI (within 24–72 hrs) should be obtained to establish a baseline before reactive enhancement develops.
2. Conventional MRI Features
| Feature | Postoperative Change | Residual Tumour |
|---|---|---|
| Enhancement pattern | Thin, linear, smooth margins along resection cavity | Nodular, thick, irregular enhancement |
| Location | Along dural/surgical margins | Away from cavity margin, within parenchyma |
| T2/FLAIR signal | Diffuse oedema, blood products; expected perilesional change | Mass-like T2 hyperintensity extending beyond expected surgical zone |
| DWI | Restricted diffusion at resection margin (cytotoxic oedema, ischaemia) | Restricted diffusion within a mass lesion (hypercellularity) |
| T1 pre-contrast | Hyperintense rim (methemoglobin from surgical haemorrhage) | Iso/hypointense mass |
| T2* (GRE/SWI) | Haemosiderin, blood products at cavity margins | Tumour blush; may have internal haemorrhage |
| Temporal evolution | Enhancement fades over 4–8 weeks | Enhancement persists or increases |
3. The 72-Hour MRI — Baseline Standard
The early postoperative MRI (within 24–72 hours) is the gold standard first step:
- Captures true residual enhancing tumour before reactive dural/parenchymal enhancement sets in
- Any nodular or thick enhancement at this stage = residual tumour until proven otherwise
- This scan forms the baseline for all subsequent response assessments (RANO criteria)
The image below illustrates this concept — note the nodular contrast enhancement (right, post-contrast) in the resection cavity that is absent on the pre-contrast T1 (left), distinguishing residual high-grade glioma from thin reactive margins:
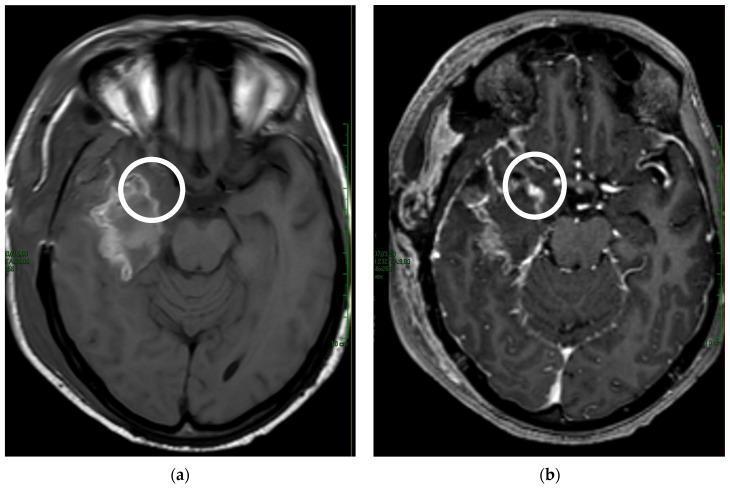
4. Advanced MRI Techniques — When Conventional MRI is Ambiguous
When conventional sequences are inconclusive (especially after the 72-hour window), the following advanced/functional MRI techniques are essential (PET/MRI in Oncology, p. 11):
A. MR Spectroscopy (MRS / MRSI)
| Metabolite | Residual Tumour | Postoperative Change |
|---|---|---|
| Cho (Choline) | Elevated (↑↑) | Normal or mildly elevated |
| NAA (N-Acetylaspartate) | Decreased (↓) | Decreased (non-specific) |
| Cho:NAA ratio | >1.8–2.0 = suspicious | <1.5 = likely benign |
| Cho:Cr ratio | Elevated | Normal/low |
| Lipid/Lactate | May be present | May be present (non-specific) |
B. Perfusion MRI
- DSC (Dynamic Susceptibility Contrast) — rCBV:
- Residual tumour: rCBV elevated (>1.75–2.0 relative to contralateral WM)
- Postoperative/radiation change: rCBV low or normal
- DCE (Dynamic Contrast Enhancement): K-trans elevated in tumour
- ASL (Arterial Spin Labelling): Non-contrast perfusion; tumour shows high CBF
C. Diffusion-Weighted Imaging (DWI) & ADC Map
- Residual tumour: low ADC (restricted diffusion = hypercellularity)
- Postoperative necrosis/oedema: high ADC (facilitated diffusion)
- ADC threshold: Values <1000–1100 × 10⁻⁶ mm²/s are suspicious for tumour
D. Diffusion Tensor Imaging (DTI)
- Helps map white matter tract involvement and identify tumour infiltration along tracts vs. surgical disruption
5. The Problem of Pseudoprogression
A major pitfall in interpretation is pseudoprogression, which mimics residual/progressive tumour:
- Occurs in 21–44% of diffuse intrinsic pontine glioma (DIPG) and 7–12% of high-grade gliomas (pHGG) (Imaging of Paediatric Gliomas, p. 3)
- Appears within the first 3 months after chemoradiotherapy (especially temozolomide + RT)
- Mechanism: increased BBB permeability → transient ↑ in oedema and contrast enhancement
- Key distinction: pseudoprogression is a diagnosis of exclusion — resolves or stabilizes on follow-up without treatment change
| Feature | Pseudoprogression | True Progression |
|---|---|---|
| Timing | <3 months post-CRT | Any time |
| Clinical status | Stable/improving | Worsening |
| rCBV | Low/normal | High |
| MRS Cho | Normal/mildly elevated | Markedly elevated |
| Follow-up MRI | Resolves spontaneously | Increases |
6. PET Imaging as Adjunct
When MRI is still inconclusive:
- FDG-PET: Limited in brain (high background glucose), but hypermetabolism = tumour
- Amino acid PET (11C-MET, 18F-FET, 18F-FDOPA): Superior to FDG for brain tumours — high uptake in viable tumour, low in postoperative/treatment changes
- PET/MRI fusion: Combines metabolic and anatomical data for highest diagnostic accuracy
7. Practical Clinical Algorithm
Post-craniotomy for brain tumour
↓
Early MRI within 24–72 hrs (baseline)
↓
Nodular/thick enhancement?
YES → Residual tumour (high probability)
NO → Likely complete resection
↓
Follow-up MRI at 4–8 weeks (post-CRT)
↓
New or increased enhancement?
↓
Add: DWI/ADC + Perfusion (DSC rCBV) + MRS
↓
rCBV high + Cho↑ + ADC low → RESIDUAL/RECURRENT TUMOUR
rCBV low + Cho normal + ADC high → POSTOPERATIVE/TREATMENT CHANGE
↓
Still ambiguous? → Amino acid PET or biopsy
Summary Table
| Parameter | Residual Tumour | Postoperative Change |
|---|---|---|
| Timing of MRI | Seen even at 24–72 hrs | Develops after 72 hrs |
| Enhancement | Nodular, thick, irregular | Thin, linear, smooth |
| ADC | Low (<1100) | High (>1200) |
| rCBV | High (>1.75) | Low/normal |
| MRS Cho:NAA | >1.8 | <1.5 |
| FET/MET-PET | High uptake | Low uptake |
| Trend over time | Grows/persists | Resolves over weeks |
Bottom line: Conventional MRI alone is insufficient beyond the 72-hour window. A multiparametric approach — early postoperative MRI + DWI + perfusion + spectroscopy, supplemented by amino acid PET when ambiguous — provides the highest diagnostic accuracy for this clinically critical distinction.
This is a shared conversation. Sign in to Orris to start your own chat.