Parkinsons disease
Reading File
search_medical_text
Parkinson's disease etiology pathophysiology clinical features diagnosis
search_medical_text
Parkinson's disease management treatment dopaminergic therapy
search_medical_image
Parkinson's disease pathology Lewy bodies substantia nigra
GradeResponse
GradeResponse
GradeResponse
Parkinson's Disease
Overview
Parkinson's disease (PD) is the second most common neurodegenerative disorder after Alzheimer's disease. It results from progressive loss of dopaminergic neurons in the substantia nigra pars compacta, leading to dopamine depletion in the striatum and the characteristic motor and non-motor features.
Pathophysiology
The hallmark pathological features are:
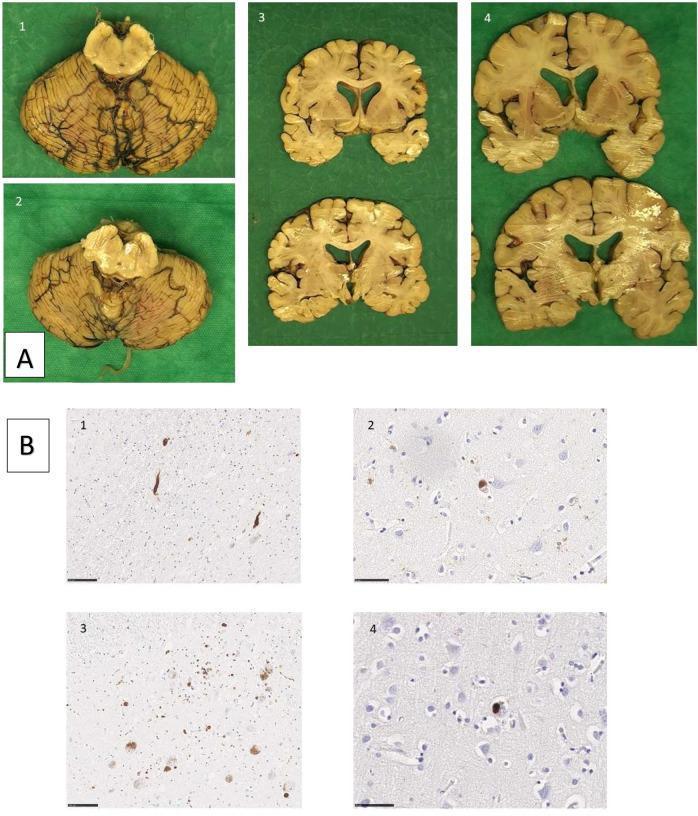
- Loss of dopaminergic neurons in the substantia nigra, causing the macroscopic finding of depigmentation of the midbrain
- Lewy bodies — intraneuronal cytoplasmic inclusions composed of aggregated alpha-synuclein protein
- Lewy neurites — thread-like alpha-synuclein deposits in axonal processes
The Braak staging hypothesis suggests pathology begins in the olfactory bulb and lower brainstem before ascending to the substantia nigra and cortex, which explains the early non-motor symptoms.
Etiology & Risk Factors
| Factor | Details |
|---|---|
| Age | Most important risk factor; incidence rises sharply after age 60 |
| Sex | Men affected ~1.5× more than women |
| Genetics | LRRK2, SNCA, Parkin, PINK1, DJ-1 mutations; ~10–15% familial |
| Environmental | Pesticide/herbicide exposure (rotenone, paraquat), well water |
| Protective | Smoking (controversial), caffeine, physical activity |
Clinical Features
Motor Symptoms (Cardinal Features)
The diagnosis rests on clinical criteria (MDS 2015 criteria):
| Feature | Description |
|---|---|
| Bradykinesia | Slowness and decrement of movement amplitude — required for diagnosis |
| Rest tremor | 4–6 Hz "pill-rolling" tremor, suppressed by movement |
| Rigidity | "Lead-pipe" or "cogwheel" (if tremor superimposed) |
| Postural instability | Late feature; leads to falls |
Classic presentation: TRAP — Tremor, Rigidity, Akinesia/bradykinesia, Postural instability
Non-Motor Symptoms (Often Precede Motor Features)
- Olfactory loss (hyposmia) — one of the earliest signs
- REM sleep behavior disorder (RBD)
- Constipation and autonomic dysfunction
- Depression and anxiety
- Cognitive impairment / Parkinson's disease dementia (late)
- Orthostatic hypotension
Diagnosis
Diagnosis is clinical, based on MDS criteria:
- Parkinsonism must be present (bradykinesia + rest tremor and/or rigidity)
- Absence of absolute exclusion criteria (e.g., cerebellar signs, supranuclear gaze palsy, rapid progression)
- Supportive criteria: unilateral onset, clear levodopa responsiveness, rest tremor, olfactory loss
Imaging: DaTscan (dopamine transporter SPECT) can support diagnosis in ambiguous cases but is not required. MRI is mainly used to exclude structural causes.
Differentials to exclude (Parkinson's Disease, p. 8):
- Essential tremor
- Drug-induced parkinsonism (neuroleptics, metoclopramide)
- Normal pressure hydrocephalus
- Vascular parkinsonism
- Atypical parkinsonian syndromes: PSP, MSA, CBD, DLB
Management
(Parkinson's Disease, p. 3)
Pharmacological
| Drug Class | Examples | Role |
|---|---|---|
| Levodopa/Carbidopa | Sinemet, Rytary | Most effective; first-line, can be used early |
| Dopamine agonists | Pramipexole, ropinirole, rotigotine | Often used in younger patients to delay levodopa |
| MAO-B inhibitors | Selegiline, rasagiline, safinamide | Mild symptomatic benefit; possible neuroprotective debate |
| COMT inhibitors | Entacapone, opicapone | Adjunct to levodopa to reduce "off" time |
| Amantadine | — | Anti-dyskinesia; mild dopaminergic effects |
| Anticholinergics | Trihexyphenidyl | For tremor, especially in younger patients |
Levodopa remains the gold standard. A diagnostic trial of levodopa is useful when diagnosis is uncertain — a robust response supports idiopathic PD.
Device-Based Therapies (Advanced Disease)
- Deep brain stimulation (DBS) — subthalamic nucleus or globus pallidus interna; highly effective for motor fluctuations and dyskinesias
- Levodopa-carbidopa intestinal gel (LCIG/Duopa) — continuous jejunal infusion; reduces "off" time
- Focused ultrasound — thalamotomy for tremor-dominant disease
Non-Pharmacological
- Exercise: proven benefit when started early; neuroprotective effects suspected
- Physiotherapy: gait, balance, fall prevention
- Speech therapy: hypophonia, dysphagia (Lee Silverman Voice Treatment)
- Occupational therapy: ADL maintenance
- Nutritional counseling: protein redistribution to minimize levodopa interactions
Impulse Control Disorders
Dopaminergic therapy — especially dopamine agonists — can cause hypersexuality, pathological gambling, binge eating, and compulsive shopping at any disease stage. Patients should be counseled proactively.
Complications & Prognosis
| Complication | Notes |
|---|---|
| Motor fluctuations | "Wearing off," "on-off" phenomenon — manage with dose adjustments, COMT inhibitors, MAO-B inhibitors |
| Dyskinesias | Peak-dose choreiform movements; amantadine, DBS |
| Dementia | ~80% lifetime risk; associated with Lewy body spread to cortex |
| Dysphagia | Aspiration pneumonia is a leading cause of death |
| Falls | Major source of morbidity; postural instability + freezing of gait |
| Psychosis | Hallucinations; use quetiapine or clozapine (avoid dopamine blockers); pimavanserin is approved |
PD is not directly fatal but significantly reduces quality of life and life expectancy. Median survival from diagnosis is approximately 10–15 years, though this varies widely.
Summary
Parkinson's disease is a progressive alpha-synucleinopathy driven by dopaminergic neurodegeneration. Diagnosis is clinical; levodopa responsiveness is both diagnostic and therapeutic. Management is multimodal — pharmacological, device-based, and rehabilitative. Early exercise and proactive management of non-motor symptoms significantly improve outcomes.
This is a shared conversation. Sign in to Orris to start your own chat.