Internal fertilization in mammals
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of internal fertilization in mammals, drawn from The Developing Human: Clinically Oriented Embryology and Langman's Medical Embryology.
Internal Fertilization in Mammals
Overview
Internal fertilization — the union of sperm and oocyte inside the female reproductive tract — is the defining reproductive strategy of mammals. It allows embryonic development in a protected, nutrient-rich environment and is essential for viviparity (live birth) in most mammalian species.
Site of Fertilization
In humans (and most mammals), fertilization normally occurs in the ampulla of the uterine tube, the widest portion of the tube lying close to the ovary. If the oocyte is not fertilized there, it passes slowly toward the uterus, where it degenerates and is resorbed. Fertilization does not normally occur in the body of the uterus. Chemical attractants (chemoattractants) secreted by the oocyte and surrounding cumulus cells guide capacitated sperm to the egg via sperm chemotaxis.
Sperm Transport and Viability
- Of 200–300 million spermatozoa deposited in the vagina at ejaculation, only 300–500 reach the ampulla.
- Only 1% of sperm deposited in the vagina enter the cervix; cervical crypts provide short-term sperm storage and gradual release.
- Transit from cervix to uterine tube can take as little as 30 minutes or up to 6 days.
- Sperm remain viable in the female genital tract for up to 48 hours (some estimates up to several days).
- The oocyte must be fertilized within 12–24 hours of ovulation before it degenerates.
Pre-Fertilization Requirements
1. Capacitation
Sperm require a period of conditioning in the female tract — capacitation — before they can fertilize. In humans this takes approximately 7 hours. During this process:
- A glycoprotein coat and seminal plasma proteins are removed from the acrosomal region of the plasma membrane.
- Capacitation primarily occurs via epithelial interactions with the mucosal lining of the uterine tube.
- Only capacitated sperm can pass through the corona radiata and undergo the acrosome reaction.
2. Acrosome Reaction
Triggered by binding to the zona pellucida (specifically the glycoprotein ZP3), the acrosome reaction releases hydrolytic enzymes — principally acrosin (a proteolytic enzyme) and trypsin-like substances — that allow penetration of the zona pellucida.
Phases of Fertilization
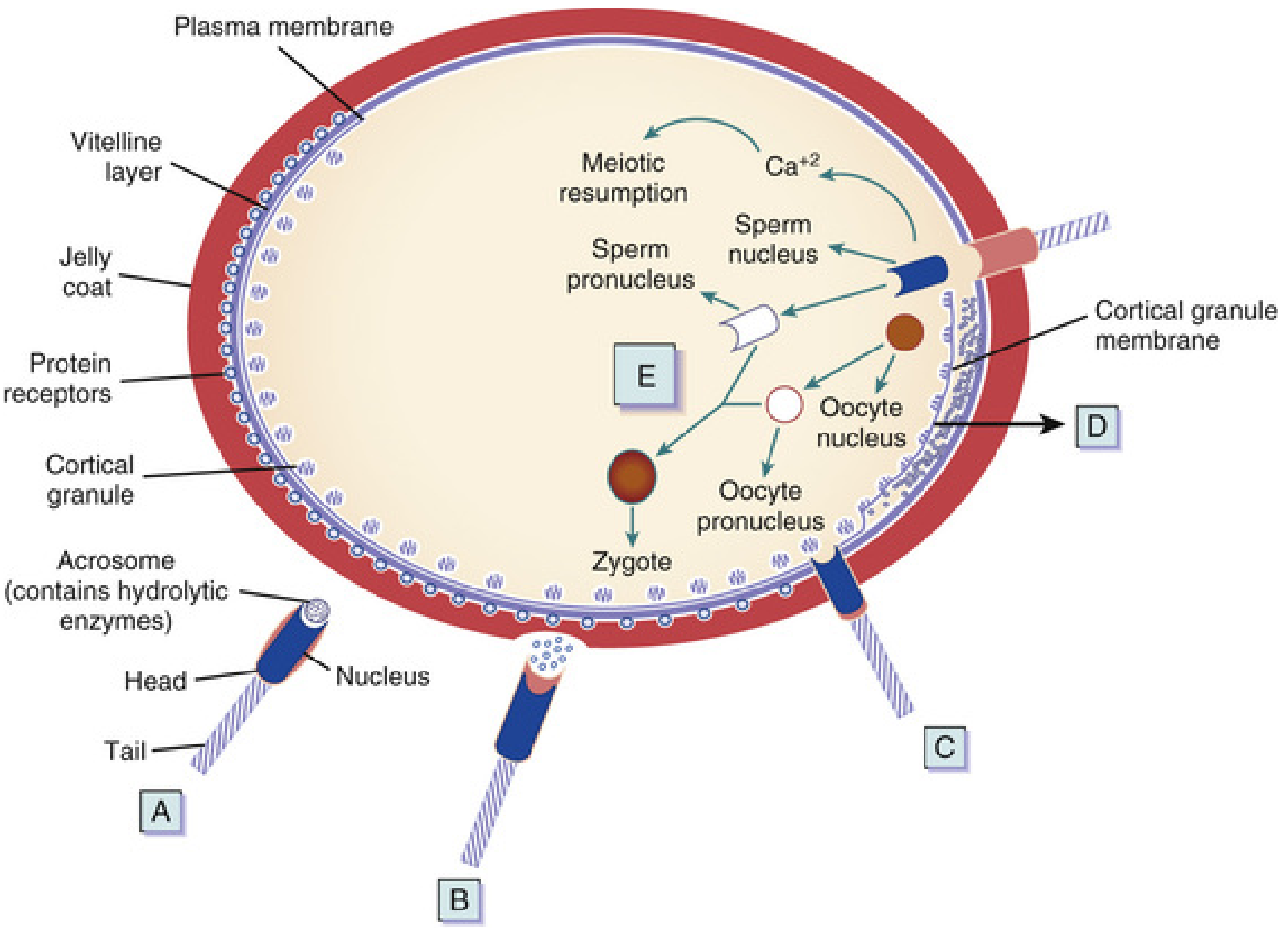
Phase 1 — Penetration of the Corona Radiata
Capacitated sperm pass freely through the follicular cells of the corona radiata. The enzyme hyaluronidase released from the acrosome, combined with tubal mucosal enzymes and the mechanical action of the sperm tail, disperses these cells.
Phase 2 — Penetration of the Zona Pellucida
The zona pellucida is a glycoprotein shell that surrounds the oocyte. Sperm bind to ZP3, which both maintains sperm binding and induces the acrosome reaction. Acrosomal enzymes — esterase, acrosin, and neuraminidase — lyse a path through the zona.
Once a single sperm penetrates, the zona reaction occurs: lysosomal enzymes released from cortical granules lining the oocyte plasma membrane alter the zona's structure, making it impermeable to additional sperm. This is the primary block to polyspermy.
Phase 3 — Fusion of Oocyte and Sperm Cell Membranes
- Initial adhesion is mediated by integrins on the oocyte and disintegrins on the sperm.
- Sperm surface proteins — IZUMO, ADAMs 1/2/3, CRISP1 — bind to oocyte receptors (Juno, integrins, CD9, CD81).
- The plasma membranes fuse (at the posterior region of the sperm head, since the acrosomal membrane was lost).
- The sperm head and tail enter the oocyte cytoplasm; the plasma membrane of the sperm remains on the oocyte surface.
Oocyte Responses After Sperm Entry
The oocyte responds in three coordinated ways:
-
Cortical and zona reactions — Lysosomal enzymes from cortical granules are released into the perivitelline space → the oocyte membrane becomes impenetrable to other sperm, and ZP2/ZP3 receptors are inactivated. This prevents polyspermy.
-
Resumption of the second meiotic division — The oocyte (arrested in metaphase II) completes meiosis II → produces the mature oocyte plus the second polar body. The oocyte nucleus becomes the female pronucleus (22 + X chromosomes).
-
Metabolic activation of the egg — The activating factor (likely phospholipase C-zeta from the sperm) triggers a Ca²⁺ wave that reactivates cell cycling and initiates the molecular events of early embryogenesis.
Pronucleus Formation and Syngamy
- The sperm nucleus decondenses and enlarges to form the male pronucleus; the tail detaches and degenerates.
- Male and female pronuclei are morphologically indistinguishable.
- Both pronuclei replicate their DNA (each haploid → 1n, 2c).
- The pronuclei come into close contact, lose their nuclear envelopes, and chromosomes organize on the cleavage spindle.
- This fusion event — syngamy — produces the zygote, restoring the diploid number (2n = 46 in humans).
- The oocyte containing two pronuclei just before this fusion is technically called an ootid.
Biological Results of Fertilization
| Outcome | Mechanism |
|---|---|
| Restoration of diploidy (46 chromosomes) | Fusion of two haploid gametes |
| Determination of chromosomal sex | X-bearing sperm → XX (female); Y-bearing sperm → XY (male) |
| Initiation of cleavage | Metabolic activation by sperm-derived factors |
| Genetic variation | Recombination + random assortment of parental chromosomes |
| Completion of meiosis II | Sperm penetration lifts meiotic arrest |
Key Molecular Players
| Molecule | Role |
|---|---|
| ZP3 | Sperm receptor on zona; induces acrosome reaction |
| ZP2 | Secondary sperm binding after acrosome reaction; altered post-fertilization |
| Acrosin | Proteolytic enzyme; lyses path through zona pellucida |
| IZUMO (sperm) / Juno (oocyte) | Essential for gamete membrane fusion |
| Phospholipase C-zeta | Sperm-derived; triggers Ca²⁺ oscillations → egg activation |
| Hyaluronidase | Disperses corona radiata cells |
Sources:
- The Developing Human: Clinically Oriented Embryology, 10th ed., pp. 116–128
- Langman's Medical Embryology, 14th ed., pp. 58–61
Internal fertilization in mammals
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
I now have comprehensive, multi-source content. Here is a complete, well-structured answer.
Internal Fertilization in Mammals
Internal fertilization — the union of sperm and oocyte inside the female reproductive tract — is the defining reproductive strategy of viviparous mammals. It protects gametes from the external environment and enables embryonic development in a stable, nutrient-rich setting.
1. Sperm Transport to the Site of Fertilization
Ejaculation deposits 200–600 million spermatozoa around the external os of the uterus and in the vaginal fornix. From there, sperm must reach the uterine tube:
- Sperms pass through the cervical canal by flagellar movement; cervical crypts store some sperm and release them gradually, prolonging the fertilization window.
- Passage through the uterus into the uterine tube occurs mainly by muscular contractions of the uterus and tube walls — not primarily by sperm propulsion alone.
- Prostaglandins in semen stimulate uterine motility at the time of intercourse.
- Sperm move 2–3 mm per minute; motile sperm have been recovered from the ampulla as early as 5 minutes after deposition at the cervix, though transport can take up to 45 minutes.
- Sperm survive up to 48 hours in the female tract; the oocyte must be fertilized within 12–24 hours of ovulation.
- Of 200–300 million spermatozoa deposited, only 300–500 reach the site of fertilization. Only one fertilizes the egg — the rest likely assist in penetrating barriers surrounding the oocyte.
The normal site of fertilization is the ampulla of the uterine tube (the widest portion, closest to the ovary). Chemoattractants secreted by the oocyte and cumulus cells guide capacitated sperm to the egg via sperm chemotaxis.
2. Pre-Fertilization Conditioning
Capacitation
Freshly ejaculated sperm cannot fertilize an oocyte immediately — they must undergo capacitation, a ~7-hour period of biochemical conditioning in the female genital tract:
- A glycoprotein coat and seminal plasma proteins are removed from the sperm acrosomal membrane.
- Cholesterol (the major inhibitor of capacitation) is stripped from the plasma membrane; phospholipid redistribution follows.
- Adenylyl cyclase activity increases → rising intracellular cAMP levels.
- Tyrosine phosphorylation (via Src kinase family) increases — used clinically as a biochemical marker of capacitation.
- Ca²⁺ channel activation → hyperpolarization → acquisition of hyperactivated motility (vigorous, whiplash-like flagellar beating driven by CatSper Ca²⁺ channels in the sperm tail).
- Capacitated sperm show no visible morphologic change but are far more active.
Acrosome Reaction
Triggered by binding to ZP3 (a zona pellucida glycoprotein), the acrosome reaction involves:
- Fusion of the sperm plasma membrane with the outer acrosomal membrane → formation of perforations (apertures).
- Release of acrosomal enzymes: hyaluronidase, acrosin (a serine protease), esterase, and neuraminidase.
- This is regulated by tyrosine kinase (Src kinase), calcium ions, prostaglandins, and progesterone.
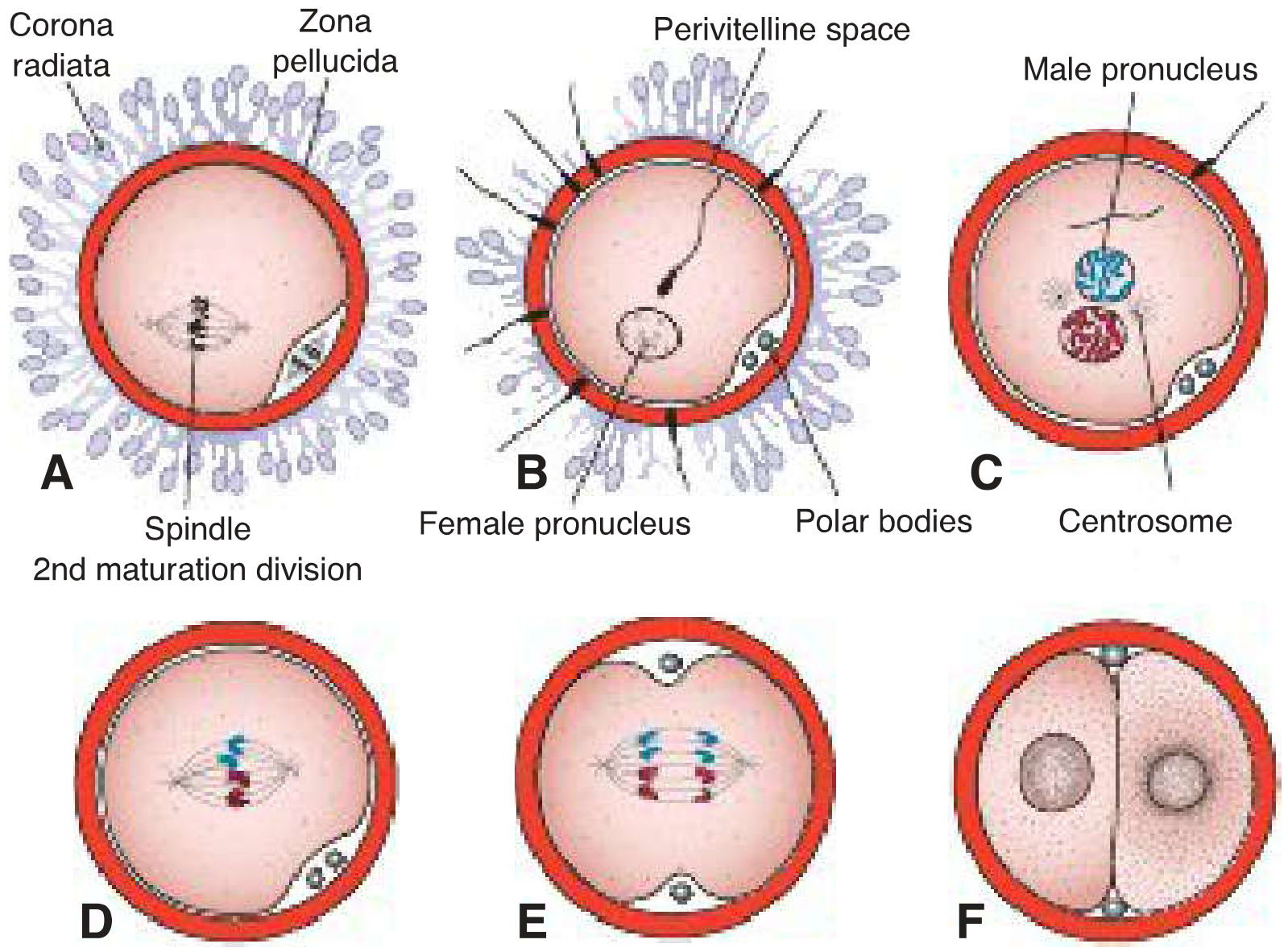
3. The Three Phases of Fertilization
Phase 1 — Penetration of the Corona Radiata
The corona radiata is the outer layer of follicular cells surrounding the oocyte. Capacitated sperm pass through it freely, aided by:
- Hyaluronidase released from the acrosome.
- Tubal mucosal enzymes.
- Mechanical action of the sperm tail.
Phase 2 — Penetration of the Zona Pellucida
The zona pellucida is a glycoprotein shell (ZP1, ZP2, ZP3, ZP4) that surrounds the oocyte. Sperm bind to ZP3, which both anchors binding and triggers the acrosome reaction. Acrosomal enzymes — chiefly acrosin — lyse a path through the zona by limited proteolysis. Hyperactivated flagellar beating provides physical force. Once one sperm penetrates:
- Zona reaction: cortical granule enzymes (proteases) cross-link zona proteins and degrade ZP2/ZP3 receptors → zona becomes impermeable to further sperm penetration → permanent block to polyspermy.
Phase 3 — Fusion of Oocyte and Sperm Cell Membranes
- Initial adhesion: integrins on the oocyte bind to disintegrins on the sperm.
- Key fusion proteins: sperm IZUMO1 binds oocyte Juno (essential for membrane fusion); also fertilin (PH-30), ADAMs 1/2/3, CRISP1 interact with oocyte CD9, CD81, and integrins.
- Fusion occurs at the posterior sperm head (the acrosomal membrane was shed during the acrosome reaction).
- The sperm head and tail enter the oocyte cytoplasm; the plasma membrane remains as a surface appendage; sperm mitochondria are excluded (all mitochondria in the embryo are maternal).
- The paternal centrosome is incorporated — it is essential for building the first and subsequent mitotic spindles.
4. Oocyte Responses After Sperm Entry
Three coordinated reactions follow immediately:
i. Block to Polyspermy (two sequential mechanisms)
| Mechanism | Timing | Event |
|---|---|---|
| Fast block | Seconds | Large oolemma depolarization via Ca²⁺-activated Cl⁻ channels — transient electrical barrier |
| Cortical reaction | Minutes | Ca²⁺ wave propagates cortically → cortical granules fuse with oolemma → enzymes released into perivitelline space |
| Zona reaction | Minutes | Granule proteases cross-link zona proteins; ZP2/ZP3 receptors degraded → permanent structural block |
Polyspermy is lethal — it causes severe chromosomal defects and embryonic mortality.
ii. Resumption of the Second Meiotic Division
The secondary oocyte (arrested at metaphase II) completes meiosis II upon sperm entry:
- Produces the mature oocyte + second polar body (expelled into the perivitelline space).
- The oocyte nucleus becomes the female pronucleus (22 + X chromosomes).
- Signaled by phospholipase C-zeta 1 (PLCζ1) from the sperm, which triggers sustained Ca²⁺ oscillations in the oocyte cytoplasm, reactivating cell cycling.
iii. Metabolic Activation
PLCζ1-driven Ca²⁺ oscillations activate the full complement of molecular events needed for embryogenesis.
5. Pronucleus Formation and Syngamy

- The sperm nucleus decondenses and enlarges → male pronucleus (23 paternal chromosomes); the sperm tail degenerates.
- Male and female pronuclei are morphologically indistinguishable.
- Both pronuclei replicate their DNA (each 1n → 1n, 2c).
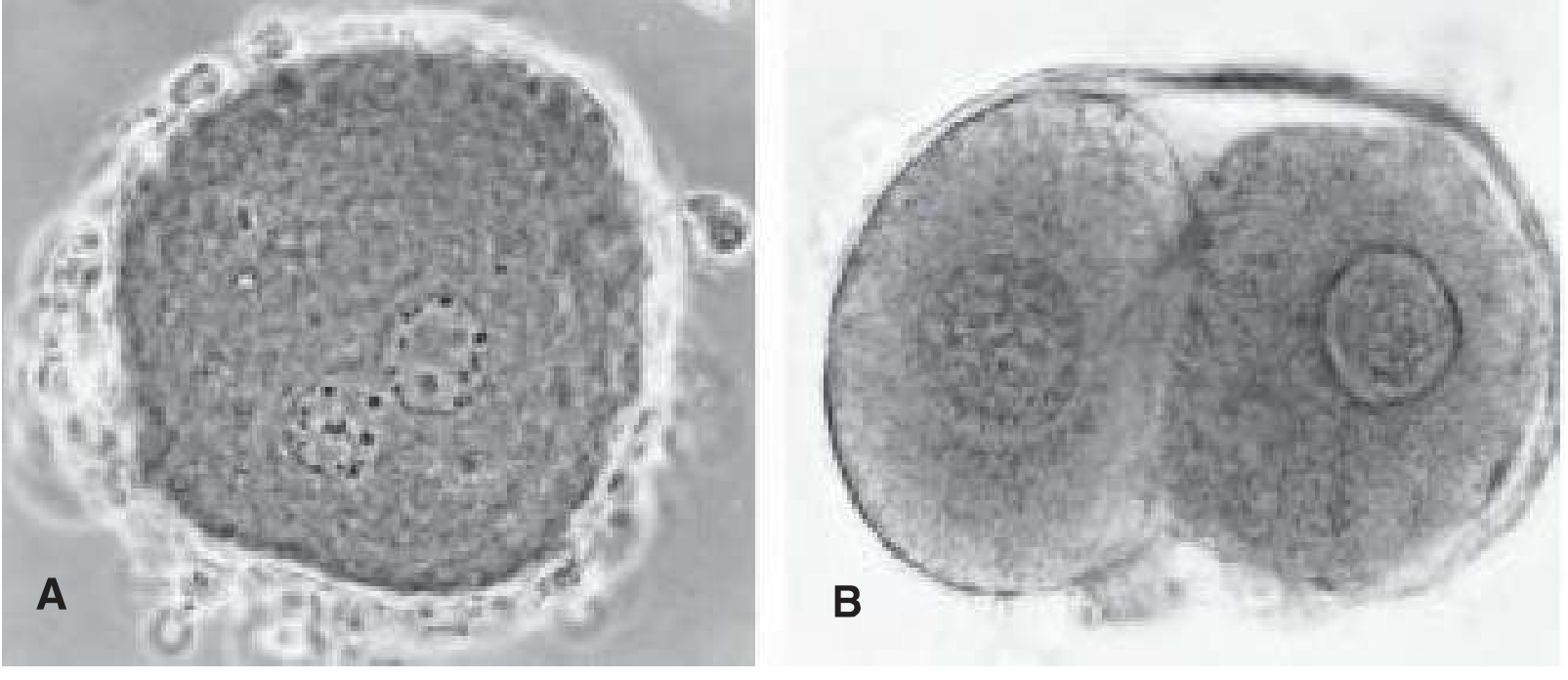
- The oocyte containing two pronuclei at this stage is technically called an ootid.
- Pronuclei migrate toward the center, nuclear envelopes dissolve (without fusing), and chromosomes align on the first mitotic (cleavage) spindle.
- This event — syngamy — produces the zygote: 2n = 46 chromosomes in humans.
6. Outcomes of Fertilization
| Outcome | Mechanism |
|---|---|
| Restoration of diploidy (46 chromosomes) | Fusion of two haploid gametes (23 + 23) |
| Sex determination | Y-bearing sperm → XY male; X-bearing sperm → XX female |
| Initiation of cleavage | Metabolic activation (Ca²⁺ oscillations via PLCζ1) |
| Genetic variation | Meiotic recombination + random chromosome assortment |
| Completion of meiosis II | Sperm entry lifts meiotic metaphase II arrest |
| Maternal mitochondria only | Sperm mitochondria excluded from oocyte |
| Paternal centrosome only | Sperm centrosome used for first mitotic spindle |
7. Key Molecular Players Summary
| Molecule | Location | Function |
|---|---|---|
| ZP3 | Zona pellucida | Primary sperm receptor; triggers acrosome reaction |
| ZP2 | Zona pellucida | Secondary binding; altered post-fertilization to block polyspermy |
| Acrosin | Acrosome | Serine protease; lyses zona pellucida |
| Hyaluronidase | Acrosome | Disperses corona radiata |
| CatSper | Sperm tail | Ca²⁺ channel; drives hyperactivated motility |
| IZUMO1 (sperm) / Juno (oocyte) | Gamete membranes | Essential for gamete membrane fusion |
| Fertilin (PH-30) | Sperm | Binds oolemma before fusion |
| PLCζ1 | Sperm (enters oocyte) | Triggers Ca²⁺ oscillations → egg activation, meiosis II resumption |
| Src kinase | Sperm | Regulates capacitation and acrosome reaction |
Sources:
- The Developing Human: Clinically Oriented Embryology, 10th ed., pp. 111–128
- Langman's Medical Embryology, 14th ed., pp. 58–61
- Histology: A Text and Atlas with Correlated Cell and Molecular Biology, pp. 2221–2226
This is a shared conversation. Sign in to Orris to start your own chat.