I need to know all about multiple myeloma from definition to prevention .
Reading File
Finding Sources
Searching PubMed
"multiple myeloma"[MeSH Terms] AND "therapy"[MeSH Terms]
Searching Images
multiple myeloma plasma cells bone marrow pathology
Reading File
Reading File
Searching PubMed
"multiple myeloma"[MeSH Terms] AND "diagnosis"[MeSH Terms]
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have all the material needed for a complete, well-illustrated answer. Here is a comprehensive guide to multiple myeloma from definition to prevention:
Multiple Myeloma: A Complete Overview
1. Definition
Multiple myeloma is a malignancy of plasma cells characterized by bone marrow infiltration and extensive skeletal destruction, resulting in anemia, bone pain, and fractures. Formally, it is defined by the simultaneous presence of:
- ≥10% clonal plasma cells on bone marrow examination
- M protein in serum or urine (absent in the rare nonsecretory form)
- One or more myeloma-defining events (MDEs)
Patients must be differentiated from those with MGUS (monoclonal gammopathy of undetermined significance) and smoldering multiple myeloma, which are precursor states that do not yet meet MDE criteria.
— Goldman-Cecil Medicine, p. 1977
2. Epidemiology
| Feature | Details |
|---|---|
| Proportion of all malignancies | ~1% |
| Proportion of hematologic malignancies | >10% |
| Annual incidence (US) | ~4 per 100,000; ~30,000 new cases/year |
| Median age at diagnosis | 65–70 years |
| Sex | Slightly more common in males |
| Race | Incidence in Black Americans is nearly twice that in White Americans |
| Rare in young adults | Only 2% are <40 years old |
— Goldman-Cecil Medicine, p. 1977; Robbins & Kumar Basic Pathology, p. 422
3. Etiology & Risk Factors
The precise cause is unknown, but the following factors are implicated:
- Radiation exposure (including prior radiotherapy)
- Chemical exposures: benzene, organic solvents, herbicides, insecticides
- Genetic predisposition: familial clustering in first-degree relatives and identical twins reported
- Precursor state: nearly all cases evolve from a preceding MGUS phase (~1% annual risk of MGUS progressing to myeloma); only a minority have clinically recognized MGUS before diagnosis
— Goldman-Cecil Medicine, p. 1977
4. Pathogenesis & Molecular Biology
4.1 Genetic Events
Multiple myeloma arises through a stepwise accumulation of genetic abnormalities in clonal plasma cells:
| Abnormality | Details |
|---|---|
| IgH translocations (chromosome 14q32) | ~40% of cases; fuse IgH locus to oncogenes (cyclin D1, cyclin D3, FGFR3/MMSET, c-MAF) |
| Hyperdiploid myeloma | ~40% of cases; trisomies without IgH translocation; generally better prognosis |
| RAS mutations | Key secondary event driving MGUS → symptomatic myeloma |
| MYC translocations | Late event; associated with aggressive disease |
| del(17p) | Loss of TP53; high-risk feature |
| del(1p) / gain(1q) | Associated with adverse prognosis; occur with disease progression |
| NF-κB pathway dysregulation | Results from above abnormalities |
4.2 The Bone Marrow Microenvironment
- IL-6 (from marrow stromal fibroblasts and macrophages) is a critical growth factor supporting myeloma cell survival and proliferation
- Angiogenesis is induced, creating a permissive niche
- Abnormal paracrine loops sustain the malignant clone
4.3 Bone Destruction Mechanism
The hallmark lytic bone disease results from a dual hit:
- Osteoclast activation: Myeloma cells upregulate RANKL on stromal cells while simultaneously reducing osteoprotegerin (OPG), causing the RANKL/OPG ratio to rise → osteoclast hyperactivation. Additional drivers: MIP-1α, SDF-α, IL-1β, IL-3, IL-6
- Osteoblast suppression: Mediated by elevated IL-3, IL-7, and DKK1 (dickkopf-1)
- Net result: pure osteolytic disease with hypercalcemia, pathologic fractures, and bone pain
— Goldman-Cecil Medicine, p. 1977–1978
5. Pathology
5.1 Gross Pathology
Multifocal destructive skeletal lesions most commonly involving the:
- Vertebral column, ribs, skull, pelvis, femur, clavicle, scapula
Lesions arise in the medullary cavity, erode cancellous bone, and progressively destroy cortex. They appear as "punched-out" defects 1–4 cm in diameter. Pathologic fractures occur most frequently in the vertebral column and femur.
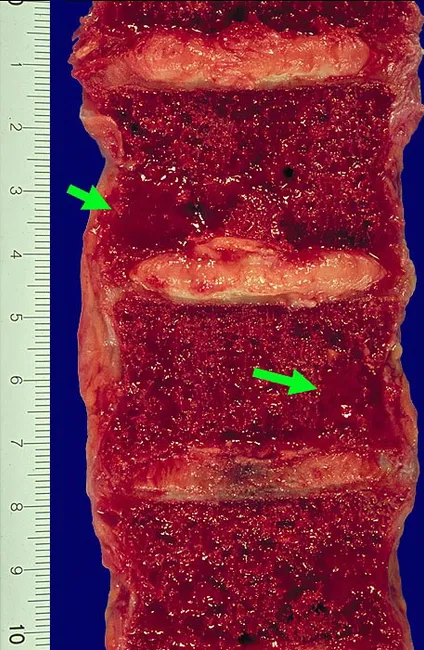
Sagittal section of vertebral bodies showing gelatinous, red-brown plasmacytomas replacing normal marrow with marked cortical thinning.
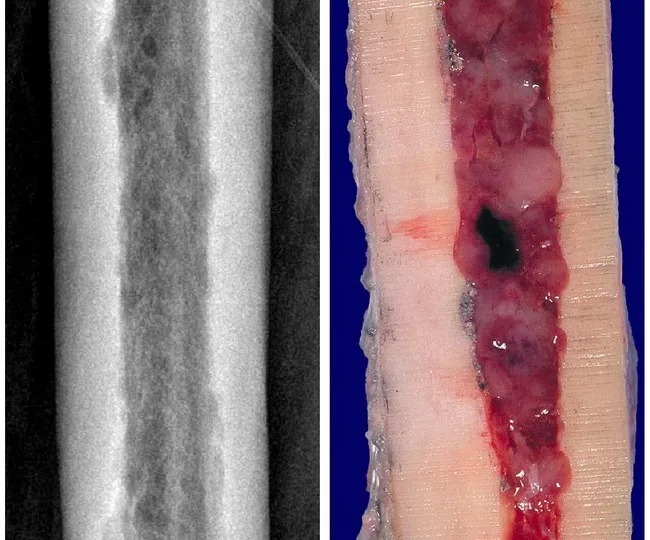
Femur: radiographic osteolysis with endosteal scalloping (left) correlating with gross marrow replacement by plasmacytoma (right).
5.2 Microscopic Pathology
Bone marrow shows >30% plasma cell cellularity. Myeloma cells may resemble normal plasma cells, but more often display:
- Prominent nucleoli
- Cytoplasmic inclusions (Russell bodies) containing immunoglobulin
- Occasional multinucleation
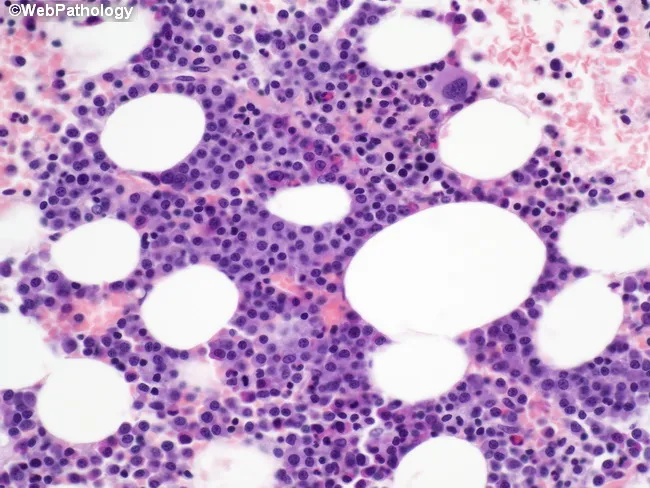
Bone marrow biopsy: islands of malignant plasma cells infiltrating among preserved hematopoietic elements.
5.3 Renal Pathology (Myeloma Kidney)
- Proteinaceous casts of Bence Jones proteins obstruct distal convoluted tubules and collecting ducts
- Macrophage-derived multinucleate giant cells surround casts
- Adjacent tubular epithelium undergoes necrosis/atrophy
- Additional: metastatic calcification, AL amyloidosis in glomeruli/vessel walls, bacterial pyelonephritis
— Robbins & Kumar Basic Pathology, p. 422
6. Immunoglobulin Profile
| M Protein Type | Frequency |
|---|---|
| IgG | ~60% |
| IgA | 20–25% |
| Free light chains only | ~20% |
| IgM, IgD, IgE | Rare |
| Nonsecretory | ~1% |
Free light chains (Bence Jones proteins) are detected in urine. Together, free light chains + M protein are present in 60–70% of cases.
7. Clinical Features
Clinical manifestations flow from four pathophysiologic consequences:
7.1 Skeletal Disease ("CRAB" + Bone-Specific)
- Bone pain — most common presenting symptom (especially back/chest)
- Pathologic fractures — vertebral compression fractures, femoral fractures
- Hypercalcemia → confusion, weakness, lethargy, constipation, polyuria, renal dysfunction
7.2 Renal Insufficiency
- Occurs in up to 50% of patients
- Second leading cause of death (after infections)
- Driven by: Bence Jones cast nephropathy, AL amyloidosis, hypercalcemia/dehydration, pyelonephritis
7.3 Anemia & Cytopenias
- Normocytic, normochromic anemia from marrow replacement
- May include leukopenia and thrombocytopenia
- Fatigue, pallor, and dyspnea
7.4 Immune Suppression
- Paradox: elevated total immunoglobulin (M protein) BUT severely depressed functional antibody production
- Results in recurrent bacterial infections (pneumonia, urinary tract infections) — leading cause of death
- Bacterial pathogens predominate (encapsulated organisms especially)
7.5 Hyperviscosity Syndrome (less common)
- More typical of IgM-producing diseases but can occur with high IgA or IgG3 myeloma
- Headache, visual disturbances, bleeding tendencies
7.6 Neurologic Manifestations
- Spinal cord or nerve root compression from vertebral collapse/plasmacytoma
- Metabolic encephalopathy from hypercalcemia
- Peripheral neuropathy (from amyloid or treatment toxicity)
8. Diagnostic Criteria
8.1 Myeloma-Defining Events (MDEs) — "CRAB + SLiM"
| Criterion | Threshold |
|---|---|
| C — Hypercalcemia | Serum calcium >0.25 mmol/L above upper normal, or >2.75 mmol/L |
| R — Renal insufficiency | Creatinine >177 µmol/L (>2 mg/dL) attributable to myeloma |
| A — Anemia | Hemoglobin >20 g/L below lower normal, or <100 g/L |
| B — Bone lesions | ≥1 osteolytic lesion on skeletal survey, CT, or PET-CT |
| S — Clonal plasma cells ≥60% on bone marrow biopsy | |
| Li — Serum FLC ratio ≥100 | |
| M — >1 focal lesion on MRI |
8.2 Laboratory Workup
- Serum protein electrophoresis (SPEP) — detects M-spike
- Serum immunofixation — identifies immunoglobulin class
- Serum free light chain (FLC) assay — κ/λ ratio
- 24-hour urine protein electrophoresis + immunofixation — Bence Jones proteinuria
- CBC — anemia, cytopenias
- Chemistry panel — creatinine, calcium, LDH, β₂-microglobulin, albumin
- Bone marrow biopsy — confirms ≥10% clonal plasma cells
- Cytogenetics/FISH — risk stratification
- Skeletal imaging — whole-body low-dose CT or PET-CT preferred over plain skeletal survey
9. Precursor States & Spectrum of Disease
| Condition | Plasma Cells | M Protein | MDEs | Management |
|---|---|---|---|---|
| MGUS | <10% | <3 g/dL | None | Observation |
| Smoldering MM | 10–59% | ≥3 g/dL | None | Observation ± clinical trial |
| Multiple Myeloma | ≥10% | Any level | Present | Active treatment |
| Solitary plasmacytoma | Single lesion | May be absent | — | Radiation ± systemic |
MGUS progresses to myeloma at ~1% per year. Risk of progression from MGUS to myeloma or related disorder at 20 years ranges from 5% (low risk) to 58% (all 3 risk factors abnormal).
10. Staging
Revised International Staging System (R-ISS)
| Stage | Criteria | Median OS |
|---|---|---|
| I | β₂-microglobulin <3.5 mg/L + albumin ≥3.5 g/dL + standard cytogenetics + normal LDH | ~Not reached (>5 yr) |
| II | Neither Stage I nor III | ~4–5 years |
| III | β₂-microglobulin ≥5.5 mg/L + high-risk cytogenetics OR elevated LDH | ~2–3 years |
High-risk cytogenetic features: del(17p), t(4;14), t(14;16), del(1p), gain(1q) per 2025 IMS/IMWG consensus guidelines (PMID 40489728).
11. Treatment
Treatment is divided into two major tracks based on transplant eligibility.
11.1 Transplant-Eligible Patients (Younger, Good Performance Status)
Step 1 — Induction (3–4 drug regimen, ~4 cycles):
- VRd: Bortezomib (proteasome inhibitor) + Lenalidomide (immunomodulator) + Dexamethasone — standard backbone
- Daratumumab (Dara) + VRd (Dara-VRd): Preferred quadruplet in fit newly diagnosed patients; superior outcomes shown in recent systematic review (PMID 39348665)
- KRd: Carfilzomib + Lenalidomide + Dexamethasone (alternative)
Step 2 — Stem cell mobilization + Autologous Stem Cell Transplantation (ASCT):
- High-dose melphalan conditioning → reinfusion of patient's own stem cells
- Significantly prolongs remission vs. chemotherapy alone
Step 3 — Consolidation (optional 2–4 cycles of original induction regimen)
Step 4 — Maintenance:
- Lenalidomide 10 mg/day (standard; improves OS post-ASCT)
- Bortezomib maintenance in high-risk disease (added to lenalidomide)
Allogeneic transplantation: Reserved for clinical trials or selected high-risk patients as salvage; limited by high treatment-related mortality.
11.2 Transplant-Ineligible Patients (~50% of newly diagnosed)
Options include:
- VRd × ~9 months → lenalidomide maintenance
- Dara + Rd (DRd) until disease progression
- Ixazomib + Rd (oral regimen for patients unable to access parenteral therapy)
- VCd (Bortezomib/Cyclophosphamide/Dexamethasone): used in severe renal failure or when lenalidomide is unavailable
11.3 Drug Classes and Mechanisms
| Drug Class | Examples | Mechanism |
|---|---|---|
| Proteasome inhibitors (PI) | Bortezomib, Carfilzomib, Ixazomib | Block proteasomal degradation → accumulation of misfolded proteins → apoptosis (myeloma cells are particularly vulnerable due to heavy immunoglobulin synthesis) |
| Immunomodulatory drugs (IMiDs) | Thalidomide, Lenalidomide, Pomalidomide | Bind cereblon → IKZF1/IKZF3 degradation → T-cell and NK-cell activation; anti-angiogenic |
| Anti-CD38 monoclonal antibodies | Daratumumab, Isatuximab | Direct plasma cell killing via ADCC, CDC, and apoptosis |
| Anti-SLAMF7 antibody | Elotuzumab | Activates NK cells against SLAMF7-expressing myeloma cells |
| Alkylating agents | Melphalan, Cyclophosphamide | DNA cross-linking |
| Glucocorticoids | Dexamethasone | Anti-inflammatory and direct myeloma cell apoptosis |
| Belantamab mafodotin | Anti-BCMA ADC | Delivers cytotoxin to BCMA-expressing plasma cells (see PMID 40143674) |
| CAR-T cell therapy | Idecabtagene vicleucel (ide-cel), Ciltacabtagene autoleucel (cilta-cel) | Autologous T cells engineered to target BCMA on myeloma cells |
| Bispecific antibodies | Teclistamab, Talquetamab | T-cell engagers targeting BCMA or GPRC5D |
11.4 Treatment of Relapsed/Refractory Myeloma
Nearly all patients eventually relapse. Principles:
- Select regimens with ≥2 drugs not previously used or not previously refractory to
- Duration of remission shortens with each line
- CAR-T cell therapy and bispecific antibodies are highly active in heavily pretreated patients
- Active clinical trial participation encouraged
11.5 Supportive Care
| Complication | Management |
|---|---|
| Bone disease | Bisphosphonates (zoledronic acid, pamidronate) — reduce skeletal-related events; denosumab (anti-RANKL) |
| Infection prophylaxis | Acyclovir (herpes zoster prophylaxis during PI therapy); PCP prophylaxis; IVIG for recurrent severe infections |
| Hypercalcemia | Hydration, bisphosphonates, corticosteroids, calcitonin |
| Renal failure | Hydration, avoid NSAIDs/contrast, dialysis if needed; some agents require dose adjustment |
| Anemia | Erythropoiesis-stimulating agents, transfusion |
| Venous thromboembolism | Aspirin or anticoagulation during IMiD therapy |
| Pain | Analgesics, radiation to painful bone lesions, vertebroplasty/kyphoplasty |
12. Prognosis
- Untreated, with multiple bony lesions: rarely survive >6–12 months
- Smoldering myeloma: may not require treatment for years
- Median overall survival with modern therapy: approximately 5–7 years; significantly longer in low-risk disease
- High-risk cytogenetics (del17p, t(4;14), t(14;16), gain 1q) confer shorter remission duration and OS
- Minimal Residual Disease (MRD) negativity is emerging as the strongest surrogate for long-term outcomes
- Advances in immunotherapy (CAR-T, bispecifics) offer unprecedented depth of remission in relapsed/refractory settings
13. Prevention
There are currently no proven primary prevention strategies for multiple myeloma, as the exact cause is unknown. However, several measures are relevant:
13.1 Modifiable Risk Factor Reduction
- Limit occupational chemical exposure: benzene, organic solvents, herbicides, pesticides — use appropriate protective equipment
- Avoid unnecessary radiation exposure: medical and occupational
- Maintain healthy weight and lifestyle: obesity has been associated with increased plasma cell dyscrasia risk in epidemiological studies
13.2 Secondary Prevention (MGUS Surveillance)
Since virtually all myeloma evolves from MGUS, early identification and monitoring of MGUS is the closest clinical strategy to true prevention of clinical myeloma:
- Low-risk MGUS (IgG, M protein <1.5 g/dL, normal FLC ratio): repeat labs at 6 months, then every 2–3 years if stable
- Intermediate/high-risk MGUS: more frequent follow-up (every 6–12 months)
- No pharmacologic intervention currently proven to prevent MGUS → myeloma progression; several clinical trials are ongoing
13.3 Smoldering Myeloma — Active Research
- Lenalidomide has shown delayed progression in some trials of smoldering myeloma, but is not yet standard of care outside clinical trials for all patients
- Daratumumab-based treatment in high-risk smoldering myeloma is under active investigation
Summary Table
| Domain | Key Points |
|---|---|
| Definition | Malignant plasma cell neoplasm; ≥10% clonal plasma cells + M protein + MDE |
| Epidemiology | 1% of all cancers; median age 65–70; 2× incidence in Black Americans |
| Pathogenesis | IgH translocations / hyperdiploid chromosomes; IL-6-driven; RANKL/OPG imbalance |
| Symptoms | Bone pain, fractures, anemia, renal failure, recurrent infections, hypercalcemia |
| Diagnosis | SPEP, FLC assay, 24h urine, bone marrow biopsy, FISH, whole-body CT/PET |
| Staging | R-ISS (I–III) using β₂-microglobulin, albumin, LDH, cytogenetics |
| Treatment | PI + IMiD + anti-CD38 ± ASCT; CAR-T and bispecifics in relapsed disease |
| Prognosis | Median OS ~5–7 years; improving with novel immunotherapy |
| Prevention | No proven primary prevention; MGUS surveillance is key; avoid chemical/radiation exposure |
This is a shared conversation. Sign in to Orris to start your own chat.