Non hodgkins lymphoma clinical features , diagnosis and management
Reading File
Finding Sources
Searching PubMed
"non-Hodgkin lymphoma"[MeSH Terms] AND ("diagnosis" OR "management")
Finding Sources
Searching PubMed
non-Hodgkin lymphoma treatment
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
I have enough comprehensive content from the textbooks. Now I'll compile a thorough answer using the Goldman-Cecil Medicine and Katzung's pharmacology content, supplemented by my medical knowledge.
Non-Hodgkin Lymphoma (NHL): Clinical Features, Diagnosis & Management
Definition
Non-Hodgkin lymphomas (NHL) are a heterogeneous group of solid tumors arising from lymphoid tissue (predominantly B cells, but also T cells and NK cells). They are distinguished from Hodgkin lymphoma by the absence of Reed-Sternberg cells and by distinct patterns of spread and behavior.
— Goldman-Cecil Medicine, Ch. 171
Epidemiology & Risk Factors
- ~80,000 new cases/year in the United States; ~20,000 deaths/year
- Account for ~5% of new cancers and ~3% of cancer deaths
- Lifetime risk: 2.4% (men), 1.9% (women)
- Incidence rises sharply with age; higher in Whites than other ethnic groups
- Highest global rates in the USA, Europe, and Australia; lower in Asia
Risk factors:
| Category | Examples |
|---|---|
| Immune deficiency | HIV/AIDS (>100-fold risk), post-transplant immunosuppression (up to 20% of solid organ recipients), congenital immunodeficiencies |
| Infectious agents | EBV (Burkitt lymphoma, post-transplant lymphoma), H. pylori (gastric MALT lymphoma), HTLV-1 (adult T-cell leukemia/lymphoma), HCV, HHV-8 |
| Autoimmune disease | Sjögren syndrome, rheumatoid arthritis, SLE (MALT lymphomas) |
| Environmental | Agricultural chemicals, organic solvents, hair dyes, radiation exposure |
| Genetic | Familial clustering; polymorphisms in TNF, IL-10, lymphotoxin-α; cytogenetic translocations |
Notable genetic translocations:
- t(14;18) — BCL2 overexpression → follicular lymphoma
- t(8;14) — c-MYC activation → Burkitt lymphoma
- t(11;14) — BCL1/cyclin D1 → mantle cell lymphoma
- t(2;5) — ALK fusion → anaplastic large cell lymphoma
WHO Classification (major subtypes)
B-cell NHL (~85% of cases):
- Diffuse large B-cell lymphoma (DLBCL) — most common, ~31%
- Follicular lymphoma — ~22%
- Marginal zone/MALT lymphoma — ~8%
- Mantle cell lymphoma — ~6%
- Small lymphocytic lymphoma/CLL — ~7%
- Burkitt lymphoma — highly aggressive
- Primary mediastinal large B-cell lymphoma
T-cell/NK-cell NHL (~15%):
- Peripheral T-cell lymphoma (PTCL-NOS)
- Anaplastic large cell lymphoma (ALCL)
- Angioimmunoblastic T-cell lymphoma
- Extranodal NK/T-cell lymphoma (nasal type)
- Adult T-cell leukemia/lymphoma (HTLV-1 associated)
Grading by clinical behavior:
| Grade | Examples | Behavior |
|---|---|---|
| Low (indolent) | Follicular, SLL, marginal zone | Slow-growing, often incurable but long survival |
| Intermediate/High (aggressive) | DLBCL, mantle cell | Rapidly progressive; often curable with treatment |
| Very high (highly aggressive) | Burkitt, lymphoblastic | Rapidly fatal if untreated; potentially curable |
Clinical Features
General (nodal) presentation
- Painless lymphadenopathy — most common presenting feature; cervical, axillary, or inguinal nodes; typically firm and rubbery
- NHL spreads in a non-contiguous (unpredictable) pattern, unlike Hodgkin lymphoma which spreads contiguously
- B symptoms (present in ~30%): fever >38°C, drenching night sweats, unexplained weight loss >10% body weight over 6 months
Extranodal involvement (more common in NHL than HL)
- GI tract — most common extranodal site; abdominal pain, bowel obstruction, GI bleeding; MALT lymphomas of stomach
- Bone marrow — pancytopenia (anemia, thrombocytopenia, neutropenia)
- Waldeyer's ring — tonsillar enlargement, dysphagia
- CNS — headache, cranial nerve palsies, meningismus (especially in highly aggressive subtypes or HIV-related NHL)
- Skin — particularly with mycosis fungoides/Sézary syndrome (cutaneous T-cell lymphoma): patches, plaques, tumors, erythroderma
- Mediastinum — superior vena cava (SVC) syndrome, particularly T-cell lymphoblastic lymphoma
- Spleen — splenomegaly with hypersplenism
- Liver — hepatomegaly, occasionally jaundice
Specific subtypes — distinctive presentations
| Subtype | Typical Features |
|---|---|
| Follicular lymphoma | Elderly patients; waxing and waning lymphadenopathy; bone marrow involvement common; advanced stage at presentation |
| DLBCL | Rapidly enlarging nodal or extranodal mass; can arise de novo or transform from follicular ("Richter transformation") |
| Burkitt lymphoma | Jaw mass (endemic, African form); abdominal mass mimicking appendicitis or intussusception (sporadic); spontaneous tumor lysis |
| Mantle cell lymphoma | Splenomegaly, bone marrow/GI polyposis; poor prognosis |
| Primary CNS lymphoma | Cognitive changes, focal neurological deficits; almost exclusively in immunosuppressed patients |
| Mycosis fungoides | Skin plaques/tumors; pruritis; Sézary syndrome with leukemic phase |
| MALT lymphoma | Dyspepsia, H. pylori infection (gastric); indolent course |
Diagnosis
Initial workup
- Excisional lymph node biopsy — gold standard; provides adequate tissue for histology, immunophenotyping, cytogenetics, and molecular studies. Core-needle biopsy acceptable in some cases. Fine-needle aspiration alone is insufficient.
- Histopathology — architecture (follicular vs. diffuse), cell morphology, mitotic index
- Immunophenotyping (flow cytometry / IHC):
- B-cell markers: CD19, CD20, CD22, CD79a
- T-cell markers: CD3, CD4, CD8
- Ki-67 (proliferation index): high in aggressive lymphomas
- BCL2, BCL6, MYC, cyclin D1 (mantle cell), CD10, CD5
- Cytogenetics/FISH — identifies characteristic translocations (e.g., t(14;18), t(8;14), t(11;14))
- Molecular studies — gene rearrangement (clonality), next-generation sequencing
Staging workup (Lugano Classification, 2014)
- PET-CT (FDG-PET) — preferred for staging FDG-avid lymphomas; highly sensitive for nodal and extranodal sites
- CT chest/abdomen/pelvis — for non-FDG-avid subtypes
- Bone marrow biopsy — especially if PET-CT negative for marrow involvement in low-grade NHL
- CBC, comprehensive metabolic panel, LDH, uric acid, β2-microglobulin
- Serum protein electrophoresis
- HIV, HBV, HCV serology (HBV reactivation risk with rituximab)
- Echocardiogram (if anthracycline therapy planned)
- Lumbar puncture — CNS prophylaxis assessment in high-risk DLBCL
Lugano Staging (modified Ann Arbor)
| Stage | Description |
|---|---|
| I | Single lymph node region or single extranodal site (IE) |
| II | Two or more nodal regions on same side of diaphragm |
| III | Nodal regions on both sides of diaphragm |
| IV | Disseminated involvement (liver, bone marrow, lung) |
Suffix E = extranodal contiguous extension; B = B symptoms
Prognostic scores
International Prognostic Index (IPI) — for aggressive NHL (especially DLBCL):
- Age >60
- Serum LDH > normal
- Performance status ≥2
- Stage III or IV
- Extranodal sites >1
Scores 0–1 = low risk (~73% 5-year OS); 4–5 = high risk (~26% 5-year OS)
Follicular Lymphoma International Prognostic Index (FLIPI) — for follicular lymphoma
Management
Principles
Treatment depends on: histologic subtype, disease stage, IPI score, performance status, and patient comorbidities.
Aggressive NHL (DLBCL — most common aggressive subtype)
First-line:
- R-CHOP (Rituximab + Cyclophosphamide, Doxorubicin, Vincristine, Prednisone) — standard of care for most patients
- Rituximab (anti-CD20 monoclonal antibody) added to CHOP significantly improves response rates, disease-free survival, and overall survival vs. CHOP alone (randomized Phase III evidence)
- Typically 6 cycles (4–8 depending on stage/response)
- Limited stage (I–II): R-CHOP × 3–4 cycles ± involved-site radiotherapy (ISRT)
- Advanced stage (III–IV): R-CHOP × 6 cycles
Relapsed/Refractory DLBCL:
- Salvage chemotherapy (R-ICE, R-DHAP) followed by autologous stem cell transplantation (auto-SCT) in eligible patients
- CAR-T cell therapy (CD19-directed):
- Axicabtagene ciloleucel (axi-cel)
- Tisagenlecleucel
- Both approved for patients who have progressed after ≥2 lines of systemic therapy
- Polatuzumab vedotin (anti-CD79b antibody-drug conjugate) + bendamustine + rituximab
- Bispecific antibodies (epcoritamab, glofitamab) — increasingly used in later lines
Recent evidence: A 2024 meta-analysis (PMID 38696731) found CAR-T cell therapy and bispecific antibodies show comparable efficacy in third-line or later large B-cell lymphoma, helping guide sequencing decisions.
Indolent NHL (Follicular lymphoma — most common indolent subtype)
Key principle: Advanced-stage indolent NHL is generally incurable with standard chemotherapy; treatment is palliative. However, PFS and OS are long (often >10 years with modern therapy).
Watch and wait (active surveillance): appropriate for asymptomatic patients with low tumor burden; no evidence that early chemotherapy improves survival
Indications to treat: symptomatic disease, B symptoms, bulky disease, organ compromise, rapid progression, cytopenias
First-line treatment options:
- Bendamustine + Rituximab (BR) — preferred for most patients
- R-CHOP or R-CVP — alternatives
- Rituximab monotherapy — for frail/elderly patients or minimal disease
- Radiotherapy — curative for localized stage I–II disease
Maintenance:
- Rituximab maintenance (every 2 months × 2 years) prolongs PFS after initial therapy
Transformation to DLBCL ("Richter transformation") — occurs in ~3% per year; requires aggressive chemotherapy (R-CHOP) ± auto-SCT
Highly Aggressive NHL (Burkitt lymphoma)
- Intensive, short-duration multi-agent chemotherapy: R-CODOX-M/IVAC, R-hyper-CVAD, or DA-EPOCH-R
- CNS prophylaxis (intrathecal chemotherapy) mandatory
- Tumor lysis syndrome prophylaxis critical (allopurinol/rasburicase, aggressive hydration)
- Cure rates ~80–90% in young patients with modern regimens
2025 Guideline: ERN-EuroBloodNet guidelines for adult Burkitt lymphoma (PMID 39909657) recommend DA-EPOCH-R or CODOX-M/IVAC as preferred regimens.
Mantle Cell Lymphoma
- R-CHOP alternating with R-DHAP → auto-SCT (for younger fit patients)
- Ibrutinib (BTK inhibitor) — highly active in relapsed/refractory disease; now used up front in combination
- Bendamustine-rituximab for older/frail patients
- Rituximab maintenance prolongs remission
T-Cell Lymphomas (PTCL)
- CHOP-based regimens are standard, but outcomes are inferior to B-cell lymphomas
- Brentuximab vedotin + CHP (for ALCL and other CD30+ PTCL)
- Auto-SCT in first remission for eligible high-risk patients
- Novel agents: romidepsin, pralatrexate, belinostat
CNS Lymphoma
- High-dose methotrexate-based regimens (not R-CHOP, which penetrates CNS poorly)
- Consolidation with autologous SCT or whole brain radiotherapy
Supportive Care & Complications
| Issue | Management |
|---|---|
| Tumor lysis syndrome | Allopurinol/rasburicase, IV hydration, electrolyte monitoring |
| Infection prophylaxis | PCP prophylaxis (trimethoprim-sulfamethoxazole), antiviral (acyclovir), antifungal in high-risk |
| HBV reactivation | Screen all patients; prophylactic antiviral if HBsAg+ or anti-HBc+ before rituximab |
| Neutropenia | G-CSF support with myelosuppressive regimens |
| CNS prophylaxis | Intrathecal MTX or high-dose systemic MTX in high-risk DLBCL |
| Fertility preservation | Discuss before chemotherapy in reproductive-age patients |
Prognosis
| Subtype | Prognosis |
|---|---|
| Follicular lymphoma (advanced) | Median survival >10 years; incurable with standard therapy |
| DLBCL | ~60–70% cured with R-CHOP (low IPI); ~30–40% in high IPI |
| Burkitt lymphoma | ~80–90% cure with intensive regimens (young adults) |
| Mantle cell lymphoma | Median OS ~5–7 years; improving with novel agents |
| PTCL (aggressive T-cell) | Poor; median OS ~2–3 years |
| Primary CNS lymphoma | Median OS ~3–4 years |
Sources: Goldman-Cecil Medicine (26th ed.), Ch. 171; Katzung's Basic and Clinical Pharmacology (16th ed.), Ch. on Hematologic Malignancies; Swanson's Family Medicine Review
Hodgkins lymphoma types
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
Reed-Sternberg cell Hodgkin lymphoma histology owl eye
Excellent — comprehensive content from both Robbins Pathology and Goldman-Cecil, plus histology images. I have everything needed for a thorough answer.
Hodgkin Lymphoma: Types (Classification)
Hodgkin lymphoma (HL) is a B-cell malignancy defined by the presence of Hodgkin-Reed-Sternberg (HRS) cells — large neoplastic cells that typically constitute only a small minority of the tumor mass, surrounded by a reactive inflammatory infiltrate.
The WHO recognizes two major categories dividing into 5 subtypes total:
The Reed-Sternberg Cell — Pathologic Hallmark
The classic RS cell is 15–45 μm in diameter with:
- Bilobed or multilobed nucleus
- Prominent "owl-eye" nucleoli — large, eosinophilic, inclusion-like, surrounded by a clear halo
- Abundant pale eosinophilic cytoplasm
Classic RS cell immunophenotype: CD15⁺, CD30⁺, CD45⁻, PAX5 (weak), negative for B-cell (CD20) and T-cell markers
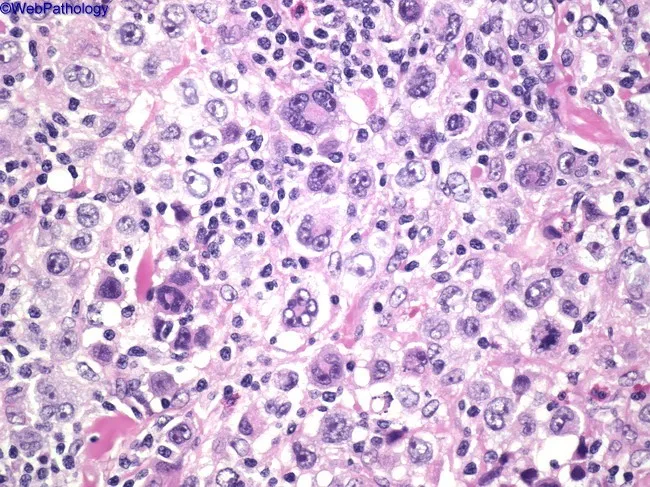
Category 1: Classic Hodgkin Lymphoma (cHL) — ~90% of all HL
All four subtypes share the classic HRS cell immunophenotype (CD15⁺, CD30⁺, CD20⁻).
1. Nodular Sclerosis (NSHL) — Most Common (~65–70%)
| Feature | Detail |
|---|---|
| Age/Sex | Adolescents & young adults; equal sex incidence |
| Sites | Lower cervical, supraclavicular, mediastinal nodes (most common HL subtype with mediastinal disease) |
| Morphology | Broad collagen bands divide lymph node into nodules; RS cell variant = "lacunar cells" — single multilobate nucleus with pale cytoplasm that retracts in formalin ("lacune") |
| Background | Lymphocytes, eosinophils, macrophages, neutrophils |
| EBV association | Low (~10–25%) |
| Prognosis | Excellent |
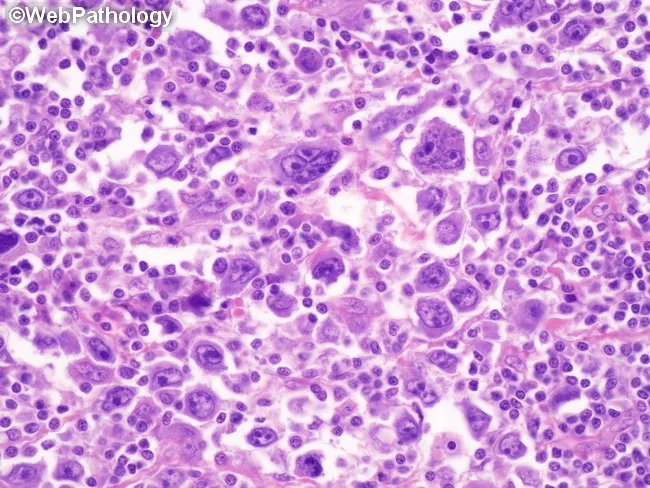
2. Mixed Cellularity (MCHL) — ~25%
| Feature | Detail |
|---|---|
| Age/Sex | Bimodal: young adults and older adults (>50 yrs); more common in males |
| Sites | Peripheral lymph nodes; often involves subdiaphragmatic nodes; less mediastinal involvement than NSHL |
| Morphology | Classic RS cells most abundant here; diffuse or vaguely nodular pattern; no fibrosis bands |
| Background | Rich mixed infiltrate: lymphocytes, eosinophils, plasma cells, histiocytes, neutrophils |
| EBV association | Highest (~70% of cases) |
| Associations | HIV, immunosuppression, developing world |
| Prognosis | Good; slightly worse than nodular sclerosis |
3. Lymphocyte-Rich (LRHL) — ~5%
| Feature | Detail |
|---|---|
| Age/Sex | Older adults; male predominance |
| Morphology | RS cells present but sparse; background is predominantly small lymphocytes (B or T cells); eosinophils and neutrophils absent or rare |
| Architecture | May be nodular or diffuse |
| EBV association | Intermediate (~40%) |
| Prognosis | Excellent — best among classic subtypes |
4. Lymphocyte Depletion (LDHL) — Rarest (~1%)
| Feature | Detail |
|---|---|
| Age/Sex | Older patients; males; HIV-positive patients |
| Morphology | Relatively few lymphocytes; abundant RS cells or variants ("pleomorphic/sarcomatous" pattern); diffuse fibrosis in reticular variant |
| Background | Sparse inflammatory cells; sheets of neoplastic cells |
| EBV association | Very high (~75%) |
| Associations | HIV, developing countries, advanced stage at presentation |
| Prognosis | Poorest of all HL subtypes; often advanced (stage III/IV) at diagnosis |
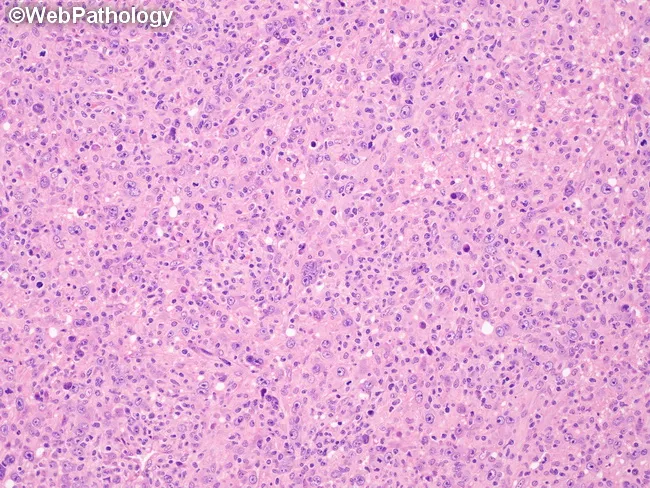
Category 2: Nodular Lymphocyte-Predominant HL (NLPHL) — ~5%
| Feature | Detail |
|---|---|
| Age/Sex | Young males; median age ~30–35 years |
| Sites | Cervical, axillary, or inguinal nodes; mediastinum rarely involved |
| Morphology | Large nodules of predominantly small B lymphocytes and histiocytes; RS cell variant = "popcorn cells" (lymphohistiocytic/L&H cells) — multilobed delicate nucleus resembling popped corn |
| Immunophenotype | CD20⁺, CD79a⁺, BCL6⁺, CD45⁺; CD15⁻, CD30⁻ (or weakly positive) — distinct from classic HL |
| EBV association | Absent (EBV negative) |
| Behavior | Indolent; late relapses common; risk of transformation to DLBCL (~3%) |
| Prognosis | Excellent; often managed with rituximab (due to CD20 expression) |
Summary Comparison Table
| Subtype | Frequency | RS Cell Variant | Background | EBV | Prognosis |
|---|---|---|---|---|---|
| Nodular Sclerosis | 65–70% | Lacunar cells | Mixed; collagen bands | 10–25% | Excellent |
| Mixed Cellularity | ~25% | Classic RS cells | Rich mixed infiltrate | ~70% | Good |
| Lymphocyte-Rich | ~5% | Rare RS cells | Predominantly lymphocytes | ~40% | Excellent |
| Lymphocyte Depletion | ~1% | Abundant RS cells | Few lymphocytes | ~75% | Poor |
| NLPHL | ~5% | Popcorn (L&H) cells | Small B cells + histiocytes | Absent | Excellent |
Key Immunophenotypic Distinction
| Marker | Classic HL | NLPHL |
|---|---|---|
| CD15 | ✅ Positive | ❌ Negative |
| CD30 | ✅ Positive | ❌/± Negative/weak |
| CD20 | ❌ Negative | ✅ Positive |
| CD45 | ❌ Negative | ✅ Positive |
| PAX5 | Weak | Strong |
| EBV | Variable | Negative |
This immunophenotypic distinction is clinically important: NLPHL can be treated with rituximab (anti-CD20), while classic HL subtypes are not routinely CD20-targeted.
— Robbins & Kumar Basic Pathology, Ch. 10; Goldman-Cecil Medicine, Ch. 172
This is a shared conversation. Sign in to Orris to start your own chat.