What is cardiogenic shock ? Explain its different types .
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
Reading File
I have comprehensive content from multiple authoritative textbooks. I can now compose a detailed, well-structured answer.
Cardiogenic Shock
Definition
Cardiogenic shock (CS) is a low cardiac output state resulting in life-threatening end-organ hypoperfusion and hypoxia, caused by primary cardiac dysfunction rather than inadequate intravascular volume. The classic hemodynamic triad (SHOCK trial criteria) is:
- Sustained systolic BP < 90 mmHg for ≥ 30 minutes (or vasopressor dependence to maintain SBP > 90 mmHg)
- Cardiac index ≤ 2.2 L/min/m²
- Pulmonary capillary wedge pressure > 15 mmHg
- Elevated arterial lactate > 2 mmol/L as a marker of tissue hypoperfusion
In-hospital mortality ranges from 40–60%, depending on shock severity and etiology. CS complicates approximately 5–10% of acute MIs and is the most common cause of in-hospital death following acute MI.
— Harrison's Principles of Internal Medicine 22E, p. 2368
Pathophysiology
The pathophysiology of CS is a vicious, self-perpetuating spiral rooted in three main pathways:
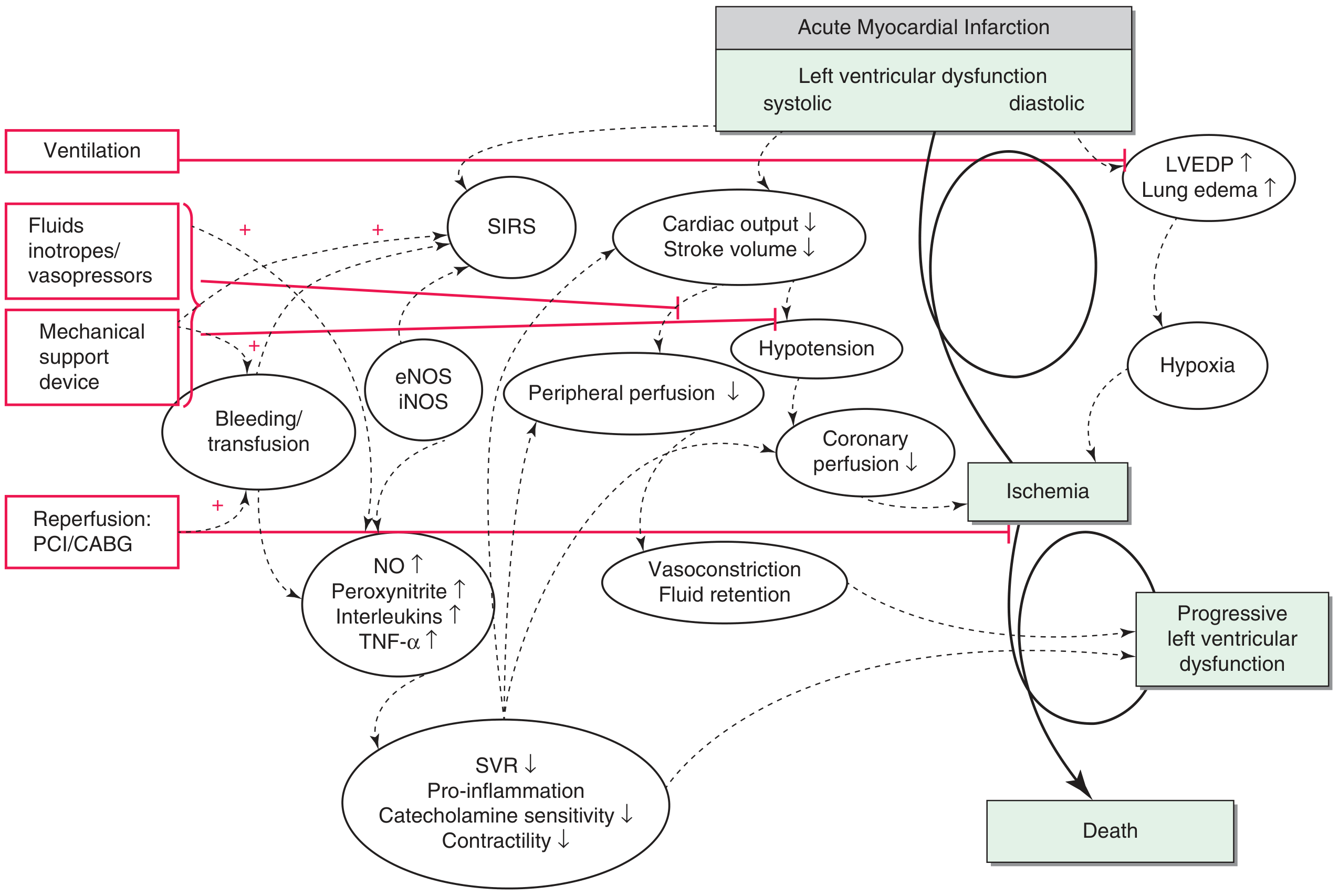
-
↑ Left ventricular end-diastolic pressure (LVEDP) → reduces coronary perfusion pressure → further ischemia + pulmonary edema → systemic hypoxia → worsened myocardial oxygen delivery
-
↓ Blood pressure → reflex compensatory peripheral vasoconstriction → ↑ afterload → ↓ cardiac output further; simultaneously reduces coronary perfusion pressure directly
-
Inflammatory response → tissue necrosis triggers release of nitric oxide, peroxynitrite (direct negative inotrope and cardiotoxin), TNF-α, and interleukins → systemic vasodilation (paradoxically worsening hypotension), ↓ catecholamine sensitivity, ↓ contractility
Lactic acidosis and hypoxemia compound the cycle further, reducing the efficacy of endogenous and exogenous catecholamines.
— Fuster and Hurst's The Heart, 15th Ed., p. 1847
SCAI Staging Classification (2019, Updated)
The Society for Cardiovascular Angiography and Interventions (SCAI) introduced a five-stage classification to standardize communication and guide treatment escalation:
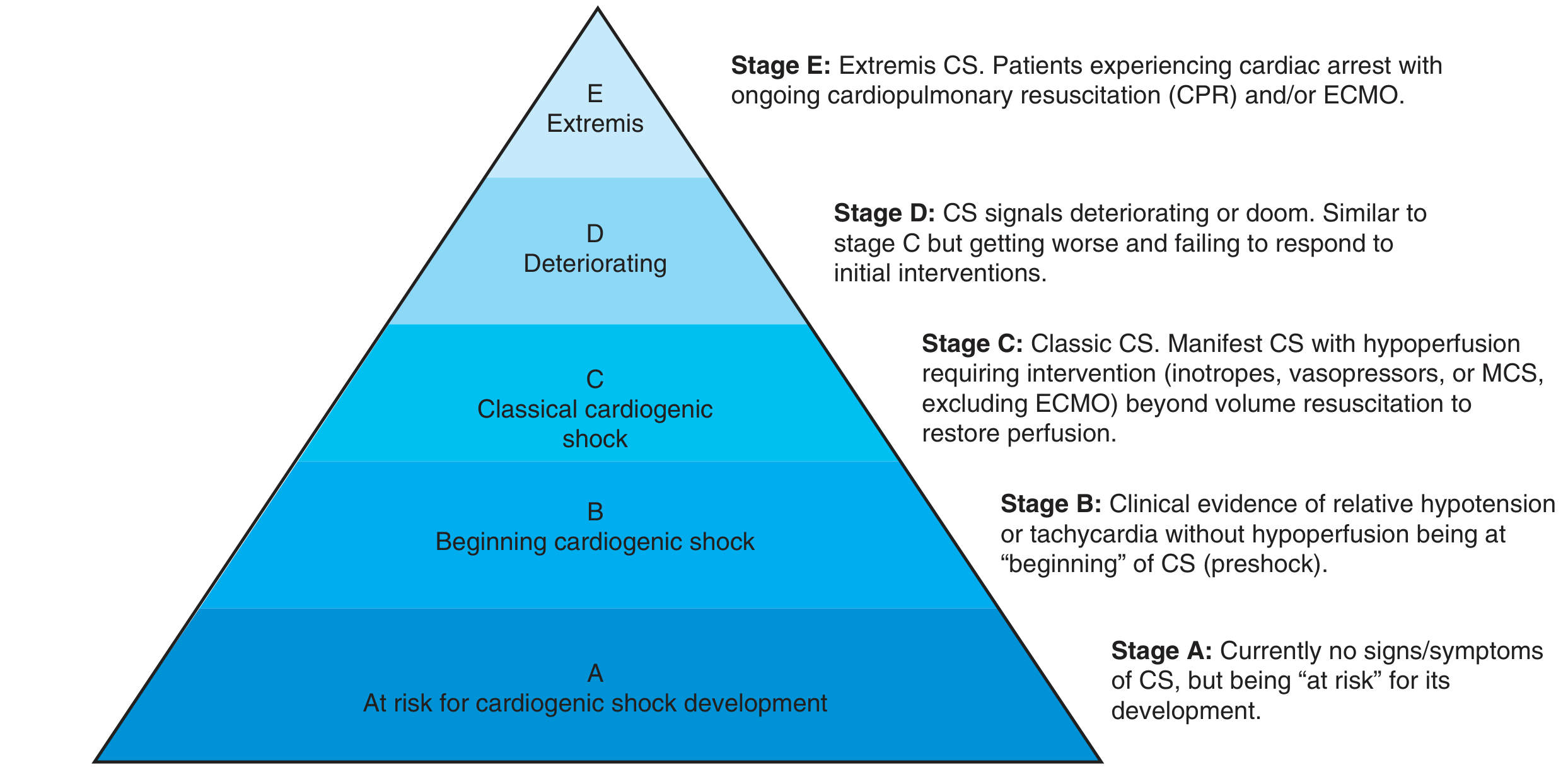
| Stage | Label | Description |
|---|---|---|
| A | At Risk | No current signs/symptoms of CS; risk factors present (e.g., large MI, severe heart failure history) |
| B | Beginning (Preshock) | Relative hypotension or tachycardia without end-organ hypoperfusion; monitor closely and treat early |
| C | Classic | Manifest CS with hypoperfusion requiring inotropes, vasopressors, or MCS (excluding ECMO) beyond volume resuscitation |
| D | Deteriorating | Similar to Stage C but failing to respond to ≥ 30 minutes of initial interventions |
| E | Extremis | Cardiovascular collapse; cardiac arrest with ongoing CPR and/or ECMO |
The updated SCAI model also incorporates a three-axis approach: (1) shock severity, (2) phenotype/etiology, and (3) risk modifiers such as cardiac arrest. Stage E prompts consideration of futility and palliative care.
— Harrison's, p. 2368–2369; Fuster's Heart, p. 1846
Types / Etiological Classification
CS is most usefully classified by the dominant ventricular dysfunction and underlying etiology.
Type 1 — CS with Predominant Left Ventricular Failure
The most common form, accounting for ~80% of CS complicating acute MI. Etiology is subcategorized as:
| Category | Examples |
|---|---|
| Primary myocardial | Acute MI (most common), end-stage cardiomyopathy, myocarditis, Takotsubo syndrome, fulminant myocarditis |
| Obstructive | Acute aortic stenosis, hypertrophic obstructive cardiomyopathy (HOCM), left ventricular outflow obstruction |
| Valvular/structural | Acute severe mitral regurgitation (e.g., papillary muscle rupture after MI), acute aortic regurgitation |
Mechanism: Loss of sufficient LV contractile mass (≥ 40% of LV necrotic or ischemic at autopsy) → ↓ stroke volume → ↓ cardiac output → ↑ LVEDP → pulmonary congestion + hypoperfusion.
Hemodynamics: Low cardiac index, elevated PCWP, elevated SVR (compensatory).
Mechanical complications of MI that cause this type include:
- Acute mitral regurgitation (papillary muscle rupture)
- Ventricular septal rupture (VSR)
- Free wall rupture / pericardial tamponade
— Schwartz's Principles of Surgery, 11th Ed., p. 2524–2526; Harrison's, p. 2369
Type 2 — CS with Predominant Right Ventricular Failure
Less common than LV failure as the primary driver, but carries similar overall mortality. Should be actively sought when an inferior MI patient presents in shock without evidence of LV dysfunction.
Etiology:
- Primary: RV infarction (usually with inferior STEMI), RV cardiomyopathy
- Obstructive: Massive or submassive acute pulmonary embolism (acute RV pressure overload)
- Severe pulmonary hypertension with acute decompensation
Mechanism: RV failure → ↓ RV output → ↓ LV preload (ventricular interdependence) → ↓ cardiac output despite preserved LV function; paradoxical septal shift may further impair LV filling.
Hemodynamics: Elevated right atrial/CVP pressure, low or normal PCWP, low cardiac output. The classic triad in RV MI is hypotension + clear lung fields + elevated JVP.
Key management distinction: Volume loading (to augment RV preload) is appropriate here; diuretics are contraindicated. Avoid vasodilators. Reperfusion of the culprit RCA is critical.
— Fuster's Heart, 15th Ed., p. 1823; Harrison's, p. 2369
Type 3 — CS with Biventricular Failure
Concurrent LV and RV failure, representing a more severe phenotype with higher mortality. Etiology is primary, obstructive, electrical, or structural.
Etiology:
- Extensive anterior MI with RV extension
- Fulminant myocarditis (global dysfunction)
- Severe, decompensated dilated cardiomyopathy
- Arrhythmias (sustained VT/VF, high-degree AV block) — "electrical" cause
- Cardiac tamponade — "obstructive" cause (external compression of both ventricles)
Mechanism: Combined failure of both ventricles creates a compounded reduction in systemic cardiac output; management is more complex as strategies targeting one ventricle may worsen the other.
— Fuster's Heart, 15th Ed., p. 1827; Goldman-Cecil Medicine, block 12
Quick Summary of Causes
| Cause | Frequency |
|---|---|
| Acute MI with LV pump failure | Most common (~80% of CS in MI) |
| Acute severe mitral regurgitation | ~7% of CS in MI |
| Ventricular septal rupture | ~4% of CS in MI |
| Predominant RV failure | ~3–5% |
| Free wall rupture/tamponade | ~2% |
| Other (myocarditis, cardiomyopathy, arrhythmia, tamponade, Takotsubo) | Remainder |
Key Clinical Points
- Diagnosis: ECG, chest X-ray, ABG, lactate, echocardiography (invaluable for identifying LV vs. RV vs. biventricular involvement and mechanical complications). Invasive hemodynamics confirm but are not mandatory.
- Vasopressors: Norepinephrine is the preferred first-line vasopressor (fewer arrhythmias vs. dopamine, better outcomes vs. epinephrine). Catecholamines should be used at the lowest effective dose for the shortest time.
- Revascularization: Early PCI of the culprit artery is the cornerstone of treatment in MI-related CS; mortality correlates directly with restoration of coronary flow (33% with complete reperfusion vs. 85% with absent reperfusion).
- Mechanical Circulatory Support (MCS): IABP is no longer routinely recommended for LV failure CS. Active MCS devices (Impella, VA-ECMO) are used for refractory or high-risk cases.
- IABP-SHOCK II trial: Routine IABP in CS complicating MI did not reduce 30-day, 12-month, or 6-year mortality.
— Harrison's, p. 2371; Schwartz's Surgery, p. 2525–2526
This is a shared conversation. Sign in to Orris to start your own chat.