apoptosis extrinsic pathway
Reading File
Finding Sources
Finding Sources
Searching Images
apoptosis extrinsic pathway death receptor caspase
Reading File
Reading File
Loading Image
Finding Sources
Reading File
Reading File
I now have comprehensive information from multiple authoritative textbooks. Here is a complete answer:
Apoptosis — Extrinsic (Death Receptor) Pathway
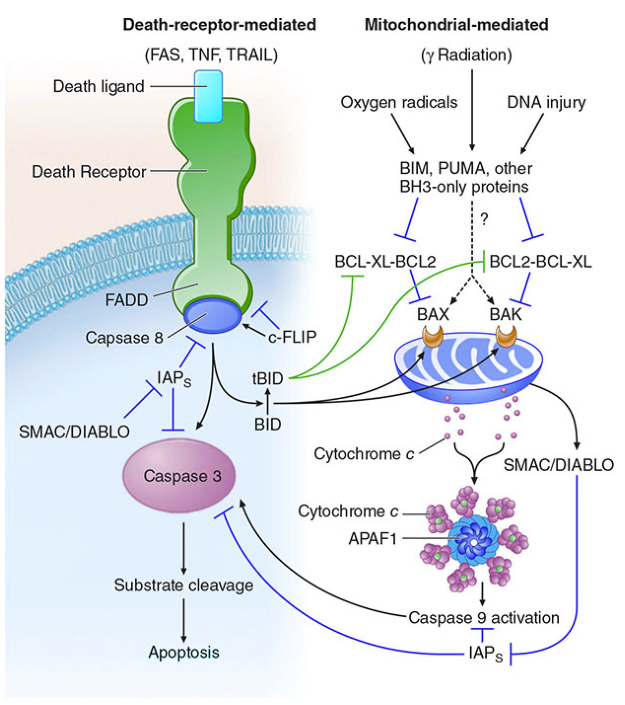
Overview
The extrinsic pathway (also called the death receptor pathway) initiates apoptosis from outside the cell via specific ligand–receptor interactions at the plasma membrane. It is distinct from the intrinsic (mitochondrial) pathway, though the two can intersect.
Step-by-Step Mechanism
1. Death Receptor Engagement
Death receptors are members of the TNF receptor superfamily that carry a cytoplasmic death domain (DD) — a ~80-amino-acid motif essential for transmitting apoptotic signals. The best-characterized examples:
| Receptor | Ligand |
|---|---|
| Fas (CD95, APO-1) | FasL (CD95L) |
| TNFR1 | TNF-α |
| DR4 / DR5 | TRAIL (TNF-related apoptosis-inducing ligand) |
When a death ligand (e.g., FasL) binds its receptor, three or more receptor molecules oligomerize, clustering their cytoplasmic death domains.
2. DISC Formation
The clustered death domains recruit the adaptor protein FADD (Fas-associated death domain protein). FADD contains both a death domain (binds receptor) and a death effector domain (DED) (binds caspase-8/10).
FADD recruits pro-caspase-8 (and/or pro-caspase-10) through DED–DED interactions. This multiprotein complex — receptor + FADD + pro-caspase-8/10 — is the Death-Inducing Signaling Complex (DISC).
3. Caspase-8 Activation
Within the DISC, multiple pro-caspase-8 molecules are brought into close proximity, enabling autocatalytic cleavage and generation of active caspase-8 (an initiator caspase).
4. Effector Caspase Cascade (Execution Phase)
Active caspase-8 directly cleaves and activates the executioner caspases — caspase-3, -6, and -7. These then:
- Cleave ICAD (inhibitor of caspase-activated DNase) → releases CAD/DFF40 → causes internucleosomal DNA fragmentation
- Proteolyze structural nuclear matrix proteins → nuclear fragmentation
- Activate other downstream substrates → membrane blebbing, apoptotic body formation, and phagocytic recognition signals (e.g., phosphatidylserine exposure)
Crosstalk with the Intrinsic Pathway
In some cell types (e.g., hepatocytes, pancreatic β-cells), caspase-8 cleaves the BH3-only protein BID → generating tBID (truncated BID), which translocates to the mitochondria, activates BAX/BAK, and amplifies the apoptotic signal through the intrinsic pathway. This amplification loop is necessary in cells where direct caspase-8 activation alone is insufficient to commit the cell to death.
Key Regulators
| Molecule | Role |
|---|---|
| c-FLIP | Competitive inhibitor; binds pro-caspase-8 and blocks DISC assembly/activation → anti-apoptotic |
| IAPs (XIAP, survivin, etc.) | Inhibit active caspases-3, -7, and -9 |
| SMAC/DIABLO | Released from mitochondria; neutralizes IAPs → promotes apoptosis |
Physiological Roles
- Peripheral tolerance: FasL on activated T cells kills self-reactive lymphocytes expressing Fas (activation-induced cell death, AICD)
- CTL-mediated killing: Cytotoxic T lymphocytes express FasL to kill virus-infected and tumor target cells (alongside the perforin/granzyme mechanism)
- Immune privilege: Tissues like the eye and testis express FasL to induce apoptosis in infiltrating immune cells
Summary Diagram (Extrinsic Pathway)
Death ligand (FasL / TNF / TRAIL)
↓
Death receptor (Fas / TNFR1 / DR4-5) ← oligomerization
↓
DISC: FADD + Pro-Caspase-8/10
↓ autocatalytic cleavage
Active Caspase-8 ←— inhibited by c-FLIP
↙ ↘
tBID (→ mito) Caspase-3/6/7 (executioners) ←— inhibited by IAPs
↓
DNA fragmentation, nuclear collapse,
membrane blebbing → APOPTOSIS
Sources:
- Robbins, Cotran & Kumar — Pathologic Basis of Disease, pp. 59–60
- Robbins & Kumar Basic Pathology, block 1
- Schwartz's Principles of Surgery, 11th Ed., p. 1644
- Harrison's Principles of Internal Medicine, 21st Ed.
This is a shared conversation. Sign in to Orris to start your own chat.