Sevoflurane vaporiser
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
sevoflurane vaporizer anaesthetic machine
Reading File
Reading File
Loading Image
I now have comprehensive content. Let me compile the full answer on the sevoflurane vaporiser.
Sevoflurane Vaporiser
Physical Basis of Vaporisation
Sevoflurane, like all volatile anaesthetics, must be vaporised before delivery to the patient. The key physical properties governing vaporiser design are:
Saturated Vapour Pressure (SVP): At 20°C and sea level, sevoflurane has an SVP of 160 mmHg, giving a saturated vapour concentration of 160/760 × 100 = ~21% — far higher than the clinically required 1–4%. The vaporiser must dilute this to a usable concentration.
Latent heat of vaporisation: Energy is consumed as liquid sevoflurane converts to vapour. Without compensation, the remaining liquid cools, decreasing SVP and reducing vaporiser output. Modern vaporisers are constructed of metals with high specific heat and thermal conductivity (e.g., copper alloys) to minimise this temperature drop.
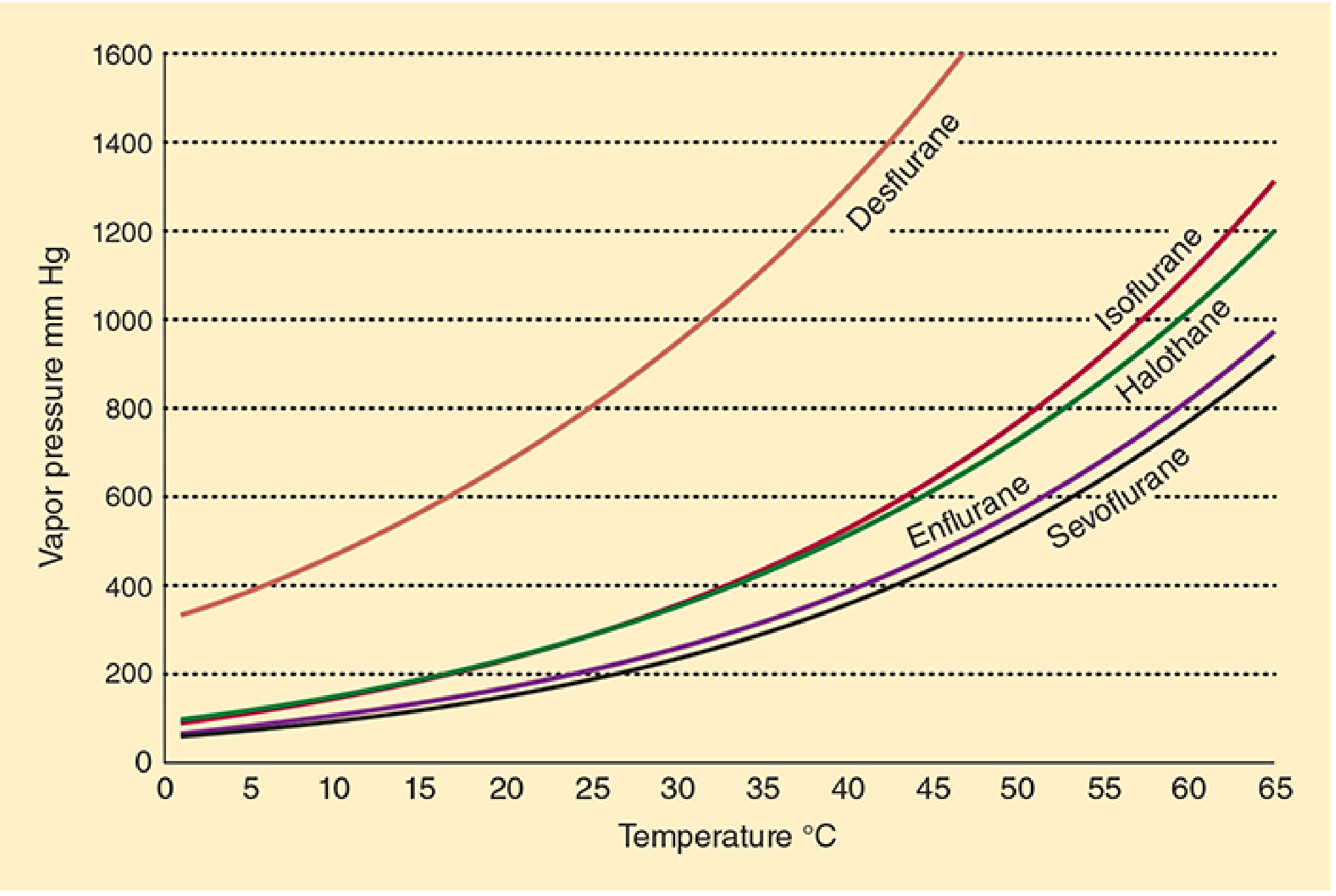
Sevoflurane has the lowest SVP among commonly used agents at room temperature — the highest curve is desflurane, which requires a heated pressurised vaporiser.
Classification of the Sevoflurane Vaporiser
Sevoflurane is delivered by a variable bypass, flow-over, temperature-compensated, agent-specific, out-of-breathing-circuit vaporiser (e.g., GE Tec 7, Dräger Vapor 2000/3000).
| Feature | Detail |
|---|---|
| Variable bypass | Concentration dial splits incoming fresh gas between bypass and vaporising chambers |
| Flow-over | Gas flows over a wick saturated with liquid sevoflurane (vs. obsolete bubble-through) |
| Temperature-compensated | Bimetallic strip or bellows adjusts bypass ratio automatically with temperature change |
| Agent-specific | Designed only for sevoflurane; keyed filling port prevents misfilling |
| Out-of-circuit | Located between flowmeters and common gas outlet, upstream of patient |
Basic Operating Principle
Fresh gas from the flowmeters enters the vaporiser inlet. The concentration control dial sets a precise ratio of gas flow:
- Bypass chamber: gas bypasses the liquid, carrying no anaesthetic
- Vaporising chamber: gas flows over wicks saturated with liquid sevoflurane, becoming saturated with vapour (~21%)
The two streams recombine at the vaporiser outlet to yield the clinically set concentration.
Worked example (Barash): To deliver 2% sevoflurane, the vaporiser creates a bypass:vaporising ratio of 9.5:1 — i.e., 21 mL of sevoflurane vapour is added to every 950 mL of bypass gas. The dial essentially controls this split ratio automatically.
Temperature Compensation
Because SVP is temperature-dependent (falling vapour pressure at lower temperatures would reduce output), the vaporiser uses:
- A bimetallic strip or aneroid bellows that expands/contracts with temperature
- This physically alters the aperture of the bypass/vaporising split to maintain a constant output regardless of ambient temperature changes
Vaporiser materials are chosen for high specific heat (to buffer temperature swings) and high thermal conductivity (to equilibrate temperature rapidly).
Why Sevoflurane ≠ Desflurane Vaporiser
Sevoflurane can be delivered in a standard variable bypass vaporiser precisely because its SVP (160 mmHg) is moderate and manageable at room temperature. Desflurane (SVP 669 mmHg at 20°C, boiling point 22.8°C) cannot — it would boil uncontrollably in a conventional vaporiser and requires a specialised electrically heated, pressurised Tec 6 / D-Vapor system.
Misfilling
Sevoflurane vaporisers use agent-specific keyed filling ports (Quik-Fil / Saf-T-Fill) to prevent wrong-agent filling. However, if misfilling occurs:
- A sevoflurane vaporiser filled with isoflurane: the isoflurane SVP (240 mmHg) is higher than sevoflurane's (160 mmHg), so the vaporiser will deliver a higher-than-set concentration — potentially dangerous.
- An isoflurane vaporiser filled with sevoflurane: lower SVP means less drug delivered than dialled — inadequate anaesthesia.
Location and Interlock
- Vaporisers must sit between the flowmeters and the common gas outlet
- Machines with multiple vaporisers must have an interlocking (exclusion) device so only one vaporiser can be active at a time
- When the dial is in the "off" position, internal leaks may go undetected — leak checks must be performed with the dial turned on
MRI Considerations
Standard sevoflurane vaporisers may contain internal ferrous components not detectable by a horseshoe magnet. Only MRI-certified non-ferrous vaporisers should be used in the MRI suite — a non-certified vaporiser poses a projectile hazard.
ICU Delivery (AnaConDa)
In the ICU, sevoflurane can be delivered via the AnaConDa (Anaesthetic Conserving Device) — a miniature vaporiser inserted directly into the ventilator circuit. It uses a syringe driver to inject liquid sevoflurane onto an activated charcoal filter that acts as a vaporising and heat-moisture exchange element, eliminating the need for a full anaesthetic workstation.
Sources: Barash, Clinical Anesthesia 9e, pp. 1970–1984; Morgan & Mikhail, Clinical Anesthesiology 7e, pp. 119–125
From dorsch and dorsch
Finding Sources
Finding Sources
Finding Sources
Unfortunately, Dorsch & Dorsch (Understanding Anesthesia Equipment) is not available in the medical textbook library. The library's anesthesiology collection contains only three titles:
- Barash, Cullen & Stoelting's Clinical Anesthesia, 9e
- Miller's Anesthesia, 2-Volume Set, 10e
- Morgan & Mikhail's Clinical Anesthesiology, 7e
Dorsch & Dorsch is the classic dedicated equipment reference, but it is not in the library database. I can offer you the sevoflurane vaporiser content from any of the three available texts — all of which cover vaporiser physics, the variable bypass design, temperature compensation, agent-specific filling, and misfilling hazards in considerable detail (as shown in the previous answer, which drew primarily from Barash and Morgan & Mikhail).
If you have a specific aspect of the sevoflurane vaporiser you'd like explored in depth from those available sources, I'm happy to go deeper.
This is a shared conversation. Sign in to Orris to start your own chat.