Q. University of Pennsylvania smell identification test in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
University of Pennsylvania Smell Identification Test UPSIT olfactory testing
smell identification test anosmia hyposmia olfaction clinical assessment
olfactory testing methods Sniffin Sticks UPSIT threshold discrimination identification
UPSIT University of Pennsylvania smell identification test booklet microencapsulated
UPSIT microencapsulated odorants scratch sniff procedure scoring interpretation
olfactory dysfunction causes nasal rhinosinusitis head trauma neurodegenerative
olfactory nerve anatomy pathway cribriform plate olfactory bulb cortex
olfaction assessment ENT rhinology clinical examination Scott Brown Cummings
UPSIT malingering detection smell loss norms percentile age gender categories
University of Pennsylvania Smell Identification Test (UPSIT)
A Comprehensive Study for 50 Marks
1. INTRODUCTION AND HISTORICAL BACKGROUND
"The most widely used olfactory test, the 40-item University of Pennsylvania Smell Identification Test (UPSIT), uses norms based on nearly 4000 normal subjects. A determination is made of both absolute dysfunction (i.e., mild loss, moderate loss, severe loss, total loss, probable malingering) and relative dysfunction (percentile rank for age and gender)."
2. ANATOMY AND PHYSIOLOGY: THE FOUNDATION OF UPSIT
2.1 Olfactory Pathway
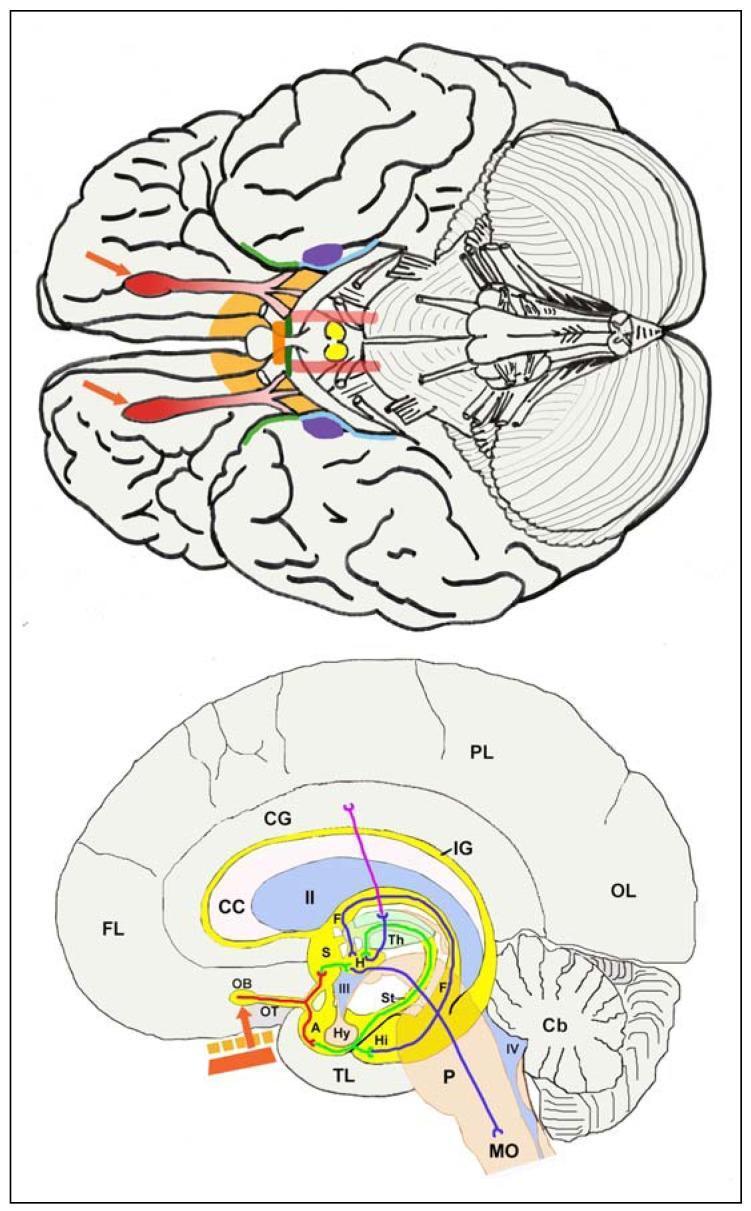
OLFACTORY PATHWAY FLOWCHART:
Odorant molecules in inspired air
↓
Olfactory epithelium (superior nasal cavity)
– Olfactory receptor neurons (ORNs) — bipolar cells
– ~10 million ORNs, ~400 functional receptor types
↓
Axons (CN I / Olfactory nerve fila)
– Pass through cribriform plate of ethmoid bone
↓
Olfactory Bulb (Glomeruli)
– Mitral & tufted cells synapse here
↓
Olfactory Tract
– Lateral stria → Piriform cortex (primary olfactory cortex)
– Medial stria → Anterior commissure
↓
Secondary projections:
→ Entorhinal cortex (Brodmann area 28)
→ Amygdala (emotional responses to smell)
→ Hypothalamus (autonomic, endocrine)
→ Thalamus → Orbitofrontal cortex (conscious perception, flavor)
→ Hippocampus (olfactory memory)
2.2 Classification of Olfactory Disorders
| Term | Definition |
|---|---|
| Normosmia | Normal smell function |
| Hyposmia | Reduced smell sensitivity |
| Anosmia | Complete loss of smell |
| Dysosmia | Distorted smell perception |
| Parosmia | Distortion in presence of odorant |
| Phantosmia | Smell perception without odorant |
| Presbyosmia | Age-related smell decline |
| Cacosmia | Everything smells unpleasant |
3. THE UPSIT IN DETAIL
3.1 Basic Description
| Parameter | Detail |
|---|---|
| Full name | University of Pennsylvania Smell Identification Test |
| Commercial name | SIT (Smell Identification Test) |
| Developer | Richard L. Doty et al., 1984 |
| Format | 4 test booklets × 10 items = 40 odorants |
| Type | Forced-choice, self-administered |
| Method | Scratch-and-sniff microencapsulated odorants |
| Time | 10–15 minutes |
| Age range | 5 years and above |
| Normative database | ~4000 normal subjects (extended to >10,000 in later studies) |
| Scoring range | 0 – 40 |
3.2 Physical Components
- Four saddle-bound booklets (Booklet 1–4), each containing 10 odorant-impregnated strips
- A scoring key / answer sheet
- Instruction sheet
- Pencil (for scratching the strips)
UPSIT BOOKLET STRUCTURE:
┌─────────────────────────────────────────────┐
│ UPSIT BOOKLET (1 of 4) │
│ │
│ Item 1: [Microencapsulated strip] │
│ ○ A. Pizza ○ B. Chocolate │
│ ○ C. Banana ○ D. Smoke │
│ │
│ Item 2: [Microencapsulated strip] │
│ ○ A. Rose ○ B. Lemon │
│ ○ C. Mint ○ D. Motor oil │
│ .... │
│ Item 10: [Microencapsulated strip] │
│ ○ A. Garlic ○ B. Soap │
│ ○ C. Peach ○ D. Gasoline │
└─────────────────────────────────────────────┘
× 4 booklets = 40 items total
3.3 The 40 UPSIT Odorants
| Booklet | Sample Odorants |
|---|---|
| 1 | Pizza, chocolate, motor oil, rose, clove, leather, strawberry, lilac, pineapple, smoke |
| 2 | Menthol, natural gas, lemon, watermelon, paint thinner, banana, cedar, soap, grape, turpentine |
| 3 | Gasoline, onion, coconut, vanilla, peanut, peach, root beer, cherry, pine, dill pickle |
| 4 | Cheddar cheese, mint, lime, skunk, orange, musk, spearmint, wintergreen, coffee, peanut butter |
3.4 Microencapsulation Technology
HOW MICROENCAPSULATION WORKS:
Odorant chemical (e.g., isoamyl acetate for banana)
↓
Encapsulated in tiny polymer microcapsules
(diameter ~10–50 micrometers)
↓
Printed onto adhesive strips in booklets
↓
At testing: patient scratches strip with pencil
↓
Microcapsules rupture → odorant released
↓
Patient sniffs and selects from 4 options
- Long shelf life (2–4 years)
- Reproducible odor concentration
- Portable and self-administered
- Standardized across centers worldwide
4. TEST ADMINISTRATION PROCEDURE
4.1 Pre-Test Conditions
PRE-TEST CHECKLIST:
□ No eating, drinking, or smoking for 30 minutes prior
□ No strong perfumes/colognes on patient or examiner
□ No nasal decongestant sprays for 24 hours
□ Quiet, well-ventilated room
□ Confirm patient can read (or read items aloud for illiterate patients)
□ Test at ambient temperature (extremes affect volatilization)
□ Record: age, sex, ethnicity, medications, nasal history
4.2 Step-by-Step Administration Flowchart
UPSIT ADMINISTRATION FLOWCHART:
START
│
▼
Patient seated comfortably; instructions read aloud
│
▼
Hand Booklet 1 to patient
│
▼
Patient opens to Item 1
│
▼
Patient uses pencil tip to FIRMLY SCRATCH the
brown strip at the designated scratch area
│
▼
Patient immediately brings booklet to nose
and sniffs once (2–3 cm from strip)
│
▼
Patient reads 4 answer options (forced-choice)
and MUST select ONE answer (even if no smell perceived)
│
▼
Patient circles/marks chosen answer
│
▼
Repeat for all 10 items in Booklet 1
│
▼
Proceed to Booklets 2, 3, 4 sequentially
│
▼
All 40 items completed
│
▼
Examiner scores using answer key
│
▼
Calculate raw score (0–40)
│
▼
Interpret using normative tables (age + sex)
│
▼
END — Generate report
4.3 Key Administration Points
- Forced-choice paradigm: The patient must choose one of four options — even if they perceive no odor. This is critical for malingering detection.
- One sniff per item: Multiple prolonged sniffs cause olfactory fatigue (adaptation).
- Hold booklet 2–3 cm from nose: Too close may miss volatilized odorant; too far reduces concentration.
- Do not assist the patient in choosing an answer.
- The test can be self-administered or administered by a technician.
- Available in multiple languages (Spanish, French, German, Chinese, Japanese, Korean, etc.).
5. SCORING AND INTERPRETATION
5.1 Raw Score Calculation
SCORING:
Total correct answers (out of 40) = Raw UPSIT Score
Each correct response = 1 point
Incorrect = 0 points
Total range: 0 – 40
5.2 Absolute Dysfunction Categories
| Score (out of 40) | Category |
|---|---|
| 35–40 | Normosmia (normal) |
| 30–34 | Microsmia / Mild loss |
| 26–29 | Moderate loss |
| 19–25 | Severe loss |
| ≤18 | Total loss / Anosmia |
| ≤5 | Probable malingering (below chance) |
5.3 Malingering Detection — A Unique Feature
MALINGERING DETECTION LOGIC:
Chance performance = 10/40 (25%)
↓
Score < 6 → Statistically impossible by chance
↓
Conclusion: Patient was aware of correct odors
but chose wrong answers deliberately
↓
= PROBABLE MALINGERER
5.4 Relative Dysfunction — Normative Percentile Ranking
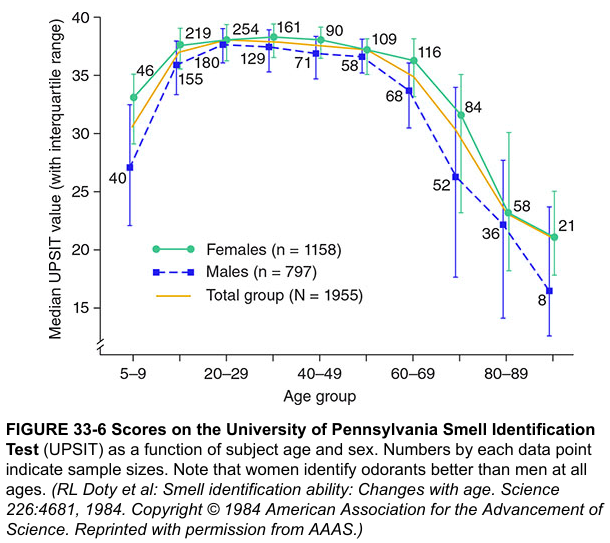
- Females consistently outperform males at all ages
- Peak scores at 20–40 years (~35–37/40)
- Progressive decline after age 60 (presbyosmia)
- By age 80+, median scores approach 25–28/40
- UPSIT scores therefore MUST be interpreted relative to age and sex norms
PERCENTILE INTERPRETATION:
─────────────────────────────────────────
Percentile Clinical significance
─────────────────────────────────────────
> 75th Normal for age/sex
26–75th Low-normal — monitor
11–25th Hyposmic range
5–10th Significantly hyposmic
< 5th Anosmia / severe loss
─────────────────────────────────────────
6. OLFACTORY DISORDERS ASSESSED BY UPSIT
6.1 Common Causes of Abnormal UPSIT Scores
CAUSES OF ABNORMAL UPSIT (in order of frequency):
1. UPPER RESPIRATORY TRACT INFECTIONS
(viral — post-COVID, post-influenza)
Mechanism: Viral damage to olfactory epithelium,
neuroepithelial destruction, mucosal edema
↓
2. HEAD TRAUMA
Mechanism: Shearing of olfactory fila through
cribriform plate → scarring (cribriform plate
fracture NOT required for smell loss)
Risk ↑ with poor GCS score + prolonged amnesia
↓
3. CHRONIC RHINOSINUSITIS
Mechanism: Obstructive (mucus, polyps block
odorant access) + sensorineural (mucosal
inflammation damages epithelium)
6.2 Conditions Evaluated by UPSIT
| Category | Conditions |
|---|---|
| Conductive | Nasal polyps, chronic rhinosinusitis, septal deviation |
| Sensorineural | Post-viral, post-traumatic, idiopathic |
| Neurodegenerative | Parkinson's disease (earliest marker), Alzheimer's disease, Lewy body dementia |
| Toxic/Drug | Cocaine abuse, tobacco, cadmium, aminoglycosides, metronidazole, carbamazepine |
| Endocrine | Hypothyroidism, Addison's disease, Cushing's |
| Congenital | Kallmann syndrome (anosmia + hypogonadism), congenital anosmia |
| Psychiatric | Schizophrenia (reduced identification scores), depression |
| Infectious | COVID-19 (sudden onset anosmia — cardinal feature), tuberculosis, syphilis |
| Neoplastic | Olfactory groove meningioma, olfactory neuroblastoma (esthesioneuroblastoma) |
7. CLINICAL APPLICATIONS OF UPSIT
7.1 Diagnostic Applications
CLINICAL APPLICATION FLOWCHART:
Patient complaint: "I can't smell properly"
↓
History + Nasal endoscopy
↓
UPSIT administered
↓
┌───────────┬───────────┬─────────────┐
↓ ↓ ↓ ↓
Normal Mild loss Severe loss Malingering
(35–40) (30–34) (≤18) (≤5)
↓ ↓ ↓ ↓
Reassure Monitor + Investigate: Legal report
Treat cause MRI, EEG,
Neurology ref
7.2 Specific Clinical Uses
-
Parkinson's Disease (PD) Screening: UPSIT is one of the earliest biomarkers of PD — olfactory loss precedes motor symptoms by up to 4–7 years. UPSIT scores in PD average 18–22/40.
-
Alzheimer's Disease: Impaired UPSIT scores correlate with hippocampal volume loss; may predict cognitive decline.
-
COVID-19 (Post-COVID anosmia): UPSIT provides objective documentation of smell loss, serial monitoring of recovery, and medicolegal certification.
-
Occupational Medicine / Legal Claims: Provides objective, reproducible documentation of smell loss for worker's compensation and personal injury claims (Harrison's, p. 1013).
-
Treatment Efficacy Monitoring: Serial UPSIT scores before and after:
- Endoscopic sinus surgery (CRS with nasal polyps)
- Olfactory training
- Corticosteroid therapy
- Zinc supplementation
-
Pre-operative baseline: Before skull base / anterior fossa surgery.
8. COMPARISON OF UPSIT WITH OTHER OLFACTORY TESTS
COMPARISON TABLE: OLFACTORY TESTS
Parameter UPSIT Sniffin' Sticks CCCRC Test Brief Smell
(USA) (Europe) (Canada) Identification
─────────────────────────────────────────────────────────────────────────
Items 40 Threshold+Disc Threshold 12 items
+ID (TDI) +ID
Method Scratch/sniff Felt-tip pens Bottles Scratch/sniff
Domains Identification Threshold Threshold Identification
tested Discrimination Identification
Identification
Administration Self-admin Examiner-led Examiner Self-admin
Time 10–15 min 30–40 min 20–30 min 5 min
Normative data ~4000+ ~4000 (Hummel) ~500 ~500
Malingering YES (4AFC) Partial No Partial
detection
Cost $$$ $$ $ $
Portability Excellent Good Moderate Excellent
Languages 12+ German, others English Limited
─────────────────────────────────────────────────────────────────────────
8.1 Sniffin' Sticks vs UPSIT
- Pen-like devices with felt-tip impregnated with odorant
- Tests Threshold (T), Discrimination (D), Identification (I)
- TDI score: Maximum 48 — normosmia >30.5
- Advantage: Tests three domains (UPSIT only tests identification)
- Disadvantage: Requires trained examiner, more time, more expensive
8.2 UPSIT Reduced Versions
| Version | Items | Use |
|---|---|---|
| B-SIT (Brief) | 12 items | Quick screening |
| CC-SIT (Cross-Cultural) | 12 items | Multiple ethnicities |
| SAN (San Diego Odor ID Test) | 8 items | Elderly screening |
| PODS (Pocket Odor Test) | 3 items | Ultra-brief bedside |
| NHANES Pocket Smell Test | 8 items | Epidemiological surveys |
9. TEXTBOOK PERSPECTIVES
9.1 Scott Brown's Otorhinolaryngology (8th Edition)
- Emphasizes two main methods: psychophysical (like UPSIT) and electrophysiological (olfactory ERPs)
- UPSIT recommended as gold standard psychophysical test for identification
- Notes that conductive causes (polyps, CRS) often show better recovery post-treatment vs. sensorineural
- Advocates serial UPSIT to monitor sinus disease treatment outcomes
- Olfactory training (Hummel protocol — 4 odors x 2×/day) recommended alongside UPSIT monitoring
9.2 Cummings Otolaryngology – Head and Neck Surgery (7th Edition)
- UPSIT described as the most extensively validated olfactory identification test
- Emphasizes age-gender normative interpretation
- Notes UPSIT utility in sinonasal malignancy workup: olfactory neuroblastoma (esthesioneuroblastoma) may present with anosmia detectable by UPSIT
- Discusses Kadish staging of olfactory neuroblastoma, where UPSIT may track functional residual olfaction
- Recommends UPSIT before and after endoscopic skull base surgery
9.3 Stell & Maran's Textbook of Head and Neck Surgery
- Advocates UPSIT for systematic pre- and post-operative olfactory assessment in sinus surgery
- Notes UPSIT can unmask bilateral anosmia in patients who attribute only nasal blockage as their complaint
- Recommends combining UPSIT with nasal endoscopy and CT sinuses
9.4 Dhingra's Diseases of Ear, Nose and Throat (8th Ed.)
- UPSIT described under "Tests of Olfaction"
- Listed alongside Elsberg & Levy's method (butanol threshold) and CCCRC battery
- Emphasizes that in Indian/Asian populations, CC-SIT (Cross-Cultural variant) with locally familiar odorants may be more appropriate
- Notes that UPSIT performance depends on odorant cultural familiarity — a limitation in non-Western populations
9.5 Hazarika's Textbook of ENT and Head & Neck Surgery
- UPSIT placed in context of rhinological examination — after anterior rhinoscopy and nasal endoscopy
- Recommended for objective documentation in cases of post-traumatic anosmia in medicolegal context
- Notes that bilateral posterior choanal atresia in adults (rare, missed diagnosis) may show severe UPSIT scores alongside normal threshold-type tests if odorants are given retronasally
9.6 Zakir Hussain / Mohan Bansal (Textbook of ENT)
- UPSIT cited as standard for documenting olfactory loss in cases of chronic sinusitis, nasal polyps
- Emphasizes its role in pre- and post-operative assessment
- Stresses patient education on its self-administered nature — important in resource-limited settings
10. LIMITATIONS OF UPSIT
LIMITATIONS OF UPSIT:
┌──────────────────────────────────────────────┐
│ 1. Tests ONLY identification domain │
│ (not threshold or discrimination) │
│ │
│ 2. Cultural bias — odorants familiar to │
│ North American populations; may │
│ underperform in Asian/African patients │
│ │
│ 3. Cognitive demand — requires reading, │
│ decision-making; impaired by dementia │
│ │
│ 4. Cannot differentiate conductive from │
│ sensorineural loss by itself │
│ │
│ 5. Single-nostril testing not standard │
│ (bilateral testing only) │
│ │
│ 6. Expensive compared to simpler tests │
│ (e.g., alcohol sniff test) │
│ │
│ 7. Olfactory fatigue if items presented │
│ too rapidly │
│ │
│ 8. Not suitable for intubated/ICU patients │
│ │
│ 9. Odor familiarity dependent on education │
│ and cultural exposure │
└──────────────────────────────────────────────┘
11. DIFFERENTIAL DIAGNOSIS OF SMELL LOSS: ALGORITHMIC APPROACH USING UPSIT
DIAGNOSTIC ALGORITHM FOR OLFACTORY LOSS:
Patient presents with smell complaint
↓
Detailed history:
- Onset (sudden vs. gradual)
- Preceding URTI / COVID-19
- Head trauma history
- Nasal symptoms (blockage, rhinorrhea, polyps)
- Drug history
- Neurological symptoms
↓
Anterior rhinoscopy + Nasal endoscopy
↓
UPSIT administered
↓
┌──────────────────────────────────┐
│ UPSIT Result │
└──────┬──────────────┬───────────┘
↓ ↓
Normal (35–40) Abnormal (< 35)
↓ ↓
Reassure + ┌──────────────────┐
Retest in │ Nasal endoscopy │
6 months │ findings? │
└───┬──────────┬──┘
↓ ↓
Structural No structural
(polyps, CRS) cause found
↓ ↓
CT Sinuses MRI brain/
+ Surgery olfactory bulbs
→ Serial UPSIT ↓
monitoring Consider:
- Parkinson's
- Alzheimer's
- Post-viral
- Kallmann's
- Malingering
12. RECENT ADVANCES
12.1 UPSIT in COVID-19 (Post-COVID Olfactory Dysfunction)
- COVID-19 caused an unprecedented global surge in anosmia/parosmia
- SARS-CoV-2 infects sustentacular (support) cells of olfactory epithelium via ACE2 receptors (not directly the ORNs)
- UPSIT became a key outcome measure in COVID-19 olfactory research
- Studies showed mean UPSIT scores of 18–24/40 in acute COVID-19 anosmia
- Serial UPSIT demonstrated that 70–80% recovered to near-normal within 6–12 months; ~10% had persistent loss
- Olfactory training (Hummel protocol) combined with corticosteroids improved UPSIT scores in post-COVID patients
12.2 Neuroimaging Correlation
- Diffusion tensor imaging (DTI) of olfactory tracts now correlates with UPSIT scores — lower UPSIT → reduced fractional anisotropy of olfactory tracts
- Olfactory bulb volume (OBV) on MRI inversely correlates with UPSIT impairment: OBV < 40 mm³ suggests sensorineural loss
- Functional MRI (fMRI): UPSIT score correlates with activation in piriform cortex and orbitofrontal cortex
12.3 Parkinson's Disease Biomarker Applications
- UPSIT score < 25/40 in a patient > 60 years — independent risk factor for developing PD within 5 years (Schenck et al., 2002)
- UPSIT integrated into PD diagnostic criteria supplementary assessment
- Combined with α-synuclein skin biopsy + DAT-SPECT — a powerful PD prodrome detection battery
12.4 Digital and AI-Assisted UPSIT
- App-based smell testing: Digital versions under development (e.g., University of Pennsylvania digital UPSIT-like tools)
- Machine learning: AI models trained on large UPSIT datasets to predict neurodegenerative disease onset
- Telemedicine adaptation: Home-based UPSIT kits mailed to patients, with results entered online — validated for remote administration
12.5 Olfactory Training + UPSIT Monitoring
4 odorants: Rose, Eucalyptus, Lemon, Clove
↓
Sniff each odorant × 20 seconds, twice daily
↓
Duration: 12–16 weeks minimum
↓
Monitor with UPSIT at 0, 8, 16 weeks
↓
Expected improvement: +3 to +6 UPSIT points
12.6 Cross-Cultural and Pediatric Adaptations
- CC-SIT (Cross-Cultural Smell Identification Test): 12 items with odorants familiar across multiple cultures
- CCSIT-3 (3-item): Ultra-brief screening for resource-limited settings
- Pediatric UPSIT norms: Established for children ≥ 5 years; developmental olfactory changes documented
- BSIT (Brief Smell Identification Test): 12 items, validated as screening tool in primary care
12.7 Genetic and Molecular Advances
- Whole exome sequencing identifying mutations in olfactory receptor genes (OR genes) in familial anosmia — UPSIT provides phenotypic documentation
- PROKR2/PROK2 gene mutations (Kallmann syndrome) validated against UPSIT anosmia scores
13. UPSIT IN MEDICOLEGAL CONTEXT
MEDICOLEGAL VALUE OF UPSIT:
Claim: "I lost my smell after workplace chemical exposure"
↓
UPSIT administered
↓
Score documented:
┌─────────────────────────────────────┐
│ Score 15/40 → Severe loss │
│ Percentile 3rd for age/sex │
│ Consistent with toxic anosmia │
│ NOT malingering (score > 10) │
└─────────────────────────────────────┘
↓
Serial testing at 3, 6 months
↓
Objective, reproducible evidence
in court / compensation tribunal
- Provides objective (not self-reported) documentation
- Detects malingerers (scores below chance)
- Serial testing allows monitoring of genuine recovery vs. persistent disability
- Normative data allows comparison with expected performance for age/sex
14. SUMMARY TABLES
Table 1: UPSIT at a Glance
| Feature | Detail |
|---|---|
| Developed by | Doty et al., 1984, Univ. of Pennsylvania |
| Number of items | 40 |
| Booklets | 4 (10 items each) |
| Method | Microencapsulated scratch-and-sniff |
| Paradigm | 4-alternative forced-choice |
| Time | 10–15 minutes |
| Self-administered | Yes |
| Languages | 12+ |
| Normative data | ~4000 subjects |
| Score range | 0–40 |
| Malingering detection | Yes (chance = 10/40) |
| Sensitivity | ~85% for anosmia |
| Specificity | ~90% for normosmia |
Table 2: UPSIT Score Interpretation
| Score | Category | Action |
|---|---|---|
| 35–40 | Normal | Reassure |
| 30–34 | Mild loss | Monitor, treat underlying cause |
| 26–29 | Moderate loss | Investigate, MRI |
| 19–25 | Severe loss | Investigate further, neurology |
| ≤18 | Anosmia | Full workup: MRI, neurology |
| ≤5 | Probable malingering | Medicolegal assessment |
Table 3: UPSIT in Selected Diseases
| Disease | Typical UPSIT Score |
|---|---|
| Normal young adult | 35–38/40 |
| Chronic rhinosinusitis | 28–32/40 |
| Post-viral (COVID-19) | 18–24/40 |
| Post-traumatic | 15–22/40 |
| Parkinson's disease | 18–22/40 |
| Alzheimer's disease | 20–26/40 |
| Kallmann syndrome | 0–5/40 |
| Malingerer (claimed anosmia) | ≤5/40 |
| Unilateral anosmia (one nostril) | ~30–33/40 |
15. CONCLUSION
Key References
- Doty RL et al. Development and standardization of the University of Pennsylvania Smell Identification Test: A standardized microencapsulated test of olfactory function. Physiology & Behavior, 1984; 32(3):489–502
- Harrison's Principles of Internal Medicine, 21st Ed., McGraw-Hill — pp. 996, 1006–1007, 1013
- Cummings Otolaryngology: Head and Neck Surgery, 7th Ed. — Chapter: Olfaction and Gustation
- Scott Brown's Otorhinolaryngology: Head and Neck Surgery, 8th Ed. — Chapter: Disorders of Smell and Taste
- Dhingra PL, Dhingra S. Diseases of Ear, Nose and Throat, 8th Ed. — Chapter: Nose and Paranasal Sinuses
- Hazarika P et al. Textbook of ENT and Head & Neck Surgery — Chapter: Olfactory Disorders
- Stell & Maran's Textbook of Head & Neck Surgery — Chapter: Olfactory Assessment
- Hummel T et al. "Effects of olfactory training in patients with olfactory loss." Laryngoscope, 2009
- Doty RL. "The olfactory system and its disorders." Seminars in Neurology, 2009; 29:74–81
- Schenck CH et al. UPSIT and Parkinson's disease prodrome. Ann Neurol, 2002
Exam Tip (50-Mark Answer): For a 50-mark question, structure your answer with: Introduction → Anatomy/Physiology → Test Description (physical components, odorants, microencapsulation) → Administration (flowchart) → Scoring (tables + malingering detection) → Clinical Applications → Comparison with other tests → Textbook perspectives → Recent advances → Limitations. Use diagrams, flowcharts, and tables throughout. The examiner expects depth in all sections — especially the normative interpretation, malingering detection logic, and neurodegenerative disease applications.