Community acquired pneumonia- etiology, common pathogens based on age group, investigations and management, as per latest 2025-26 guidelines
community acquired pneumonia etiology pathogen age group classification
community acquired pneumonia investigations diagnosis severity scoring CURB-65 PSI
community acquired pneumonia chest X-ray radiograph findings
community acquired pneumonia antibiotic treatment management guidelines 2024 2025 empirical therapy
Community-Acquired Pneumonia (CAP)
Etiology | Pathogens by Age | Investigations | Management
1. Definition & Classification
| Category | Description |
|---|---|
| Non-severe (outpatient) | Mild symptoms, no hypoxia, ambulatory |
| Moderate (inpatient, non-ICU) | Requires hospitalization, not meeting severe criteria |
| Severe (ICU) | Meets ATS major/minor severity criteria (see below) |
2. Etiology
General Causative Agents
| Category | Pathogens |
|---|---|
| Typical bacteria | Streptococcus pneumoniae (most common), Haemophilus influenzae, Staphylococcus aureus (incl. MRSA), Klebsiella pneumoniae, Moraxella catarrhalis, Pseudomonas aeruginosa |
| Atypical bacteria | Mycoplasma pneumoniae, Chlamydophila pneumoniae, Legionella pneumophila |
| Viruses | Influenza A/B, RSV, SARS-CoV-2, Rhinovirus, Adenovirus, Metapneumovirus, Parainfluenza |
| Fungi (endemic) | Histoplasma capsulatum, Coccidioides immitis, Blastomyces dermatitidis |
| Aspiration-related | Oral anaerobes (Peptostreptococcus, Bacteroides, Fusobacterium) |
S. pneumoniae remains the single most common identifiable bacterial pathogen across all age groups (Harrison's, p. 3799).
3. Common Pathogens by Age Group
| Age Group | Most Common Pathogens |
|---|---|
| Neonates (0–28 days) | Group B Streptococcus (GBS), E. coli, Listeria monocytogenes, CMV |
| Infants (1–3 months) | Chlamydia trachomatis (afebrile pneumonia), RSV, S. pneumoniae, H. influenzae |
| Children (3 months – 5 years) | RSV, Parainfluenza, Rhinovirus, S. pneumoniae, H. influenzae type b |
| Children (5–15 years) | Mycoplasma pneumoniae (most common), Chlamydophila pneumoniae, S. pneumoniae |
| Young adults (15–40 years) | Mycoplasma pneumoniae, Chlamydophila pneumoniae, S. pneumoniae |
| Adults (40–65 years) | S. pneumoniae (predominant), H. influenzae, M. pneumoniae, Influenza viruses |
| Elderly (>65 years) | S. pneumoniae, H. influenzae, Gram-negative bacilli, S. aureus, Aspiration, Influenza, RSV |
| Immunocompromised | Pneumocystis jirovecii (PCP), Aspergillus, CMV, Nocardia, atypicals + all above |
Key point: "Walking pneumonia" in school-age children and young adults is predominantly Mycoplasma pneumoniae. In elderly and aspiration-prone patients, Gram-negatives and anaerobes gain prominence.
4. Investigations
4.1 Minimum Baseline Workup (All Patients)
| Investigation | Rationale |
|---|---|
| Chest X-ray (PA + lateral) | Confirm diagnosis, assess extent, identify complications (effusion, abscess, multilobar) |
| Pulse oximetry / ABG | Assess oxygenation; ABG if SpO₂ <92% or severe disease |
| CBC with differential | Leukocytosis/leukopenia; neutrophilia in bacterial, lymphopenia in viral |
| CRP / ESR | Inflammatory markers; CRP useful for monitoring response |
| Procalcitonin (PCT) | Helps distinguish bacterial from viral; guides antibiotic stewardship |
| Serum electrolytes, BUN, creatinine | Part of CURB-65/PSI; assess for end-organ involvement |
| LFTs | Legionella can cause hepatitis; drug toxicity monitoring |
| Blood glucose | Diabetes is a key risk factor |
| Blood cultures × 2 | Before antibiotics — mandated in moderate-severe/ICU CAP |
4.2 Severity Assessment Tools
| Variable | Points |
|---|---|
| Confusion (new) | 1 |
| Urea >7 mmol/L (BUN >20 mg/dL) | 1 |
| Respiratory rate ≥30/min | 1 |
| Blood pressure: systolic <90 or diastolic ≤60 mmHg | 1 |
| 65 years of age or older | 1 |
| Score | Mortality | Disposition |
|---|---|---|
| 0–1 | <3% | Outpatient |
| 2 | ~9% | Consider admission |
| 3–5 | 15–40% | Hospitalize (ICU if ≥4) |
Per ACEP 2025 guidelines: "The PSI and CURB-65 can support clinical judgement by identifying patients at low risk of mortality who may be appropriate for outpatient treatment. Although both are acceptable, the PSI is supported by a larger body of evidence and is preferred by ATS/IDSA." (ACEP CAP Guideline, p. 4)
- CLASS I–II: Outpatient
- CLASS III: Short stay/observation
- CLASS IV–V: Hospitalization (Class V = ICU consideration)
- RR ≥30, PaO₂/FiO₂ <250, multilobar infiltrates, confusion/disorientation, BUN ≥20, leukopenia (WBC <4000), thrombocytopenia (<100,000), hypothermia (<36°C), hypotension requiring aggressive fluid resuscitation
- Invasive mechanical ventilation, septic shock requiring vasopressors
4.3 Microbiology Workup — When to Order
| Test | Indication |
|---|---|
| Blood cultures ×2 | Moderate-severe, ICU, empirically immunocompromised, asplenic |
| Sputum C&S + Gram stain | Moderate-severe; productive cough; all ICU patients |
| Urinary Legionella antigen | Severe CAP, travel to endemic areas, outbreak setting |
| Urinary Pneumococcal antigen | Moderate-severe CAP (sensitivity 70–80%) |
| Respiratory viral PCR panel | All hospitalized patients (influenza, RSV, SARS-CoV-2, atypicals) |
| Mycoplasma/Chlamydia PCR | Young adults, atypical presentation, cluster outbreaks |
| Bronchoscopy + BAL | ICU, non-responding CAP, immunocompromised, diagnostic uncertainty |
| HIV serology | First episode pneumonia in young adults, recurrent/unusual CAP |
| Beta-D-glucan / Galactomannan | Suspected fungal pneumonia (immunocompromised) |
4.4 Radiological Findings
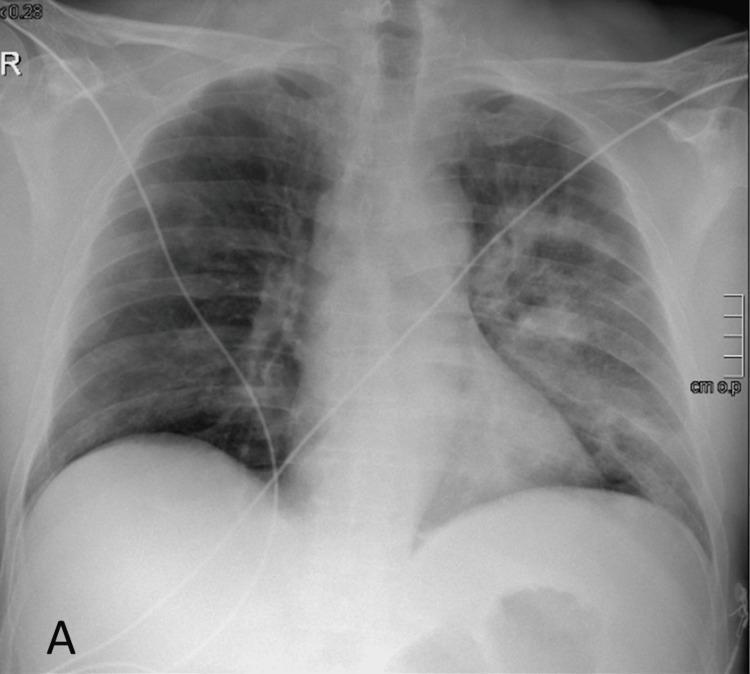
| Pattern | Likely Pathogen |
|---|---|
| Lobar/segmental consolidation | S. pneumoniae, K. pneumoniae |
| Bilateral interstitial / ground-glass | Viral (COVID-19, Influenza), PCP, Mycoplasma |
| Cavitation | S. aureus (MRSA), anaerobes, K. pneumoniae, TB |
| Patchy bronchopneumonia | H. influenzae, S. aureus, aspiration |
| Unilateral pleural effusion | S. pneumoniae (parapneumonic), anaerobes |
- CXR normal but clinical suspicion high
- Non-resolving pneumonia (>6 weeks)
- Recurrent pneumonia in same segment (exclude malignancy)
- Suspected complications (abscess, empyema)
5. Management
5.1 General Principles
- Risk stratify using PSI/CURB-65 before deciding on setting of care
- Start antibiotics within 4 hours of diagnosis (within 1 hour if septic shock)
- Oral bioequivalent agents may be substituted for IV once clinical stability achieved (temperature <37.8°C, HR <100, RR <24, SBP ≥90, SpO₂ ≥90% on room air, tolerating orals)
- Review and de-escalate based on culture results (antibiotic stewardship)
5.2 Empirical Antibiotic Therapy
OUTPATIENT (Non-severe, no comorbidities)
| Patient Type | Preferred Regimen | Alternative |
|---|---|---|
| Healthy, no recent antibiotics | Amoxicillin 1 g PO TDS × 5 days | Doxycycline 100 mg PO BD × 5 days |
| With comorbidities (COPD, DM, heart/liver/renal disease, malignancy, immunosuppression) | Amoxicillin-clavulanate 875/125 mg PO BD + Azithromycin/Clarithromycin | Respiratory fluoroquinolone (Levofloxacin 750 mg OD × 5d OR Moxifloxacin 400 mg OD × 5d) |
| Atypical suspected (young, walking pneumonia) | Azithromycin 500 mg day 1, then 250 mg OD × 4 days | Doxycycline 100 mg BD × 5 days |
INPATIENT — NON-ICU (Moderate)
| Preferred | Alternative |
|---|---|
| Beta-lactam + Macrolide (Amoxicillin-clavulanate/Ceftriaxone 1–2 g IV OD + Azithromycin 500 mg OD) | Respiratory fluoroquinolone monotherapy (Levofloxacin 750 mg OD or Moxifloxacin 400 mg OD) |
INPATIENT — ICU (Severe)
| Regimen | Indication |
|---|---|
| Ceftriaxone 2 g IV OD + Azithromycin 500 mg IV OD | Standard severe CAP |
| Ceftriaxone 2 g IV OD + Levofloxacin 750 mg IV OD | If macrolide contraindicated (e.g., QT prolongation) |
| Add Vancomycin 15–20 mg/kg IV Q8–12h OR Linezolid 600 mg IV BD | Suspected MRSA (post-influenza, cavitation, necrotizing pneumonia, skin/soft tissue MRSA history, nasal MRSA screen positive) |
| Add Piperacillin-tazobactam 4.5 g IV Q6h OR Cefepime 2 g IV Q8h | Suspected Pseudomonas (structural lung disease, bronchiectasis, recent hospitalization, prior Pseudomonas infection, immunocompromised) |
2024–2025 Update (IDSA/ATS): Procalcitonin-guided therapy is now recommended to guide duration and de-escalation. A PCT <0.25 µg/L at 48–72 hours supports antibiotic discontinuation in stable patients.
5.3 Specific Pathogen-Directed Therapy
| Pathogen | Drug of Choice | Alternative |
|---|---|---|
| S. pneumoniae (penicillin-sensitive) | Amoxicillin / Penicillin G | Ceftriaxone, Levofloxacin |
| S. pneumoniae (penicillin-resistant) | Ceftriaxone 2 g IV OD | Levofloxacin / Moxifloxacin |
| M. pneumoniae / C. pneumoniae | Azithromycin or Doxycycline | Levofloxacin / Moxifloxacin |
| Legionella pneumophila | Levofloxacin 750 mg OD × 5 days | Azithromycin (moderate disease) |
| MRSA | Vancomycin or Linezolid | Clindamycin (susceptible strains) |
| H. influenzae | Amoxicillin-clavulanate | Ceftriaxone, Levofloxacin |
| K. pneumoniae | Ceftriaxone or Cefepime | Piperacillin-tazobactam |
| Pseudomonas aeruginosa | Anti-pseudomonal beta-lactam ± fluoroquinolone/aminoglycoside | — |
| Influenza-associated | Oseltamivir 75 mg BD × 5 days (regardless of symptom duration in hospitalized) + antibacterials | — |
| PCP (P. jirovecii) | TMP-SMX (15–20 mg/kg/day of TMP IV/PO) ± Prednisolone if PaO₂ <70 mmHg | Pentamidine, Atovaquone |
5.4 Duration of Antibiotic Therapy
| Setting | Duration |
|---|---|
| Outpatient, non-severe | 5 days (clinical stability end point) |
| Inpatient, non-severe | 5–7 days |
| Severe/ICU (typical pathogens) | 7 days |
| Legionella | 5–7 days (fluoroquinolone) or 10–14 days (azithromycin) |
| S. aureus / MRSA | 7–21 days depending on bacteremia/empyema |
| Pseudomonas | 10–14 days |
| Lung abscess/aspiration | 3–6 weeks |
ATS/IDSA 2019 (reaffirmed 2023) emphasizes: minimum 5 days for most outpatient CAP if clinical stability is met — longer courses are NOT superior and increase resistance.
5.5 Supportive & Adjunctive Management
| Measure | Details |
|---|---|
| Oxygen therapy | Target SpO₂ ≥94% (92–95% in COPD); use HFNC if escalating O₂ needs |
| IV fluids | Judicious resuscitation in septic shock; avoid fluid overload |
| Corticosteroids | Methylprednisolone 0.5 mg/kg IV BD × 5 days in severe CAP (CRP >150 mg/L); reduces treatment failure and ICU stay (IDSA 2023 conditional recommendation) |
| DVT prophylaxis | LMWH in all hospitalized patients |
| Chest physiotherapy | Indicated in lobar collapse, mucus plugging |
| Nutrition | Early enteral nutrition in ventilated patients |
| Bronchodilators | If wheeze / bronchospasm (especially in COPD) |
5.6 Patients Not Responding to Treatment
- Confirm diagnosis (re-CXR, CT chest)
- Broaden coverage (MRSA, Pseudomonas, Legionella)
- Consider non-infectious causes (cryptogenic organising pneumonia, vasculitis, malignancy)
- Bronchoscopy + BAL for microbiological sampling
- Exclude parapneumonic effusion / empyema (USS / CT; drain if needed)
6. Criteria for ICU Admission (ATS/IDSA 2019)
- Invasive mechanical ventilation required
- Septic shock requiring vasopressors
7. Prevention
| Strategy | Details |
|---|---|
| Pneumococcal vaccine | PCV20 (preferred) or PCV15 + PPSV23 for all adults ≥65, and younger adults with high-risk conditions (ACIP 2024) |
| Influenza vaccine | Annual for all ≥6 months |
| COVID-19 vaccine | Updated seasonal boosters as per local guidelines |
| RSV vaccine | Abrysvo/mRESVIA — recommended for adults ≥60 years (FDA approved 2023–2024) |
| Smoking cessation | Reduces mucociliary dysfunction and aspiration risk |
| Aspiration precautions | Head elevation 30–45°, oral hygiene in high-risk patients |
8. Special Populations
| Population | Key Consideration |
|---|---|
| HIV/AIDS | Always consider PCP; atypical presentations common |
| Post-influenza | High risk of S. aureus (MRSA) — cover empirically |
| Alcoholism | K. pneumoniae, aspiration anaerobes |
| Structural lung disease | Anti-Pseudomonas coverage required |
| Splenectomia/asplenic | Encapsulated organisms (S. pneumoniae, H. influenzae, N. meningitidis) — rapid fatal course |
| Pregnancy | Avoid fluoroquinolones, tetracyclines; use macrolide + beta-lactam |
| Elderly | Atypical presentations (confusion, falls, no fever); high aspiration risk; GNR more common |
Key Takeaways
- CURB-65 / PSI guide disposition; PSI preferred by ATS/IDSA
- Standard outpatient CAP: Amoxicillin (healthy) or beta-lactam + macrolide / respiratory FQ (comorbidities) × 5 days
- Severe CAP: Ceftriaxone + Azithromycin; add MRSA or Pseudomonas cover based on risk factors
- Duration: 5 days for most cases when clinical stability is achieved — do not over-treat
- Procalcitonin: use to guide stewardship and de-escalation
- Corticosteroids: consider in severe CAP with CRP >150 — conditional IDSA 2023 recommendation
- Vaccination: PCV20 + annual influenza + RSV vaccine (≥60 yrs) for prevention
Sources: Harrison's Principles of Internal Medicine, 21st ed. (p. 3799); ACEP Critical Issues in Adult CAP Management (Emergency Department), 2025 (p. 4); ATS/IDSA Community-Acquired Pneumonia Guidelines 2019 (reaffirmed 2023); IDSA Practice Guidelines 2024 updates; ACIP Immunization Schedule 2024.
Give a complete summary of Asthma
Asthma: Comprehensive Overview
1. Definition & Epidemiology
- ~300 million people worldwide are affected
- Increasingly prevalent in developing countries
- 96% of asthma deaths occur in low- and middle-income countries (LMICs)
- In primary care, 25–35% of patients labeled as asthma may not actually have it — confirmation with objective tests is essential
2. Pathophysiology
- Bronchoconstriction — airways narrow
- Airway wall thickening due to inflammation and edema
- Increased mucus production
- Viral respiratory infections (most common)
- Allergens (house dust mite, pets, pollens, cockroach)
- Tobacco smoke/vaping
- Exercise, laughter, cold air
- Stress
- Beta-blockers, NSAIDs/aspirin (in susceptible individuals)
- Occupational sensitizers
3. Diagnosis
3.1 History of Variable Respiratory Symptoms
- Wheeze, breathlessness, chest tightness, cough
- Symptoms vary in frequency and intensity
- Worse at night or on waking
- Triggered by exercise, allergens, cold air, laughter
- Often worse during viral infections
3.2 Evidence of Variable Expiratory Airflow
| Test | Adults | Children |
|---|---|---|
| Bronchodilator reversibility (FEV₁ or FVC) | ↑ ≥12% AND ≥200 mL | ↑ ≥12% of predicted |
| PEF variability (2 weeks) | >10% | >13% |
| Response to 4 weeks ICS | ↑ FEV₁ ≥12% + ≥200 mL | ↑ FEV₁ ≥12% |
| Positive bronchial challenge | (methacholine, mannitol, exercise) | |
| Variation between visits | ≥12% and ≥200 mL | ≥12% |
3.3 Type 2 Biomarkers (Supportive)
- FeNO >50 ppb (adults) / >35 ppb (children) supports Type 2 asthma
- Blood eosinophils above reference range supports Type 2 asthma
- Lower levels do not rule out asthma
3.4 Special Diagnostic Populations
| Population | Key Point |
|---|---|
| Cough-variant asthma | Cough only; confirm with bronchial challenge; treat with ICS |
| Occupational asthma | Refer urgently; remove exposure immediately |
| Pregnancy | Continue ICS; defer bronchial challenge until post-delivery |
| Elderly | May be underdiagnosed; exclude heart failure, COPD |
| Asthma + COPD overlap | Worse outcomes; treat with ICS-containing regimen |
| LMICs | Use PEF if spirometry unavailable; exclude TB, HIV-related disease |
4. Assessment of Asthma Control (GINA Table 2)
4A. Symptom Control (past 4 weeks)
- Daytime symptoms >2×/week?
- Night waking due to asthma?
- SABA reliever needed >2×/week?
- Any activity limitation?
| Score | Level |
|---|---|
| None of these | Well controlled |
| 1–2 | Partly controlled |
| 3–4 | Uncontrolled |
4B. Risk Factors for Exacerbations
- Uncontrolled symptoms
- SABA over-use (≥3 × 200-dose canisters/year; ≥12/year = markedly increased death risk)
- Inadequate ICS use / poor adherence / incorrect technique
- Obesity, GERD, chronic rhinosinusitis, confirmed food allergy
- Allergen exposure, smoking, vaping, air pollution
- FEV₁ <60% predicted
- Elevated blood eosinophils or high FeNO
- Prior intubation or ICU admission for asthma
4C. Severity Classification
| Severity | Definition |
|---|---|
| Mild | Controlled with as-needed low-dose ICS-formoterol, or low-dose daily ICS |
| Moderate-severe | Requires medium–high dose ICS-LABA |
| Difficult-to-treat | Uncontrolled despite medium/high ICS + second controller |
| Severe | Still uncontrolled despite high-dose ICS-LABA with good adherence |
5. Management: General Principles
6. Pharmacological Treatment: Adults & Adolescents (≥12 years)
Two Treatment Tracks
| Feature | Track 1 (Preferred) | Track 2 (Alternative) |
|---|---|---|
| Reliever | Low-dose ICS-formoterol (AIR) | SABA or ICS-SABA |
| Advantage | Reduces severe exacerbations by ~65% vs SABA; simpler (1 inhaler, all steps) | Use if ICS-formoterol unavailable or patient stable and adherent on current regimen |
Track 1 — Preferred (ICS-Formoterol as Reliever)
| Step | Treatment |
|---|---|
| Steps 1–2 | AIR-only: as-needed low-dose ICS-formoterol |
| Step 3 | MART: low-dose ICS-formoterol once–twice daily + as-needed |
| Step 4 | MART: medium-dose ICS-formoterol twice daily + as-needed |
| Step 5 | Refer for expert phenotyping + add-on biologics/LAMA |
Track 2 — Alternative (SABA or ICS-SABA Reliever)
| Step | Treatment |
|---|---|
| Step 1 | No daily ICS; take low-dose ICS whenever SABA is taken |
| Step 2 | Low-dose daily ICS + as-needed SABA |
| Step 3 | Low-dose ICS-LABA daily + SABA/ICS-SABA as needed |
| Step 4 | Medium-dose ICS-LABA daily + SABA/ICS-SABA as needed |
| Step 5 | Refer for phenotyping + add-on treatment |
Step 5: Severe Asthma Add-on Options
| Agent | Indication | Age |
|---|---|---|
| Omalizumab (anti-IgE) | Severe allergic asthma | ≥6 years |
| Mepolizumab (anti-IL-5) | Severe eosinophilic asthma | ≥6 years |
| Benralizumab (anti-IL-5Rα) | Severe eosinophilic asthma | ≥12 years |
| Reslizumab (anti-IL-5) | Severe eosinophilic asthma | ≥18 years |
| Dupilumab (anti-IL-4Rα) | Severe eosinophilic / Type 2 / OCS-dependent | ≥6 years |
| Tezepelumab (anti-TSLP) | Severe asthma (any phenotype) | ≥12 years |
| LAMA (e.g. tiotropium) | Add-on to ICS-LABA; small improvement in lung function/exacerbations | ≥6 years |
7. Pharmacological Treatment: Children 6–11 Years
| Step | Preferred Treatment |
|---|---|
| Step 1 | Low-dose ICS whenever SABA taken |
| Step 2 | Daily low-dose ICS + as-needed SABA |
| Step 3 | Low-dose ICS-LABA, OR medium-dose ICS, OR very-low-dose ICS-formoterol MART |
| Step 4 | Medium-dose ICS-LABA OR low-dose ICS-formoterol MART; refer to specialist |
| Step 5 | Refer for phenotyping; biologics (omalizumab, mepolizumab, dupilumab); consider LAMA add-on |
8. Stepping Down Treatment
- Reduce ICS dose by 25–50%
- Do not completely stop ICS in adults/adolescents
- Review every 2–3 months before further step-down
- Ensure patient has a written action plan before stepping down
9. Non-Pharmacological Management
- Smoking/vaping cessation — at every visit
- Physical activity — encouraged; may slightly improve asthma control
- Allergen avoidance — only for confirmed sensitized patients
- Weight reduction — even 5–10% loss improves asthma control in obesity
- Manage comorbidities — allergic rhinitis (intranasal corticosteroids), GERD, OSA, anxiety/depression
- Occupational asthma — eliminate exposure; refer urgently
- Aspirin-exacerbated respiratory disease (AERD) — avoid all NSAIDs; consider LTRA add-on
- Written asthma action plan — every patient; include daily medications, when to step up, when to use OCS, when to seek emergency care
10. Managing Exacerbations
Self-Management (written action plan)
- Track 1 (ICS-formoterol): take extra doses as needed; seek care if not improving in 2–3 days or if needing >12 inhalations/day
- Track 2 (SABA): use SABA + quadruple ICS dose for 1–2 weeks; seek care if needing SABA again within 3 hours
Oral Corticosteroids (in action plan)
- Adults: prednisolone 40–50 mg/morning × 5–7 days
- Children: prednisolone 1–2 mg/kg/day (max 40 mg) × 3–5 days
- No taper needed if course <2 weeks
Primary Care / Emergency Management
| Severity | Features | Management |
|---|---|---|
| Mild–Moderate | Talks in phrases, SpO₂ 90–95%, PEF >50% | SABA 4–10 puffs by pMDI + spacer q20min × 3; OCS for moderate; controlled O₂ (target 93–95%) |
| Severe | Talks in words, SpO₂ <90%, PEF ≤50%, RR >30, HR >120 | Transfer to acute care; give SABA + ipratropium bromide + IV/IM corticosteroid; consider IV magnesium sulfate |
| Life-threatening | Drowsy, confused, silent chest | Immediate ICU transfer |
- Prescribe ICS-containing controller (prefer ICS-formoterol MART in adults)
- Check inhaler technique and adherence
- Provide written action plan
- Follow-up within 2–7 days (1–3 days for children)
11. Asthma in Specific Contexts
| Context | Key Point |
|---|---|
| Pregnancy | Monitor every 4–6 weeks; continue ICS (reduces preterm birth, low birth weight, perinatal mortality); ICS and β₂-agonists safe for fetus |
| COVID-19 | Well-controlled asthma → no increased risk of severe COVID-19; continue ICS; avoid nebulizers during infection |
| Elderly | Consider arthritis, eyesight, inspiratory flow when choosing inhaler; exclude cardiac causes of breathlessness |
| Surgery | Ensure good control pre-op; continue ICS peri-operatively; give hydrocortisone peri-operatively if on long-term high-dose ICS or recent OCS |
| Food allergy | Specialist testing; ensure anaphylaxis plan with epinephrine auto-injector |
12. Key Safety Points (GINA 2025)
- Never prescribe SABA alone — increases exacerbation risk and mortality
- SABA overuse is a red flag: ≥3 canisters/year → higher exacerbation risk; ≥12 canisters/year → markedly increased death risk
- Montelukast (LTRA) — associated with serious neuropsychiatric effects including suicidal ideation; counsel patients/caregivers
- LABA should never be used without ICS in asthma
- Maintenance OCS — last resort only; multiple short courses increase long-term risk of diabetes, osteoporosis, cataract, heart failure
- ICS side effects minimized by: rinsing mouth after use (candidiasis), using spacer with pMDI, lowest effective dose
Summarise the key changes in 2025 GInA updtae
GINA 2025 key changes updates asthma guidelines new recommendations
GINA 2025 new additions revisions inhaler biologic track management
GINA 2025: Key Changes at a Glance
1. Tezepelumab Added to Step 5 for All Phenotypes
- Tezepelumab (anti-TSLP) is now more prominently featured as a Step 5 add-on biologic, notable because it is effective regardless of eosinophil count or allergic status — the only approved biologic for unselected severe asthma phenotypes
- Age indication extended: recommended from ≥12 years
2. Dupilumab Expanded to Children ≥6 Years
- Dupilumab (anti-IL-4Rα/IL-13) indication broadened to children ≥6 years (previously ≥12 years in earlier GINA versions)
- Particularly relevant in OCS-dependent and Type 2 high severe asthma
3. Stronger Warnings Against SABA Monotherapy
- GINA 2025 reinforces that SABA-alone reliever is no longer recommended at any step
- Updated data confirms that patients using ≥12 SABA canisters/year have a markedly increased risk of asthma-related death
- Messaging strengthened: every prescription of SABA should be accompanied by an ICS-containing controller
4. ICS-Formoterol AIR Strategy Elevated at Steps 1–2
- As-needed ICS-formoterol (AIR: Anti-Inflammatory Reliever) is re-affirmed as the preferred reliever at Steps 1 and 2 for adults and adolescents ≥12 years
- Evidence cited: the GINA-endorsed SYGMA 1 & 2, Novel START, and PRACTICAL trials demonstrating superior exacerbation reduction (~65% fewer severe exacerbations vs SABA monotherapy)
- Reinforced as superior even for patients with infrequent symptoms
5. Revised SABA Overuse Thresholds Emphasised
- ≥3 × 200-dose canisters/year = increased exacerbation risk (previously less prominently stated)
- ≥12 canisters/year = markedly increased mortality risk — both figures now explicitly highlighted in risk factor tables
6. Updated Guidance on Oral Corticosteroids (OCS)
- GINA 2025 strengthens the "OCS-sparing" framing at Step 5
- Biologics are now positioned as the preferred Step 5 escalation before maintenance OCS
- Renewed emphasis on cumulative OCS burden and its systemic effects (diabetes, osteoporosis, cardiovascular disease, adrenal suppression)
- Clinicians are encouraged to actively track total lifetime OCS exposure
7. Inhaled Corticosteroid–LABA Safety
- Continued emphasis that LABAs must never be used without ICS in asthma
- Fixed-dose ICS-LABA combinations remain recommended; free-combination LABA remains contraindicated as monotherapy
8. Montelukast (LTRA) Neuropsychiatric Safety
- Strengthened language around FDA/EMA black box warnings for montelukast
- All patients and caregivers must be counselled about neuropsychiatric side effects (behavioral changes, depression, suicidal ideation) before initiating
- Montelukast no longer a preferred add-on at any step; reserved as an alternative when other options are unsuitable
9. COVID-19 and Respiratory Virus Guidance Updated
- Patients with well-controlled asthma on ICS do not have increased risk of severe COVID-19
- ICS should not be stopped during COVID-19 infection
- Nebulizers should be avoided during acute respiratory infections due to aerosol transmission risk — pMDI + spacer preferred
10. Environmental and Climate Change Framing
- New section highlighting the impact of climate change, air pollution, and wildfire smoke on asthma burden
- Clinicians encouraged to counsel patients on air quality index monitoring and avoidance during pollution events
- Thunderstorm asthma acknowledged as an emerging public health concern
11. FeNO and Blood Eosinophils — Refined Cut-offs
| Biomarker | Threshold | Interpretation |
|---|---|---|
| FeNO | >50 ppb (adults), >35 ppb (children) | Supports Type 2 / eosinophilic asthma |
| Blood eosinophils | ≥300 cells/µL | Predicts biologic response (IL-5 pathway) |
| Blood eosinophils | ≥150 cells/µL | Lower threshold for considering dupilumab |
12. Pediatric Asthma (6–11 years) — Clarifications
- Low-dose ICS-formoterol MART now explicitly listed as an alternative at Step 3 for children 6–11 years (not just adults)
- Greater emphasis on confirming diagnosis before escalating in children, given high rate of misdiagnosis
- Spacer use mandatory with pMDI in all children
Summary Table: What's New vs. Unchanged
| Area | GINA 2025 Change |
|---|---|
| Tezepelumab | Added Step 5; broader phenotype indication |
| Dupilumab | Expanded to ≥6 years |
| SABA monotherapy | Explicitly prohibited at all steps |
| AIR strategy (Steps 1–2) | Strengthened as preferred track |
| OCS | Biologics preferred before maintenance OCS |
| Montelukast | Black box warning counselling mandatory |
| FeNO/eosinophil thresholds | Refined cut-offs for biologic selection |
| Climate/environment | New dedicated guidance |
| COVID-19 | Updated — continue ICS, avoid nebulizers |
| Pediatric MART | Extended to children 6–11 at Step 3 |
Which antihistamine is safe to prescribe generlly
antihistamine safety profile second generation H1 antihistamine prescribing
cetirizine loratadine fexofenadine antihistamine comparison sedation cardiac safety
Antihistamines: Which is Safe to Prescribe Generally?
First-Generation vs. Second-Generation
| Feature | 1st Generation (e.g. chlorpheniramine, diphenhydramine, hydroxyzine) | 2nd Generation (e.g. cetirizine, loratadine, fexofenadine) |
|---|---|---|
| Sedation | High (crosses BBB) | Low to none |
| Anticholinergic effects | Yes (dry mouth, urinary retention, constipation, blurred vision) | Minimal |
| Performance impairment | Significant (driving, operating machinery) | Minimal |
| Sleep quality | Disrupted | Not affected |
| Cardiac safety | QTc prolongation risk (esp. high doses) | Generally safe; fexofenadine safest |
| Preferred in clinical use | No — avoid routinely | Yes |
The Key Second-Generation Options
| Drug | Sedation | Notes |
|---|---|---|
| Fexofenadine | None | Most non-sedating; no QTc risk; safest overall for driving/work |
| Loratadine / Desloratadine | Minimal | Once daily; good safety profile; safe in pregnancy (Category B) |
| Cetirizine / Levocetirizine | Mild (in some patients) | Slightly more sedating than loratadine; levocetirizine is the active isomer with cleaner profile |
| Bilastine | None | Newer; does not cross BBB; good for allergic rhinitis and urticaria |
| Rupatadine | Minimal | Anti-PAF activity as well; useful in urticaria |
General Recommendation
Fexofenadine 120–180 mg OD or Loratadine 10 mg OD are the safest choices for the general adult population — non-sedating, no anticholinergic effects, no significant drug interactions, and no QTc concerns.
Special Populations
| Population | Recommended Choice |
|---|---|
| Elderly | Fexofenadine or loratadine (avoid 1st gen — fall risk, urinary retention, confusion) |
| Pregnancy | Loratadine (most data; Category B) or cetirizine |
| Children | Cetirizine syrup (≥2 yrs), loratadine syrup (≥2 yrs), fexofenadine (≥6 yrs) |
| Drivers / pilots | Fexofenadine (only antihistamine approved in some aviation contexts) |
| Renal impairment | Fexofenadine — dose reduce; avoid cetirizine in severe renal failure |
| Hepatic impairment | Fexofenadine preferred (mainly renal excretion) |
| Cardiac patients | Fexofenadine or loratadine (avoid 1st gen entirely) |
When to Use First-Generation Antihistamines
- Acute urticaria/anaphylaxis adjunct (hydroxyzine, promethazine IM/IV)
- Night-time sedation in pruritic conditions where sedation is desirable
- Motion sickness (promethazine)
- Avoid routinely in elderly, drivers, or any patient requiring cognitive performance