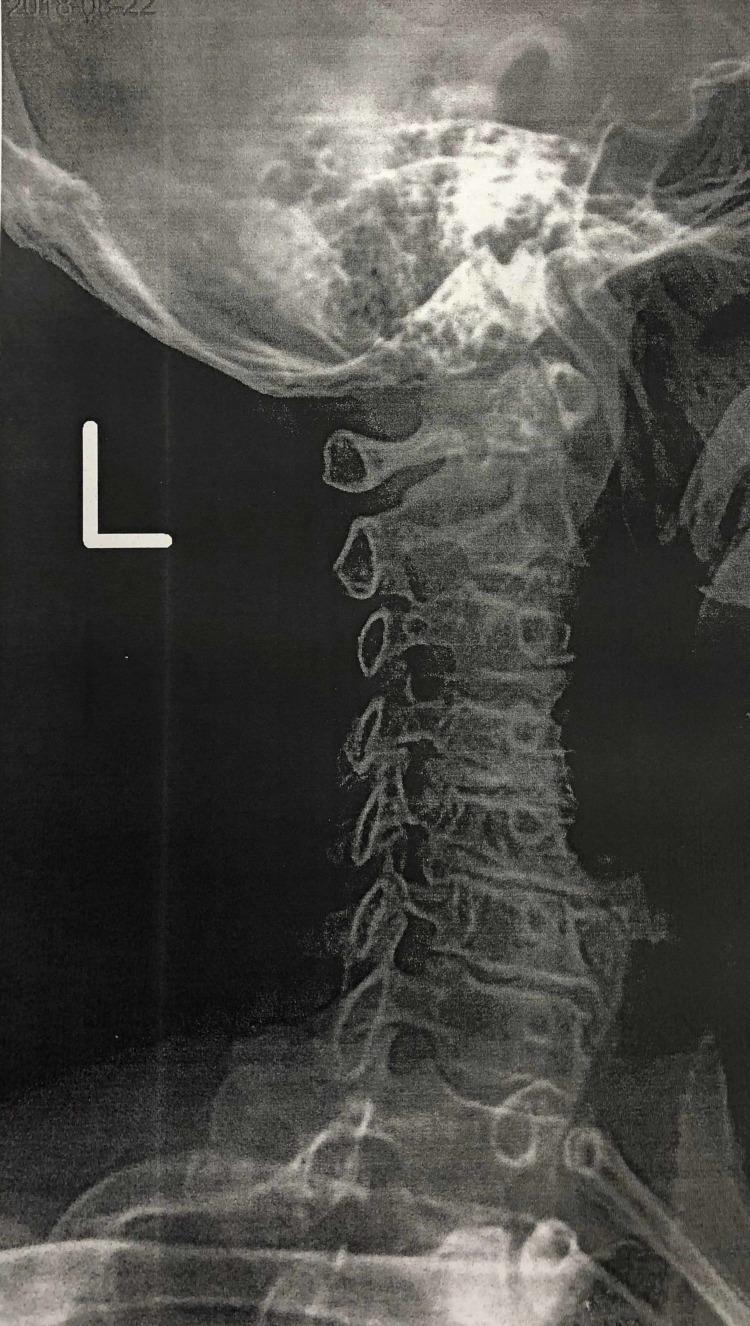
Potential findings in this X ray cervical spine in a 65 year old female of indian origin
cervical spine X-ray findings elderly degenerative changes spondylosis
cervical spine X-ray degenerative spondylosis osteophytes elderly
Cervical Spine X-Ray Analysis — 65-Year-Old Indian Female
Disclaimer: This is an educational analysis only. Formal radiological reporting must be performed by a qualified radiologist with the original digital images.
Image Overview
Probable Radiographic Findings
1. Cervical Spondylosis (Most Prominent Finding)
- Osteophyte formation (bone spurs) at multiple levels, particularly C4–C7 — visible as bony projections off vertebral endplates
- Intervertebral disc space narrowing — reduced height between vertebral bodies, most pronounced at C5/C6 and C6/C7 (the most commonly affected levels)
- Subchondral sclerosis — increased opacity/whitening of vertebral endplates
Degenerative cervical changes are present in ~95% of individuals by age 65 (Epidural Interventions in the Management of Chronic Spinal Pain, p. 36)
2. Reduction/Loss of Cervical Lordosis
- The normal "C-shaped" forward curve of the cervical spine may appear straightened or even reversed (kyphotic), a common finding in degenerative disease and muscle spasm
3. Foraminal Narrowing
- Lateral osteophytes encroaching on the neural foramina (uncovertebral joint hypertrophy), potentially causing cervical radiculopathy
4. Facet Joint Changes
- Posterior element arthropathy — sclerosis and narrowing of facet joints, visible in the lateral view
5. Osteoporosis/Osteopenia (Age/demographic-specific)
- Indian post-menopausal women have a high prevalence of osteoporosis
- Radiographic signs: generalized decreased bone density, biconcave ("fish vertebrae") or wedge-shaped vertebral bodies
- Vertebral compression fracture at any level should be excluded
6. DISH (Diffuse Idiopathic Skeletal Hyperostosis) (Differential)
- Flowing ossification along the anterior aspect of ≥4 contiguous vertebrae
- Relatively preserved disc height (distinguishes from spondylosis)
- Common in older South Asian patients with metabolic syndrome/diabetes
7. OPLL (Ossification of the Posterior Longitudinal Ligament) (Important differential in Asian population)
- Higher prevalence in East/South Asian patients
- Appears as a dense linear opacity posterior to the vertebral bodies on lateral view
- Can cause significant cord compression
Reference Image — Cervical Spondylosis
Summary Table
| Finding | Likelihood | Clinical Significance |
|---|---|---|
| Cervical spondylosis (C5–C7) | Very High | Neck pain, radiculopathy, myelopathy |
| Loss of lordosis | High | Postural/muscular component |
| Foraminal narrowing | High | Radiculopathy (arm pain/numbness) |
| Osteoporosis/osteopenia | High | Fracture risk |
| Facet arthropathy | High | Axial neck pain |
| OPLL | Moderate (Asian) | Cord compression risk |
| DISH | Low–Moderate | Usually incidental |
Clinical Correlation Suggested
- MRI cervical spine for cord/nerve root compression assessment
- DEXA scan for bone mineral density (high-risk demographic)
- Neurology/orthopedics referral if radicular or myelopathic symptoms are present
Causes and pharmacological management of binge eating, more at night in a 25 year old female
binge eating disorder causes etiology young female pathophysiology
pharmacological treatment binge eating disorder medications
night eating syndrome nocturnal binge eating causes management
binge eating disorder SSRIs topiramate lisdexamfetamine pharmacotherapy evidence
Binge Eating (Nocturnal Pattern) in a 25-Year-Old Female
Distinguishing the Syndrome
| Feature | Binge Eating Disorder (BED) | Night Eating Syndrome (NES) |
|---|---|---|
| Timing | Any time, but often evenings | After dinner / nocturnal awakenings |
| Loss of control | Core feature | Less prominent |
| Awareness during episode | Present | May be reduced |
| Morning anorexia | Absent | Characteristic |
| DSM-5 recognition | Yes | Not a formal DSM-5 diagnosis |
| Stress/mood link | Strong | Strong |
Causes & Contributing Factors
1. Neurobiological
- Dysregulation of the mesolimbic dopamine reward system — binge eating activates the same reward circuitry as substance use; cravings are reinforced by dopamine release
- Serotonin deficiency — reduced 5-HT activity impairs satiety signaling and mood regulation, driving carbohydrate/sugar craving particularly at night
- Circadian rhythm disruption — nocturnal eating is linked to phase-delayed circadian clocks; cortisol and melatonin dysregulation alter hunger hormones
- Leptin/ghrelin imbalance — ghrelin (hunger hormone) peaks are dysregulated; leptin resistance impairs satiety
2. Psychological
- Depression and dysthymia — higher depression scores correlate with more severe binges (IFSO-WGO Obesity Guidelines, p. 65); lowered mood and energy are the most common binge triggers
- Anxiety disorders — eating as emotional regulation / anxiolytic behavior
- Negative affect cascade — anger, loneliness, guilt, frustration, irritability are strongly associated with BED episodes, particularly in interpersonal contexts (IFSO-WGO Obesity Guidelines, p. 65)
- Emotional dysregulation — deficits in identifying and tolerating emotions (alexithymia-spectrum)
- Body image disturbance — pervasive concerns about weight/shape drive restrict-binge cycles
3. Behavioral / Lifestyle
- Daytime dietary restriction — skipping meals or under-eating during the day leads to compensatory hyperphagia at night (the "restrict-binge cycle")
- Screen time / late-night stimulation — delays sleep onset, prolongs exposure to food cues
- Stress and poor sleep — sleep deprivation elevates ghrelin and reduces leptin, dramatically increasing appetite and impulsivity
4. Hormonal (Female-specific)
- Premenstrual phase — progesterone and estrogen fluctuations in the luteal phase significantly increase carbohydrate cravings and binge likelihood
- Polycystic ovary syndrome (PCOS) — highly prevalent in young Indian women; insulin resistance and androgen excess drive appetite dysregulation and BED comorbidity
- Hypothyroidism — subclinical hypothyroidism increases appetite, fatigue, and mood disturbance
5. Psychiatric Comorbidities
- 64–79% of patients with BED have a concurrent psychiatric disorder (IFSO-WGO Obesity Guidelines, p. 65)
- Most common: Major Depressive Disorder, Generalized Anxiety Disorder, ADHD
- Substance use disorders (including nicotine) are also common
Pharmacological Management
First-Line
Lisdexamfetamine (Vyvanse) — Only FDA-approved drug for BED
- Dose: 50–70 mg once daily (morning)
- Mechanism: Prodrug of dextroamphetamine; reduces impulsivity and food reward salience via dopamine/norepinephrine reuptake inhibition
- Evidence: Significantly reduces binge days/week vs. placebo in RCTs
- Cautions: Controlled substance (Schedule II), cardiovascular monitoring, abuse potential, not suitable if anxiety is predominant; avoid in hypertension
Second-Line / Off-Label
Topiramate
- Dose: 25 mg/day titrated to 100–300 mg/day
- Mechanism: Glutamate antagonism + GABA potentiation; reduces reward salience and food craving
- Evidence: Reduces binge frequency and weight; one of the better-evidenced options
- Cautions: Cognitive side effects ("topamax stupidity"), teratogenic (contraception mandatory in women of childbearing age), kidney stones, paresthesias
SSRIs — Fluoxetine, Sertraline, Escitalopram
- First choice when depression/anxiety is a prominent comorbidity
- Fluoxetine 60 mg/day has the most evidence for reducing binge frequency in BED
- Sertraline 50–200 mg/day — also reduces binge episodes and weight
- Onset: 4–6 weeks; generally well tolerated
- Particularly useful in this age group given the safety profile
SNRIs — Venlafaxine, Duloxetine
- Useful when depression + pain or anxiety comorbidity present
- Reduces binge frequency; modest weight-neutral to weight-loss effect
Naltrexone/Bupropion (Contrave)
- Mechanism: Bupropion activates POMC neurons; naltrexone blocks opioid-mediated reward from eating
- Reduces binge frequency and promotes weight loss
- Avoid in patients with seizure risk or bulimia nervosa history
Melatonin (specifically for nocturnal/NES pattern)
- 3–5 mg at bedtime
- Addresses circadian dysregulation contributing to nighttime eating
- Safe, adjunctive role
Drugs to Avoid
| Drug | Reason |
|---|---|
| Benzodiazepines | Disinhibit eating behavior, dependence risk |
| Olanzapine/Quetiapine | Significant weight gain, worsens BED |
| Tricyclic antidepressants | Weight gain, carbohydrate craving |
Practical Pharmacotherapy Algorithm
Step 1: Assess comorbidities
├── Depression/Anxiety prominent → SSRI (Fluoxetine 60mg or Sertraline)
├── ADHD comorbid → Lisdexamfetamine (addresses both)
├── Obesity + binge → Topiramate or Naltrexone-Bupropion
└── Predominantly nocturnal → Add Melatonin adjunctively
Step 2: Reassess at 8–12 weeks
└── Inadequate response → Augment or switch class
Step 3: Always combine with psychological therapy (CBT is gold standard)
Key Non-Pharmacological Measures (Essential Adjuncts)
- CBT (Cognitive Behavioral Therapy) — strongest evidence base; targets the restrict-binge cycle and emotional dysregulation
- Structured meal timing — regular breakfast/lunch prevents compensatory evening hyperphagia
- Sleep hygiene — treat insomnia; inadequate sleep is a major driver of nighttime eating
- Rule out PCOS, hypothyroidism — basic labs: TSH, fasting insulin, testosterone, DHEA-S
- Food diary — identify emotional triggers
Clinical note: In a 25-year-old female, before initiating pharmacotherapy, screen for PCOS and thyroid dysfunction (both common in young Indian women and both drive disordered eating), assess for depression/anxiety formally, and confirm the diagnosis (BED requires ≥1 binge/week for 3 months with marked distress). Lisdexamfetamine remains the only FDA-approved agent, but SSRIs with concurrent CBT are often the most practical and safe first step in this demographic.
Plain MRI and contrast MRI findings in early Parkinson disease
MRI findings early Parkinson disease conventional plain MRI brain
MRI brain Parkinson disease substantia nigra findings
substantia nigra MRI neuromelanin sensitive imaging Parkinson nigrosome
Parkinson disease brain MRI dopaminergic neurodegeneration imaging
MRI Findings in Early Parkinson's Disease
Key principle: Conventional CT/MRI brain should not be used routinely to diagnose Parkinson's disease (Parkinson Disease guidelines, p. 3). Standard MRI is often normal in early PD — its primary roles are to exclude mimics and to detect specific advanced sequences that support diagnosis.
Plain (Conventional) MRI Findings
Standard Sequences (T1, T2, FLAIR) — Early PD
| Structure | Finding | Clinical Significance |
|---|---|---|
| Substantia nigra (SN) | Loss of normal T2 hypointensity (iron signal); reduced volume | Reflects dopaminergic neuron loss in SNc |
| Nigrosome-1 | Loss of "swallow tail" sign on SWI/T2* | Highly specific for PD (see below) |
| Midbrain | Normal or mildly reduced midbrain area | Atrophy more prominent in atypical parkinsonism |
| Locus coeruleus | Subtle volume loss on neuromelanin-sensitive sequences | Early PD involvement |
| Cortex/white matter | Normal in early PD | Differentiates from DLB, vascular parkinsonism |
| Ventricles | Normal | Hydrocephalus excludes NPH as mimic |
The "Swallow Tail" Sign — Most Important Plain MRI Finding
- Normal brains show a bilateral comma/swallow-tail shaped hyperintensity in the posterior SN — representing nigrosome-1, a cluster of dopaminergic neurons rich in neuromelanin
- In early PD, this hyperintensity is absent or reduced unilaterally or bilaterally — called "loss of the swallow tail sign"
- Sensitivity ~90%, Specificity ~90% for PD vs. controls
- Can be detected even in early/prodromal disease
Neuromelanin-Sensitive MRI (NM-MRI) — Emerging Sequence
- Uses T1-weighted inversion recovery sequence tuned to neuromelanin's paramagnetic properties
- In healthy controls: bright (hyperintense) SNc due to neuromelanin accumulation in dopaminergic neurons
- In early PD: progressive reduction in NM-MRI signal in SNc, correlating with neuron loss
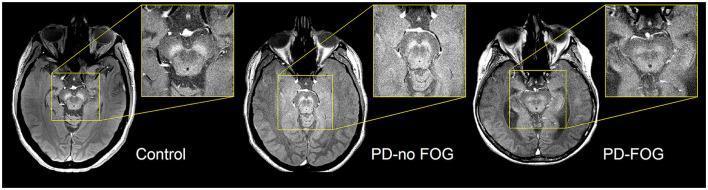
Contrast-Enhanced MRI Findings
Early PD — Contrast MRI
| Finding on Contrast MRI | Interpretation |
|---|---|
| No enhancement in SN or basal ganglia | Consistent with PD (no blood-brain barrier breakdown) |
| Leptomeningeal/parenchymal enhancement | Suggests encephalitis, CNS vasculitis — NOT PD |
| Ring-enhancing lesion in basal ganglia | Abscess, toxoplasmosis, metastasis — excludes PD |
| White matter enhancement | Inflammatory demyelination (MS mimicking PD) |
| No diffusion restriction | DWI normal in PD; restriction seen in CJD (important mimic) |
Advanced/Specialized MRI Techniques in Early PD
| Technique | Finding in Early PD | Utility |
|---|---|---|
| SWI (Susceptibility Weighted Imaging) | Loss of swallow-tail sign; increased iron deposition in SN | Best available conventional biomarker |
| Neuromelanin MRI (NM-MRI) | Reduced SNc signal volume | Correlates with dopaminergic neuron count |
| DTI (Diffusion Tensor Imaging) | Reduced fractional anisotropy in SN and nigrostriatal tract | Research; detects microstructural degeneration |
| MRI volumetry | Mild SN volume reduction | Less reliable in early disease |
| MRS (MR Spectroscopy) | Reduced NAA/Cr ratio in SN | Reflects neuronal loss/dysfunction |
| fMRI (resting state) | Altered default mode network connectivity | Research tool; not clinical standard |
| Quantitative SWI (R2)* | Elevated R2* (iron) in SN | Quantitative biomarker of SN iron accumulation |
Role of MRI in Differentiating PD from Mimics
| Condition | Key MRI Finding Differentiating from PD |
|---|---|
| Multiple System Atrophy (MSA-P) | Putaminal hypointensity + "hot cross bun" sign in pons |
| Progressive Supranuclear Palsy (PSP) | Midbrain atrophy — "hummingbird sign" on sagittal; reduced midbrain:pons ratio |
| Corticobasal Degeneration (CBD) | Asymmetric parietal cortical atrophy, high T2 signal in SN |
| Vascular Parkinsonism | Periventricular/subcortical white matter hyperintensities (FLAIR) |
| Normal Pressure Hydrocephalus | Ventriculomegaly disproportionate to sulcal atrophy |
| Wilson's Disease | T2 hyperintensity in basal ganglia, putamen, thalamus |
| DLB | Relatively preserved hippocampal volume (vs. AD); clinical overlap |
Summary
Early PD on MRI:
├── Conventional T1/T2/FLAIR → Often NORMAL
├── SWI/T2* at 3T → Loss of "swallow tail" (nigrosome-1) ← Most useful
├── Neuromelanin MRI → Reduced SNc hyperintensity
├── Contrast MRI → No enhancement (used to exclude mimics only)
├── DWI → Normal (abnormal DWI → think CJD)
└── Advanced (DTI, MRS, fMRI) → Research/specialist use