Latest guidelines in Bipolar disorder
Reading File
Checking Date & Time
Finding Sources
Searching PubMed
"bipolar disorder" AND "guidelines"
Searching the Web
bipolar disorder guidelines 2024 2025 APA CANMAT WFSBP latest recommendations
Finding Sources
Searching PubMed
"bipolar disorder" AND "treatment" AND "recommendations"
Reading File
Reading Article
Searching PubMed
"bipolar disorder" AND "CANMAT OR WFSBP OR APA"
Reading File
Reading Article
Reading Article
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Here is a comprehensive synthesis of current guidelines and evidence for the management of bipolar disorder.
Bipolar Disorder: Current Guidelines & Evidence
Synthesized from The Maudsley Prescribing Guidelines (15th ed.), Goldman-Cecil Medicine, Kaplan & Sadock's Comprehensive Textbook of Psychiatry, Goodman & Gilman's Pharmacology, and recent PubMed literature (2023–2024).
1. Diagnosis & Classification (DSM-5-TR)
The core diagnostic requirement for Bipolar I is ≥1 manic episode lasting ≥7 days (or any duration requiring hospitalization), with:
- Elevated/expansive/irritable mood plus increased goal-directed activity or energy
- ≥3 additional symptoms: grandiosity, decreased sleep need, pressured speech, flight of ideas, distractibility, psychomotor agitation, reckless behavior
Bipolar II requires at least one hypomanic episode (≥4 days, not requiring hospitalization) and at least one major depressive episode — never a full manic episode.
12-month prevalence: ~0.6% (BD-I); collectively the bipolar spectrum affects >2% of the population.
2. Acute Mania / Hypomania
First-line treatment
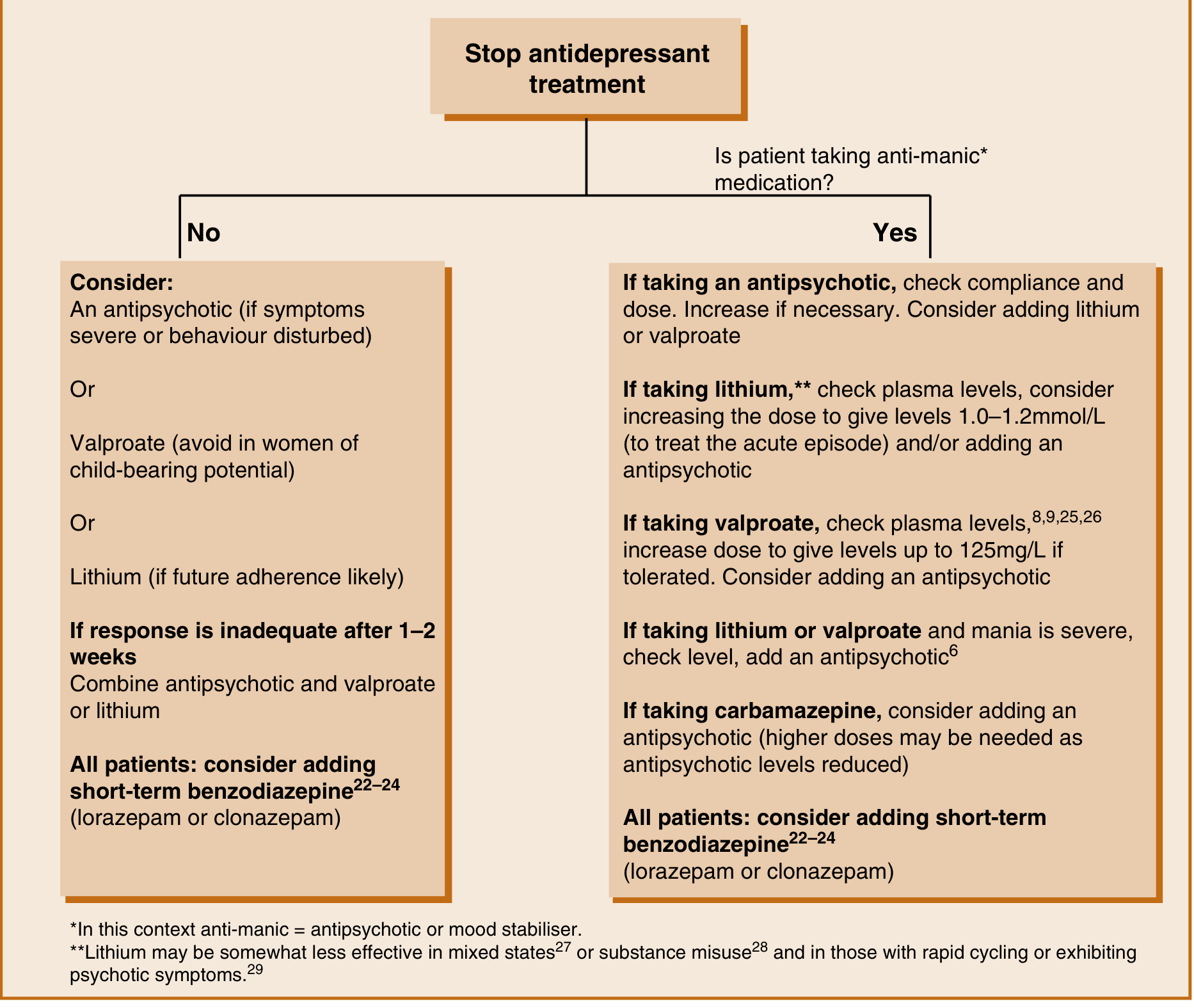
- Stop any antidepressants — this is the first step upon onset of mania (see flowchart below)
- SGAs are more rapidly efficacious than mood stabilizers for acute mania; efficacy across agents is broadly similar in network meta-analyses, with risperidone possibly showing a slight edge
- Adjunctive SGA + mood stabilizer is more effective than monotherapy with either alone
Monotherapy options (approved for acute mania)
| Drug | Notes |
|---|---|
| Aripiprazole | Effective alone or as add-on; nausea, akathisia common |
| Asenapine (sublingual) | Effective in mania; lower metabolic burden |
| Cariprazine | Effective in mania + mixed features; low weight gain |
| Olanzapine | Effective; superior to lithium in some studies; significant metabolic effects |
| Quetiapine | Robust efficacy; low EPSEs; metabolic effects |
| Risperidone | Particularly effective in combination; LAI not suitable for acute phase |
| Ziprasidone | FDA-approved for mania |
| Haloperidol | FGA; still used; higher risk of tardive dyskinesia and depression |
| Lithium (600–1,500 mg/day; target 1.0–1.2 mmol/L for acute episode) | Somewhat less effective in mixed states or rapid cycling |
| Valproate (target 50–125 mg/L) | Useful; avoid in women of childbearing potential |
| Carbamazepine (400–1,200 mg/day) | Slightly less effective than lithium |
- Add short-term benzodiazepine (lorazepam or clonazepam) for agitation in all patients
- If inadequate response after 1–2 weeks: combine antipsychotic + valproate or lithium
3. Bipolar Depression
Bipolar depression accounts for the majority of symptomatic illness over a lifetime and carries a ~15% suicide rate. It is more severe, more frequent, and more treatment-resistant than unipolar depression.
Guideline-specific recommendations
| Guideline | First-Line | Second-Line | Notes |
|---|---|---|---|
| NICE (UK) | Quetiapine monotherapy OR olanzapine + fluoxetine | Lamotrigine | Assumes no antipsychotic already prescribed |
| BAP | Lamotrigine, lurasidone, quetiapine | + mood stabiliser for mania protection | Caveat: need mood stabiliser cover |
| CANMAT/ISBD 2023 | Quetiapine, lurasidone (adjunct), lithium, lamotrigine | Olanzapine+fluoxetine, lumateperone, valproate (2nd-line) | Olanzapine demoted to 3rd-line |
| RANZCP 2020 | Lithium, lamotrigine, valproate, quetiapine, lurasidone, cariprazine | Olanzapine, carbamazepine |
Drugs with strong evidence for bipolar depression (FDA-licensed or guideline-endorsed)
| Drug | Mechanism/Notes |
|---|---|
| Quetiapine | Most evidence; robust in mania + depression + maintenance; metabolic side effects |
| Lurasidone | FDA-licensed; monotherapy or adjunct to Li/valproate; minimal weight gain, nausea/akathisia |
| Cariprazine | FDA-licensed; effective in mixed features; low weight gain |
| Lumateperone | Newer FDA-licensed agent; effective in bipolar I and II depression |
| Olanzapine + fluoxetine | Highest effect size in 2023 network meta-analysis (101 RCTs, Lancet Psychiatry [PMID 37595997]); weight gain concern |
| Lamotrigine | Effective as add-on in acute depression (SMD −0.30) and maintenance (RR 0.84 vs. placebo); titrate slowly to minimize SJS risk (start 25 mg/day, max 200 mg/day) |
| Lithium | Proven anti-suicidal effect; useful especially as adjunct |
⚠️ Antidepressants: May precipitate mania/rapid cycling. Use at minimum dose for minimum duration, only when a mood stabilizer is already in place. Not recommended as monotherapy.
Efficacy ranking (2023 network meta-analysis, 101 RCTs, Lancet Psychiatry [PMID 37595997]):
Olanzapine/fluoxetine > Quetiapine > Olanzapine > Lurasidone > Lumateperone > Cariprazine > Lamotrigine
4. Maintenance / Prophylaxis
Goal: prevent recurrence of both manic and depressive episodes.
Evidence hierarchy for maintenance
- Lithium — gold standard; best evidence for preventing mania and depression; proven anti-suicide effect (Cipriani et al.); lithium + valproate combination is superior to valproate alone (BALANCE trial)
- Quetiapine — robust evidence for prevention of both poles
- Valproate — evidence for both poles, but monotherapy inferior to lithium (BALANCE); avoid in women of childbearing potential
- Lamotrigine — primarily protects against depressive recurrence; 2024 meta-analysis (PMID 38750644): RR 0.84 vs. placebo; comparable to lithium in relapse prevention
- Aripiprazole — FDA-approved for maintenance; LAI effective (predominantly prevents mania)
- Olanzapine — effective; metabolic burden limits long-term use
- Carbamazepine — somewhat less effective than lithium; significant drug interactions
Key principle: Continue the acute-phase regimen that worked. Withdrawing antipsychotics from a Li/valproate combination may worsen relapse risk.
Long-acting injectables (LAIs)
- Aripiprazole LAI (FDA-approved for maintenance): reduces relapses significantly vs. placebo (26.5% vs. 51.1%); predominantly protects against mania
- Risperidone LAI: effective for manic/hypomanic episodes; does not protect against depressive relapse
- Consider LAIs when adherence is a concern
5. Rapid Cycling
Defined as ≥4 mood episodes per year. Management:
- Lithium may be less effective; combination therapy often needed
- Valproate, lamotrigine, olanzapine used
- Identify and correct precipitants: hypothyroidism, antidepressant use, substance misuse, sleep disruption
6. Special Populations
Pregnancy & Postpartum (ACOG CPG No. 5, CANMAT 2024 [PMID 39936923])
- Valproate: contraindicated (teratogenicity, neurodevelopmental effects)
- Lithium: use with caution (Ebstein anomaly risk is lower than historically thought but still monitor; fetal echocardiography recommended)
- Lamotrigine: relatively safer option
- ECT: considered for severe, refractory cases
- Risk of relapse postpartum is very high — do not abruptly discontinue mood stabilizers
Late-onset Bipolar
- New-onset mania in midlife/later life is often secondary to medical conditions (CNS tumors, steroids, thyroid disease, vascular events) — investigate thoroughly before diagnosing idiopathic bipolar disorder
7. Non-Pharmacological Treatments
- Psychotherapy: CBT, family-focused therapy, interpersonal and social rhythm therapy (IPSRT), and psychoeducation are adjunctive standard of care — they support adherence, reduce relapse triggers, and improve psychosocial functioning
- ECT: Effective for refractory mania or depression, catatonia, or when medication is contraindicated (e.g., pregnancy)
- Lifestyle: Regular sleep-wake cycles are critical — phase advance of circadian rhythms can precipitate mania; light exposure management, sleep hygiene, exercise
- rTMS / dTMS: Emerging evidence (systematic review [PMID 38759496]); not yet guideline-endorsed as primary treatment
8. Physical Monitoring (Maudsley 15th ed.)
| Monitoring | Lithium | Valproate | Carbamazepine | Antipsychotics |
|---|---|---|---|---|
| Thyroid (TFTs) | Baseline + every 6 months | — | — | — |
| Renal (eGFR) | Baseline + every 6 months | — | — | — |
| LFTs | — | Every 3 months × 1yr, then annual | Monthly × 3 months, then annual | — |
| FBC | If indicated | Every 3 months × 1yr, then annual | Monthly × 3 months, then annual | — |
| Plasma levels | Target 0.6–1.2 mmol/L (up to 1.4 in acute mania) | Target 50–100 μg/mL (up to 125 in acute mania) | Target 4–12 μg/mL | — |
| Glucose | — | — | — | Baseline, then every 4–6 months |
| Lipids | — | — | — | Baseline + 3 months |
| ECG | If cardiovascular risk | If cardiovascular risk | If cardiovascular risk | If cardiovascular risk |
| BMI/waist | Annual | Annual | Annual | Monthly × first 3 months |
Key Recent Evidence Updates
| Study | Finding |
|---|---|
| Lancet Psychiatry 2023 (NMA, 101 RCTs) [PMID 37595997] | Olanzapine/fluoxetine, quetiapine, olanzapine, lurasidone, lumateperone, cariprazine, and lamotrigine all superior to placebo for bipolar depression with moderate-confidence evidence; antidepressants carry higher manic switch risk |
| Bipolar Disord. 2024 (meta-analysis, PMID 38750644) | Lamotrigine effective as add-on for acute depression (SMD −0.30) and maintenance (RR 0.84 vs. placebo); comparable to lithium for relapse prevention |
| CANMAT/ISBD 2023 update [PMID 38695002] | Updated first-line recommendations; lumateperone added; valproate demoted to 2nd-line for bipolar depression |
| CANMAT Perinatal Guidelines 2024 [PMID 39936923] | Specific guidance for managing BD during pregnancy/postpartum |
Sources: The Maudsley® Prescribing Guidelines in Psychiatry (15th ed.) | Goldman-Cecil Medicine | Goodman & Gilman's Pharmacological Basis of Therapeutics | Kaplan & Sadock's Comprehensive Textbook of Psychiatry | Lancet Psychiatry 2023 [PMID 37595997] | Bipolar Disord. 2024 [PMID 38750644] | CANMAT/ISBD 2023 [PMID 38695002]
What does CANMAT 2023 guidelines say
Checking Date & Time
Searching PubMed
CANMAT ISBD 2023 bipolar disorder guidelines
Reading Article
Fetching Full Text
Finding Sources
Reading File
Reading File
Searching Content
Finding Sources
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.