Explain
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Your notes cover glycogenolysis (glycogen breakdown) and its regulation. Here's a full explanation of everything on the page:
Top Section — Glycogenolysis Pathway (in Liver)
This is the end-stage of glycogen breakdown converting stored glycogen to free glucose for release into the blood:
| Step | Enzyme | Product |
|---|---|---|
| Glycogen (linear branches) | Glycogen phosphorylase | Glucose 1-Phosphate |
| Glucose 1-P | Phosphoglucomutase | Glucose 6-Phosphate |
| Glucose 6-P | Glucose 6-phosphatase (liver only) | Free Glucose |
Key point: Muscle lacks glucose 6-phosphatase, so it cannot release free glucose into the blood — glucose stays trapped in muscle for local use. Only the liver can complete this pathway to release free glucose.
Regulation — The Core Concept
Your notes outline 3 layers of regulation:
(1) Two Key Enzymes
- Glycogen synthase — builds glycogen (glycogenesis)
- Glycogen phosphorylase — breaks down glycogen (glycogenolysis)
These two enzymes are reciprocally regulated — when one is ON, the other is OFF.
(2) Two States: Dephosphorylated (DP) vs. Phosphorylated (P)
| Enzyme | Dephosphorylated (DP) state | Phosphorylated (P) state |
|---|---|---|
| Glycogen synthase | Active (builds glycogen) | Inactive |
| Glycogen phosphorylase | Inactive | Active (breaks down glycogen) |
This is the crux: phosphorylation activates phosphorylase but inactivates synthase — the opposite effects on the two enzymes ensure they are never both ON at the same time.
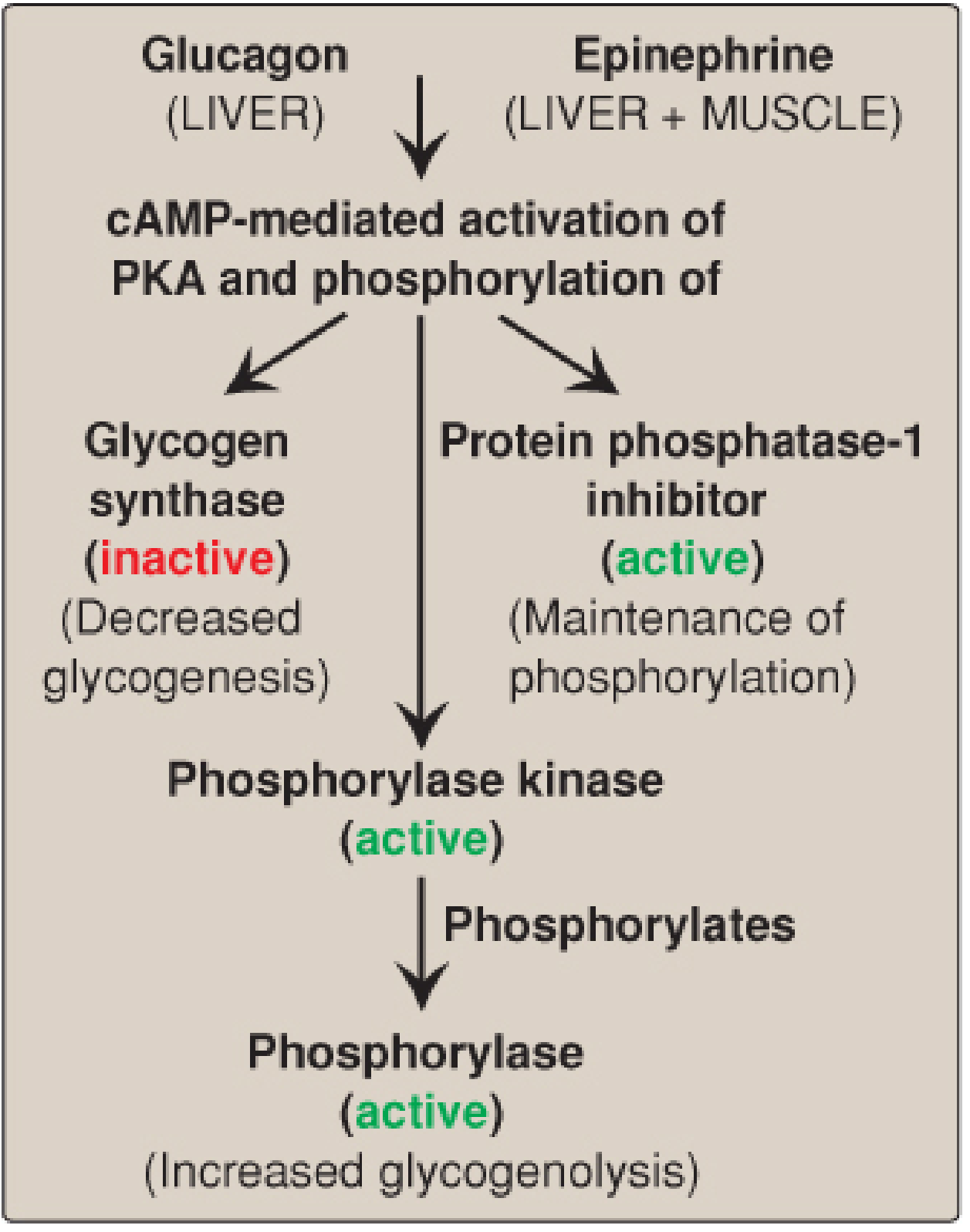
(3) Hormonal Regulation — Insulin & Glucagon
The cAMP Cascade (Fasting / Stress State)
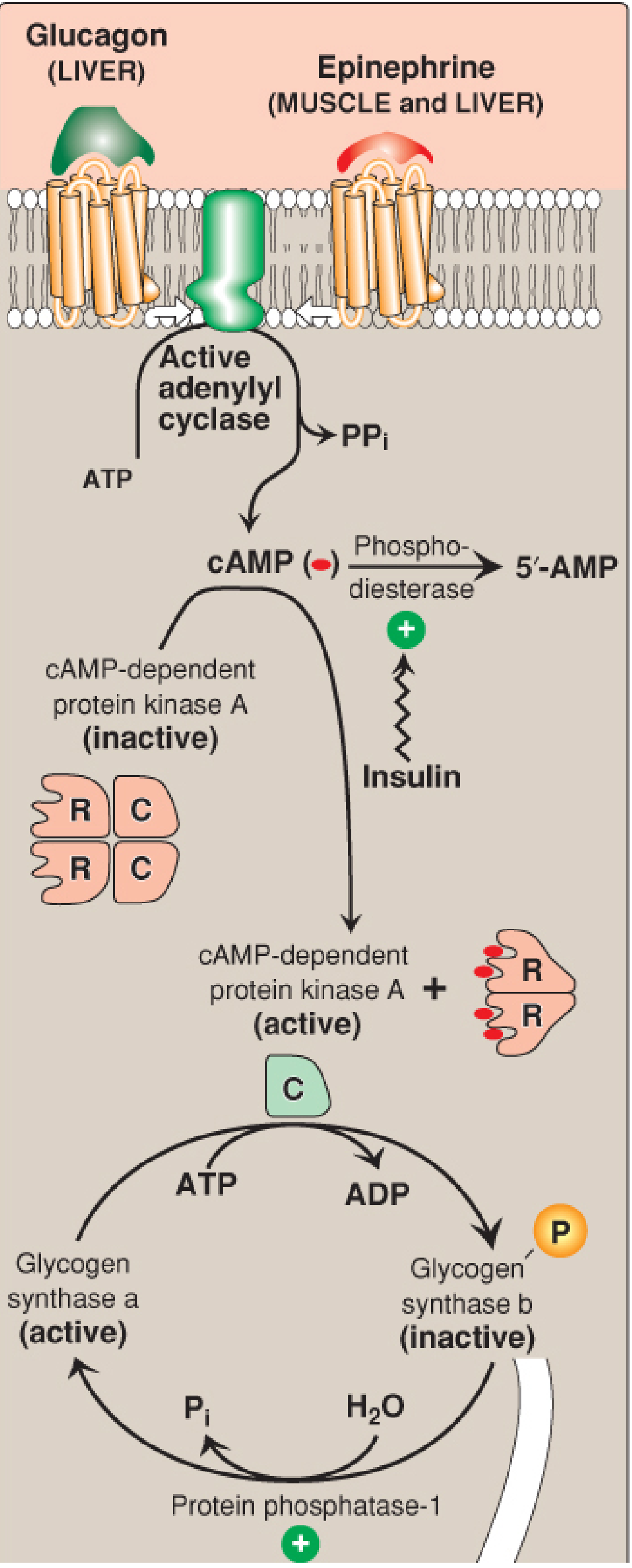
Glucagon (acts on liver) and Epinephrine (acts on both liver & muscle) signal low blood glucose or stress:
- Both hormones bind receptors → activate adenylyl cyclase → ATP → ↑ cAMP
- cAMP activates Protein Kinase A (PKA)
- PKA phosphorylates two targets simultaneously:
- Glycogen synthase → becomes inactive (stops glycogen synthesis)
- Phosphorylase kinase → becomes active
- Active phosphorylase kinase uses Ca²⁺ as a cofactor → phosphorylates glycogen phosphorylase b → phosphorylase a (active)
- Active glycogen phosphorylase drives glycogenolysis: Glycogen → Glucose 1-P → (via phosphoglucomutase) → Glucose 6-P → (via glucose 6-phosphatase in liver) → free glucose released into blood
Insulin (fed state — high blood glucose) does the opposite:
- Activates phosphodiesterase → degrades cAMP → ↓ cAMP
- PKA becomes inactive → phosphorylase kinase stays inactive → glycogen phosphorylase stays inactive
- Protein phosphatase-1 is activated → dephosphorylates glycogen synthase → synthase becomes active → glycogen synthesis resumes
Allosteric Regulation (Left Side of Your Notes)
In addition to hormonal/covalent control, these enzymes respond to metabolite levels directly:
| Effector | Effect on Glycogen Synthase | Effect on Glycogen Phosphorylase |
|---|---|---|
| Glucose 6-P ⊕ | Activates (even in "b"/dephosphorylated form) | Inhibits (even in active "a" form) |
| ATP ⊖ | — | Inhibits |
| Free glucose ⊖ (liver only) | — | Inhibits |
| AMP ⊕ (muscle only) | — | Activates phosphorylase b without phosphorylation |
This allosteric layer allows cells to respond instantly to energy levels, even before hormonal signals arrive.
Summary in One Sentence
During fasting/stress, glucagon/epinephrine → ↑cAMP → PKA → phosphorylates and inactivates glycogen synthase while activating phosphorylase kinase → activates glycogen phosphorylase → glycogenolysis → glucose released. During feeding, insulin → ↓cAMP → reversal of all the above → glycogen synthesis resumes.
Basic Medical Biochemistry, 6e; Biochemistry (Lippincott Illustrated Reviews), 8e
This is a shared conversation. Sign in to Orris to start your own chat.