causes of stroke in deatil. compae ishemic and hemorhaic stroke in detail
causes and etiology of stroke ischemic hemorrhagic
ischemic vs hemorrhagic stroke comparison pathophysiology clinical features diagnosis management
ischemic hemorrhagic stroke CT scan brain imaging comparison
Stroke: Causes & Detailed Comparison
What Is a Stroke?
PART 1 — Causes of Stroke (All Types)
A. Causes of Ischemic Stroke (~85% of all strokes)
1. Thrombotic (Large-Vessel Disease)
- Atherosclerosis — the most common cause; plaque buildup in carotid or intracranial arteries narrows the lumen, causing in-situ thrombosis
- Carotid artery stenosis — plaques at the carotid bifurcation are a major source
- Intracranial atherosclerosis — more prevalent in Asian, African, and Hispanic populations
- Arterial dissection — spontaneous or traumatic tearing of the arterial wall (carotid or vertebral), forming an intramural hematoma that occludes flow; common in young adults
2. Embolic
- Cardioembolic — clots form in the heart and travel to the brain:
- Atrial fibrillation (AF) — #1 cardiac cause; left atrial appendage thrombus
- Acute myocardial infarction with mural thrombus
- Dilated cardiomyopathy
- Valvular disease (rheumatic mitral stenosis, prosthetic valves, infective endocarditis)
- Patent foramen ovale (PFO) — paradoxical embolism from venous clot
- Artery-to-artery embolism — fragments from atherosclerotic plaques in proximal vessels
3. Small-Vessel Disease (Lacunar Stroke)
- Lipohyalinosis — hypertension causes degenerative changes in small perforating arteries
- Microatheroma — small plaque in perforating arteries
- Results in small (<15 mm) deep infarcts in the basal ganglia, thalamus, internal capsule, pons
4. Other / Less Common Causes
| Cause | Mechanism |
|---|---|
| Hypercoagulable states | Antiphospholipid syndrome, Factor V Leiden, protein C/S deficiency, malignancy |
| Vasculitis | CNS vasculitis, giant cell arteritis, SLE, polyarteritis nodosa |
| Sickle cell disease | Vaso-occlusion in cerebral vessels |
| Moyamoya disease | Progressive stenosis of the internal carotid arteries |
| Oral contraceptives | Prothrombotic state, especially with migraines with aura |
| Drugs | Cocaine, amphetamines (vasospasm + vasculitis) |
| Migraine with aura | Cortical spreading depression, rarely causes true infarction |
| Venous sinus thrombosis | Cerebral venous outflow obstruction causing venous infarction |
| Iatrogenic | Post-cardiac surgery, TEVAR, carotid endarterectomy (as noted in Reporting Standards for Type B Aortic Dissections, p. 16) |
B. Causes of Hemorrhagic Stroke (~15% of all strokes)
1. Intracerebral Hemorrhage (ICH) — Bleeding INTO Brain Tissue
| Cause | Notes |
|---|---|
| Hypertension | #1 cause; affects deep perforating arteries → basal ganglia, thalamus, cerebellum, pons |
| Cerebral amyloid angiopathy (CAA) | Amyloid deposits weaken vessel walls; lobar hemorrhages in elderly |
| Anticoagulation/Thrombolytics | Warfarin, NOACs, tPA — impair clotting; risk increases with INR >3 |
| Arteriovenous malformations (AVM) | Congenital tangles of vessels; rupture risk ~2–4%/year |
| Cavernous malformations | Clusters of abnormal capillaries |
| Tumors | Primary (glioblastoma) or metastatic (melanoma, renal cell, choriocarcinoma) |
| Cocaine/sympathomimetics | Acute BP surge ruptures vessels |
| Vasculitis | Inflammatory destruction of vessel walls |
| Hemorrhagic transformation of ischemic stroke | Reperfusion into infarcted tissue |
2. Subarachnoid Hemorrhage (SAH) — Bleeding into the Subarachnoid Space
| Cause | Notes |
|---|---|
| Berry (saccular) aneurysm rupture | ~85% of non-traumatic SAH; occurs at arterial bifurcations |
| AVM rupture | ~10% of SAH |
| Trauma | Most common cause of SAH overall |
| Cocaine use | Vasospasm and acute hypertension |
| Coagulopathy | Anticoagulants |
C. Shared / Common Risk Factors for All Strokes
| Modifiable | Non-Modifiable |
|---|---|
| Hypertension (most important) | Age (risk doubles every decade after 55) |
| Atrial fibrillation | Male sex |
| Diabetes mellitus | Family history / genetics |
| Hyperlipidemia | Race (Black > White > Asian for ICH) |
| Smoking | Prior stroke or TIA |
| Obesity & metabolic syndrome | |
| Physical inactivity | |
| Heavy alcohol use | |
| Sleep apnea |
PART 2 — Ischemic vs Hemorrhagic Stroke: Detailed Comparison
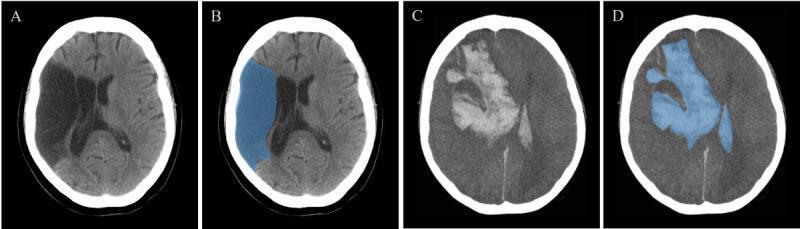
1. Basic Overview
| Feature | Ischemic Stroke | Hemorrhagic Stroke |
|---|---|---|
| Frequency | ~85% of all strokes | ~15% of all strokes |
| Subtypes | Thrombotic, embolic, lacunar | ICH, SAH |
| Core mechanism | Arterial occlusion → infarction | Vessel rupture → bleeding |
| Tissue effect | Ischemic necrosis (pale infarct) | Mass effect + ischemia from compression |
2. Pathophysiology
| Ischemic | Hemorrhagic | |
|---|---|---|
| Initiating event | Thrombosis or embolism blocks artery | Vessel wall failure → rupture |
| Ischemic penumbra | Present — salvageable tissue surrounding core infarct | Surrounding tissue compressed, not primarily ischemic |
| Cytotoxic edema | Primary early mechanism | Secondary to blood breakdown products |
| Vasogenic edema | Develops later (BBB breakdown) | Early and prominent |
| Mass effect | Mild in early hours; may worsen with large MCA infarcts | Often immediate and severe |
| Herniation risk | In large hemispheric infarcts (malignant MCA) | High — hematoma expands |
| Reperfusion injury | Yes — hemorrhagic transformation possible | N/A (bleeding is primary) |
3. Clinical Presentation
| Feature | Ischemic | Hemorrhagic (ICH) | Hemorrhagic (SAH) |
|---|---|---|---|
| Onset | Often abrupt; maximal at onset or stepwise | Sudden, during activity | Sudden — "thunderclap headache" |
| Headache | Absent or mild | Present (~50%), moderate | Severe ("worst headache of life") |
| Consciousness | Usually preserved initially | Often depressed (elevated ICP) | May lose consciousness at onset |
| Nausea/vomiting | Uncommon | Common (ICP↑) | Very common |
| Blood pressure | Variable, often normal-high | Markedly elevated | Often elevated |
| Focal deficits | Yes — territory-specific | Yes — often deep structures | Variable; meningism prominent |
| Seizures | Uncommon acutely | More common | Common |
| Meningismus | Absent | Absent | Present (blood in CSF) |
| Symptom progression | Often maximal at onset or fluctuates | May worsen over hours (hematoma expansion) | May worsen from vasospasm (days 3–14) |
4. Neurological Deficits by Territory
| Territory | Ischemic Syndrome | Notes |
|---|---|---|
| MCA (dominant) | Contralateral hemiplegia, aphasia, hemineglect | Most common |
| MCA (non-dominant) | Contralateral hemiplegia, hemispatial neglect | |
| ACA | Contralateral leg weakness > arm weakness, abulia | |
| PCA | Homonymous hemianopia, memory loss | |
| Vertebrobasilar | Diplopia, dysphagia, ataxia, crossed deficits | Locked-in syndrome if basilar occlusion |
| Lacunar | Pure motor, pure sensory, ataxic hemiparesis |
| Location | Hemorrhagic Syndrome |
|---|---|
| Putamen (most common) | Contralateral hemiplegia, gaze deviation toward lesion |
| Thalamus | Contralateral sensory/motor loss, vertical gaze palsy |
| Cerebellum | Ataxia, vomiting, gaze palsy — can herniate rapidly |
| Pons | Pinpoint pupils, quadriplegia, hyperthermia — often fatal |
| Lobar (CAA) | Variable depending on lobe; seizures common |
5. Diagnosis
| Investigation | Ischemic Stroke | Hemorrhagic Stroke |
|---|---|---|
| Non-contrast CT (first-line) | May be normal early; hypodensity develops over hours | Hyperdense (bright) blood immediately visible |
| MRI DWI | Gold standard — detects ischemia within minutes | Less sensitive for acute blood vs CT |
| MRI GRE/SWI | Can show microbleeds | Best for detecting old blood/microbleeds |
| CT Angiography | Identifies occlusion site; collateral flow | Identifies aneurysm, AVM, "spot sign" (active bleeding) |
| LP (lumbar puncture) | Not usually indicated | Essential for SAH when CT is negative — xanthochromia |
| ECG | AF, MI detection | Can show ST changes from autonomic surge |
| Echocardiogram | Cardioembolic workup | Less relevant |
| Coagulation studies | PT/INR/PTT — baseline | Critical — guides reversal strategy |
| CBC, metabolic panel | Rule out mimics, baseline | Rule out thrombocytopenia, coagulopathy |
6. Management
Ischemic Stroke
| Intervention | Details |
|---|---|
| IV Alteplase (tPA) | Within 4.5 hours of onset; improves outcomes if no hemorrhage |
| Mechanical thrombectomy | For large vessel occlusion (LVO); up to 24 hours with good penumbra |
| Aspirin | 325 mg within 24–48 hrs (not within 24 hrs of tPA) |
| Dual antiplatelet (DAPT) | Aspirin + clopidogrel for 21 days in minor stroke/TIA |
| Anticoagulation | For AF-related stroke; timing debated (usually 4–14 days) |
| BP management | Allow permissive hypertension up to 220/120 mmHg (pre-tPA: lower to <185/110) |
| Statins | High-intensity statin for all ischemic strokes |
| Neuroprotection | Avoid hyperthermia, hypoglycemia, hyperglycemia |
| Hemicraniectomy | For malignant MCA infarction with edema/herniation |
Hemorrhagic Stroke (ICH)
| Intervention | Details |
|---|---|
| BP control | Target SBP <140 mmHg (AHA 2022) — reduces hematoma expansion |
| Reversal of anticoagulation | Vitamin K + PCC/FFP for warfarin; specific reversal agents (idarucizumab, andexanet) for NOACs |
| Surgical evacuation | For cerebellar hematoma >3 cm, or hydrocephalus; conventional ICH — selected cases |
| Minimally invasive surgery | Emerging — stereotactic aspiration, MIS craniotomy |
| ICP management | HOB 30°, mannitol/hypertonic saline, EVD for hydrocephalus |
| Seizure prophylaxis | Treat clinical seizures; prophylactic AEDs not routinely recommended |
| No tPA/thrombolytics | Absolutely contraindicated |
Subarachnoid Hemorrhage (SAH)
| Intervention | Details |
|---|---|
| Nimodipine | 60 mg q4h for 21 days — reduces vasospasm and improves outcome |
| Aneurysm securing | Surgical clipping or endovascular coiling within 24–72 hours |
| Triple-H therapy | Hypertension, hypervolemia, hemodilution — for vasospasm (controversial now) |
| Transcranial Doppler | Monitor for vasospasm (days 4–14) |
7. Complications
| Complication | Ischemic | Hemorrhagic |
|---|---|---|
| Cerebral edema | Delayed (peaks 3–5 days) | Early, severe |
| Hemorrhagic transformation | Yes (especially with tPA or large infarct) | N/A |
| Vasospasm | Uncommon | Major complication of SAH (days 4–14) |
| Hydrocephalus | Rare | Common (intraventricular blood) |
| Re-occlusion/re-stroke | Yes | Hematoma re-expansion (up to 24 hrs) |
| Seizures | ~5–10% | ~20–30% |
| Dysphagia/aspiration | Common | Common |
| DVT/PE | High risk (immobility) | High risk |
8. Prognosis
| Ischemic | ICH | SAH | |
|---|---|---|---|
| 30-day mortality | ~8–12% | ~35–50% | ~25–50% |
| Functional independence at 3 months | ~40–50% | ~20% | ~30–40% |
| Recurrence risk (1 year) | ~10–15% | ~2–4% | Low if aneurysm secured |
| Predictors of poor outcome | Large infarct size, age, baseline NIHSS | Hematoma volume, deep location, IVH, GCS | Hunt-Hess grade, Fisher grade |
Summary Table
| Feature | Ischemic | Hemorrhagic |
|---|---|---|
| Frequency | 85% | 15% |
| Mechanism | Occlusion | Rupture |
| CT appearance | Hypodense (dark) | Hyperdense (bright) |
| Headache | Rare | Common (severe in SAH) |
| LOC at onset | Usually preserved | Often impaired |
| tPA eligible | Yes (strict criteria) | Absolutely contraindicated |
| BP target (acute) | Permissive (<220/120) | Aggressive reduction (<140) |
| Anticoagulation | Eventual use | Generally avoided |
| Surgery | Thrombectomy (LVO) | Evacuation (selected), aneurysm coiling/clipping |
| Mortality | Lower | Higher |
- Harrison's Principles of Internal Medicine, 21st Edition (p. 12128)
- Reporting Standards for Type B Aortic Dissections (p. 16)
- AHA/ASA Guidelines for Early Management of Acute Ischemic Stroke (2019/2022 updates)
- AHA/ASA Guidelines for Management of Spontaneous ICH (2022)
expain TIA ACUTE MANAGEMNET OF STROKE I DETAIL
TIA transient ischemic attack definition diagnosis ABCD2 score management
acute ischemic stroke management tPA thrombolysis thrombectomy guidelines
TIA ABCD2 score stroke acute management algorithm
TIA workup investigations imaging risk stratification ABCD2 score emergency department
acute stroke initial assessment blood pressure airway neuroprotection general management
Transient Ischemic Attack (TIA) & Acute Stroke Management
PART 1 — TRANSIENT ISCHEMIC ATTACK (TIA)
Definition
"A transient episode of neurological dysfunction caused by focal brain, spinal cord, or retinal ischemia, without acute infarction." (Adult Patients With Suspected TIA in the Emergency Department, p. 2)
- The old definition used 24 hours as the cutoff — if symptoms resolved within 24 hours, it was called a TIA
- This was abandoned because ~1/3 of all TIAs show evidence of infarction on MRI despite symptom resolution
- Most TIAs last fewer than 1–2 hours clinically
- If imaging is unavailable and symptoms last >24 hours → classified as clinical stroke
Why TIA Is a Medical Emergency
| Time After TIA | Stroke Risk |
|---|---|
| 48 hours | 3–10% |
| 7 days | 5–10% |
| 90 days | 10–15% |
Causes of TIA (Same as Ischemic Stroke)
| Mechanism | Examples |
|---|---|
| Large vessel atherosclerosis | Carotid stenosis, intracranial atherosclerosis |
| Cardioembolic | Atrial fibrillation, valvular disease, LV thrombus |
| Small vessel / lacunar | Hypertensive lipohyalinosis |
| Arterial dissection | Carotid/vertebral dissection (young adults) |
| Hypercoagulable states | Antiphospholipid syndrome, Factor V Leiden |
| Cryptogenic | No identified cause despite full workup |
Clinical Features of TIA
Carotid (Anterior Circulation) TIA
- Contralateral hemiplegia or hemiparesis (face/arm > leg)
- Contralateral hemisensory loss
- Aphasia (dominant hemisphere)
- Amaurosis fugax — transient monocular blindness ("curtain descending") from ophthalmic artery ischemia — classic TIA symptom
Vertebrobasilar (Posterior Circulation) TIA
- Vertigo, diplopia, dysphagia, dysarthria
- Ataxia, drop attacks
- Bilateral weakness or sensory loss
- Hemianopia or cortical blindness
Important: Isolated dizziness, isolated syncope, amnesia, or confusion alone are not TIA until proven otherwise.
ABCD2 Score — Risk Stratification
| Parameter | Criteria | Score |
|---|---|---|
| A — Age | ≥60 years | 1 |
| B — Blood Pressure | SBP ≥140 or DBP ≥90 | 1 |
| C — Clinical features | Unilateral weakness | 2 |
| Speech disturbance without weakness | 1 | |
| Other | 0 | |
| D — Duration of symptoms | ≥60 minutes | 2 |
| 10–59 minutes | 1 | |
| <10 minutes | 0 | |
| D — Diabetes | Present | 1 |
| Total | 0–7 |
Score Interpretation
| Score | Risk Category | 2-Day Stroke Risk |
|---|---|---|
| 0–3 | Low | ~1% |
| 4–5 | Moderate | ~4% |
| 6–7 | High | ~8–12% |
Per the meta-analysis by Wardlaw et al (2015) cited in Adult Patients With Suspected TIA in the Emergency Department (p. 33):
- ABCD2 ≥4 at 7 days: stroke risk 5.2%; at 90 days: 8.9%
- ABCD2 <4 at 7 days: stroke risk 1.4%; at 90 days: 2.4%
- Sensitivity of ABCD2 ≥4 for 7-day stroke: 86.7%
Investigations for TIA
Immediate (within 24 hours)
| Investigation | Purpose |
|---|---|
| Non-contrast CT brain | Exclude hemorrhage, early infarct |
| MRI brain (DWI) | Gold standard — detects acute infarction even with resolved symptoms; ~50% of TIAs show DWI lesion |
| CT Angiography / MR Angiography | Identify carotid stenosis, intracranial stenosis, dissection |
| ECG (12-lead) | Detect atrial fibrillation, MI |
| Blood glucose | Exclude hypoglycemia mimicking TIA |
| CBC, coagulation, metabolic panel | Baseline, rule out hematological causes |
| Carotid Doppler ultrasound | Stenosis at carotid bifurcation |
Within 48–72 Hours
| Investigation | Purpose |
|---|---|
| Echocardiogram (TTE/TEE) | Cardioembolic source: LV thrombus, PFO, valvular disease |
| Prolonged cardiac monitoring (Holter, implantable loop recorder) | Paroxysmal AF (may need 30-day or longer monitoring) |
| Lipid panel, HbA1c | Vascular risk factors |
| Hypercoagulable workup | If young, cryptogenic, or recurrent (protein C/S, Factor V Leiden, antiphospholipid antibodies) |
Management of TIA
Immediate (Emergency Department)
- Aspirin 300–325 mg loading dose immediately (if no hemorrhage on CT)
- Dual antiplatelet therapy (DAPT): Aspirin + Clopidogrel (75 mg)
- For high-risk TIA (ABCD2 ≥4) or minor ischemic stroke (NIHSS ≤3)
- Continue for 21 days, then switch to monotherapy
- Based on POINT and CHANCE trials — reduces 90-day stroke risk by ~25%
- Do NOT use anticoagulation as first-line for non-AF TIA
- Start anticoagulation (NOAC preferred over warfarin) once hemorrhage excluded
- Timing after TIA: can start immediately (no significant infarct burden)
- Target INR 2–3 if warfarin used
- Target <130/80 mmHg long-term (AHA 2021)
- In the acute phase: do not lower aggressively if BP <220/120 (unless hypertensive emergency)
- Start or resume antihypertensives within 24–48 hours
- High-intensity statin (atorvastatin 40–80 mg) for all non-cardioembolic TIA
- Target LDL <70 mg/dL (or <55 mg/dL in high-risk)
Hospitalization vs. Outpatient?
| Risk | Disposition |
|---|---|
| ABCD2 ≥4 OR DWI lesion OR cardiac source suspected OR crescendo TIA | Admit for monitoring and urgent workup |
| ABCD2 0–3, no DWI lesion, low-risk features | Expedited outpatient workup within 24–48 hrs (TIA clinic) |
Carotid Revascularization
- Carotid endarterectomy (CEA) or carotid artery stenting (CAS) for ipsilateral carotid stenosis ≥50%
- Greatest benefit with stenosis 70–99%
- Should be performed within 2 weeks of TIA (ideally within 48–72 hours if stable)
Secondary Prevention (Long-term)
| Intervention | Details |
|---|---|
| Antiplatelet or anticoagulation | Based on mechanism |
| BP control | ACE inhibitor + thiazide diuretic (PROGRESS trial) |
| Statin | High-intensity |
| Smoking cessation | Reduces risk by ~50% |
| Glycemic control | HbA1c <7% |
| Lifestyle modification | Exercise, diet, weight loss, alcohol moderation |
PART 2 — ACUTE MANAGEMENT OF STROKE (DETAILED)
The "Time = Brain" Principle
Every minute during stroke, approximately 1.9 million neurons die. Every hour without treatment = 3.6 years of accelerated brain aging.
Step 1 — Pre-Hospital / EMS
| Action | Details |
|---|---|
| FAST/BE-FAST recognition | Balance, Eyes, Face drooping, Arm weakness, Speech difficulty, Time to call 911 |
| Cincinnati Prehospital Stroke Scale | Facial droop, arm drift, speech abnormality — any 1 = 72% probability of stroke |
| Time of last known well (LKW) | Critical — determines eligibility for tPA |
| Notify receiving hospital | "Stroke alert" activation before arrival |
| Do NOT lower BP in field | Unless hypertensive emergency with end-organ damage |
| Blood glucose check | Hypoglycemia is the #1 stroke mimic |
| IV access, ECG, oxygen | O2 only if SpO2 <94% |
Step 2 — Emergency Department — Initial Assessment (Target: Door-to-CT <25 min)
Primary Survey — ABCs
| System | Action |
|---|---|
| Airway | Protect if GCS ≤8 or loss of gag reflex → intubate |
| Breathing | SpO2 ≥94%; supplemental O2 only if hypoxic |
| Circulation | IV access ×2, continuous BP/cardiac monitoring |
| Disability (Neuro) | GCS, NIHSS, pupils, glucose |
| Exposure | Fever? (treat aggressively — worsens outcomes) |
Rapid History
- Exact time of symptom onset (or last known well)
- Recent surgery, trauma, bleeding history
- Medications (anticoagulants, antiplatelets)
- Prior stroke/TIA
- Contraindications to tPA
NIHSS (NIH Stroke Scale)
| Score | Severity |
|---|---|
| 0 | No stroke |
| 1–4 | Minor |
| 5–15 | Moderate |
| 16–20 | Moderate-severe |
| 21–42 | Severe |
Step 3 — Emergency Imaging (Target: Door-to-CT-read <45 min)
| Imaging | What It Shows |
|---|---|
| Non-contrast CT brain | Hemorrhage (excludes tPA use); early ischemic signs (ASPECTS score) |
| CT Angiography (CTA) head & neck | Large vessel occlusion (LVO) for thrombectomy eligibility |
| CT Perfusion (CTP) | Ischemic core vs penumbra — extends thrombectomy window to 24 hrs |
| MRI DWI + FLAIR | Most sensitive for acute ischemia; FLAIR-DWI mismatch estimates time of onset |
Early CT Signs of Ischemia
- Hyperdense MCA sign — clot in MCA visible as bright vessel
- Loss of insular ribbon — loss of gray-white differentiation in insula
- ASPECTS score — 0–10; score <6 = large infarct, poor prognosis, consider excluding from thrombectomy
Step 4 — IV Thrombolysis (tPA / Alteplase)
Mechanism
Eligibility Criteria
| Criteria | Details |
|---|---|
| Time window | Within 4.5 hours of symptom onset |
| Age | ≥18 years (no upper age limit per AHA 2019) |
| CT | No hemorrhage, no large established infarct |
| BP before tPA | Must be <185/110 mmHg (treat with labetalol, nicardipine, or clevidipine) |
| Blood glucose | 50–400 mg/dL |
Absolute Contraindications to tPA
| Contraindication |
|---|
| Hemorrhagic stroke / any intracranial hemorrhage |
| CT showing >1/3 MCA territory involvement |
| Significant head trauma in past 3 months |
| Prior intracranial surgery within 3 months |
| Active internal bleeding (not menstruation) |
| Suspected aortic dissection |
| Intracranial neoplasm, AVM, aneurysm |
| Platelets <100,000; INR >1.7; aPTT elevated on heparin |
Relative Contraindications (Risk-Benefit Discussion)
- Minor or rapidly improving symptoms
- Seizure at stroke onset
- Major surgery within 14 days
- GI/urinary bleeding within 21 days
- Recent MI within 3 months
- Pregnancy (relative — can be used if benefit outweighs risk)
Dosing
- 0.9 mg/kg IV (max 90 mg)
- 10% as IV bolus over 1 minute, remainder over 60 minutes
- Admit to ICU/stroke unit post-tPA; monitor BP every 15 minutes
Post-tPA Monitoring
- No antiplatelets or anticoagulants for 24 hours after tPA
- Watch for symptomatic intracranial hemorrhage (sICH) — ~6%
- BP target after tPA: <180/105 mmHg for 24 hours
- Repeat CT/MRI at 24 hours before starting antiplatelets
Tenecteplase — Newer Alternative
- Single IV bolus (0.25 mg/kg, max 25 mg)
- Easier to administer; similar efficacy to alteplase in recent trials (AHA 2022 guidelines now include as alternative)
Step 5 — Mechanical Thrombectomy (Endovascular Therapy)
Eligible Vessels
- Internal carotid artery (ICA)
- M1 and M2 segments of MCA
- Basilar artery
- Vertebral artery
Time Windows
| Window | Criteria |
|---|---|
| 0–6 hours | Clinical + imaging criteria; treat all eligible LVOs |
| 6–16 hours | DAWN/DEFUSE-3 criteria: penumbra-to-core mismatch on CTP; age ≥80 + NIHSS ≥10 |
| 16–24 hours | DAWN criteria; strict mismatch profile required |
Bridging Strategy
- If patient is eligible for both tPA AND thrombectomy → give tPA first without waiting to assess response
- Do NOT delay thrombectomy to assess tPA response (Endovascular Therapy of Acute Ischemic Stroke, p. 3)
- Rate of tPA-induced recanalization before thrombectomy is <10% — don't wait
Direct Thrombectomy (No tPA)
- When tPA is contraindicated (hemorrhage risk, >4.5 hours, INR elevated, recent surgery)
- Proceed directly to catheter-based clot retrieval
Technique
- Stent retriever (Solitaire, Trevo) — mesh device captures and extracts clot
- Aspiration thrombectomy — direct suction catheter
- Combined approach — both techniques
- Goal: TICI 2b/3 reperfusion (≥50% territory reperfused)
Outcomes
- mTICI 2b-3 reperfusion → 50–60% achieve functional independence at 90 days
- Number needed to treat (NNT) to prevent disability: ~2.6
Step 6 — General Stroke Unit Care (Neuroprotection)
Blood Pressure Management
| Situation | BP Target |
|---|---|
| Ischemic stroke, NOT tPA eligible | Permissive — allow up to 220/120 mmHg (do not treat unless hypertensive emergency) |
| Pre-tPA | Must lower to <185/110 before giving tPA |
| Post-tPA (24 hrs) | Keep <180/105 mmHg |
| After thrombectomy | Keep <180/105 mmHg |
| Hemorrhagic stroke | Target SBP <140 mmHg aggressively |
Glucose Management
- Hyperglycemia (>180 mg/dL) worsens infarct — treat with insulin
- Hypoglycemia mimics stroke and worsens outcome — treat immediately with dextrose
- Target: 140–180 mg/dL in acute phase
Temperature
- Fever (>37.5°C) dramatically worsens outcomes — use acetaminophen aggressively
- Therapeutic hypothermia: investigational, not standard of care
Oxygen
- Supplemental O2 only if SpO2 <94%
- Routine O2 supplementation does NOT improve outcomes (IST-3)
Positioning
- Head of bed flat (0°) for ischemic stroke — improves cerebral perfusion
- HOB 30° for hemorrhagic stroke, risk of aspiration
Dysphagia Screening
- Screen before any oral intake
- Nil by mouth until formal swallow assessment
- NG tube / PEG if persistent dysphagia
Cardiac Monitoring
- Continuous telemetry for ≥24 hours (ideally 72 hours)
- Detect paroxysmal AF — found in ~15% of cryptogenic strokes
- Neurogenic cardiac injury (Takotsubo, ST changes, troponin elevation) — common post-stroke
Step 7 — Acute Hemorrhagic Stroke Management
Intracerebral Hemorrhage (ICH)
| Intervention | Details |
|---|---|
| BP control | Target SBP <140 mmHg within 1 hour (AHA 2022); use nicardipine or labetalol IV |
| Reversal of anticoagulation | Warfarin → Vitamin K 10 mg IV + 4-factor PCC (Kcentra); NOACs → specific reversal agents |
| Reversal agents | Dabigatran → Idarucizumab (Praxbind); Factor Xa inhibitors → Andexanet alfa |
| Platelet transfusion | Only if platelets <100,000 or patient on antiplatelet agents pre-operatively |
| ICP management | HOB 30°, isotonic fluids, avoid hypotonic solutions, mannitol or hypertonic saline |
| Seizure | Treat clinical seizures; prophylactic AEDs not routinely recommended |
| Surgery | Cerebellar hematoma >3 cm or causing hydrocephalus → urgent surgical evacuation; EVD for hydrocephalus |
| NO tPA | Absolutely contraindicated |
Subarachnoid Hemorrhage (SAH)
| Intervention | Details |
|---|---|
| Secure the aneurysm | Surgical clipping or endovascular coiling within 24–72 hours |
| Nimodipine | 60 mg PO/NG q4h × 21 days — reduces vasospasm and improves neurological outcome |
| BP management | Before securing aneurysm: SBP <160 mmHg to prevent re-bleeding |
| Vasospasm monitoring | Transcranial Doppler (TCD) daily from days 3–14; CT perfusion if vasospasm suspected |
| Induced hypertension | For symptomatic vasospasm (triple-H therapy — now mainly targeted euvolemia + hypertension) |
| EVD | For hydrocephalus (common with intraventricular blood) |
| Anti-fibrinolytics | Tranexamic acid short-term (pre-operative only) to reduce re-bleeding |
Step 8 — Secondary Prevention (Post-Stroke)
| Intervention | Details |
|---|---|
| Antiplatelet therapy | Aspirin 75–100 mg; or clopidogrel; DAPT ×21 days for minor stroke/TIA |
| Anticoagulation | For AF — start within 4–14 days (NOAC preferred) |
| Statin | High-intensity atorvastatin 40–80 mg |
| BP control | Target <130/80 mmHg; start ACEi + thiazide |
| Carotid revascularization | CEA/CAS for ipsilateral carotid stenosis ≥50% |
| PFO closure | In selected young cryptogenic stroke patients with PFO |
| Lifestyle | Smoking cessation, exercise, Mediterranean diet, alcohol moderation |
Step 9 — Stroke Unit Care & Rehabilitation
| Component | Details |
|---|---|
| Dedicated stroke unit | Reduces mortality and disability by ~25% vs general ward |
| Multidisciplinary team | Stroke neurologist, nurses, physiotherapy, OT, speech therapy, social work |
| Early mobilization | Within 24–48 hours (very early mobilization <24 hrs may worsen outcomes — AVERT trial) |
| Dysphagia rehab | Speech therapy; PEG if needed |
| DVT prophylaxis | Pneumatic compression devices immediately; low-dose LMWH after 24–48 hrs |
| Urinary catheter | Avoid unless necessary — infection risk |
| Pressure ulcers | Regular repositioning, skin care |
| Psychological support | Post-stroke depression in ~30–40%; start SSRIs if needed |
Acute Stroke Management Summary Algorithm
SYMPTOM ONSET / LAST KNOWN WELL
↓
EMS: BE-FAST, glucose, IV access, alert hospital
↓
ED ARRIVAL → Stroke Alert
↓
ABCs + NIHSS + Blood glucose
↓
STAT Non-contrast CT Brain (<25 min)
↓
┌─────────────────────────────────────┐
│ HEMORRHAGE? │
│ YES → ICH/SAH protocol │
│ NO → Ischemic stroke pathway │
└─────────────────────────────────────┘
↓ (Ischemic)
CTA Head & Neck → LVO?
↓
┌─────────────────────────────────────────────────────────┐
│ tPA ELIGIBLE? (≤4.5 hrs, no contraindications) │
│ YES → Give tPA immediately (Door-to-Needle <60 min) │
│ + If LVO → proceed to thrombectomy (don't wait) │
│ NO + LVO → Direct thrombectomy (≤24 hrs with mismatch) │
└─────────────────────────────────────────────────────────┘
↓
STROKE UNIT ADMISSION
BP / Glucose / Temp / O2 / Dysphagia screen / Cardiac monitor
↓
SECONDARY PREVENTION + REHABILITATION
- Adult Patients With Suspected TIA in the Emergency Department (p. 2, p. 33)
- Endovascular Therapy of Acute Ischemic Stroke (p. 3)
- Harrison's Principles of Internal Medicine, 21st Edition (p. 12128)
- AHA/ASA Guidelines for Early Management of Acute Ischemic Stroke (2019)
- AHA/ASA Guidelines for Management of Spontaneous ICH (2022)
- DAWN, DEFUSE-3, POINT, CHANCE, AVERT trials
TRICKS FOR MANGEMNT FOR STROKE FOR 10 MARKER ALL POINTS SHOULD CCOVER
MANAGEMENT OF STROKE — 10-Marker Answer
🧠 Master Mnemonic: "FAST BE SAFE RECOVER"
Use this to remember ALL domains of stroke management in sequence.
TRICK 1 — "TIME = BRAIN" 🕐
Every 1 minute of stroke = 1.9 million neurons lost Every 1 hour = 3.6 years of accelerated brain aging
| Target | Time |
|---|---|
| Door-to-CT | <25 minutes |
| Door-to-CT read | <45 minutes |
| Door-to-Needle (tPA) | <60 minutes |
| Door-to-Groin (thrombectomy) | <90 minutes |
TRICK 2 — Pre-Hospital: Mnemonic "BE-FAST"
| Letter | Meaning |
|---|---|
| B | Balance loss |
| E | Eyes (vision change) |
| F | Face drooping |
| A | Arm weakness |
| S | Speech abnormality |
| T | Time — call emergency NOW |
- Check blood glucose immediately (hypoglycemia = #1 stroke mimic)
- Do NOT lower BP in field
- Pre-notify hospital → Stroke Alert
TRICK 3 — ED Initial Assessment: Mnemonic "ABCDE"
| Letter | Action |
|---|---|
| A | Airway — intubate if GCS ≤8 |
| B | Breathing — O₂ only if SpO₂ <94% |
| C | Circulation — 2 IV lines, BP monitoring, ECG |
| D | Disability — GCS, NIHSS score, blood glucose |
| E | Exposure/Everything — fever? bleeding? medications? |
- 0 = Normal | 1–4 = Minor | 5–15 = Moderate | 16–20 = Mod-Severe | 21–42 = Severe
TRICK 4 — Emergency Imaging: Mnemonic "CT FIRST, MRI CONFIRMS"
| Scan | Purpose |
|---|---|
| Non-contrast CT | Exclude hemorrhage (mandatory before tPA) |
| CT Angiography | Find large vessel occlusion (LVO) for thrombectomy |
| CT Perfusion | Core vs penumbra — extends window to 24 hrs |
| MRI DWI | Gold standard for ischemia detection |
- Hyperdense MCA sign = clot in MCA (visible as bright vessel on CT)
- Hypodense area = ischemic infarct
- Hyperdense area = hemorrhage
TRICK 5 — Separate Ischemic from Hemorrhagic: Mnemonic "HICH"
Hemorrhage → Image first → Contraindicated for tPA → Hypertension control
| Feature | Ischemic | Hemorrhagic |
|---|---|---|
| CT appearance | Dark (hypodense) | Bright (hyperdense) |
| tPA | ✅ Give | ❌ Absolutely NO |
| BP target | Permissive ≤220/120 | Aggressive <140 mmHg |
| Headache | Mild/absent | Severe |
| LOC | Usually preserved | Often impaired |
TRICK 6 — IV tPA (Thrombolysis): Mnemonic "4.5 HOURS, 0.9 mg/kg, 90 MAX"
- 0.9 mg/kg IV (max 90 mg)
- 10% as bolus over 1 min → rest over 60 min
Contraindications Trick: "BRAIN BLEED"
| Letter | Contraindication |
|---|---|
| B | Bleeding — active internal bleeding |
| R | Recent surgery/trauma (within 3 months) |
| A | Anticoagulants — INR >1.7, elevated aPTT |
| I | Intracranial lesion (AVM, aneurysm, tumor) |
| N | No hemorrhage confirmed — must have CT first |
| B | BP >185/110 (untreated) |
| L | Low platelets (<100,000) |
| E | Evidence of large infarct (>1/3 MCA territory) |
| E | Earlier stroke within 3 months |
| D | Dissection (aortic) suspected |
TRICK 7 — Mechanical Thrombectomy: Mnemonic "LVO = GO"
If Large Vessel Occlusion found → GO for thrombectomy
| Time Window | Criteria |
|---|---|
| 0–6 hours | Any LVO, eligible patient |
| 6–16 hours | DEFUSE-3 criteria: penumbra > core on CT perfusion |
| 16–24 hours | DAWN criteria: clinical-imaging mismatch |
Give tPA first + proceed to thrombectomy WITHOUT WAITING for tPA response Direct thrombectomy if tPA contraindicated
TRICK 8 — General Stroke Unit Care: Mnemonic "GOFTS"
| Letter | Meaning | Target |
|---|---|---|
| G | Glucose | 140–180 mg/dL; treat hypo AND hyperglycemia |
| O | Oxygen | SpO₂ ≥94%; do NOT give routine O₂ |
| F | Fever | Treat aggressively; target <37.5°C (paracetamol) |
| T | Temperature/Positioning | HOB flat (ischemic) / HOB 30° (hemorrhagic) |
| S | Swallowing | Screen before ANY oral intake; NBM until cleared |
TRICK 9 — BP Management: Mnemonic "Three Targets"
| Situation | BP Target |
|---|---|
| Ischemic stroke (no tPA) | Allow up to 220/120 — permissive |
| Before giving tPA | Must be <185/110 |
| After tPA (24 hrs) | Keep <180/105 |
| Hemorrhagic stroke | SBP <140 immediately |
Drug of choice for BP lowering: IV Labetalol, IV Nicardipine, or IV Clevidipine
TRICK 10 — Hemorrhagic Stroke — Mnemonic "REVERSE BLEED"
| Step | Action |
|---|---|
| R | Reverse anticoagulation (PCC for warfarin; idarucizumab for dabigatran; andexanet alfa for Xa inhibitors) |
| E | Emergency BP control (SBP <140 mmHg) |
| V | Vitamin K 10 mg IV (if warfarin-related) |
| E | EVD (External ventricular drain) for hydrocephalus |
| R | Remove clot surgically if cerebellar hematoma >3 cm |
| S | Seizure management (treat clinical seizures) |
| E | Elevate head of bed to 30° |
| B | Blood pressure drugs IV |
| L | Lab monitoring (INR, platelets, coag) |
| E | Extra monitoring for ICP |
| E | No thrombolytics EVER |
| D | DVT prophylaxis (pneumatic compression) |
TRICK 11 — SAH Specific: Mnemonic "ANNS"
| Letter | Action |
|---|---|
| A | Aneurysm — clip or coil within 24–72 hours |
| N | Nimodipine 60 mg q4h × 21 days (vasospasm prevention) |
| N | Neurocritical monitoring (TCD for vasospasm days 3–14) |
| S | "Worst headache of LIFE" = SAH until proven otherwise → CT then LP |
TRICK 12 — Secondary Prevention: Mnemonic "ABCDS"
| Letter | Intervention |
|---|---|
| A | Antiplatelet (aspirin ± clopidogrel) OR Anticoagulation (if AF) |
| B | Blood pressure control (<130/80 mmHg) — ACEi + thiazide |
| C | Carotid revascularization (CEA/CAS if stenosis ≥50%) |
| D | Diabetes control (HbA1c <7%) + Diet + Dyslipidemia (statin) |
| S | Smoking cessation + Statin (atorvastatin 40–80 mg) |
- PFO closure — in young cryptogenic stroke with PFO
- AF management — cardioversion, rate control, anticoagulation
- Long-term cardiac monitoring — implantable loop recorder for cryptogenic stroke
TRICK 13 — Rehabilitation: Mnemonic "MOSS"
| Letter | Meaning |
|---|---|
| M | Multidisciplinary team (neuro, physio, OT, speech, SW) |
| O | Occupational therapy + early mobilization (24–48 hrs) |
| S | Speech therapy for aphasia/dysphagia |
| S | Psychological support — post-stroke depression in 30–40% (SSRIs) |
COMPLETE 10-MARK ANSWER OUTLINE
| # | Domain | Key Point |
|---|---|---|
| 1 | Pre-hospital | BE-FAST, glucose, stroke alert, time recording |
| 2 | Initial ED assessment | ABCs, NIHSS, IV access, ECG |
| 3 | Emergency imaging | NCCT brain — exclude hemorrhage; CTA for LVO |
| 4 | Differentiate stroke type | Ischemic vs hemorrhagic (CT: dark vs bright) |
| 5 | IV thrombolysis | tPA 0.9 mg/kg within 4.5 hrs, BP <185/110 pre-dose |
| 6 | Mechanical thrombectomy | LVO, 0–24 hrs with mismatch, don't delay for tPA response |
| 7 | Supportive care (GOFTS) | Glucose, O₂, Fever, positioning, Swallow screen |
| 8 | Hemorrhagic stroke Rx | Reverse anticoagulation, BP <140, surgery if needed |
| 9 | Secondary prevention (ABCDS) | Antiplatelet/anticoag, BP, statin, carotid, smoking |
| 10 | Rehabilitation | Early mobilisation, MDT, speech, psychology |
One-Page Visual Summary
STROKE MANAGEMENT — QUICK RECALL
PRE-HOSPITAL ED ARRIVAL IMAGING
BE-FAST ABCs + NIHSS NCCT Brain
Glucose check IV access + ECG → No bleed? = Ischemic
Stroke alert BP monitoring → Bleed? = Hemorrhagic
Time = Brain Glucose control CTA → LVO?
ISCHEMIC HEMORRHAGIC
↓ ↓
tPA (≤4.5 hrs) Reverse anticoag (PCC/Vit K)
0.9 mg/kg, max 90 mg SBP <140 mmHg
BP <185/110 before Surgery if cerebellar >3cm
LVO → Thrombectomy NO tPA EVER
(0–24 hrs, don't wait) Nimodipine if SAH
SUPPORTIVE CARE (GOFTS)
Glucose 140–180 | O₂ if <94% | Fever treat | HOB flat | Swallow screen
SECONDARY PREVENTION (ABCDS)
Antiplatelet/AC | BP <130/80 | Carotid Rx | Diabetes/Diet/Dyslipid | Smoking
REHAB (MOSS)
MDT | OT + Mobilise | Speech | Support (psychology)
DESCRIBE IN DEATIL LACUNAR STROKE
lacunar stroke definition pathophysiology small vessel disease lipohyalinosis
lacunar infarct clinical syndromes pure motor pure sensory ataxic hemiparesis diagnosis management
lacunar infarct MRI brain basal ganglia internal capsule
lacunar stroke risk factors hypertension diabetes small vessel pathology prognosis treatment
LACUNAR STROKE — Detailed Description
1. DEFINITION
"Lacunar infarction refers to infarction following atherothrombotic or lipohyalinotic occlusion of a small artery in the brain. The term small-vessel stroke is now the preferred term."
Key Defining Features:
| Feature | Detail |
|---|---|
| Size | <15 mm in diameter (most <10 mm) |
| Location | Deep brain structures only |
| Vessel involved | Single small penetrating artery (50–200 µm diameter) |
| Frequency | ~20–25% of all ischemic strokes |
| Mechanism | Small vessel disease (NOT embolism or large vessel atherosclerosis) |
2. ANATOMY — WHERE DO LACUNAR INFARCTS OCCUR?
| Artery | Brain Region Supplied | Common Lacunar Location |
|---|---|---|
| Lenticulostriate arteries (from MCA) | Basal ganglia, internal capsule | Putamen, caudate, internal capsule |
| Thalamoperforating arteries (from PCA/basilar) | Thalamus | Ventral thalamus |
| Paramedian pontine perforators (from basilar) | Pons | Basis pontis |
| Anterior choroidal artery branches | Internal capsule, thalamus | Posterior limb of internal capsule |
Most Common Sites (Memorize):
- Putamen (most common)
- Thalamus
- Pons (basis pontis)
- Internal capsule (posterior limb and genu)
- Caudate nucleus
- Cerebellum (less common)
Lacunar infarcts do NOT occur in the cortex — cortical involvement always suggests embolism or large vessel disease.
3. PATHOPHYSIOLOGY
Two Main Mechanisms:
A. Lipohyalinosis (Primary Small Vessel Disease)
- The most common mechanism
- Chronic hypertension and diabetes cause degenerative changes in the wall of small penetrating arteries
- The normal vessel wall is replaced by lipid deposits and hyaline material → vessel wall thickens → lumen narrows → occlusion
- Affects arteries 40–200 µm in diameter
- Leads to fibrinoid necrosis in acute hypertensive crises
B. Microatheroma
- Small atherosclerotic plaque forms at the origin of the penetrating artery (where it branches off the parent vessel)
- Causes in-situ thrombotic occlusion
- More common in larger penetrating arteries (200–800 µm)
- Associated with diabetes mellitus and dyslipidemia
C. Other (Less Common) Mechanisms
| Mechanism | Notes |
|---|---|
| Cardiac embolism | Rare — small emboli can occasionally occlude penetrating arteries |
| Cerebral amyloid angiopathy | Amyloid deposits in small vessel walls |
| Vasculitis | Inflammatory small vessel destruction |
| Hypercoagulable states | Thrombophilia in young patients |
Pathological Sequence:
Hypertension / Diabetes
↓
Lipohyalinosis / Microatheroma
↓
Lumen narrowing of penetrating artery
↓
Thrombotic occlusion
↓
Small deep infarct (<15 mm)
↓
Necrosis → macrophage infiltration
↓
Cavitation → small fluid-filled cavity ("lacune")
4. RISK FACTORS
Primary (Strongest):
| Risk Factor | Mechanism |
|---|---|
| Hypertension | #1 cause — drives lipohyalinosis; risk increases proportionally with BP level |
| Diabetes mellitus | Promotes microatheroma and lipohyalinosis; doubles stroke risk |
Secondary:
| Risk Factor | Notes |
|---|---|
| Smoking | Accelerates small vessel disease |
| Hyperlipidemia | Promotes microatheroma |
| Age | Risk doubles every decade after 55 |
| Male sex | Higher baseline risk |
| African/Asian ancestry | Higher prevalence of intracranial small vessel disease |
| Metabolic syndrome | Clustering of risk factors |
| Obstructive sleep apnea | Nocturnal BP surges damage small vessels |
Key distinction: Lacunar strokes are NOT primarily caused by atrial fibrillation or carotid atherosclerosis — those cause cortical/embolic infarcts. If a "lacunar" infarct is found with AF, a cardioembolic cause must be excluded.
5. CLASSICAL CLINICAL SYNDROMES
Syndrome 1 — Pure Motor Hemiparesis (PMH)
Most common lacunar syndrome (~50% of lacunar strokes)
| Feature | Detail |
|---|---|
| Deficit | Weakness of face + arm + leg on the SAME side (all three involved) |
| Sensation | Normal — no sensory loss |
| Location | Posterior limb of internal capsule OR basis pontis |
| Vessel | Lenticulostriate OR pontine perforator |
| Key point | Cortical features absent: no aphasia, no neglect, no visual field defect, no cortical sensory loss |
Syndrome 2 — Pure Sensory Stroke (PSS)
| Feature | Detail |
|---|---|
| Deficit | Hemisensory loss of face + arm + leg (all modalities — pain, temperature, touch, proprioception) |
| Motor | Normal — no weakness |
| Location | Ventral posteromedial/posterolateral thalamus |
| Vessel | Thalamoperforating artery |
| Key point | Classic "cheiro-oral" syndrome — tingling around mouth + ipsilateral hand |
Syndrome 3 — Ataxic Hemiparesis (AH)
| Feature | Detail |
|---|---|
| Deficit | Ipsilateral cerebellar ataxia + contralateral hemiparesis (leg > arm) — on the SAME side |
| Location | Ventral pons OR posterior limb of internal capsule |
| Vessel | Pontine perforator |
| Key point | Combination of pyramidal + cerebellar findings on same side is unique to this syndrome |
Syndrome 4 — Dysarthria-Clumsy Hand Syndrome (DCHS)
| Feature | Detail |
|---|---|
| Deficit | Dysarthria (slurred speech) + clumsiness/weakness of hand (fine motor difficulty) |
| Location | Ventral pons OR genu of internal capsule |
| Vessel | Pontine perforator |
| Key point | Facial weakness may be present; no aphasia (speech content normal, only articulation impaired) |
Summary Table of Classic Syndromes:
| Syndrome | Motor | Sensory | Ataxia | Dysarthria | Location |
|---|---|---|---|---|---|
| Pure Motor Hemiparesis | ✅ Face+Arm+Leg | ❌ | ❌ | ± | Post. limb IC / Pons |
| Pure Sensory Stroke | ❌ | ✅ All modalities | ❌ | ❌ | Ventral thalamus |
| Ataxic Hemiparesis | ✅ Leg > arm | ❌ | ✅ Same side | ❌ | Pons / IC |
| Dysarthria-Clumsy Hand | ✅ Hand (mild) | ❌ | ❌ | ✅ | Pons / Genu IC |
Additional / Less Common Syndromes:
- Sensorimotor stroke — combined motor + sensory; thalamocapsular infarct
- Hemichorea-hemiballismus — involuntary movements; subthalamic nucleus infarct
- Thalamic dementia — bilateral thalamic infarcts; severe memory loss
- Locked-in syndrome — bilateral pontine base infarcts (rare but important)
6. WHAT LACUNAR STROKE DOES NOT CAUSE
| Absent Feature | Reason |
|---|---|
| Aphasia | Cortical language areas spared |
| Neglect / agnosia | Parietal cortex spared |
| Visual field defects (hemianopia) | Occipital cortex / optic radiation may be spared |
| Seizures at onset | No cortical irritation |
| Loss of consciousness | Reticular activating system usually spared |
| Cortical sensory loss (agraphesthesia, astereognosis) | Parietal cortex spared |
Presence of ANY cortical feature in a suspected lacunar stroke → reconsider the diagnosis → look for embolic or large vessel cause.
7. IMAGING
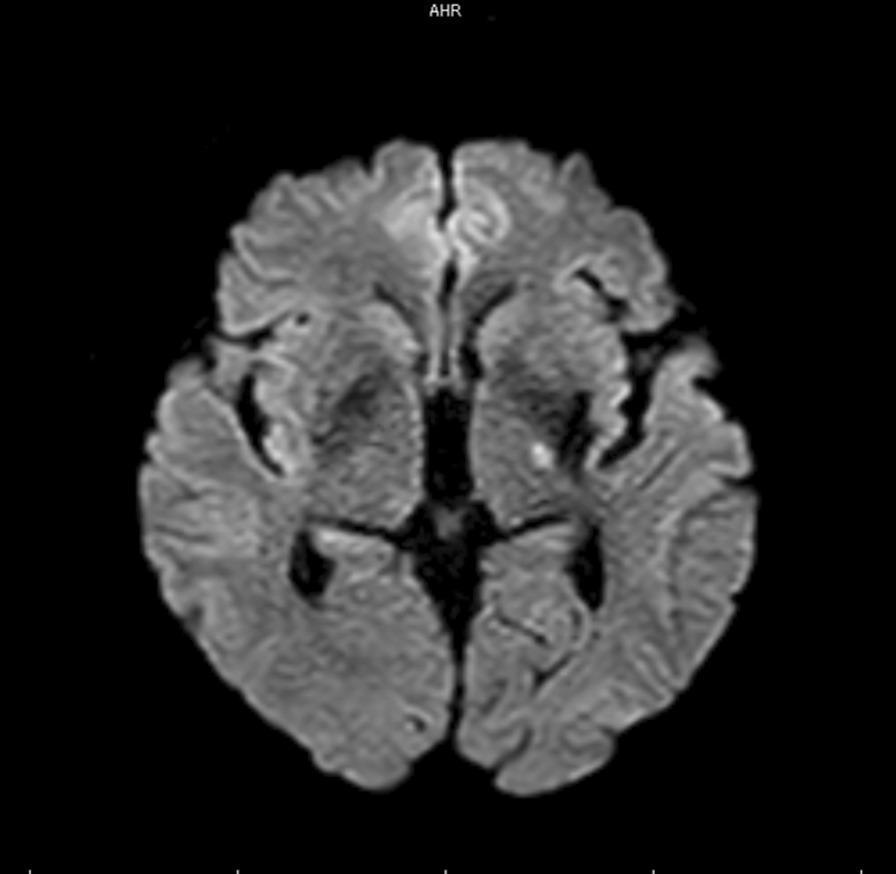
CT Brain
| Time | Finding |
|---|---|
| Acute (<24 hrs) | Often normal on CT — lesion too small to detect |
| Subacute (24–72 hrs) | Small hypodense (dark) lesion in deep structures |
| Chronic | Small fluid-filled cavity (hypodense) = "lacune" |
MRI Brain
| Sequence | Finding |
|---|---|
| DWI (Diffusion-Weighted) | Gold standard — bright spot within minutes of ictus; detects small lesions CT misses |
| ADC map | Dark (restricted diffusion) — confirms acute infarct |
| FLAIR | Lesion visible as bright signal; also shows white matter hyperintensities (leukoaraiosis) |
| T1 | Old lacunes appear as dark CSF-signal cavities |
| T2 | Bright signal in subacute/chronic lesions |
White Matter Changes (Leukoaraiosis):
- Seen on FLAIR/T2 as diffuse white matter hyperintensities
- Marker of widespread small vessel disease
- Associated with cognitive decline, gait disturbance, and recurrent lacunar strokes
- Graded by Fazekas scale (0–3)
8. DIAGNOSIS
Diagnostic Criteria for Lacunar Stroke:
- Clinical syndrome consistent with one of the classic lacunar syndromes
- No cortical signs
- Lesion <15 mm on imaging in a deep location
- No large vessel source (carotid stenosis, cardioembolic)
- Risk factors present (hypertension, diabetes)
Investigations:
| Investigation | Purpose |
|---|---|
| MRI DWI (urgent) | Confirms acute lacunar infarct |
| Non-contrast CT | Exclude hemorrhage; may miss acute lacunar |
| MRA/CTA | Exclude large vessel occlusion |
| ECG + 24-hr Holter | Exclude atrial fibrillation |
| Echocardiogram | Exclude cardioembolic source |
| Carotid Doppler | Exclude carotid stenosis |
| Fasting glucose, HbA1c | Diabetes assessment |
| Lipid profile | Dyslipidemia |
| BP monitoring (24-hr ABPM) | Ambulatory BP — assess true BP burden |
| CBC, coagulation | Baseline; exclude hematological cause |
Differential Diagnosis:
| Condition | Differentiating Feature |
|---|---|
| Cortical ischemic stroke | Cortical signs present (aphasia, neglect, seizures) |
| Intracerebral hemorrhage | Hyperdense on CT; often headache + LOC |
| Brain tumor | Subacute onset, surrounding edema, mass effect |
| Demyelination (MS) | Young patient, white matter lesions, relapsing-remitting course |
| Metabolic (hypoglycemia) | Glucose level confirms |
| Cerebral abscess | Fever, ring-enhancing lesion, diffusion restriction in core |
9. PROGNOSIS
| Feature | Lacunar vs Cortical Stroke |
|---|---|
| 30-day mortality | Lower (~2–5%) vs cortical (~15%) |
| Functional recovery | Generally better — smaller lesion, no cortical involvement |
| Recurrence risk | ~10% at 1 year; ~30% at 5 years |
| Cognitive decline | Progressive if small vessel disease advances → vascular dementia |
| Post-stroke depression | Common (~30%) |
| Gait disturbance | Progressive with recurrent lacunes — lacunar state |
Lacunar State (État Lacunaire):
- Multiple recurrent lacunar infarcts over years
- Pseudobulbar palsy — bilateral UMN lesions → emotional lability, dysarthria, dysphagia
- Marche à petits pas — short-stepped, shuffling gait
- Vascular dementia — stepwise cognitive decline
- Urinary incontinence
- Mimics Parkinson's disease but lacks tremor; rigidity is symmetrical
10. MANAGEMENT
Acute Phase
A. Thrombolysis (tPA)
- Lacunar strokes CAN receive IV tPA if within 4.5 hours and no contraindications
- However, benefit may be less clear than for large vessel strokes (smaller penumbra)
- Still recommended by AHA guidelines if eligible
B. General Supportive Care
- BP management: permissive hypertension acutely (do not lower unless >220/120)
- Glucose: target 140–180 mg/dL
- Temperature: treat fever aggressively
- Swallowing screen before oral intake
Antiplatelet Therapy (Key Treatment)
| Regimen | Details |
|---|---|
| Aspirin 75–100 mg daily | First-line long-term antiplatelet |
| Aspirin + Clopidogrel (DAPT) | For 21 days post-minor stroke/TIA (CHANCE/POINT trials) — reduces recurrence by ~25% |
| Clopidogrel 75 mg | If aspirin intolerant |
| Ticagrelor | Alternative in high-risk patients |
Anticoagulation (warfarin/NOAC) is NOT superior to antiplatelet therapy for small vessel lacunar stroke (SPS3 trial) and increases bleeding risk — do NOT use unless AF is present.
Risk Factor Control (Most Important Long-term Intervention)
| Risk Factor | Target | Drug of Choice |
|---|---|---|
| Hypertension | BP <130/80 mmHg | ACE inhibitor + thiazide (PROGRESS trial) |
| Diabetes | HbA1c <7% | Metformin ± SGLT2 inhibitor |
| Dyslipidemia | LDL <70 mg/dL | Atorvastatin 40–80 mg |
| Smoking | Complete cessation | NRT, varenicline |
| Obesity | BMI <25 | Diet + exercise |
Intensive BP lowering is the single most important intervention to prevent recurrent lacunar strokes — target SBP <130 mmHg (SPS3 trial showed benefit).
Secondary Prevention Summary:
| Intervention | Lacunar Stroke Specific |
|---|---|
| Antiplatelet | ✅ Yes (aspirin or clopidogrel) |
| Anticoagulation | ❌ No (unless AF proven) |
| Statin | ✅ Yes (high-intensity) |
| BP control | ✅ Most important — SBP <130 |
| Carotid surgery | ❌ Not indicated (no large vessel disease) |
| PFO closure | Not standard (cause is small vessel, not embolic) |
Rehabilitation
- Physiotherapy — strength training, gait rehabilitation
- Occupational therapy — ADL retraining
- Speech therapy — dysarthria, dysphagia
- Cognitive rehabilitation — for vascular mild cognitive impairment
- Psychological support — post-stroke depression (SSRIs effective)
11. QUICK EXAM SUMMARY TABLE
| Feature | Lacunar Stroke |
|---|---|
| Definition | Small deep infarct <15 mm from penetrating artery occlusion |
| Frequency | ~20–25% of ischemic strokes |
| Mechanism | Lipohyalinosis / microatheroma |
| Main risk factors | Hypertension (#1), Diabetes |
| Location | Basal ganglia, internal capsule, thalamus, pons |
| Classic syndromes | Pure motor, pure sensory, ataxic hemiparesis, dysarthria-clumsy hand |
| Absent features | No aphasia, neglect, hemianopia, seizures, LOC |
| Best imaging | MRI DWI (CT often normal acutely) |
| Acute treatment | tPA if eligible; supportive care |
| Key long-term Rx | BP control (<130/80) + antiplatelet + statin |
| Avoid | Anticoagulation (without AF) |
| Prognosis | Good short-term; vascular dementia long-term if recurrent |
- Harrison's Principles of Internal Medicine, 21st Edition (p. 12146, 12148)
- AHA/ASA Ischemic Stroke Guidelines (2019)
- SPS3 Trial (Secondary Prevention of Small Subcortical Strokes)
- CHANCE and POINT Trials (dual antiplatelet therapy)
- PROGRESS Trial (perindopril + indapamide for BP)
B;OOD SUPPLY OF BRAIN IN DEATIL
blood supply of brain arterial anatomy carotid vertebral basilar arteries
circle of Willis anatomy anterior middle posterior cerebral arteries territories
circle of Willis brain arterial supply diagram anatomy
middle cerebral artery branches territory internal carotid artery segments venous drainage brain dural sinuses
cerebral arterial territories MCA ACA PCA cortical supply brain map
BLOOD SUPPLY OF THE BRAIN — Complete Detail
OVERVIEW
| System | Artery | Supplies |
|---|---|---|
| Anterior (Carotid) | Internal Carotid Artery (ICA) × 2 | ~80% of cerebral blood flow — cerebral hemispheres |
| Posterior (Vertebrobasilar) | Vertebral Artery × 2 → Basilar Artery | ~20% — brainstem, cerebellum, occipital lobe, thalamus |
PART 1 — ANTERIOR CIRCULATION (CAROTID SYSTEM)
A. Common Carotid Artery (CCA)
- Right CCA — arises from the brachiocephalic trunk (innominate artery)
- Left CCA — arises directly from the aortic arch
- Bifurcates at the level of C4 (upper border of thyroid cartilage) into:
- External carotid artery (ECA) → face, scalp, meninges
- Internal carotid artery (ICA) → brain
B. Internal Carotid Artery (ICA) — Segments
| Segment | Name | Location | Key Branches |
|---|---|---|---|
| C1 | Cervical | Neck | None |
| C2 | Petrous | Petrous bone (carotid canal) | Caroticotympanic, vidian artery |
| C3 | Lacerum | Foramen lacerum | None |
| C4 | Cavernous | Cavernous sinus | Meningohypophyseal trunk, inferolateral trunk |
| C5 | Clinoid | Anterior clinoid process | None |
| C6 | Ophthalmic | Intradural | Ophthalmic artery, superior hypophyseal artery |
| C7 | Communicating | Cistern | Anterior choroidal artery, Posterior communicating artery (PCoA) |
ICA Terminates by dividing into:
- Anterior Cerebral Artery (ACA)
- Middle Cerebral Artery (MCA)
C. Ophthalmic Artery
- First intracranial branch of ICA (C6 segment)
- Enters orbit via optic canal
- Branches: Central retinal artery (supplies retina), posterior ciliary arteries, supraorbital, supratrochlear arteries
- Central retinal artery occlusion → sudden painless monocular blindness (like "curtain" descending) — amaurosis fugax in TIA
D. Anterior Choroidal Artery (AChA)
- Arises from ICA (C7 segment)
- Small but critically important
- Supplies:
- Posterior limb of internal capsule (genu and retrolenticular part)
- Optic tract
- Lateral geniculate body
- Hippocampus
- Amygdala
- Choroid plexus of lateral ventricle
- Occlusion → Hemiplegia + hemianesthesia + hemianopia (three H's)
E. Anterior Cerebral Artery (ACA)
| Segment | Name | Details |
|---|---|---|
| A1 (Precommunal) | Stem | From ICA to anterior communicating artery (ACoA); gives deep penetrating branches |
| A2 (Postcommunal) | Distal ACA | Beyond ACoA; gives cortical branches |
A1 Deep Branches (Medial Lenticulostriate Arteries):
- Anterior limb of internal capsule
- Anterior perforated substance
- Amygdala
- Anterior hypothalamus
- Inferior head of caudate nucleus
A2 Cortical Branches:
| Branch | Territory Supplied |
|---|---|
| Orbitofrontal artery | Orbital surface of frontal lobe |
| Frontopolar artery | Frontal pole |
| Callosomarginal artery | Cingulate gyrus, paracentral lobule |
| Pericallosal artery | Corpus callosum, medial frontal/parietal lobes |
ACA Cortical Territory:
- Medial surface of frontal and parietal lobes
- Superior 1 cm of lateral cortex (parasagittal strip)
- Paracentral lobule (motor/sensory for leg and foot)
- Anterior corpus callosum
- Anterior cingulate gyrus
ACA Occlusion Syndrome:
| Feature | Explanation |
|---|---|
| Contralateral leg weakness > arm (foot/leg most affected) | Paracentral lobule |
| Contralateral sensory loss in leg | Sensory paracentral lobule |
| Abulia (lack of initiative, apathy) | Anterior cingulate |
| Urinary incontinence | Medial frontal lobe |
| Contralateral grasp reflex | Frontal lobe |
| Alien hand syndrome | Corpus callosum disconnection |
F. Middle Cerebral Artery (MCA)
Segments:
| Segment | Location | Key Branches |
|---|---|---|
| M1 (Sphenoidal) | From ICA to Sylvian fissure | Lenticulostriate arteries (deep perforators); early MCA branches |
| M2 (Insular) | Within Sylvian fissure (insula) | Superior and inferior trunk division |
| M3 (Opercular) | Opercular cortex | Cortical branches emerge |
| M4 (Cortical) | Lateral convexity surface | Named cortical branches |
M1 Deep Branches — Lenticulostriate Arteries (LSA):
- Arise at right angles from M1
- Penetrate anterior perforated substance
- Supply: Putamen, caudate nucleus, globus pallidus, posterior limb of internal capsule, corona radiata
- These are the vessels most commonly involved in hypertensive hemorrhage and lacunar stroke
MCA Cortical Branches (M2–M4):
| Branch | Territory |
|---|---|
| Superior trunk | Frontal and parietal operculum, lateral frontal cortex |
| Orbitofrontal | Lateral orbital frontal |
| Prefrontal | Lateral prefrontal |
| Pre-Rolandic (precentral) | Premotor cortex, face/arm motor area |
| Rolandic (central) | Primary motor AND sensory cortex (face + arm) |
| Anterior parietal | Supramarginal gyrus |
| Inferior trunk | Temporal and posterior parietal lobes |
| Posterior parietal | Angular gyrus (dominant) |
| Temporal branches (ant, mid, post) | Superior/middle temporal gyrus |
MCA Cortical Territory:
- Entire lateral surface of cerebral hemisphere EXCEPT:
- Superior 1 cm strip (ACA)
- Occipital pole and inferior temporal (PCA)
- Primary motor cortex (face + arm > leg)
- Primary sensory cortex (face + arm > leg)
- Broca's area (dominant inferior frontal gyrus — speech production)
- Wernicke's area (dominant superior temporal gyrus — speech comprehension)
- Angular gyrus (reading, writing)
- Insula
- Basal ganglia, internal capsule (via LSA)
MCA Occlusion Syndromes:
| Deficit | Cause |
|---|---|
| Contralateral hemiplegia (face + arm >> leg) | Motor cortex + posterior IC |
| Contralateral hemisensory loss | Sensory cortex |
| Gaze deviation TOWARD lesion | Frontal eye fields (ipsilateral) |
| Contralateral homonymous hemianopia | Optic radiation |
| Aphasia (dominant hemisphere) | Broca's + Wernicke's areas |
| Hemispatial neglect (non-dominant) | Right parietal lobe |
| Anosognosia (unawareness of deficit) | Non-dominant hemisphere |
- Broca's (expressive) aphasia — dominant
- Contralateral face + arm weakness (leg spared)
- Contralateral face + arm sensory loss
- Wernicke's (receptive) aphasia — dominant
- Contralateral homonymous superior quadrantanopia
- Hemispatial neglect — non-dominant
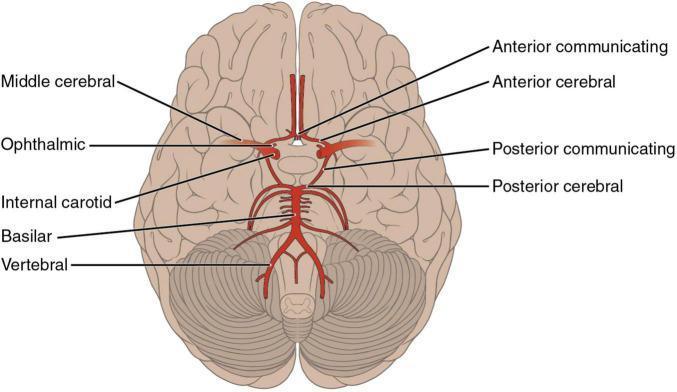
PART 2 — POSTERIOR CIRCULATION (VERTEBROBASILAR SYSTEM)
A. Vertebral Arteries (VA)
- Arise from subclavian arteries bilaterally
- Enter skull through foramen magnum
- Travel along anterior surface of medulla
- Unite at pontomedullary junction to form the Basilar Artery
Branches of Vertebral Artery:
| Branch | Supplies |
|---|---|
| Posterior Inferior Cerebellar Artery (PICA) | Lateral medulla + inferior cerebellum (most important branch) |
| Anterior Spinal Artery | Anterior 2/3 of spinal cord |
| Posterior Spinal Artery | Posterior columns of spinal cord |
| Medullary perforators | Medulla oblongata |
PICA Occlusion — Lateral Medullary Syndrome (Wallenberg Syndrome):
| Feature | Structure Affected |
|---|---|
| Ipsilateral facial pain + temperature loss | Descending trigeminal tract |
| Contralateral body pain + temperature loss | Spinothalamic tract |
| Ipsilateral Horner's syndrome (ptosis, miosis, anhidrosis) | Descending sympathetic fibers |
| Ipsilateral limb ataxia | Inferior cerebellar peduncle / cerebellum |
| Dysphagia, dysarthria, hoarseness | Nucleus ambiguus (CN IX, X) |
| Vertigo, nausea, vomiting | Vestibular nuclei |
| Hiccups | Reticular formation |
| No limb weakness | Corticospinal tract (pyramids) spared |
Memory trick: In Wallenberg — CROSS pattern — ipsilateral face, contralateral body for pain/temp loss
B. Basilar Artery
| Group | Number | Supplies |
|---|---|---|
| Paramedian perforators | 7–10 | Wedge of pons on either side of midline (corticospinal tract, MLF, CN VI nucleus) |
| Short circumferential | 5–7 | Lateral two-thirds of pons, middle and superior cerebellar peduncles |
| Long circumferential (AICA, SCA) | Bilateral | Cerebellar hemispheres |
Named Branches of Basilar Artery:
- Supplies: Lateral lower pons + inferior cerebellum + inner ear (labyrinthine artery often a branch)
- Occlusion → Lateral inferior pontine syndrome (similar to Wallenberg but at pontine level):
- Ipsilateral facial weakness (CN VII nucleus)
- Ipsilateral hearing loss, tinnitus, vertigo (CN VIII)
- Ipsilateral facial numbness (CN V)
- Ipsilateral Horner's syndrome
- Ipsilateral limb ataxia
- Contralateral body pain/temp loss
- Occlusion → Medial pontine syndromes:
- CN VI palsy (ipsilateral) + contralateral hemiplegia = Millard-Gubler syndrome
- Bilateral pontine base infarct → Locked-in syndrome (quadriplegia + preserved consciousness)
- Internuclear ophthalmoplegia (INO) — MLF lesion
- Supplies: Superior cerebellum + dorsolateral upper pons
- Occlusion:
- Ipsilateral limb ataxia, dysmetria
- Contralateral pain/temp loss (body + face)
- Ipsilateral Horner's syndrome
- Ipsilateral partial CN IV palsy
C. Posterior Cerebral Artery (PCA)
| Segment | Location |
|---|---|
| P1 (Precommunal) | From basilar to posterior communicating artery |
| P2 (Postcommunal) | Beyond PCoA; cortical branches |
P1 Deep Branches — Thalamoperforating Arteries:
- Supply thalamus (anterolateral, posteromedial nuclei)
- Posterior perforated substance
- Subthalamic nucleus
- Midbrain (cerebral peduncle, red nucleus, substantia nigra)
P2 Cortical Branches:
| Branch | Territory |
|---|---|
| Calcarine artery | Primary visual cortex (V1) — occipital pole |
| Posterior temporal artery | Inferior temporal gyrus, fusiform gyrus |
| Parieto-occipital artery | Cuneus, precuneus |
| Posterior choroidal arteries | Choroid plexus of 3rd ventricle, posterior thalamus, hippocampus |
PCA Cortical Territory:
- Occipital lobe (primary + association visual cortex)
- Inferior and medial temporal lobe (fusiform, parahippocampal gyrus, hippocampus)
- Posterior thalamus
- Midbrain
- Splenium of corpus callosum
PCA Occlusion Syndromes:
| Syndrome | Features |
|---|---|
| Complete PCA (P2) | Contralateral homonymous hemianopia WITH macular sparing (macular cortex has dual supply from MCA) |
| Bilateral PCA | Cortical blindness (Anton's syndrome — patient unaware of blindness); Balint's syndrome |
| Dominant PCA | Visual object agnosia, alexia without agraphia (can write but can't read), anomic aphasia |
| Non-dominant PCA | Contralateral visual neglect, prosopagnosia (can't recognize faces), topographagnosia |
| P1 + thalamic | Thalamic syndrome (Dejerine-Roussy): contralateral hemianesthesia + burning dysesthetic pain |
| Midbrain (top of basilar) | Weber syndrome — CN III palsy + contralateral hemiplegia; vertical gaze palsy |
PART 3 — CIRCLE OF WILLIS
ACA (left) ——— ACoA ——— ACA (right)
| |
ICA (left) ICA (right)
| |
PCoA (left) PCoA (right)
| |
PCA (left) ——— Basilar ——— PCA (right)
|
Vertebral ×2
Components:
| Vessel | Full Name | Connects |
|---|---|---|
| ACoA | Anterior Communicating Artery | Left ACA ↔ Right ACA |
| PCoA (×2) | Posterior Communicating Artery | ICA ↔ PCA (anterior ↔ posterior circulations) |
| ACA A1 segment (×2) | Anterior Cerebral Artery proximal | ICA → ACoA |
| PCA P1 segment (×2) | Posterior Cerebral Artery proximal | Basilar → PCoA |
Function of Circle of Willis:
- Collateral circulation — if one artery is occluded, blood can reach the territory via the circle
- Provides pressure equalization across cerebral vasculature
- Only 20–25% of people have a complete, functional Circle of Willis — many have hypoplastic or absent segments
Clinical Importance:
| Anomaly | Clinical Significance |
|---|---|
| Hypoplastic A1 segment | ACA territory at risk if ICA occludes |
| Fetal PCA (PCA arising from ICA rather than basilar) | Posterior territory at risk with ICA disease |
| Hypoplastic PCoA | No collateral between anterior and posterior circulations |
| Aneurysms at communicating arteries | ACoA = most common site for berry aneurysm (~30%); PCoA = CN III palsy when ruptures |
PART 4 — INTERNAL CAPSULE BLOOD SUPPLY
| Region of Internal Capsule | Blood Supply |
|---|---|
| Anterior limb | Medial lenticulostriate (ACA) + lateral lenticulostriate (MCA) |
| Genu | Lateral lenticulostriate (MCA) + anterior choroidal artery |
| Posterior limb | Lateral lenticulostriate (MCA) + anterior choroidal artery |
| Retrolenticular | Anterior choroidal artery + PCA |
| Sublenticular | Anterior choroidal artery |
A small infarct in the posterior limb of IC → pure motor hemiparesis (entire body — face + arm + leg) — classic lacunar syndrome
PART 5 — BRAINSTEM BLOOD SUPPLY SUMMARY
| Level | Artery | Key Structures Supplied |
|---|---|---|
| Midbrain | PCA, SCA, basilar perforators | Cerebral peduncles (CN III, corticospinal tract), red nucleus, substantia nigra, PAG |
| Pons (upper) | SCA, basilar perforators | Corticospinal + corticobulbar tracts, CN IV, V nuclei, MLF |
| Pons (mid/lower) | AICA, basilar perforators | CN VI, VII nuclei, MLF, PPRF, spinothalamic tract |
| Medulla (lateral) | PICA | Vestibular nuclei, CN IX/X, descending trigeminal, spinothalamic, descending sympathetics |
| Medulla (medial) | Anterior spinal + vertebral perforators | Pyramid (CST), CN XII nucleus, medial lemniscus |
Key Brainstem Stroke Syndromes:
| Syndrome | Vessel | Key Deficits |
|---|---|---|
| Wallenberg (Lateral medullary) | PICA/VA | Ipsilateral face + contralateral body pain/temp; Horner; dysphagia; vertigo; NO weakness |
| Medial medullary | Anterior spinal/VA | Contralateral hemiplegia; ipsilateral CN XII palsy; contralateral loss of position/vibration |
| Millard-Gubler (Lateral pontine) | AICA/basilar | Ipsilateral CN VI + VII palsy; contralateral hemiplegia |
| Raymond syndrome | Basilar perforator | Ipsilateral CN VI palsy + contralateral hemiplegia (no CN VII) |
| Weber (Midbrain) | PCA/basilar | Ipsilateral CN III palsy + contralateral hemiplegia |
| Benedikt | PCA | Ipsilateral CN III + contralateral tremor/ataxia (red nucleus) |
| Locked-in | Basilar (bilateral pontine) | Quadriplegia + bulbar palsy; consciousness preserved; eye blinking only |
| Top of basilar | Bilateral PCA + SCA | Bilateral visual loss + altered consciousness + vertical gaze palsy |
PART 6 — VENOUS DRAINAGE OF THE BRAIN
Superficial (Cortical) Veins:
| Vein | Drains |
|---|---|
| Superior cerebral veins (8–12) | Superior lateral + medial hemisphere → superior sagittal sinus |
| Superficial middle cerebral vein | Sylvian fissure region → cavernous sinus |
| Vein of Trolard (superior anastomotic) | Connects superficial MCV to superior sagittal sinus |
| Vein of Labbé (inferior anastomotic) | Connects superficial MCV to transverse sinus |
Deep (Central) Veins:
| Vein | Drains |
|---|---|
| Internal cerebral veins (×2) | Thalamus, basal ganglia, corpus callosum → unite to form Great Cerebral Vein |
| Basal vein of Rosenthal | Basal structures (hippocampus, basal ganglia) → Great Cerebral Vein |
| Great Cerebral Vein of Galen | All deep veins → straight sinus |
Dural Venous Sinuses:
| Sinus | Location | Drains Into |
|---|---|---|
| Superior Sagittal Sinus (SSS) | Upper border of falx cerebri | Confluence of sinuses (torcular Herophili) |
| Inferior Sagittal Sinus (ISS) | Lower border of falx | Straight sinus |
| Straight Sinus | Junction of falx + tentorium | Confluence |
| Confluence of Sinuses (Torcular) | Internal occipital protuberance | Transverse sinuses bilaterally |
| Transverse Sinuses | Groove on occipital bone | Sigmoid sinuses |
| Sigmoid Sinuses | S-shaped groove | Internal Jugular Vein (IJV) |
| Cavernous Sinuses | Each side of sella turcica | Via petrosal sinuses → IJV |
| Occipital Sinus | Falx cerebelli | Confluence |
Cavernous Sinus — Special Importance:
- CN III, IV, V1, V2, VI in its walls/lumen
- ICA (with sympathetic plexus) traversing it
- Communicates across midline via intercavernous sinuses
Cerebral Venous Sinus Thrombosis (CVST):
- Clotting in dural venous sinuses
- Causes: hypercoagulability, OCP, pregnancy, dehydration, infection, malignancy
- SSS most commonly affected
- Presents: headache, seizures, focal deficits, papilledema, venous infarction (often hemorrhagic)
- Treatment: anticoagulation (even with hemorrhagic infarct)
COMPLETE SUMMARY TABLE
| Artery | Origin | Territory | Key Occlusion Syndrome |
|---|---|---|---|
| Ophthalmic | ICA (C6) | Retina, orbit | Amaurosis fugax (TIA) |
| AChA | ICA (C7) | Post. limb IC, optic tract, hippocampus | Hemiplegia + hemianesthesia + hemianopia |
| ACA | ICA | Medial frontal/parietal, paracentral lobule | Contralateral LEG weakness, abulia |
| MCA | ICA | Lateral hemisphere, BG, IC | Contralateral face+arm > leg, aphasia, neglect |
| Lenticulostriate | MCA (M1) | Basal ganglia, IC | Lacunar stroke, hypertensive hemorrhage |
| PICA | Vertebral | Lateral medulla, inf. cerebellum | Wallenberg syndrome |
| AICA | Basilar | Lateral pons, inf. cerebellum, inner ear | Lateral inferior pontine syndrome |
| SCA | Basilar | Sup. cerebellum, dorsolateral pons | Ipsilateral ataxia + contralateral pain/temp |
| PCA | Basilar | Occipital, inf. temporal, thalamus | Contralateral homonymous hemianopia |
| Basilar | VA union | Pons, midbrain (via branches) | Locked-in, top-of-basilar, coma |
| PCoA | ICA/PCA | Thalamus, midbrain | CN III palsy (aneurysm); connects circulations |
- Harrison's Principles of Internal Medicine, 21st Edition (p. 12109, 12115, 12148)
- Gray's Anatomy for Students
- Neuroanatomy: An Illustrated Colour Text
- AHA/ASA Stroke Guidelines (2019)