Spinal muscular atrophy in detail
Reading File
search_medical_text
spinal muscular atrophy genetics pathophysiology SMN1 SMN2
search_medical_text
spinal muscular atrophy types classification clinical features diagnosis treatment
search_medical_image
spinal muscular atrophy motor neuron degeneration
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive overview of Spinal Muscular Atrophy (SMA).
Spinal Muscular Atrophy (SMA)
Definition
SMA is a family of hereditary lower motor neuron diseases characterized by progressive degeneration of anterior horn cells in the spinal cord (and sometimes brainstem motor nuclei), leading to muscle weakness and atrophy. It is the leading genetic cause of infant death and one of the most common autosomal recessive disorders.
Genetics & Molecular Pathophysiology
| Gene | Chromosome | Role |
|---|---|---|
| SMN1 (Survival Motor Neuron 1) | 5q13.2 | Produces full-length, functional SMN protein |
| SMN2 | 5q13.2 (inverted duplication) | Produces ~10–15% functional SMN protein; remainder is truncated (exon 7 skipped) |
- ~95% of SMA cases: Homozygous deletion or mutation of SMN1 (loss of function)
- SMN2 copy number is the primary disease modifier — more copies = milder phenotype
- SMN protein is essential for the formation and trafficking of RNA complexes (snRNPs) across the nuclear membrane, critical for pre-mRNA splicing in all cells, but motor neurons are uniquely vulnerable (Harrison's, p. 12410)
- Deficiency leads to selective degeneration of large motor neurons → denervation atrophy of skeletal muscle
Importantly, SMN2 cannot fully compensate for SMN1 loss because of a single nucleotide difference (C→T at position +6 in exon 7) that causes exon 7 skipping during splicing, yielding a truncated, unstable protein (SMNΔ7).
Classification by Type
| Type | Onset | Motor Milestones | SMN2 Copies | Natural Course |
|---|---|---|---|---|
| Type 0 (prenatal) | In utero | No movement at birth | 1 | Fatal within weeks |
| Type 1 (Werdnig-Hoffmann) | < 6 months | Never sit independently | 2 | Death by 2 yrs (if untreated) |
| Type 2 (intermediate) | 6–18 months | Sit but never stand | 3 | Survives to adulthood; wheelchair-bound |
| Type 3 (Kugelberg-Welander) | > 18 months | Walk independently | 3–4 | Near-normal lifespan; may lose ambulation |
| Type 4 (adult-onset) | > 21 years | Walk throughout adult life | 4+ | Near-normal lifespan |
Clinical Features
Type 1 (Most Common, Most Severe)
- Profound hypotonia ("floppy infant")
- Symmetric proximal > distal weakness
- Tongue fasciculations
- Areflexia
- Paradoxical breathing (diaphragm relatively spared → bell-shaped chest)
- Bulbar weakness → poor suck, swallowing difficulties
- Preserved sensation and cognition
- Alert, bright facial expression (classic finding)
Type 2
- Achieve sitting, never standing/walking
- Scoliosis (universal), joint contractures
- Respiratory compromise (restrictive lung disease)
- Tremor of outstretched hands (minipolymyoclonus)
Type 3
- Walk independently but fall frequently
- Proximal limb weakness (Gowers' sign)
- Waddling gait
- May lose ambulation during 2nd–3rd decade
- CK mildly elevated
Type 4 (Adult-Onset)
- Slowly progressive proximal weakness
- Fasciculations
- Minimal respiratory involvement
Diagnosis
1. Genetic Testing (First-Line)
- MLPA or real-time PCR for homozygous deletion of exon 7 of SMN1 — confirms diagnosis in ~95% of cases
- SMN2 copy number should be determined simultaneously (prognostic)
- Full SMN1 sequencing for compound heterozygotes
2. Supportive Investigations
| Test | Finding |
|---|---|
| EMG/NCS | Fibrillations, positive sharp waves, large polyphasic motor unit potentials; NCS normal |
| Muscle biopsy | Grouped denervation atrophy (large groups of atrophic fibers), hypertrophic fibers; rarely needed now |
| CK | Normal to mildly elevated |
| Newborn screening | Now included in many national programs (dried blood spot PCR) |
Differential Diagnosis
- Duchenne/Becker muscular dystrophy
- Congenital myopathies (nemaline, centronuclear)
- Myotonic dystrophy (congenital)
- Pompe disease
- Congenital myasthenic syndromes
- Botulism (infantile)
- X-linked spinobulbar muscular atrophy (Kennedy disease) — adults
Treatment
Disease-Modifying Therapies (DMTs)
| Drug | Mechanism | Route | Approval |
|---|---|---|---|
| Nusinersen (Spinraza) | Antisense oligonucleotide (ASO) — modifies SMN2 splicing to include exon 7, increasing functional SMN protein | Intrathecal (every 4 months after loading) | FDA 2016 |
| Onasemnogene abeparvovec (Zolgensma) | AAV9 gene therapy — delivers functional SMN1 copy | Single IV infusion | FDA 2019 (< 2 yrs) |
| Risdiplam (Evrysdi) | Small molecule SMN2 splicing modifier | Oral daily | FDA 2020 |
Key notes (Harrison's, p. 13374):
- Zolgensma is a true disease-modifying gene therapy; requires pre-treatment and 30-day post-treatment corticosteroids to prevent hepatotoxicity (liver transaminase monitoring essential)
- Nusinersen efficacy data from trials (ENDEAR, CHERISH) showed significant motor milestone gains, especially when started early; requires repeat intrathecal dosing every 4 months
- Risdiplam offers the advantage of oral administration, with efficacy comparable to nusinersen in several types
- Pre-symptomatic treatment (via newborn screening) yields dramatically superior outcomes vs. symptomatic treatment
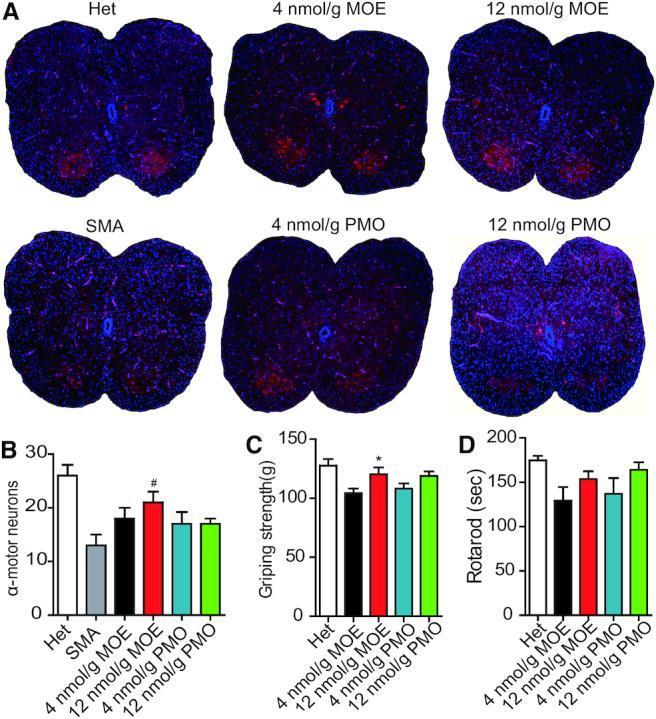
Supportive & Multidisciplinary Care
| Domain | Intervention |
|---|---|
| Respiratory | Non-invasive ventilation (BiPAP), cough assist, airway clearance; tracheostomy in severe cases |
| Nutritional | Gastrostomy feeding (Type 1/2), anti-reflux measures |
| Orthopedic | Scoliosis surveillance and surgery, joint contracture management, seating/positioning |
| Rehabilitation | Physical therapy, occupational therapy, speech-language pathology |
| Neurology | Regular monitoring of motor function (HFMSE, RULM, MFM scales) |
| Genetics | Carrier testing of family members; genetic counseling |
Prognosis
- Untreated Type 1: median survival < 2 years; death from respiratory failure
- Treated (pre-symptomatic) Type 1: majority achieve sitting and many achieve walking milestones — a transformative change from the natural history
- Types 2–3: lifespan near-normal with good respiratory management; quality of life depends on functional preservation
- Type 4: slow progression, near-normal lifespan
Newborn Screening
SMA is now included in the Recommended Uniform Screening Panel (RUSP) in the US and many other countries. Early identification before symptom onset allows treatment initiation when the largest number of motor neurons remain viable — this is the single most important prognostic factor.
Key Takeaways
- SMA is caused by SMN1 loss → deficient SMN protein → motor neuron death
- SMN2 copy number is the main disease severity modifier
- Four major types (0–4) defined by onset age and motor milestones
- Three approved DMTs: nusinersen (intrathecal ASO), onasemnogene (gene therapy), risdiplam (oral)
- Pre-symptomatic treatment via newborn screening produces far better outcomes than treating symptomatic patients
- Multidisciplinary care (respiratory, nutritional, orthopedic) remains essential alongside DMTs
This is a shared conversation. Sign in to Orris to start your own chat.