explain in detail about diabetic neuropathy in deatikl
diabetic neuropathy pathophysiology classification types symptoms
diabetic neuropathy diagnosis treatment management guidelines
diabetic neuropathy nerve damage diagram
Diabetic Neuropathy — A Detailed Overview
Definition
Pathophysiology
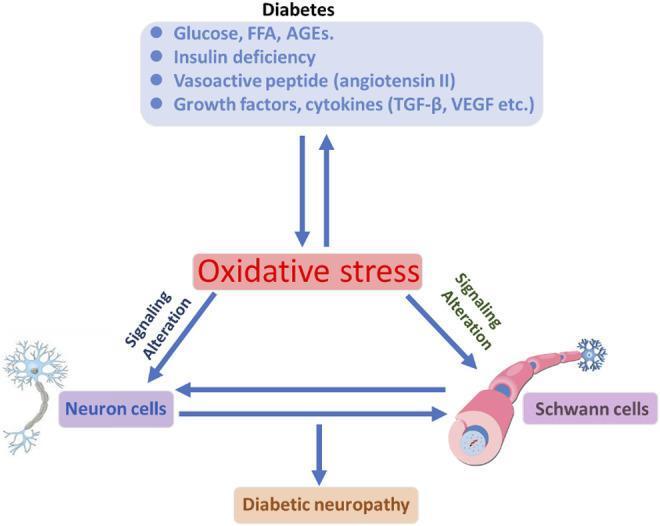
1. Hyperglycemia-Induced Oxidative Stress
- Chronic hyperglycemia leads to excessive production of reactive oxygen species (ROS)
- ROS cause oxidative damage to neurons and Schwann cells (which form the myelin sheath)
- This creates a bidirectional cycle of cellular signaling dysfunction
2. Polyol Pathway Activation
- Excess glucose is converted to sorbitol by aldose reductase
- Sorbitol accumulates inside nerve cells, causing osmotic damage and depleting myoinositol and NADPH
- This reduces nitric oxide production and impairs nerve conduction
3. Advanced Glycation End Products (AGEs)
- Glucose non-enzymatically binds to proteins, forming AGEs
- AGEs modify myelin proteins, nerve connective tissue, and cross-link structural proteins, impairing nerve function
4. PKC (Protein Kinase C) Activation
- Hyperglycemia activates PKC, which disrupts vascular tone regulation
- Leads to reduced endoneurial blood flow and ischemia of peripheral nerves
5. Free Fatty Acids, Insulin Deficiency, Vasoactive Peptides (Angiotensin II)
- These factors synergize with oxidative stress to amplify neuronal and Schwann cell injury
6. Microvascular Disease
- Thickening of endoneurial capillary basement membrane
- Reduced nerve blood flow → ischemia → axonal degeneration
Classification of Diabetic Neuropathy
| Type | Description |
|---|---|
| Distal Symmetric Polyneuropathy (DSPN) | Most common; "stocking-glove" pattern of sensory loss |
| Diabetic Autonomic Neuropathy | Affects autonomic nerves (cardiovascular, GI, GU systems) |
| Diabetic Focal/Multifocal Neuropathy | Mononeuropathy, cranial neuropathy, radiculoplexopathy |
| Diabetic Proximal Motor Neuropathy | Also called diabetic amyotrophy; proximal lower limb weakness |
1. Distal Symmetric Polyneuropathy (DSPN)
Symptoms (Sensory-Predominant)
- Numbness and tingling — begins in the toes and feet, ascends proximally ("stocking-glove distribution")
- Burning pain — usually worse at rest and at night
- Hyperesthesia — heightened sensitivity to touch
- Paresthesia/Dysesthesia — abnormal, often unpleasant sensations
- Sharp or shooting pain in the feet/legs
- Up to 50% of patients are asymptomatic, discovered only on examination
Two Subtypes of Pain
| Subtype | Features |
|---|---|
| Acute painful DN | Lasts <12 months; may follow rapid improvement in glycemic control ("insulin neuritis") |
| Chronic painful DN | Persists >12 months; difficult to treat |
Clinical Progression
- Early: Sensory symptoms (tingling, burning)
- Middle: Pain subsides but sensory loss persists; loss of protective sensation
- Late: Motor deficits develop (weakness, wasting of small foot muscles)
- End-stage: Severe sensory loss → foot ulcers, Charcot arthropathy
Physical Examination Findings
- Loss of sensation to 10-g monofilament (Semmes-Weinstein)
- Loss of vibration sense (128 Hz tuning fork)
- Loss of ankle deep-tendon reflexes
- Abnormal position sense (proprioception)
- Small muscle wasting in the feet
2. Diabetic Autonomic Neuropathy (DAN)
Cardiovascular
- Resting tachycardia (HR >100 bpm)
- Orthostatic hypotension (BP drop >20 mmHg systolic on standing)
- Reduced heart rate variability
- Silent myocardial ischemia (no chest pain due to cardiac denervation)
- Increased risk of sudden cardiac death
Gastrointestinal
- Gastroparesis diabeticorum — delayed gastric emptying; nausea, early satiety, vomiting, bloating
- Diabetic enteropathy — nocturnal diarrhea (alternating with constipation)
- Esophageal dysmotility — dysphagia
Genitourinary
- Neurogenic bladder — incomplete emptying, overflow incontinence, recurrent UTIs
- Erectile dysfunction — present in 30–75% of diabetic males
- Retrograde ejaculation
- Female sexual dysfunction — decreased vaginal lubrication
Sudomotor (Sweat Gland)
- Anhidrosis (decreased sweating) distally → compensatory hyperhidrosis proximally
- Dry, cracked skin in feet → entry point for infections
Pupillary
- Reduced pupillary dilation in dark
- Impaired adaptation to darkness
3. Focal and Multifocal Neuropathies
Cranial Neuropathy
- Most commonly affects CN III (oculomotor nerve)
- Presents with painful ptosis and ophthalmoplegia, with pupil sparing (distinguishes from posterior communicating artery aneurysm)
- Also CN IV, VI, VII may be affected
Mononeuropathy (Entrapment)
- Diabetics are predisposed to nerve compression:
- Carpal tunnel syndrome (median nerve) — most common
- Ulnar neuropathy
- Peroneal nerve palsy → foot drop
- Lateral femoral cutaneous nerve → meralgia paresthetica
Diabetic Radiculoplexopathy (Diabetic Amyotrophy / Bruns-Garland Syndrome)
- Affects older T2DM patients
- Sudden onset severe proximal leg pain, followed by weakness and wasting (quadriceps most affected)
- Unilateral or asymmetric
- Mechanism: microvasculitis of lumbosacral plexus/nerve roots
- Gradual recovery over months to years
Diagnosis
Clinical Assessment Tools
| Test | What It Detects |
|---|---|
| 10-g Semmes-Weinstein monofilament | Loss of protective sensation |
| 128-Hz tuning fork | Vibration sense (large fiber) |
| Pinprick sensation | Small fiber (pain pathway) |
| Temperature discrimination | Small fiber |
| Ankle reflexes | Early loss in DSPN |
| Proprioception testing | Posterior column function |
Scoring Systems
- Michigan Neuropathy Screening Instrument (MNSI)
- Toronto Clinical Neuropathy Score
- Neuropathy Symptom Score (NSS) and Neuropathy Disability Score (NDS)
Electrophysiological Testing
- Nerve Conduction Studies (NCS) — gold standard for large fiber neuropathy; detects reduced amplitudes and slowed conduction velocity
- Electromyography (EMG) — detects denervation in muscles
Specialized Tests
- Quantitative Sensory Testing (QST) — thresholds for vibration, temperature (small and large fiber)
- Skin punch biopsy — measures intraepidermal nerve fiber density (IENFD); best test for small fiber neuropathy
- Quantitative Sudomotor Axon Reflex Test (QSART) — for autonomic sudomotor function
- Tilt table test, heart rate variability — for cardiovascular autonomic neuropathy
Screening Recommendations (ADA)
- All T2DM patients: screen at diagnosis, then annually
- T1DM patients: screen 5 years after diagnosis, then annually
Treatment and Management
A. Disease-Modifying (Addressing the Cause)
| Strategy | Benefit |
|---|---|
| Intensive glycemic control | Most effective in T1DM (DCCT trial); modest benefit in T2DM |
| HbA1c target <7% | Slows progression |
| Cardiovascular risk factor control | BP, lipids, smoking cessation |
| Foot care and offloading | Prevents ulceration |
B. Symptomatic Pharmacological Treatment (Painful DSPN)
| Drug | Class | Notes |
|---|---|---|
| Duloxetine (60–120 mg/day) | SNRI | FDA-approved; also treats depression |
| Pregabalin (150–600 mg/day) | α2δ ligand | FDA-approved; also treats anxiety/epilepsy |
| Drug | Class |
|---|---|
| Gabapentin (300–3600 mg/day) | α2δ ligand |
| Tricyclic antidepressants (amitriptyline, nortriptyline) | TCA |
| Venlafaxine | SNRI |
| Carbamazepine | Anticonvulsant |
| Tramadol | Weak opioid |
| Agent | Notes |
|---|---|
| Capsaicin 0.075% cream | OTC; requires repeated application |
| Capsaicin 8% patch | Must be applied by healthcare provider |
| Lidocaine patches | Local analgesia |
- Tapentadol (centrally acting opioid/NRI) — FDA-approved for painful DPN
- Opioids — generally avoided as long-term therapy due to dependence risk
C. Non-Pharmacological
- Physical therapy — balance training, strengthening
- Transcutaneous Electrical Nerve Stimulation (TENS)
- Spinal cord stimulation — for refractory cases
- Cognitive behavioral therapy (CBT) — for chronic pain coping
D. Management of Autonomic Neuropathy
| Manifestation | Treatment |
|---|---|
| Gastroparesis | Metoclopramide, domperidone, erythromycin, small frequent meals |
| Orthostatic hypotension | Fludrocortisone, midodrine, compression stockings |
| Erectile dysfunction | PDE-5 inhibitors (sildenafil), vacuum devices |
| Neurogenic bladder | Timed voiding, anticholinergics or bethanechol, catheterization |
| Diarrhea | Loperamide, clonidine, antibiotics if SIBO |
Complications
| Complication | Details |
|---|---|
| Diabetic Foot Ulcer | Loss of protective sensation → unnoticed trauma → ulcer → infection → amputation |
| Charcot Arthropathy | Neuropathic joint destruction; painless, warm, swollen foot |
| Lower Limb Amputation | DM is the leading cause of non-traumatic amputation globally |
| Falls and Fractures | Loss of proprioception + balance → increased fall risk |
| Sudden Cardiac Death | Due to cardiac autonomic neuropathy |
| Infection | Impaired wound healing + loss of sensation = serious soft tissue infections |
Prognosis
- DSPN is largely irreversible once established; treatment is aimed at slowing progression and controlling symptoms
- Acute painful neuropathy (treatment-induced) generally resolves within 12 months
- Focal neuropathies (mononeuropathy, amyotrophy) often partially or fully recover spontaneously
- Autonomic neuropathy significantly worsens prognosis — cardiovascular autonomic neuropathy increases mortality risk by 2–5 fold
- Early detection and strict glycemic control remain the most effective preventive strategies
Key Takeaways
- Diabetic neuropathy is the most common complication of diabetes
- DSPN is the most common subtype — stocking-glove sensory loss, worse at night
- Pathogenesis involves oxidative stress, polyol pathway, AGEs, and microvascular ischemia
- 50% of patients are asymptomatic — routine annual screening is essential
- Duloxetine and pregabalin are first-line FDA-approved treatments for painful DPN
- Autonomic neuropathy can affect every organ system and increases mortality
- Foot care is critical — diabetic neuropathy is the leading cause of preventable lower limb amputation
toracic outlet synfrome in deatil
thoracic outlet syndrome types pathophysiology anatomy etiology
thoracic outlet syndrome clinical features diagnosis treatment surgery
thoracic outlet syndrome anatomy diagram scalene triangle costoclavicular
Thoracic Outlet Syndrome (TOS) — Detailed Overview
Definition
Anatomy of the Thoracic Outlet
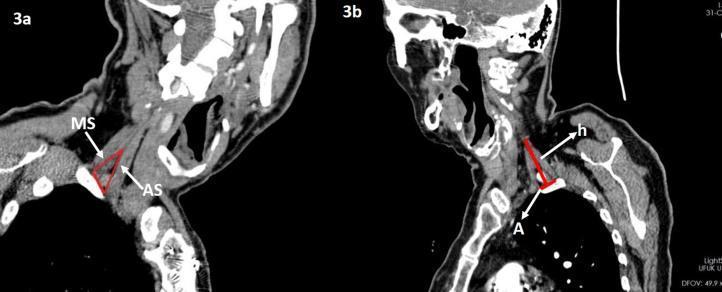
1. Interscalene Triangle
- Boundaries: Anterior scalene muscle (anteriorly), middle scalene muscle (posteriorly), first rib (inferiorly)
- Contents: Brachial plexus and subclavian artery pass through here
- Most common site of neurogenic and arterial TOS
2. Costoclavicular Space
- Boundaries: Clavicle (superiorly), first rib (inferiorly), superior border of scapula (posteriorly)
- Contents: Subclavian vein, artery, and brachial plexus
- Compressed during shoulder depression or retraction
3. Subcoracoid (Retropectoralis Minor) Space
- Boundaries: Pectoralis minor tendon (anteriorly), coracoid process (superiorly), ribs (posteriorly)
- Compression occurs when the arm is raised overhead
Etiology and Risk Factors
Structural/Anatomical Causes
| Cause | Details |
|---|---|
| Cervical rib | Rudimentary extra rib arising from C7 vertebra; present in ~0.5% of population |
| Elongated C7 transverse process | Acts as anchor for anomalous fibrocartilaginous band; key cause of true neurogenic TOS |
| Anomalous fibrocartilaginous band | Connects C7 transverse process to first rib; compresses lower brachial plexus |
| Abnormal scalene muscles | Hypertrophy, fibrous bands, or anomalous insertion of scalenus anticus |
| Clavicle fracture malunion | Callus or displaced fragment narrows costoclavicular space |
| First rib abnormalities | Exostoses or fusion anomalies |
| Pectoralis minor abnormality | Anomalous insertion compresses neurovascular bundle |
Acquired/Positional Causes
- Poor posture — forward head, rounded shoulders narrow the outlet
- Repetitive overhead activities — swimmers, baseball pitchers, painters, electricians
- Rapid weight gain — fat deposition narrows spaces
- Neck/shoulder muscle hypertrophy — bodybuilders
- Trauma — whiplash injury, clavicle or first rib fractures
- Effort thrombosis — repetitive upper limb exertion causing venous TOS (Paget-Schroetter syndrome)
Predisposing Demographics
- More common in women (3:1 female-to-male ratio for neurogenic TOS)
- Peak age: 20–50 years
- Common in athletes and manual workers
Classification
| Type | Structure Compressed | Frequency |
|---|---|---|
| Neurogenic TOS (nTOS) | Brachial plexus | ~95% of all TOS |
| Venous TOS (vTOS) | Subclavian/axillary vein | ~3–4% |
| Arterial TOS (aTOS) | Subclavian artery | ~1–2% |
1. Neurogenic TOS (nTOS) — Most Common (~95%)
Subtypes
| Subtype | Description |
|---|---|
| True (Classic) Neurogenic TOS | Objective neurological deficit; caused by anomalous band |
| Disputed (Non-specific) Neurogenic TOS | Symptoms without objective findings; controversial diagnosis |
Pathophysiology
- Compression of the lower trunk of the brachial plexus (C8–T1 nerve roots) by an anomalous fibrocartilaginous band
- The elongated C7 transverse process anchors this band, creating a tether that distorts and compresses the lower trunk
- Results in denervation of intrinsic hand muscles (ulnar and median innervated)
Clinical Features — True Neurogenic TOS
- Pain: Mild or absent (distinguishes it from disputed TOS)
- Weakness and wasting: Intrinsic muscles of the hand — particularly thenar (median) and hypothenar (ulnar) muscles
- Sensory loss: Palmar aspect of the 5th digit and medial forearm (T1 distribution)
- Insidious progression: Gradual muscle atrophy over months to years
- The classic presentation: Gilliatt-Sumner hand — wasting of thenar eminence > hypothenar
Clinical Features — Disputed Neurogenic TOS
- Shoulder and arm pain — diffuse, aching
- Paresthesias — tingling in arm and hand, often in ulnar distribution
- Neck pain — radiating to occipital region
- Headache — especially occipital
- Weakness — subjective, without objective muscle atrophy
- Symptoms reproduced or worsened by overhead activities, carrying bags, certain head positions
2. Venous TOS (vTOS) — Paget-Schroetter Syndrome (~3–4%)
Pathophysiology
- Compression of the subclavian and axillary vein in the costoclavicular space
- Repetitive arm activity → microtrauma → perivenous fibrosis → acute thrombosis
- Often precipitated by an episode of unusual or strenuous arm effort ("effort thrombosis")
Clinical Features
- Sudden onset of arm swelling, heaviness, and cyanosis
- Pitting edema of the entire upper extremity
- Dilated superficial collateral veins visible over shoulder and chest wall
- Arm pain and fatigue with use
- Usually affects the dominant arm
- Risk of pulmonary embolism (less common than DVT of lower extremity but documented)
- Common in young athletes — swimmers, rowers, baseball pitchers, weightlifters
3. Arterial TOS (aTOS) — Rarest (~1–2%)
Pathophysiology
- Compression of the subclavian artery between the cervical rib (or anomalous first rib) and the clavicle
- Chronic compression → post-stenotic dilatation → aneurysm formation → thrombus → distal embolization
Clinical Features
- Arm claudication — pain with upper limb exertion, relieved by rest
- Raynaud's phenomenon — episodic color changes in fingers (white → blue → red)
- Digital ischemia — cold, pale, painful fingers
- Ischemic tissue loss — ulceration of fingertips
- Gangrene in severe/advanced cases
- Pulsatile mass in the supraclavicular fossa (if subclavian artery aneurysm present)
- Absent or diminished radial pulse with provocative maneuvers
Clinical Provocative Tests
| Test | Technique | Positive Result | Best For |
|---|---|---|---|
| Adson's Test | Extend neck, rotate head to affected side, deep inspiration, abduct arm | Obliteration of radial pulse | Arterial TOS |
| Wright's Test (Hyperabduction) | Abduct arm to 180°, externally rotate | Pulse obliteration or symptom reproduction | Arterial/neurogenic |
| Roos Test (EAST) | Arms abducted 90°, elbows flexed 90°, open/close fists x 3 min | Arm fatigue, heaviness, paresthesias | Neurogenic TOS |
| Costoclavicular Test | Shoulders drawn back and down (military posture) | Pulse obliteration | Costoclavicular compression |
| Upper Limb Tension Test | Cervical side-bend + arm abduction/extension | Neurogenic reproduction | Neurogenic TOS |
Important: Pulse obliteration alone on these tests is not diagnostic — it occurs in up to 50% of asymptomatic individuals. Clinical correlation is mandatory.
Diagnosis
History and Physical Examination
- Detailed occupational and activity history
- Neurological examination: muscle strength, sensory testing, deep tendon reflexes
- Vascular examination: pulse assessment, blood pressure in both arms, capillary refill
Imaging
| Modality | Findings |
|---|---|
| Plain X-ray (AP cervical spine) | Elongated C7 transverse process, cervical rib, first rib abnormalities |
| Chest X-ray | Cervical rib, lung apex pathology (Pancoast tumor DDx) |
| CT Angiography / MR Angiography | Arterial stenosis, aneurysm, post-stenotic dilatation, venous thrombosis |
| Duplex Ultrasound | Venous thrombosis, arterial flow in provocative positions |
| MRI of brachial plexus | Neural compression, fibrous bands, soft tissue abnormalities |
| CT scan (sagittal) | Interscalene triangle dimensions (AS and MS muscles), surgical planning |
Electrodiagnostic Studies
- Nerve Conduction Studies (NCS): In true neurogenic TOS — reduced amplitude of medial antebrachial cutaneous sensory nerve; reduced ulnar sensory/motor amplitudes
- EMG: Denervation in C8–T1 muscles (first dorsal interosseous, abductor pollicis brevis)
- NCS may be normal in disputed neurogenic TOS
Vascular Studies
- Venography: Gold standard for diagnosing subclavian vein thrombosis in vTOS
- Arteriography: For evaluation of arterial stenosis/aneurysm in aTOS
Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| Cervical disc herniation (C7–T1) | Dermatomal pain, Spurling's sign, MRI disc pathology |
| Carpal tunnel syndrome | Median nerve distribution; positive Tinel's/Phalen's at wrist |
| Cubital tunnel syndrome | Ulnar nerve at elbow; positive Tinel's at elbow |
| Pancoast tumor | Apical lung mass on imaging; Horner's syndrome |
| Brachial neuritis (Parsonage-Turner) | Acute onset, severe pain then weakness, no positional component |
| Rotator cuff pathology | Localized shoulder pain; MRI shoulder findings |
| Raynaud's disease | Bilateral, no vascular lesion, no arm claudication |
| DVT / upper extremity thrombosis | Confirmed on Doppler/venography |
| Multiple sclerosis | CNS signs, MRI brain/spine lesions |
Treatment
Conservative Management (First-line for Neurogenic TOS)
- Postural correction — strengthen neck and shoulder stabilizers, correct forward head posture
- Scalene and pectoralis minor stretching — reduce compression
- Shoulder girdle strengthening — trapezius, rhomboids, serratus anterior
- Breathing exercises — avoid accessory muscle overuse
- Duration: 6–12 weeks minimum; success rate ~60–70% in disputed nTOS
- NSAIDs — for inflammatory/myofascial pain component
- Muscle relaxants — cyclobenzaprine, baclofen (short-term)
- Neuropathic pain agents — gabapentin, pregabalin (for neurogenic pain)
- Anticoagulation — for venous TOS with thrombosis (heparin → warfarin or DOAC)
- Thrombolytics — catheter-directed thrombolysis in acute vTOS
- Injected into anterior scalene muscle
- Temporarily relieves scalene muscle compression
- Useful diagnostically and therapeutically; effects last 3–6 months
Surgical Management
- True neurogenic TOS — surgical resection of the anomalous band is definitive treatment
- Failed conservative therapy (6–12 months) in neurogenic TOS
- All arterial TOS — needs vascular reconstruction
- Venous TOS (after thrombolysis) — to prevent recurrent thrombosis
| Approach | Description | Best Indication |
|---|---|---|
| Transaxillary First Rib Resection | Rib removed via axillary incision; no scalenectomy | Costoclavicular/venous TOS |
| Supraclavicular Approach | Direct access to scalene muscles, anomalous band, brachial plexus | True neurogenic TOS; arterial TOS |
| Infraclavicular Approach | Access to distal subclavian/axillary vessels | Arterial reconstruction |
| Combined Approaches | When multiple pathologies present | Complex cases |
- Resection of anomalous fibrocartilaginous band — for true nTOS (halts progression)
- Scalenectomy / scalenotomy — release of anterior (and sometimes middle) scalene
- First rib resection — decompresses costoclavicular space
- Cervical rib excision — when present and causative
- Vascular reconstruction — bypass or patch angioplasty for subclavian artery aneurysm/stenosis in aTOS
- Catheter-directed thrombolysis + first rib resection — staged management of Paget-Schroetter syndrome
Prognosis
| Type | Prognosis |
|---|---|
| True neurogenic TOS | Surgery halts progression; muscle wasting and weakness typically do not recover (Harrison's, p. 629) |
| Disputed neurogenic TOS | 60–80% improve with PT and conservative care; ~10–15% require surgery |
| Venous TOS | Good with timely thrombolysis + decompression; recurrence rare after rib resection |
| Arterial TOS | Good with surgery; untreated → high risk of digit loss/gangrene |
Complications
| Complication | Details |
|---|---|
| Permanent muscle atrophy | Thenar/hypothenar wasting in true nTOS — irreversible even after surgery |
| Upper extremity gangrene | From subclavian artery aneurysm and distal embolization |
| Pulmonary embolism | From subclavian/axillary vein thrombosis |
| Post-thrombotic syndrome | Chronic arm swelling and pain after venous TOS |
| Phrenic nerve injury | Rare surgical complication (supraclavicular approach) |
| Pneumothorax | Rare surgical complication |
| Brachial plexus injury | Rare but serious surgical complication |
Key Takeaways
- TOS = compression of neurovascular bundle (brachial plexus, subclavian artery/vein) at the thoracic outlet
- Neurogenic TOS (95%) is the most common — true (objective) vs. disputed (no objective findings)
- True neurogenic TOS is caused by an anomalous fibrocartilaginous band from the C7 transverse process to the first rib
- Cervical rib is the most recognizable anatomical risk factor; present in <1% of population
- Provocative tests (Roos, Adson, Wright) support diagnosis but pulse obliteration alone is not diagnostic
- Physical therapy is first-line for neurogenic TOS; surgery for true nTOS, arterial TOS, and refractory cases
- True neurogenic TOS: surgery stops progression but does not reverse muscle wasting
- Venous TOS (Paget-Schroetter) = effort thrombosis in young athletes — treat with thrombolysis + rib resection
- Arterial TOS is rare but most dangerous — risk of gangrene if untreated
expain in deatil boyulism in deatil
botulism pathophysiology Clostridium botulinum toxin mechanism types
botulism clinical features diagnosis treatment management antitoxin
botulinum toxin neuromuscular junction mechanism diagram
Botulism — Detailed Overview
Definition
The Causative Organism
Clostridium botulinum
| Feature | Detail |
|---|---|
| Gram stain | Gram-positive |
| Morphology | Rod-shaped bacillus |
| Oxygen requirement | Obligate anaerobe |
| Spore formation | Yes — forms highly resistant endospores |
| Spore resistance | Survives boiling (100°C); destroyed by autoclaving (121°C for 3 min) |
| Habitat | Ubiquitous in soil, dust, marine sediment, animal intestines |
| Toxin serotypes | 7 serotypes (A through G); human disease caused by A, B, E, F |
Related Species (Rare Causes)
- C. baratii — produces type F toxin; causes infant botulism
- C. butyricum — produces type E toxin; reported in Italy and India
The Toxin
Potency
- Estimated lethal oral dose (70 kg man): ~70 µg
- Estimated lethal inhaled dose (70 kg man): ~0.80–0.90 µg
- Classified as a Category A bioterrorism agent by the CDC
Toxin Structure
- A 150 kDa di-chain protein consisting of:
- Heavy chain (100 kDa) — binds to presynaptic nerve terminal receptors (e.g., SV2C for BoNT/A)
- Light chain (50 kDa) — zinc-dependent endopeptidase (the toxic component)
Toxin Serotypes and Clinical Relevance
| Serotype | Main Route | Key Association |
|---|---|---|
| Type A | Foodborne, wound | Most severe; longest duration of paralysis; Western USA |
| Type B | Foodborne, infant | Eastern USA; home-preserved vegetables |
| Type E | Foodborne | Fish products; Alaska, Great Lakes region |
| Type F | Infant, rare foodborne | Rare; C. baratii associated |
Pathophysiology
Conditions Required for Toxin Production
- Anaerobic environment
- Low acidity (pH > 4.5)
- Low salt and sugar content
- Temperature: 3°C–37°C (37°F–99°F)
Mechanism of Action — Step by Step
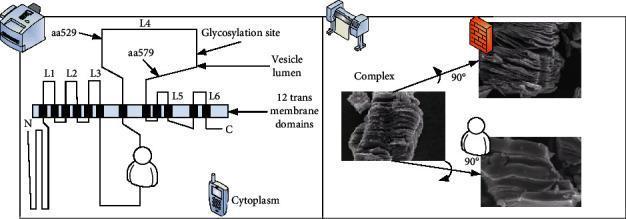
- The heavy chain of the toxin binds with high affinity to specific receptors on the presynaptic membrane of peripheral cholinergic nerve terminals (neuromuscular junctions, autonomic ganglia, parasympathetic nerve endings)
- BoNT/A and E bind SV2 (synaptic vesicle glycoprotein 2)
- BoNT/B, D, F, G bind synaptotagmin
- Toxin-receptor complex is endocytosed into an acidified endosome
- Acidic pH triggers conformational change in the heavy chain, forming a pore in the endosomal membrane
- The light chain is translocated through the pore into the cytoplasm
- The light chain acts as a zinc-dependent endopeptidase
- It cleaves SNARE proteins — the molecular machinery required for acetylcholine (ACh) vesicle docking and fusion:
| Toxin Serotype | SNARE Target Cleaved |
|---|---|
| A, C, E | SNAP-25 (synaptosomal-associated protein 25) |
| B, D, F, G | Synaptobrevin/VAMP (vesicle-associated membrane protein) |
| C (also) | Syntaxin |
- SNARE cleavage prevents ACh vesicle fusion with the presynaptic membrane
- Acetylcholine release is blocked at the neuromuscular junction
- Result: flaccid paralysis (lower motor neuron pattern — weakness without spasticity)
- Also affects autonomic cholinergic synapses → autonomic dysfunction
Types / Forms of Botulism
Overview Table
| Type | Source of Toxin | Primary Population | Mechanism |
|---|---|---|---|
| Foodborne | Preformed toxin ingested | All ages | Toxin absorbed from GI tract |
| Infant | In vivo toxin production | Infants < 12 months | Spores germinate in intestine |
| Wound | In vivo toxin production | IV drug users, trauma | Spores germinate in wound |
| Adult intestinal (Adult infant) | In vivo toxin production | Immunocompromised adults | Rare; intestinal colonization |
| Iatrogenic | Therapeutic/cosmetic injection | Any age | Overdose or spread of BoNT |
| Inhalational (Bioterrorism) | Weaponized aerosol | Any age | Airborne exposure |
1. Foodborne Botulism
Epidemiology
- Most recognized form in outbreaks
- In the USA: ~15–25 cases/year
- Type A and B from home-canned vegetables, meats
- Type E from fermented fish (Alaska Natives) and smoked fish
Common Food Sources
| Food | Toxin Type |
|---|---|
| Home-canned low-acid vegetables (green beans, corn, beets) | A, B |
| Home-canned meats, fish | A, B |
| Smoked or fermented fish (salmon, whitefish) | E |
| Cheese sauce, garlic-in-oil preparations | A |
| Prison pruno (homemade alcohol) | A |
| Fermented marine mammals (Alaska) | E |
Mechanism
- Preformed toxin is ingested in contaminated food
- Toxin is absorbed through the small intestine
- Enters systemic circulation → targets peripheral cholinergic nerve terminals
Clinical Course
- Incubation: 6 hours to 10 days (typically 12–36 hours)
- Shorter incubation = larger toxin dose = more severe illness
- Initial GI symptoms: nausea, vomiting, abdominal cramps, diarrhea
- Followed by descending flaccid paralysis (see Clinical Features below)
2. Infant Botulism
Epidemiology
- Most common form in the USA (~65–85% of all cases)
- ~100–150 cases/year in the USA
- Ages: 3 weeks to 12 months (peak: 2–4 months)
- Spores ingested from:
- Honey (most famous source — should NEVER be given to infants < 1 year)
- Soil (tracked in on shoes, clothing)
- Corn syrup (historical association)
- Environmental dust
Mechanism
- Ingested spores germinate in the intestinal lumen (infant gut lacks competing flora and protective acid)
- Toxin produced in situ → absorbed into bloodstream
Clinical Features
- "Floppy baby" syndrome:
- Constipation (often first sign — poor colonic motility)
- Weak cry
- Poor feeding, weak suck
- Hypotonia ("rag doll" appearance)
- Ptosis, expressionless face
- Loss of head control
- Decreased deep tendon reflexes
- Progression to respiratory failure if untreated
- Sudden infant death (SIDS) has been linked to undiagnosed infant botulism
Treatment
- BabyBIG (Botulism Immune Globulin Intravenous — Human) — FDA-approved specifically for infant botulism
- Antitoxin (equine) is NOT used in infants
- Supportive care; hospitalization
3. Wound Botulism
Epidemiology
- Rising incidence due to injection drug use (particularly black tar heroin)
- Also: traumatic wounds, compound fractures, crush injuries
- Type A predominates
- Incubation: 4–14 days from wound contamination
Mechanism
- Spores introduced into wound → anaerobic environment → germination → in vivo toxin production → toxin enters bloodstream
Key Difference from Foodborne
- No GI symptoms (toxin not ingested)
- Wound may appear deceptively benign or even well-healed
- Fever may be present if co-infected
High-Risk Groups
- Black tar heroin users (subcutaneous/intramuscular injection "skin popping")
- Patients with sinusitis from intranasal cocaine use (rare)
4. Adult Intestinal Toxemia (Adult Infant Botulism)
- Very rare; mirrors infant botulism in mechanism
- Occurs in adults with altered GI anatomy (bowel surgery, inflammatory bowel disease) or immunosuppression
- Spores colonize the adult intestine and produce toxin in situ
5. Iatrogenic Botulism
- Results from therapeutic or cosmetic use of botulinum toxin injections (Botox, Dysport, Xeomin)
- Usually from:
- Excessive doses
- Unintended spread to adjacent muscles
- Treatment of conditions like spasticity, hyperhidrosis, dystonia, cosmetic wrinkle reduction
- Symptoms: localized or regional weakness beyond intended area, dysphagia, diplopia
Clinical Features — The Classic Presentation
The "Four D's" + Descending Paralysis
- Diplopia (double vision)
- Dysarthria (slurred speech)
- Dysphonia (hoarse voice)
- Dysphagia (difficulty swallowing)
- Followed by descending symmetric flaccid paralysis
Cranial Nerve Involvement (Earliest Signs)
| Nerve | Manifestation |
|---|---|
| CN III, IV, VI | Diplopia, ptosis, ophthalmoplegia |
| CN VII | Facial weakness, expressionless face |
| CN IX, X | Dysphagia, dysphonia |
| CN XII | Dysarthria, tongue weakness |
Autonomic Features
- Dry mouth (xerostomia) — from blocked salivary gland cholinergic innervation
- Mydriasis (dilated, poorly reactive pupils) — key finding
- Constipation — reduced GI motility
- Urinary retention
- Orthostatic hypotension
- Anhidrosis (decreased sweating)
Motor Involvement — Descending Pattern
- Cranial nerve muscles (face, eyes, throat)
- Neck muscles
- Upper limbs
- Respiratory muscles (diaphragm, intercostals)
- Lower limbs
Respiratory Failure
- The primary cause of death in botulism
- Results from weakness of the diaphragm and accessory respiratory muscles
- Requires mechanical ventilation — may last weeks to months
- Patients must be monitored with serial vital capacity (VC) measurements
Key Features That DISTINGUISH Botulism from Other Disorders
| Feature | Botulism |
|---|---|
| Consciousness | Fully preserved (alert) |
| Fever | Absent (unless wound co-infection) |
| Sensory deficits | Absent (pure motor/autonomic) |
| Reflexes | Decreased/absent |
| Pupils | Dilated, poorly reactive (key!) |
| Pattern of paralysis | Descending, symmetric, flaccid |
Diagnosis
Clinical Diagnosis — High Suspicion Required
"Diagnosis of botulism depends on high clinical suspicion and a thorough neurologic examination. The timeliness of diagnosis is crucial to successful treatment."
Laboratory Confirmation
| Test | Method | Notes |
|---|---|---|
| Mouse bioassay | Inject patient serum/stool/food into mice; observe for paralysis | Gold standard; takes 24–96 hours; performed at CDC and state labs |
| ELISA (Endopeptidase assay) | Detects toxin activity | Faster; increasingly available |
| Stool culture | Isolation of C. botulinum | Positive in ~60% of foodborne, ~100% infant botulism |
| Serum toxin assay | Detects toxin in blood | Best within 24 hrs of symptom onset; negative result does not exclude diagnosis |
| Food culture | Culture suspected food item | Confirms foodborne outbreak |
Electrodiagnostic Studies (EMG/NCS)
| Finding | Significance |
|---|---|
| Normal nerve conduction velocity | Axons intact; demyelination absent |
| Reduced compound muscle action potential (CMAP) amplitude | Reduced ACh release at NMJ |
| Incremental response to rapid repetitive nerve stimulation (RRNS) | "Facilitation" at high-frequency stimulation (>50 Hz) — distinguishes from myasthenia gravis |
| Small, short motor unit potentials | Seen on EMG |
| Absence of sensory deficits on NCS | Confirms motor/autonomic process |
The EMG pattern of botulism resembles Lambert-Eaton Myasthenic Syndrome (LEMS) — both show facilitation at high-frequency stimulation — unlike myasthenia gravis, which shows decremental response.
Lumbar Puncture
- Normal CSF — helps exclude Guillain-Barré syndrome (which shows elevated CSF protein)
Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| Guillain-Barré Syndrome (GBS) | Ascending paralysis; sensory involvement; elevated CSF protein; absent pupil changes |
| Miller Fisher Syndrome | Triad: ophthalmoplegia, ataxia, areflexia; sensory changes; anti-GQ1b antibodies |
| Myasthenia Gravis | Fatigable weakness; acetylcholine receptor antibodies; DECREMENTAL response on RRNS; pupil spared |
| Lambert-Eaton Syndrome | Proximal limb weakness; facilitation on RRNS; VGCC antibodies; associated with malignancy |
| Stroke | Focal CNS signs; imaging findings; altered consciousness possible |
| Tick paralysis | Ascending paralysis; tick found on body; resolves after tick removal |
| Organophosphate poisoning | SLUDGE syndrome (salivation, lacrimation, urination, defecation); miosis (not mydriasis) |
| Eaton-Lambert | See above; paraneoplastic |
| Diphtheria | Palatal palsy first; demyelinating neuropathy; Corynebacterium history |
Treatment
1. Antitoxin (Cornerstone of Specific Therapy)
| Antitoxin | Type | Use |
|---|---|---|
| Heptavalent Botulinum Antitoxin (HBAT) | Equine-derived; covers types A–G | Adults and children ≥1 year; foodborne and wound botulism |
| BabyBIG (Botulism Immune Globulin IV — Human) | Human-derived; covers types A and B | Infant botulism ONLY; FDA-approved 2003 |
- In the USA, HBAT is available free of charge, 24/7 through the CDC Strategic National Stockpile
- Anaphylaxis risk with equine antitoxin — skin test before administration
2. Supportive Care (Critical)
- Mechanical ventilation — primary intervention for respiratory failure; may be required for weeks to months
- Close monitoring: serial forced vital capacity (FVC), negative inspiratory force (NIF)
- Intubate early if FVC < 30% predicted or declining
- Nasogastric/PEG tube feeding — for dysphagia and nutritional support
- Urinary catheterization — for urinary retention
- Bowel regimen — for constipation
- DVT prophylaxis — for immobile patients
- ICU care — monitoring for complications
3. Wound Botulism — Additional Management
- Surgical debridement of the wound to eliminate ongoing toxin production
- Antibiotics: Penicillin G or metronidazole
- Aminoglycosides are CONTRAINDICATED — they can worsen neuromuscular blockade
- Antitoxin administered before debridement (to prevent bacteremia-related toxin release)
4. Infant Botulism
- BabyBIG — given as a single IV infusion; reduces hospital stay by ~3 weeks
- Breast milk encouraged (provides immunological benefit)
- Antibiotics NOT routinely recommended — may lyse intestinal C. botulinum, causing sudden toxin release
5. No Role For
- Cathartics/enemas in foodborne botulism — may accelerate toxin absorption
- Guanidine hydrochloride — historically tried; not effective
- Edrophonium (Tensilon test) — negative in botulism (unlike MG)
Public Health and Reporting
- Botulism is a notifiable disease — immediately reportable to local/state health departments
- Foodborne botulism cases trigger food source investigation to prevent outbreak spread
- CDC and state health departments provide 24/7 emergency consultation and antitoxin delivery
- All suspected cases must be reported even before laboratory confirmation
Bioterrorism Potential
- Botulinum toxin is classified as a CDC Category A bioterrorism agent — highest priority
- Potential delivery: aerosolization or food/water contamination
- Inhalational botulism:
- Does not naturally occur
- Incubation: 12–36 hours after aerosol exposure
- Clinical picture identical to foodborne botulism
- Would present as a cluster of unexplained descending paralysis cases
- Strategic national stockpile of antitoxin maintained for mass casualty events
Prognosis
| Factor | Outcome |
|---|---|
| Treated cases (modern ICU care) | Mortality <5% |
| Untreated cases | Mortality up to 40–60% |
| Recovery | Slow — weeks to months; requires sprouting of new nerve terminals |
| Respiratory support | May be needed for months |
| Residual fatigue | Common for up to 1 year |
| Infant botulism | Excellent prognosis with BabyBIG; <2% mortality |
| Type A | More severe and longer duration than Type B or E |
Mechanism of Recovery
- Recovery does not occur by reversal of SNARE cleavage
- The nerve terminal sprouts new axonal branches with new SNARE proteins
- Original damaged terminal eventually recovers as well
- Process takes weeks to months
Prevention
| Strategy | Details |
|---|---|
| Proper canning technique | Pressure canning (121°C) destroys spores; boiling (100°C) does NOT |
| Boiling home-canned foods 10 min before eating | Destroys performed toxin (heat-labile) |
| No honey for infants < 1 year | Most important public health message |
| Refrigeration | Inhibits spore germination (below 3°C) |
| Acidification of foods | pH <4.5 prevents toxin production |
| Proper wound care | Prompt debridement of contaminated wounds |
| Avoid skin popping | Eliminate wound botulism risk from injection drug use |
Key Takeaways
- Botulinum toxin is the most potent biological toxin known — blocks ACh release at NMJ by cleaving SNARE proteins
- Results in descending flaccid paralysis + autonomic dysfunction (dry mouth, dilated pupils, constipation)
- Infant botulism is the most common form in the USA — never give honey to infants < 1 year
- Foodborne botulism is from preformed toxin in improperly preserved food; wound botulism increasingly from IV drug use
- Key clinical clues: alert patient + descending paralysis + dilated pupils + dry mouth + NO sensory loss + NO fever
- Antitoxin (HBAT) must be given as early as possible — it cannot reverse internalized toxin
- Respiratory failure is the primary cause of death — proactive mechanical ventilation is life-saving
- Aminoglycosides are contraindicated in wound botulism
- Recovery occurs through new nerve terminal sprouting — takes weeks to months
- All cases must be immediately reported to public health authorities
Write the V cranial nerve examination Write the assessment for VII cranial nerve Tests for visual acuity and visual field Explain glascom coma scale. Explain 3 higher mental functions. Mention 3 uses of lumbar puncture Mention 3 complications of lumbar puncture Internuclear ophthalmoplegia Mention 3 causes of ptosis Mention 3 causes of papilledema List 3 differences between UMN & LMN lesion. List three causes of Bell’s palsy Explain trigeminal neuralgia What is dysphagia? List 3 symptoms of ischaemic stroke. Explain autonomic dysreflexia. List the risk factors of stroke. Name three causes of stroke in young population Define cerebrovascular accident List 3 complications of head injury. What is postural hypotension? Name three causes of dementia List the causes of coma. List 3 types of epilepsy Name 3 common causes of seizures in elderly individuals Name three causes of dementia. List and briefly explain any 3 perceptual disorders Name 3 drugs used in the management of generalised seizures. Name three classical signs of Parkinson’s disease Name 3 drugs used in Parkinson’s disease Describe Chorea Define chorea Define ballismus List 3 causes of ataxia. What is Ataxia Talengiectasia Name 3 diseases affecting posterior column of spinal cord List the types of multiple sclerosis. List 3 clinical features multiple sclerosis List 3 clinical features of axillary nerve palsy. Mention 3 causes of wasting of small muscles of hand Mention 3 symptoms of radial nerve palsy Describe 3 complications of diabetic autonomic neuropathy List 3 clinical features of brain tumors Name three classical signs of Parkinson’s disease Name 3 drugs used in Parkinson’s disease List any 3 motor neuron diseases. Add a note on neuromyotonia Explain the etiology and pathophysiology of Myasthenia gravis Mention the clinical features of Radial nerve palsy What is Encephalitis Explain about transverse myelitis Define Cerebral Palsy and list its types List 3 clinical features of muscular dystrophy What is Hydrocephalus? List its types List any 3 complications of craniotomy List 3 indications of craniotomy Mention any 3 complications of Shunting Name 3 complications of laminectomy. List the complications of stereotactic surgery. What is shunting List 3 indications of shunting
trigeminal nerve CN5 examination sensory motor corneal reflex
facial nerve CN7 examination assessment motor taste
visual acuity visual field examination Snellen chart confrontation perimetry
Glasgow Coma Scale GCS eyes verbal motor score
higher mental functions orientation memory cognition neurological examination
lumbar puncture indications complications CSF
Quick Index
| # | Topic |
|---|---|
| 1 | CN V (Trigeminal) Examination |
| 2 | CN VII (Facial) Assessment |
| 3 | Visual Acuity & Visual Field Tests |
| 4 | Glasgow Coma Scale (GCS) |
| 5 | Three Higher Mental Functions |
| 6 | Three Uses of Lumbar Puncture |
| 7 | Three Complications of Lumbar Puncture |
| 8 | Internuclear Ophthalmoplegia (INO) |
| 9 | Three Causes of Ptosis |
| 10 | Three Causes of Papilloedema |
| 11 | UMN vs. LMN — Three Differences |
| 12 | Three Causes of Bell's Palsy |
| 13 | Trigeminal Neuralgia |
| 14 | Dysphagia |
| 15 | Three Symptoms of Ischaemic Stroke |
| 16 | Autonomic Dysreflexia |
| 17 | Risk Factors of Stroke |
| 18 | Three Causes of Stroke in Young |
| 19 | Define CVA |
| 20 | Three Complications of Head Injury |
| 21 | Postural Hypotension |
| 22 | Three Causes of Dementia |
| 23 | Causes of Coma |
| 24 | Three Types of Epilepsy |
| 25 | Three Causes of Seizures in Elderly |
| 26 | Three Perceptual Disorders |
| 27 | Three Drugs for Generalised Seizures |
| 28 | Three Classical Signs of Parkinson's |
| 29 | Three Drugs for Parkinson's Disease |
| 30 | Describe & Define Chorea |
| 31 | Define Ballismus |
| 32 | Three Causes of Ataxia |
| 33 | Ataxia Telangiectasia |
| 34 | Three Diseases of Posterior Column |
| 35 | Types of Multiple Sclerosis |
| 36 | Three Features of MS |
| 37 | Three Features of Axillary Nerve Palsy |
| 38 | Three Causes of Small Muscle Wasting |
| 39 | Three Symptoms of Radial Nerve Palsy |
| 40 | Three Complications of Diabetic Autonomic Neuropathy |
| 41 | Three Features of Brain Tumours |
| 42 | Three MNDs + Neuromyotonia |
| 43 | Myasthenia Gravis — Etiology & Pathophysiology |
| 44 | Radial Nerve Palsy — Clinical Features |
| 45 | Encephalitis |
| 46 | Transverse Myelitis |
| 47 | Cerebral Palsy — Definition & Types |
| 48 | Three Features of Muscular Dystrophy |
| 49 | Hydrocephalus — Definition & Types |
| 50 | Three Complications of Craniotomy |
| 51 | Three Indications for Craniotomy |
| 52 | Three Complications of Shunting |
| 53 | Three Complications of Laminectomy |
| 54 | Complications of Stereotactic Surgery |
| 55 | What is Shunting? |
| 56 | Three Indications for Shunting |
Answer is missing
- ✅ CN V (Trigeminal) examination
- ✅ CN VII (Facial) assessment
- ✅ Visual acuity and visual field tests
- ✅ Glasgow Coma Scale
- ✅ Three higher mental functions
- ✅ Three uses of lumbar puncture
- ✅ Three complications of lumbar puncture
- ✅ Internuclear ophthalmoplegia
- ✅ Three causes of ptosis
- ✅ Three causes of papilloedema
- ✅ Three differences UMN vs LMN
- ✅ Three causes of Bell's palsy
- ✅ Trigeminal neuralgia
- ✅ Dysphagia
- ✅ Three symptoms of ischaemic stroke
- ✅ Autonomic dysreflexia
- ✅ Risk factors of stroke
- ✅ Three causes of stroke in young
- ✅ Define CVA
- ✅ Three complications of head injury
- ✅ Postural hypotension
- ✅ Three causes of dementia (mentioned twice in your list — answered)
- ✅ Causes of coma
- ✅ Three perceptual disorders
- ✅ Three types of epilepsy
- ✅ Three causes of seizures in elderly
- ✅ Three drugs for generalised seizures
- ✅ Three classical signs of Parkinson's (mentioned twice — answered)
- ✅ Three drugs for Parkinson's (mentioned twice — answered)
- ✅ Describe Chorea
- ✅ Define Chorea
- ✅ Define Ballismus
- ✅ Three causes of ataxia
- ✅ Ataxia Telangiectasia
- ✅ Three diseases of posterior column
- ✅ Types of multiple sclerosis
- ✅ Three features of multiple sclerosis
- ✅ Transverse myelitis
- ✅ Three features of axillary nerve palsy
- ✅ Three causes of small muscle wasting
- ✅ Three symptoms of radial nerve palsy
- ✅ Clinical features of radial nerve palsy
- ✅ Three complications of diabetic autonomic neuropathy
- ✅ Three features of brain tumours
- ✅ Three motor neuron diseases + neuromyotonia
- ✅ Myasthenia gravis — etiology & pathophysiology
- ✅ Encephalitis
- ✅ Cerebral palsy — definition and types
- ✅ Three features of muscular dystrophy
- ✅ Hydrocephalus — definition and types
- ✅ Three complications of craniotomy
- ✅ Three indications for craniotomy
- ✅ Three complications of shunting
- ✅ Three complications of laminectomy
- ✅ Complications of stereotactic surgery
- ✅ What is shunting
- ✅ Three indications for shunting